Abstract
Background
Stroke patients with coexisting intracranial artery stenosis (ICAS) and white matter lesions (WML) usually have a poor outcome. However, how WML affects stroke prognosis has not been determined.
Objective
To investigate the quantitative forward flow at the middle cerebral artery in ICAS patients with different degrees of WML using 4D flow.
Design
Single-center cross-sectional cohort study.
Methods
Ischemic stroke patients with symptomatic middle cerebral artery (MCA) atherosclerosis were included, and they were divided into 2 groups based on Fazekas scale on Flair image (mild group = Fazekas 0-2, and severe group = Fazekas >2), TOF-MRA and 4D flow were performed to quantify the stenosis degree and forward flow at the proximal of stenosis. The flow parameters were compared between different white matter hyperintensity (WMH) groups, as well as in different MCA stenosis groups, logistic regression was used to validate the association between forward flow and WMH.
Results
A total of 66 patients were included in this study (mean age 56 years old, 68.2% male). 77.3% of them presented with WMH (Fazekas 1-5). Comparison of flow index between mild and severe WMH groups found a significantly lower forward flow (2.34 ± 1.09 vs 3.04 ± 1.35), higher PI (0.75 ± 0.43 vs 0.66 ± 0.32), and RI (0.49 ± 0.19 vs 0.46 ± 0.15) at ipsilateral infarction MCA in the severe WMH group, all P-values <0.05. After adjusting for other covariates, forward mean flow at ipsilateral infarction MCA is still associated with severe WMH independently, OR = 0.537, 95% CI (0.294, 0.981), P = 0.043.
Conclusion
Intracranial artery stenosis patients with coexisting severe WMH suffer from significantly decreased flow, which could explain the poor clinical outcome in this population, and also provide some insight into recanalization therapy in the future.
Plain language summary
Why was the study done? stroke patients with intracranial artery stenosis (ICAS) have a high prevalence of white matter hyperintensities (WMH), a surrogate biomarker of small vessel disease (SVD), and patients with coexisting ICAS and WMH are more likely to have unfavorable clinical outcomes and higher stroke recurrence risk. However, how WMH affects stroke outcomes has been unknown.
What did the researchers do? In this study, we compared the flow and perfusion index between different WMH groups, as well as in different ICAS groups using 4D flow combined with ASL, to obtain the quantitative flow relationship in this population.
What did the researchers find? As a result, we found that both the degree of intracranial artery stenosis and WMH burden is associated with decreased flow, and the flow decrease is more significant at the ipsilateral of infarct.
What do the findings mean? This is the first study investigating the complicated hemodynamic status using 4D flow combined with ASL in stroke patients with coexisting ICAS and WMH. The results in this study could not only provide some evidence for unfavorable clinical outcomes in ICAS patients with severe WMH burden but also give us some insight into recanalization therapy in this population.
Introduction
White matter hyperintensities (WMH), as a surrogate biomarker of small vessel disease (SVD), has a high prevalence in stroke patients with intracranial artery stenosis (ICAS), and patients with coexisting ICAS and WMH are more likely to have unfavorable clinical outcomes and higher stroke recurrence risk.1-3 However, a post hoc analysis of the Stenting and Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis (SAMMPRIS) study indicated that 49.5% patients coexisted with severe ICAS and SVD, but the presence of SVD has no significant effect on stroke recurrence in this population. 4 Thus, the effect of SVD on ICAS and potential mechanism is still inconclusive.
Both ICAS and SVD contribute to ischemic stroke risk, ICAS and SVD may represent different vessel pathology and play distinct roles in stroke occurrence. A deeply exploration and elucidation the mechanism behind the coexistence is critically important for ICAS management. Previous study demonstrated the association between intracranial large artery pulsatility and SVD. 5 SVD could possibly affect stroke prognosis by hemodynamic mechanism, thus the objective of this study was to investigate the association between the quantitative forward flow at MCA and the severity of WMH in stroke patients with ICAS.
Methods
Study design and subjects
This is a cross-sectional analysis of prospectively included subjects in a cohort study. The purpose of that cohort was to investigate the etiology and mechanism of intracranial artery disease. To be brief, quantitative flow at intracranial artery by 4D flow, luminal stenosis on MRA, and perfusion by Arterial spin labeling (ASL) were all obtained on one-stop MR scanner in this population. The protocol of this study was approved by the local ethics committee (22028-0-02), and all of the subjects participated signed the written informed consent.
Subjects were included into analysis if: (1) ischemic stroke confirmed by MR on DWI; (2) the infarct located at middle cerebral artery (MCA) supplying area, and was caused by MCA atherosclerosis. All included patients were on standard stroke care.
Subjects were excluded if: (1) patients with cardiac embolism; (2) intracranial artery disease caused by dissection, vasculitis, or Moyamoya disease. (2) patients with MCA occlusion, coexisting proximal extracranial artery stenosis or contralateral MCA stenosis; (3) subjects with atrial fibrillation and heart failure were also excluded; (4) patients with incomplete images or poor image quality.
The demographic characteristics, cardiovascular risk factors, NIHSS at admission, as well as the cognitive status (Mini-mental State Examination, MMSE) at admission were all recorded.
MR imaging protocol
A GE 3.0 T Discovery 750 MRI scanner (GE Healthcare, Milwaukee, USA) with an 8-channel head coil was used to scan patients
Image analysis
Two neuroradiologists with more than 5 years’ experience evaluated the image characteristics independently, in case of inconsistency, a third senior neuroradiologist would involve to come a consensus.
WMH was evaluated according to Fazekas scale, which has been established have a good consistency in WMH assessment.6,7 Both periventricular (pWMH) and deep white matter hyperintensities (dWMH) were evaluated separately, the scores range from 0 to 3 according the severity and WMH numbers (pWMH: 0 = no lesion, 1 = caps or pencil-thin lining, 2 = smooth halo; 3 = irregular, extending into the deep white matter; dWMH: 0 = no lesions, 1 = punctate foci, less than 4 lesions; 2 = beginning confluent foci, 5-9 lesions, 3 = confluent lesions, more than 9 lesions). We divided the WMH as mild (≤2 points) and severe (>2 points) groups based on total WMH score in this analysis. Patients with WMH were further sub grouped as pWMH-dominant or dWMH-dominant according to the severity and location of white matter lesion.
MCA stenosis degree measurement was diagnosed on TOF-MRA based on WASID criteria, 8 and were divided into different groups (<30%, 30%–49%, 50%–69%, and ≥70%) in this analysis.
Quantitative flow (maximum, minimum, and average flow) at the proximal of stenosis was obtained from 4D flow, contralateral flow at the symmetric location were acquired simultaneously (Figure 1). Pulsatility index (PI) and Resistance index (RI) of each side were calculated based on the formula defined, with PI = (systolic max flow-diastolic min flow)/(mean flow) and RI=(systolic max flow-diastolic min flow)/(systolic max flow).
9
The 4D flow processing including preprocessing (including correction for eddy currents, noise, and velocity aliasing or phase unwinding, then calculate a three-dimensional phase-contrast MRA and vascular segmentation), visualization, and quantification, which is a standard flow processing protocol using GT Flow, version 2.2.15 (Gyro Tools, Zurich, Switzerland), the corresponding processing details were also provided in supplemental materials. Quantitative flow measurement by 4D flow (pathline of 4D flow, the color denotes the quantitative flow in A, and the contour of measurement is set at the bilateral symmetrical position , shown in B and C).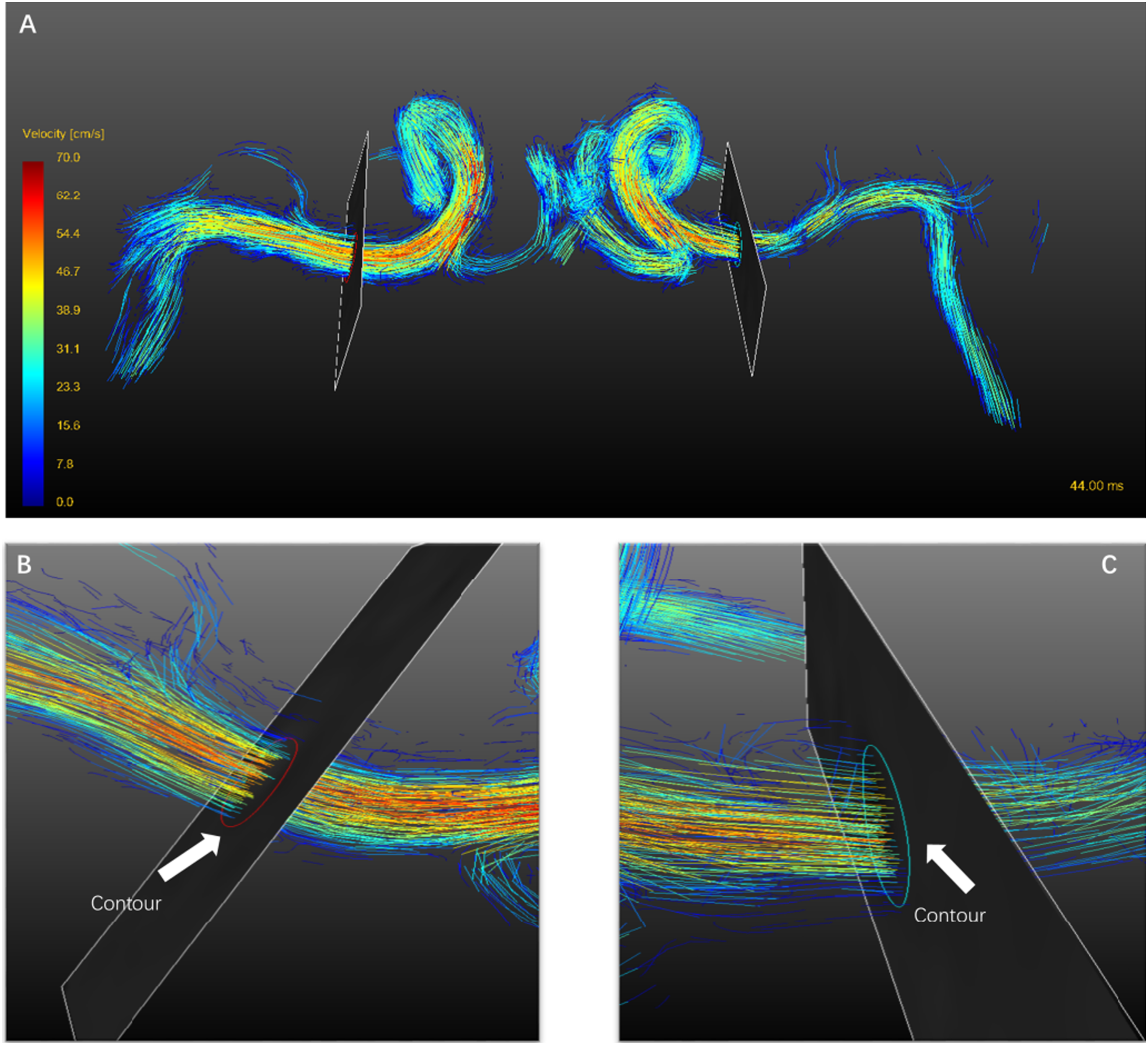
Several symmetrical ROIs (after excluded infarct area) at basal ganglia and semioval centers of the middle cerebral artery supply region were used to calculate mean CBF in each hemisphere using the GE workstation.
Collaterals were evaluated from the TOF-MRA source images as established criteria, which is a semi-quantitative scale ranges from zero to 3 (0 = almost no vascular signals in the lesion MCA territory; 1 = decreased collaterals with vascular signal intensity in the lesion MCA territory <50% than the contralateral side; 2 = decreased collateral but vascular signal intensity ≥50% of the reference side; and 3 = the vascular signal intensity is equal to or more than the reference side). We defined 0-1 as poor collaterals and grade 2-3 as good collaterals in this analysis.
Statistically analysis
Continuous variables with normal distribution were described as Mean ± SD, or Median (Inter-quartile range, IQR) if skewed distribution. t test was used to compare age, blood pressure index, flow index and CBF between WMH groups, and Mann-Whitney U test were used to test the statistical differences in NIHSS, MMSE between groups, one-way ANOVA was used to compare the flow index among different stenosis degree groups, LSD for multiple comparisons correction in post-hoc analysis; Categorical variables (sex, and cardiovascular risk factors) were presented as number and percentages, chi-square test was used to comparison between groups. In logistic analysis, boxplot was used to describe the continuous variables distribution, and collinearity diagnosis was tested to make sure the model reliability, Box-Tidwell was used to test the linearity of the logit between continuous independent variables (age and ipsilateral average flow) and the logit transformation of the dependent variable, then binary logistic regression analysis was used to adjust confounding factors and locate the association between forward flow and WMH, we adjusted confounding variables with P < 0.1 in univariate analysis. All statistical analysis were performed on SPSS 22.0. P values <0.05 were considered as statistically significant.
Result
From March 2017 to Oct 2020, a total of 107 patients were included in this cohort, of which, 66 patients satisfied the criteria above were included into analysis, the mean time from stroke onset to MR imaging was 12 (range from 9 to 17) days. The flow chart of subjects screening was shown in Figure 2. Subjects included and excluded demonstrated no significant difference in demographic characteristics, cardiovascular risk factors, and MCA stenosis degree (supplemental Table 1). Flow chart of subjects screening.
Basic characteristics of subjects included
The mean age of included subjects was 56 years old, 68.2% of them were male. WMH scores distribution were shown in Figure 3. 31.8% (21/66) included patients presented with severe WMH (score >2). Basic characteristics of subjects with mild and severe WMH were compared between groups (Table 1). As the results shown, subjects with severe WMH are much older (62 vs 53 years old in each group, P < 0.05), more likely to have hypertension (76.2% vs 48.9% in each group, P < 0.05). No other significant difference in cardiovascular risk factors, stroke severity (NIHSS), and cognitive scale (MMSE) was detected. WMH score distribution (x-axis is the total score of WMH, y-axis is the number of patients in each WMH score groups). Baseline characteristics of subjects with different WMH groups. SBP: systolic blood pressure; DBP: diastolic blood pressure. aZ value in Mann-Whitney test. bMedian (IQR), Mann-Whitney test.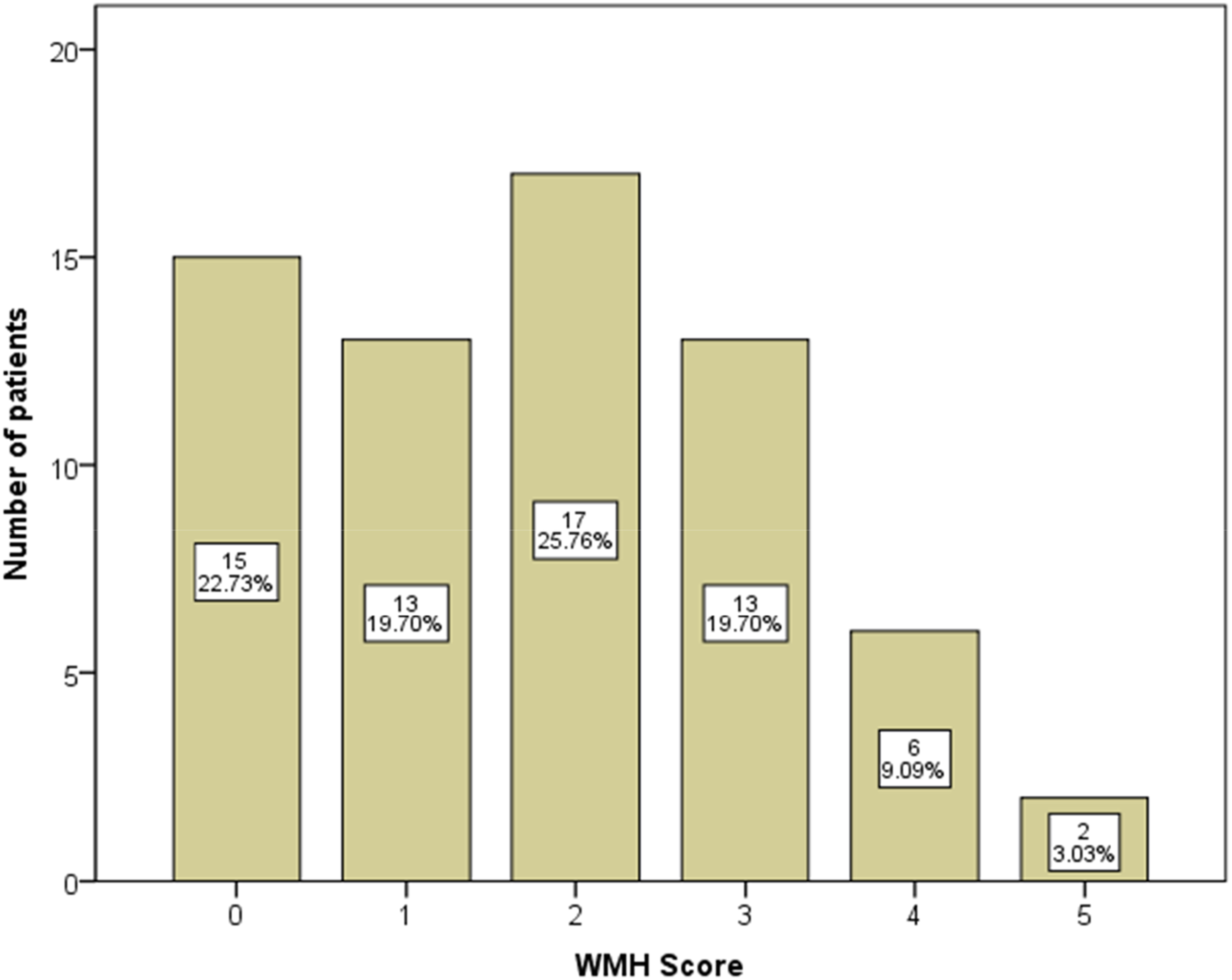
Association between quantitative forward flow and WMH
We compared the degree of MCA stenosis, quantitative forward flow at MCA, brain tissue perfusion, and collaterals between different WMH groups. The results suggested a significantly decreased flow, increased PI and RI at ipsilateral infarction MCA in patients with severe WMH compared with those with mild WMH (P < 0.05, shown in Figure 4). Despite a slightly decreased flow at contralateral MCA in patients with severe WMH, no significant statistical significance was detected. Nor any difference in the degree of stenosis, CBF, and collaterals, were found between the 2 groups (Table 2). Ipsilateral flow (A), PI (B), and RI (C) in different WMH groups. Stenosis, blood flow and perfusion parameters in different WMH groups. PI: pulsatility index; RI: resistance index.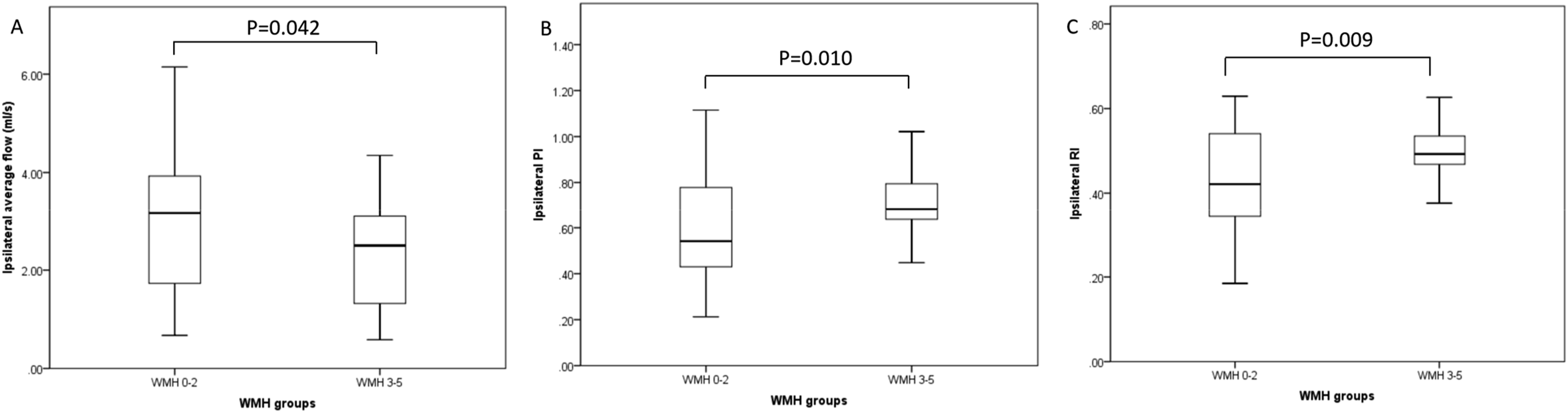

Multivariate logistic regression about forward flow and WMH.

Association between forward flow and large artery stenosis in different WMH groups
Figure 5 demonstrated the quantitative flow in patients with different degrees of MCA stenosis (F = 2.926, P = 0.041), we can see a significant decreased flow in patients with severe stenosis (>70%), post hoc tests demonstrated that the flow decrease in patients with moderate stenosis (50%-69%) is not so significant compared with patients with mild stenosis (30%-49%), P = 0.863, and no significant stenosis (<30%), P = 0.305. Moreover, we also compared the correlation between stenosis and flow in different WMH subgroups, as shown in Table 4, a decreased flow in severe MCA stenosis can be seen in both WMH groups, but the proportion of reduction is much greater in patients with severe WMH group compared with the other group, with 63.0% vs 29.7% in each group. Association between MCA stenosis and quantitative flow. Association between forward flow and stenosis in different WMH groups.

Decreased flow and WML location
Association between flow parameters and WMH distribution.

Discussion
In this study, we investigated the association between quantitative forward flow at MCA and WMH (both severity and location) in subjects with symptomatic MCA atherosclerosis, as well as the association between quantitative flow and the degree of MCA stenosis. And the main findings were: (1) a high prevalence of WMH could be seen in ischemic stroke with ICAS; (2) the decreased forward flow at symptomatic vessel is associated with both the severity of ICAS and WMH burden; (3) no significant association between decreased flow and WMH location was found.
ICAS coexisted with WMH
We detected a high prevalence of WMH in patients with ICAS, this is consistent with previous study about SVD burden in ICAS. 4 Despite the increasingly recognized association between SVD and large artery atherosclerosis in different populations, the potential mechanism behind has not been well understood. Most of them attributed it as shared cardiovascular risk factors. However, the presumed distinct pathological mechanism between large artery atherosclerosis and SVD are significantly different, whether the WMH coexisting with ICAS is an isolated phenomenon different from SVD in pathology, or have similar pathological mechanism with SVD deserve further study.
Quantitative MCA flow at ipsilateral infarction and WMH
One of the most important findings in our study was that we can see a significantly decreased flow at intracranial artery in patients with severe WMH, especially at ipsilateral infarction, which has never been investigated before. Previous studies on the association between cerebral blood flow and WMH volume are quite conflicting. 10 A meta-analysis about cerebral blood flow and SVD demonstrated that CBF is lower in subjects with severe WMH cross-sectionally, but evidence for falling CBF predating increasing WMH is conflicting. 11 Moreover, the CBF derived from perfusion could also be affected by other factors, which is also different from forward flow at MCA. WMH, as 1 of the most common SVD markers, could have a variety of pathology mechanism. The most widely recognized view is that WMH are caused by chronic hypoperfusion, impaired cerebrovascular reactivity (CVR) or blood-brain barrier (BBB) leakage. 12 However, both human and experimental studies suggest that hypoperfusion is mainly a consequence of decreased metabolic demand and not the cause of tissue damage. Our cross-sectional analysis validated the association between decreased forward flow in MCA and WMH after adjusted for recognized risk factors, but only in ipsilateral infarction, not contralateral side, which indicated potential BBB and microcirculation dysfunction at ipsilateral infarction. Further study is still needed to validate the causality relationship.
An increased PI and RI at ipsilateral infarction MCA could also be seen in the severe WMH group. Previous studies found a significant correlation between cerebroarterial pulsatility and WMH in the older population, 9 as well as in acute stroke patients. 13 Rotterdam study also demonstrated a positive association between increased arterial stiffness and larger volume of WML. 14 We did find the difference of RI and PI in patients with different WMH severity at ipsilateral infarction MCA, but not at contralateral infarction. However, only stroke patients with ICAS were included in our study, the PI and RI could also be affected by the degree of stenosis, their clinical significance can be interpreted by multiple factors.
Intracranial large artery stenosis and quantitative flow
The results of this study demonstrated a significantly decreased flow in patients with severe MCA stenosis (≥70%), this could explain the correlation between the degree of stenosis and stroke recurrence in symptomatic ICAS population.8,15 In addition, patients with severe WMH have a considerably higher proportion of flow decrease. It’s established that WMH is a microcirculatory disease, and was associated with an increased risk of stroke, dementia, and death. 16 The forward net flow depends on the pressure difference before and after stenosis, both the stenosis degree and distal small vessel resistance contribute to the pressure balance. Hypoxia could result in microcirculation dysfunction, increased endothelial cell contraction under hypoxia contribute to increased microcirculation resistance, which lead to a decreased flow at ipsilateral infarction.
Intracranial large artery flow and WMH location
We also try to explore the correlation between ipsilateral infarction MCA forward flow and WMH location, but no remarkable findings. Previous study investigated cerebral blood flow and WMH lesion location in SVD patients using perfusion imaging, and the results suggested that patients with SVD have lower blood flow, and pWMH but not dWMH were associated with decreased regional cortical blood flow. 17 While their studies were conducted mostly in the elderly, some other cardiovascular risk factors could be confounded. As for the negative results in our study, we speculated that the general WML in our study were not so severe as in previous study. And obviously, the mean age of included subjects in our study were much younger. Lastly, the limited number of cases in each group could also possibly underestimate the association to some extent.
Implications and limitations
Our study could provide some insights into the hemodynamic mechanism in ICAS, also reveal some inner relationship between ICAS and SVD. Firstly, our study validated the feasibility of 4D flow in quantitative intracranial artery flow evaluation. Secondly, SVD characteristics could also have effects on intracranial large artery forward flow to some degree; Last but not least, the significantly decreased flow at ipsilateral infarction in ICAS patients with severe WMH could possibly explain the unfavorable clinical prognosis.
There are indeed some limitations about our study. Firstly, the sample size is not large enough, confounding factors could be existed in this analysis; Secondly, cause-effect relationship between large artery flow and WMH could not be concluded from the cross-sectional analysis. Further longitudinal study is still needed in the future. Third, no other SVD markers were involved due to the predefined imaging protocol. Lastly, no normalized brain volume and total cerebral blood flow was used to diminish the individual difference.
Conclusion
In patients with symptomatic MCA atherosclerosis, both the degree of stenosis and SVD burden is associated with the decreased forward flow at proximal of symptomatic vessel, the results in this study could not only provide some evidence for unfavorable clinical outcomes in ICAS patients with severe WMH burden but also give us some insight into recanalization therapy in this population.
Supplemental Material
Supplemental Material - Decreased flow in ischemic stroke with coexisting intracranial artery stenosis and white matter hyperintensities
Supplemental Material for Decreased flow in ischemic stroke with coexisting intracranial artery stenosis and white matter hyperintensities by Xiaowei Song, Wenwen Chen, Xihai Zhao, Zhuozhao Zheng, Zhenhua Sang, Rui Li, and Jian Wu in Journal of Central Nervous System Disease.
Footnotes
Acknowledgments
We thank those patients and their relatives for their participation, also those research coordinators who help the conduction of this study.
Author contributions
Xiaowei Song: conceptualization, study design, data acquisition, interpretation, and original draft preparation; Wenwen Chen: data acquisition, interpretation, and analysis; Xihai Zhao: methodology, image interpretation; Zhuozhao Zheng: methodology, imaging protocol, data interpretation; Zhenhua Sang: formal data analysis; Jian Wu: study supervision, manuscript review and editing, final approval of the submission, funding support; Rui Li: data interpretation, and funding support.
Ethical statement
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Key R&D Program of China (2023YFC2506600), Beijing Municipal Science Technology Commission (Z171100001017019), Tsinghua Precision Medicine Foundation (20219990033), Capital's Funds for Health Improvement and Research (2020-1-2241), High-level public health technical talents construction and training project (2022-1-006).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
