Abstract
Background
After standard care, 55%-75% of patients after stroke show a persistent paresis of the upper limb (UL). Assistive devices are developed to increase the patients’ level of independence in daily life.
Objectives
To investigate the potential of Functional Electrical Stimulation (FES) to assist object manipulation in activities of daily life.
Design
Seventeen patients after stroke were tested and analyzed in a randomized cross-over design.
Methods
Functional grasping was assessed by means of the Action Research Arm Test (ARAT) and the modified Box and Block Test (mBBT), in one session with and another without FES assistance. The order of sessions was randomized. Patients’ motivation was assessed after each session. Task performance and motivation were compared between conditions using the Wilcoxon test and subgroup analyses were performed for impairment severity by distribution-based mixed-factor analyses.
Results
When analyzing the total ARAT, FES did not effectively assist the overall performance (P = .142), but did assist the performance of objects of the Grasp category (P = .020). Impairment severity showed an interaction with the orthotic effect (P = .012), as severely impaired patients profited from FES assistance and mild-moderately impaired did not. When focusing on the more functional items of the ARAT (i.e., excluding scores from thumb-middle and thumb-ring finger combinations), there was a significant orthotic effect of FES on task performance (P = .023). Further, there was an orthotic effect for the number of transported blocks in the mBBT (P = .033), exclusively prominent in the group of severely impaired patients. Functional Electrical Stimulation did not increase the patients’ motivation (P = .959), which was high after both conditions.
Conclusion
Functional Electrical Stimulation has the potential to support object manipulation, but is dependent on impairment severity and object type. To observe a consistent orthotic effect, features of the stimulator should be further developed to generate appropriate grasps and forces across subjects and objects.
Trial Registration
The trial was registered with the German Clinical Trials Register (DRKS00025889).
Introduction
After stroke, the most frequent symptom is impairment of motor function, e.g., a paresis of the upper limb (UL) with a prevalence of 63% (when averaging existing prevalence studies1-7). Of these, 64% suffer from severe and 36% from mild-moderate UL impairments. 7 Over the rehabilitation course, improvements in UL structures and functions are observed (e.g., motor control, dexterity, muscle tone and proprioception).8,9 Typically, patients who achieve partial recovery show improvements in proximal joints (shoulder and elbow) rather than at the distal part of the UL (wrist and fingers). 10 This might be one reason why recovery of functioning often does not lead to more frequent use of the arm and hand during activities of daily living (ADLs), 8 and consequently leads to a learned non-use. 11 However, there is a great proportion of patients (55%-75%) showing a persistent paresis after participating in standard care.8,12 Such a remaining hemiparesis restricts the use of the UL in ADLs and thereby reduces the patient’s level of independence at home. Six months after stroke, only one in ten patients shows full recovery of UL functions, in terms of unrestricted use of the UL in ADLs. 9
Conclusively, there is a high number of patients whose UL function is insufficient for ADL-use, and another group of patients who partially recover, but experience a decline in UL function due to acquired non-use of the paretic limb. Assistive devices are developed to enable the former group and promote the latter group to use the paretic limb for ADLs. Existing assistive devices for hand movements incorporate mostly exoskeletons (e.g., soft robotic gloves13,14 and hand exoskeletons 15 ). However, technical advances of other rehabilitation devices, such as Functional Electrical Stimulation (FES), augment the possibilities of assisting UL functions. Functional Electrical Stimulation is a well-established method used in the context of stroke rehabilitation to regain impaired sensorimotor functions. 16 Typically, an electrical current is applied to the peripheral nerve in order to elicit a muscle contraction. This bypasses the neural structures impaired by stroke that would initiate or transmit the signal to induce movement. 17 Current advances of existing stimulators include a wireless connection between stimulator and control unit as well as the augmentation of single to multi-field electrodes. These 2 aspects form the basis for use of FES as an assistive system in everyday life. On the one hand, one’s movements are not obstructed by wires. On the other hand, different types of hand functions (e.g., power grasp, pinch grip) can be stimulated by means of the multi-field electrodes. The use of surface electrodes is generally less precise in recruiting the desired muscles, in particular in the region of the forearm where there is a high density of muscles. However, the possibility to stimulate controlled movements has been shown to be increased by surface multi-field electrodes. 18
In general, FES applications can be divided into orthotic and therapeutic applications, depending on their intention and possibility to replace or retrain functions, respectively.19,20 Therefore, FES could induce orthotic effects (task performance with FES support vs unsupported) or therapeutic effects (task performance before vs after FES intervention). Nevertheless, the orthotic use of assistive devices throughout the day could still induce therapeutic effects on motor recovery. Preliminary results of the application of an assistive robotic glove at home indicated that both the orthotic and the therapeutic use of the glove resulted in comparable gains in UL function. 13 Furthermore, it has been suggested that the orthotic application of FES might play a role in patients’ motivation, 21 which could positively influence motor learning. 22
The therapeutic effects of FES are well studied, showing positive effects on UL activity. 23 Up to now, evidence about the orthotic effectiveness of FES after stroke has been limited to the lower limb. 24 Studies addressing the orthotic effectiveness for the UL are lacking, with the exception of one single-case investigation on the assistive potential of an implanted stimulation electrode. 25 To fill this gap, the potential of FES to assist patients after stroke in the functional use of the hand was studied in the following trial with the additional aim of (1) characterizing potential responders to the orthotic effect, (2) determining which object manipulations can be effectively assisted, and (3) investigating the role of FES as a motivator. As a great proportion of patients after stroke with an UL paresis need assistance to hold objects due to reduced strength and impaired hand opening, 26 we hypothesize an orthotic effect of FES on functional object manipulation.
Methods
This trial is reported according to the CONSORT 2010 reporting guidelines. Ethical approval was attained from the Ethics Committee of the Ludwig-Maximilians University (LMU) Munich, Germany (registration number: 21-0270). Prior to study conduction, the trial was registered with the German Clinical Trials Register (drks.de). The trial protocol can be accessed via the registration number DRKS00025889.
Trial Design
Applying a cross-over design (Figure 1), patients served as their own controls for testing the orthotic effect of FES during grasping. Participants attended 2 sessions which included functional grasping, tested by means of the Action Research Arm Test (ARAT) and the modified Box and Bock Test (mBBT). Both sessions were identical, but FES support was provided in only one session. In order to reduce sequence and learning effects between sessions, the starting condition (FES support vs no support) was randomized with an allocation ratio of 1:1. After functional grasping, participants answered questions on their motivation during the performed tasks by means of the Intrinsic Motivation Inventory (IMI). Scheme of the randomized cross-over study design.
In addition to the sessions, demographic and clinical data were assessed once, and 3 appointments were scheduled, one for the individual FES calibration and 2 for familiarization with FES-assisted grasping. During the familiarization sessions, patients repeatedly practiced the interaction with differently sized and shaped real-world objects while receiving the assistive stimulation. Hereby, patients became familiar with the ramping up time of the stimulation, the feeling of being guided by the stimulation and the stimulated end position of the fingers. The aim of these sessions was to reduce the influence of the cognitive abilities during first-time FES-assisted grasping, such as the anticipation of the position of hand closure.
Participants
The inclusion and exclusion criteria specified the eligible study population and took device-related contraindications into account. Study-related inclusion criteria were (i) diagnosis of an ischemic or hemorrhagic stroke, (ii) functional impairments in wrist or fingers (MRC ≤4), (iii) age ≥18 years, (iv) cognitively able to follow instructions, (v) no or low level of pain in wrist or fingers of the impaired limb (Numeric Rating Scale <4), (vi) low to moderate level of spasticity in the affected limb (Modified Ashworth Scale ≤3), and (vii) ability to sit in a chair for the duration of the session. Study-related exclusion criteria were (i) pregnancy, (ii) severe psychiatric disorder, and (iii) no motion resulting from FES (e.g., due to atrophy or polyneuropathy), which were augmented by device-related exclusion criteria such as (iv) active implantable devices or other metal implants within the stimulated area, (v) severe epilepsy or frequent epileptic seizures in the past, (vi) cancer, and (vii) wounds in the application area of the electrodes. Prior to study inclusion, written informed consent was obtained from the subjects. Demographic and clinical data was partly routine clinical data and partly collected in a testing lab at the Schoen Clinic Bad Aibling.
Testing Conditions
Clinical tests to assess the activity capacity of the hand (i.e., ARAT and mBBT) were performed in 2 different support conditions: (1) FES support of hand opening and closing, and (2) no FES support. In the supported condition, FES was used as a neuroprosthesis, applying surface electrical stimulation to assist patients after a stroke with a hemiparesis during grasping activities.
Functional Electrical Stimulation was delivered by the Fesia Grasp system (Fesia Technology S.L., Spain), which includes a wireless stimulator, a fixation garment to keep the electrode in position, and a multi-field electrode (Figure 2). The multi-field electrode incorporates 32 different stimulation channels which can be activated in different combinations and with different intensities (amplitude in mA). The electrode is attached around the patient’s forearm and fixed by the garment. The stimulator is plugged into the respective component of the electrode and connected to a tablet via Bluetooth. The Fesia Grasp including (1) a wireless stimulator, (2) the fixation garment, and (3) the multi-field electrode.
With respect to the patient’s maximum tolerance level, the individual stimulation intensity was set as high as needed to observe the required motion. The stimulation frequency and pulse width were kept constant at 35 Hz and 250 µs, respectively, which was found to be appropriate to stimulate fatigue-resistant functional grasping.27-29 The ramp-up time of FES was fixed in the software and could not be set individually. During the calibration session, the required electrode configuration for each motion primitive (wrist, thumb, index and finger flexion/extension) was selected by activating 1 or 2 cathodes per motion primitive in the tablet app. In addition to choosing the location of the stimulation by activating a specific cathode, the intensity of each cathode could selectively be modified in order to stimulate the forearm nerves which innervate the target muscles in the desired way to observe different types of hand closure, such as the palmar grasp and pinch grip, and hand opening. The sequence of each movement (opening (3 sec) – closing (7 sec) – opening (3 sec)) was manually started by the assessor when the patient was ready to perform the task. The patient was also verbally guided by the assessor through the 3 sequences of the grasping protocol.
The assistive potential of FES to support hand functions in ADLs was investigated by comparing the patients’ performance in functional grasping supported by FES with functional grasping without hand support. The term activity (i.e., the ability to execute functional tasks) can be separated into the capacity for activity and the performance of activity in daily life.
30
(Functional) capacity describes the activity someone can do, and is typically measured by standardized assessments in laboratory settings. The performance of activity in daily life instead pronounces how someone actually behaves in the unstructured environment. Since assisting hand function after stroke by FES is a new approach, standardized assessments were chosen over the performance of tasks in the unstructured environment, where even more challenges need to be tackled (e.g., user’s intention detection). The ARAT (Figure 3) and the mBBT are both tests in a standardized, clinical environment that include ADL-like components of an activity such as grasping real-world objects and releasing or placing them. Independent of whether distal FES support was provided, participants were allowed to perform the functional task while compensating impaired elbow and shoulder functions of the paretic side by the non-paretic side (Figures 3B-D). Thus, the focus was narrowed to investigating the hand functions. However, the non-paretic hand did not support wrist or hand function by grasping the paretic forearm proximal of 2 markers, located at the radial and ulnar styloid processes. The action research arm test, (A) with all the objects that need to be grasped, reached and placed including (B) a cube from the Grasp category, (C) stacking a tube from the Grip category, and (D) a marble from the Pinch category. In pictures B-D, hand function is supported by FES and proximal function is assisted by the guidance of the unimpaired side.
In the ARAT, the patient’s ability to grasp an object, lift it and place it is tested. Several different objects are used which can be attributed to the categories Grasp (i.e., differently sized cubes, cricket ball, whetstone), Grip (i.e., stacking of tubes, glass, washer) and Pinch (i.e., differently sized marbles), depending on the configuration of the fingers during object manipulation.
As second test to evaluate functional grasping, the mBBT was used. The original Box and Block Test (BBT) is a validated test for UL dexterity. 31 In the modified version of the BBT, 32 16 blocks are arranged in 4 rows of 4 blocks on the paretic side (pictured in Figure 1). The instruction was to grasp one block at a time, and transfer it over the partition to the other compartment as fast as possible, but without dropping the block before the divider.
Outcomes
Primary outcome parameters were assessed during (ARAT and mBBT) or after (IMI) both support conditions. The 16 items of the ARAT were rated by the assessor on a four-point ordinal scale, ranging from 0 (no part of the task can be performed) to 3 (normal task performance). 33 Overall, 48 points can be achieved. Patients who improved by at least one point in the ARAT while receiving FES assistance were defined as responders. Items are grouped into the Grasp, Grip and Pinch categories by totaling the scores (maximum scores of 18, 12 and 18 points, respectively). Since 4 items of the Pinch category are not prominent in daily life and cannot be stimulated by the current device (grasping marbles with thumb and middle finger and with thumb and ring finger), the orthotic effect of FES on the ARAT score was additionally analyzed after excluding these 4 items (referred to as ARAT-4 from now on). In the mBBT, the main interest was in assessing the number of successfully transferred blocks. Further, the number of errors, in terms of unsuccessful grasping attempts or dropping the block before crossing the partition, and the time needed for task completion were recorded. The IMI is a reliable multidimensional questionnaire, used to assess the patient’s perception during the performance of an activity in a laboratory environment. For this trial, a total of 25 items from the dimensions Interest/Enjoyment, Perceived Competence, Effort/Importance, Pressure/Tension and Value/Usefulness were selected, each rated on a seven-point Likert scale ranging from 1 (not at all true) to 7 (very true). The overall score as well as the score of each dimension was calculated by averaging the values of each included item.34,35
As secondary outcomes, demographic and clinical data were collected either from the patient records or assessed directly prior to the sessions. Assessments performed included the Edinburgh Handedness Inventory to determine handedness, the MRC of wrist extensor and flexor muscles (ranging from 0 = no contraction to 5 = normal power) to reliably assess muscle power 36 according to the protocol of the Medical Research Council, 37 and the maximum grip strength of each hand (paretic and non-paretic) measured by a hand dynamometer (Kern MAP 130K1, KERN & SOHN GmbH, Germany).
Categorization of impairment level as mild-moderate and severe.

Additionally, the occurrence of any adverse events was documented.
Sample Size
There are no comparable effect sizes available to perform a sample size calculation. The planned sample size was set to 15-20 patients. This number is thought to be sufficient for statistical analyses necessary for a cross-over study on the orthotic effect of FES, while keeping the cost of running multi-array FES at a minimum.
Randomization
Using a random number table, randomization of the order of testing conditions was performed with an allocation ratio of 1:1 in order to compensate for potential carry-over effects. Before the first testing, each patient picked one of the sequentially numbered pieces of paper to determine the order of conditions.
Blinding
Due to nature of the trial, blinding was not possible. Since the stimulation was above sensory threshold, the patients could not be blinded by a sham stimulation. Further, the stimulation had to be manually triggered. As the orthotic effect was tested while performing the grasping tasks, the assessor could thus not be blinded either.
Statistical Methods
Demographic and clinical data is descriptively reported by mean (M) and standard deviation (SD) or median (Mdn) and 25th – 75th quartile (Q1-Q3). Ordinal data, such as the ARAT and IMI scores, were inferentially compared between support conditions using the Wilcoxon test. The same test was applied for the outcomes of the mBBT including the number of successfully transferred blocks, the relative error (i.e., number of ineffective grasping attempts divided by number of transferred blocks), and the relative transport duration in seconds per block (i.e., time needed for task completion divided by number of successfully transported blocks), which did not follow a normal distribution. Patients who did not transport any block or less than 25% (≙ 4 blocks) were not considered in the analysis of the transport duration because of the observation period was too short.
To investigate the orthotic effect of FES under consideration of the severity of the paresis, a non-parametric, distribution-based mixed-factor analysis 40 was applied with the factors severity (mild-moderate vs severe) and support condition (with FES vs without FES). Characteristics of responders to the orthotic effect were described.
Data was analyzed and visualized using IBM SPSS Statistics 27 and R Studio.
Results
Participant Flow
The participants’ flow through the study, including the enrollment, randomization and analysis, is visualized in Figure 4. The participants’ flow through the study.
Recruitment
Between September 2021 and April 2022, patients after a stroke admitted to the Schoen Clinic Bad Aibling were screened and recruited for study participation. The recruitment was terminated after the inclusion of the 20th patient.
Baseline Data
Baseline demographic and clinical characteristics.

Orthotic Effectivity and Responders
Functional Grasping in the ARAT
In the overall ARAT performance, median performance scores during FES support (14.0 (10.0-35.5) points) were equal to median scores without FES support (14.0 (2.5-36.0) points). There was no significant difference between support conditions (Z = −1.47, P = .142). Still, the median performance for items of the Grasp category was significantly improved from 5.0 (.0-16.5) to 8.0 (3.5-17.5) points (Z = −2.33, P = .020). For objects of the Grip and Pinch category, no statistically significant performance change resulted from FES support (Z = −1.52, P = .127 and Z = −1.32, P = .189, respectively). In the Grip category, the median score increased from 5.0 (2.5-9.5) to 6.0 (4.5-9.5) points by FES assistance, while the performance decreased from 5.0 (.0-10.0) to 1.0 (.0-9.0) point in the Pinch category. An analysis of which particular objects were effectively assisted showed that FES support was effective in 4 items of the Grasp category, while it rather hindered the grasping of 1 item of the Pinch category. 41
Regarding the orthotic effect of FES in ARAT-4, patients achieved a median score of 14.0 (9.5-30.5) points under FES support, while the performance was rated 2 points lower (Mdn = 12.0 (2.5-30.5)) when assessed without support. This FES-induced performance increase was statistically significant (Z = −2.28, P = .023).
As expected, subgroup analyses revealed that patients with a different level of UL weakness performed significantly different in the ARAT (factor severity for total ARAT score and all subscales: P < .001). Figure 5 shows that the median performance of patients with a mild-moderate hemiparesis (displayed in bright (blue)) was better than the scores of severely impaired patients (displayed in dark (blue)). This observation applies to the subscales of the ARAT (A-C) as well as to the total ARAT score (D). Importantly, Figure 5 shows an increase in the median score when supported by FES in the group of severely impaired patients, while the median performance of mild-moderately impaired patients did not improve when assisted by FES. On the contrary, the score even decreased for the total ARAT and for objects of the Grip and Pinch subscale for mild-moderately impaired patients. The participants’ performance in the Action Research Arm Test shown for mild-moderately impaired in blue and severely impaired patients in dark blue. Scores are displayed for the Grasp (A), Grip (B) and Pinch category (C) as well as for the total ARAT (D). Top y-axis ticks indicate the respective maximum score.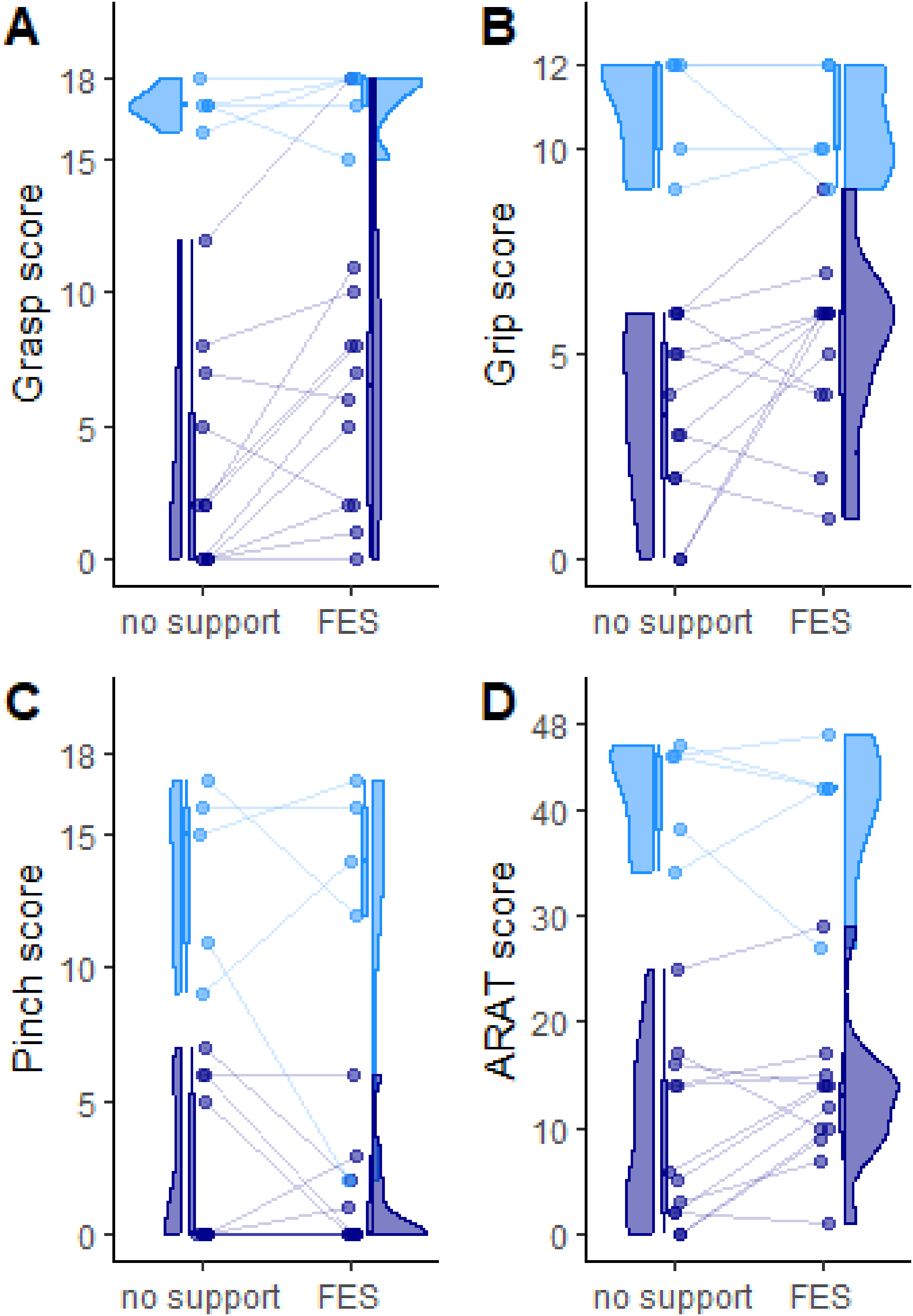
This observation was statistically analyzed by testing the interaction of the factors severity and condition. In the total ARAT score, the between-group factor condition, which defines whether or not FES support was provided, showed no significant performance increase (F(1) = 1.38, P = .241). The interaction between the factors severity and condition tended to significantly affect the total ARAT score (F(1) = 3.49, P = .062). Post-hoc Wilcoxon tests revealed no significant orthotic effect for mild-moderately impaired patients (MdnDiff = −3.0 points, P > .500), but a significant performance increase for severely impaired patients in the overall ARAT (MdnDiff = +7.5 points, Z = −2.16, P = .031, Figure 5D).
A similar trend in the interaction effect was observed for objects of the Grasp category (F(1) = 2.89, P = .089). Just as in the total ARAT score, post-hoc testing showed no significant orthotic effect in the Grasp score for mild-moderately impaired patients (MdnDiff = +1.0 points, P > .500), but a significant performance increase for severely impaired patients (MdnDiff = +4.5 points, Z = −2.36, P = .018, Figure 5A). Overall, the between-group factor condition led to a significant performance increase through the support by FES of items of the Grasp scale (F(1) = 9.08, P = .003).
In the Grip subscale, the factor condition was not statistically significant (F(1) = 2.37, P = .123). However, a significant interaction between severity and condition was found (F(1) = 4.43, P = .035). With a median increase of 2.5 points, patients with a severe hemiparesis experienced a positive orthotic effect of the FES, while the performance of mild-moderately impaired patients slightly decreased when supported by FES (MdnDiff = −2.0 points, Figure 5B). Post-hoc comparisons revealed a trend towards a significant orthotic effect of FES on grasping items of the Grip subscale in severely impaired patients (Z = −1.86, P = .063).
There was no significant interaction effect for objects of the Pinch category (F(1) = .01, P = .906, Figure 5C), nor was the factor condition statistically significant (F(1) = .68, P = .410).
In the ARAT-4 score, the severity of the paresis as well as the support condition turned out to be a significant factor that affected overall task performance (F(1) = 54.41, P < .001, F(1) = 3.94, P = .047, respectively). Further, there was a statistically significant interaction effect, indicating that FES support had a differential effect in mild-moderately impaired patients (Mdndiff = −3.0) compared to severely impaired patients (Mdndiff = +6.0; F(1) = 6.38, P = .012).
Characteristics of responders and non-responders according to the ARAT score changes.

Note. MRC: Medical Research Council.
Functional Grasping in the mBBT
Overall, patients successfully transported a median of all 16 blocks (0-16, n = 9 with the maximum of 16) in the unsupported condition as well as in the FES-supported condition, but in the latter with less variance between subjects (9-16, n = 12 with the maximum of 16). Patients with a severe hemiparesis increased their performance from a median number of 7.5 (0-16) blocks in the unsupported task to 16 blocks (.8-16) when assisted by FES. All mild-moderately impaired patients achieved the full 16 points in both conditions. There was a significant difference between the impairment levels (F(1) = 14.62, P < .001), with mild-moderately impaired patients scoring higher, as well as between the different support conditions (F(1) = 4.54, P = .033), evident in a higher number of transported blocks when FES support was provided. Further, there was a significant interaction effect between both factors, indicating that severely impaired patients transported a significantly higher number of blocks when FES support was provided (Mdndiff = +8.5) while mild-moderately impaired patients showed a ceiling effect (Mdndiff = +.0; F(1) = 4.54, P = .033), i.e., the median score of transported blocks reached the maximum of 16 blocks even without FES support.
Errors in task performance, such as improper grasping or losing the block before passing the partition, occurred in both support conditions. When performing the mBBT with FES support, patients showed a median relative erroneous task performance in 12.5% (6.3%-43.8%) of trials, while with a median proportion of 6.3% (.0%-31.3%) of trials, patients made less errors in the unsupported condition. The median relative erroneous number of trials was not significantly different (Z = −.67, P = .506). In severely impaired patients, a median of 40.7% (14.1%-54.7%) of transports were unsuccessful when grasping was assisted by FES and in mild-moderately impaired patients, the median proportion of unsuccessful transports was 6.3% (3.2%-12.5%). The median proportion of relative errors was insignificantly lower in the unsupported condition for patients with a severe hemiparesis (31.3%, 9.1%-74.3%, Z = −.11, P = .917), while it tended to be significantly lower for patients with a mild-moderate hemiparesis (.0%, .0%-.0%, Z = −1.86, P = .063).
Since a higher number of errors results in more grasping attempts to achieve the task completion, the relative transport duration is reduced. Thus, the following inferential analysis of transport duration includes the number of errors to a certain degree. The median duration for the transport per block was 8.4 (3.4-18.4) seconds per block when supported by FES. Without support, the median transport duration was longer with 10.1 (1.9-19.1) seconds per block. This prolongation was not statistically significant (Z = −.45, P = .657). While there was no significant FES-induced change in the relative transport duration in severely impaired patients (from 16.7 (10.8-26.1) to 16.3 (8.4-20.1) seconds per block, Z = −.52, P = .600), mild-moderately impaired patients showed a significantly longer relative transport time when supported by FES, apparent in a median increase from 1.9 (1.8-2.0) to 3.4 (2.3-9.6) seconds per block (Z = −2.02, P = .043).
The Role of FES as a Motivator
With a median value of 5.3 (4.1-5.6) in the FES condition and 5.5 (3.8-5.8) in the unsupported condition, the patients’ intrinsic motivation was high in both sessions and did not differ significantly (Z = −.52, P = .959). Specifically, FES support had no influence on the patients’ enjoyment of the task (Z = −.42, P = .678), the perceived competence (Z = −.62, P = .535), pressure (Z = −.43, P = .670), and task value (Z = −.18, P = .859). However, the FES support significantly lowered the perceived effort of patients while performing the functional tasks by a median of .5 points (Z = −2.24, P = .025). Especially, items on the enjoyment and value scale were rated exceptionally high. Patients enjoyed performing the tasks very much independent of the support condition (MdnFES = 6.8 (4.9-7.0), Mdnnone = 6.7 (5.3-7.0)) and expected the tasks to be of value for UL recovery (MdnFES = 6.2 (4.7-7.0), Mdnnone = 7.0 (3.6-7.0)).
Subgroup analyses (displayed in Figure 6) revealed a trend towards a significantly higher intrinsic motivation in mild-moderately impaired patients (F(1) = 3.14, P = .076), specifically prominent in significantly increased enjoyment (F(1) = 9.45, P = .002) and perceived competence (F(1) = 7.62, P = .006) as well as a significantly lower feeling of pressure (F(1) = 7.34, P = .007) compared to severely impaired patients. There was no significant interaction effect (P > .172). The participants’ responses to the Intrinsic Motivation Inventory shown for mild-moderately impaired in blue and severely impaired patients in dark blue. Scores are displayed for the subscales of the Intrinsic Motivation Inventory (A-E) as well as for the total scale (F).
Harms
There were no FES-related (serious) adverse events throughout the trial. Although patients with allergies to adhesive material were not recruited, 1 patient showed a skin rash within the stimulation area 1 day after setting the stimulation parameters. Therefore, study participation was paused until the irritation of the skin disappeared. After re-uptake of FES therapy, no more skin rash was observed, so it is questionable whether this adverse event was induced by the FES-application or was unrelated to study participation.
Discussion
Interpretation and Conclusion
FES support effectively assisted the performance of the ARAT-4 items. When analyzing the ARAT in full, including the ‘artificial’ pinch grips that could not be stimulated, patients did not show an orthotic effect in their performance. While there was an insignificant increase of the score in the Grasp category and an insignificant decrease in the Pinch category, FES showed the potential to effectively assist the manipulation of objects of the Grasp category. The finding that assistance hinders the interaction with small objects that need to be pinched has also been observed when assisting impaired fine motor functions with a soft-robotic glove after stroke 13 and after spinal cord injury. 42 Thus, assistive devices with the current state of technical development are more useful for gross motor activities and show difficulties in properly assisting fine motor functions.
In general, mild-moderately impaired patients performed significantly better in the ARAT than severely impaired patients. Regarding the ARAT-4 score, the use of FES as a neuroprosthesis was effective in the severely impaired group, while mild-moderately impaired patients did not profit from the FES support. This finding is consistent with another trial from our lab, in which the orthotic effect of FES in assisting patients after stroke during the performance of serious gaming therapy was investigated. 43 Taking all items of the ARAT into account, there was no interaction between severity and support. Still, severely impaired patients showed an orthotic effect while the performance of mild-moderately impaired patients decreased insignificantly. This finding applies to objects of the Grasp category. FES support failed in improving the performance on the Grip and Pinch scale for both impairment groups.
In conclusion, items of the Grasp category were supported most effectively and patients with a severe hemiparesis profited most from the use of FES. The responders to the orthotic effect consist of a higher proportion of patients from the group of severely impaired, in particular those with a lower MRC and less grip strength.
Based on observations that were documented by the assessor, there are 2 reasons for the inconsistent orthotic effect across items and patients. First, FES-generated movements were sometimes limited in the amount of grip force produced. For some patients and for some items, the generated force was not sufficient to lift the object or to close the hand far enough. Literature confirms the high variability between subjects in setting an appropriate stimulation intensity and selecting the location of the electrode, especially in patients after stroke. 44 Individual properties, such as the electrode-neuron distance and the conductivity of the tissue in between, or the diameter of the neuron in the muscle nerve, are factors that influence the resulting movement. 45 Sometimes, setting higher intensities would have been required for a better grasp generation, which was not possible due to painful sensations. Also, setting the stimulation parameters in a neutral position of the forearm changed the appropriateness of the stimulation for grasping movements in a pronated or supinated position, as described previously. 46 Second, the stabilization of objects using the thumb is crucial for grasping. However, functional thumb flexion was the most difficult motion primitive to program due to the absence of an electrode on the M. opponens pollicis for thumb opposition. Literature also indicates the difficulty to stimulate an appropriate and forceful movement of the thumb, 44 which leads to an unequal force distribution between the thumb and other fingers followed by the reduced possibility to stabilize objects when lifting them.
The potential of FES to assist the functional use of the hand was further investigated in the performance of the mBBT. There was a significant orthotic effect for the number of transferred blocks. With the support of FES, patients were facilitated to transport a higher amount of blocks, which is in line with the orthotic effect of a soft robotic glove in the BBT. 14 This orthotic effect was exclusively present in the group of severely impaired patients, predominantly obvious in three of these patients who were enabled to transport the maximum number of blocks only during FES support. Due to a ceiling effect in the group of mild-moderately impaired patients, who were all able to transport all blocks even without stimulation, FES could logically not increase their performance further. However, patients, especially those with a mild-moderate impairment, made more relative errors when supported by FES. Hence, they needed more grasping attempts resulting in a longer relative test duration in the FES support condition, which was significant in mild-moderately impaired patients. Another reason for the prolongation of the relative test duration was that the timing of the FES support for the different phases of the grasp was not adjustable for each individual. Therefore, patients were limited in increasing the speed in the FES condition.
Thus, FES has the potential to effectively support severely impaired patients during the mBBT, with the downside of more grasping attempts and the prolongation of time needed to perform the desired activity with FES support in patients with less severe deficits. Longer performance durations for tasks that are assisted by technologies have also been observed in previous studies, where, for example, robotic hand support was provided during the BBT14,47 or during ADL 48 for elderly participants, or cognitive support was given by Augmented Reality during the performance of ADL in patients with Alzheimer’s disease. 49
Observing an orthotic effect when using a device which was developed to support hand functions might not sound particularly interesting. However, the orthotic effect of FES has not been studied before, meaning that previous intervention studies investigated therapeutic effects of assistive devices which were not tested for their assistive capacity. The results of this trial provide insights regarding further improvements required for assistive FES, which should be considered before investigating therapeutic effects. Furthermore, severely impaired patients are not always target of orthotic applications. 13 The results of this trial should encourage the development of wearables for severely impaired patients, as they were responders to FES assistance.
Self-reported motivation was high in both conditions, with values above 5. The questions that address enjoyment and value of task performance were rated remarkably high, with values close to the maximum of 7. These values are almost identical to our previous investigation of the role of FES as motivator during serious gaming. 43 However, FES support did not increase the motivation of patients. It seemed that it is difficult for patients to discriminate between both conditions and they instead rated their motivation in the overall study participation. Nevertheless, the perceived effort was significantly lower when task performance was supported by FES. Interestingly, the subgroup analyses revealed that FES played a role as a motivator in the group of mild-moderately impaired patients, especially on the enjoyment, perceived competence and pressure subscale. Even though using the device did not increase their performance, the motivation was significantly higher when supported by FES. High motivation is important for intervention compliance 50 and thus, to potentially induce a therapeutic effect.
Generalizability
The characteristics of the analyzed patients allow the findings to be generalized to a broad scope of patients after stroke. In addition to an equal distribution of sex, the population was heterogeneous in terms of age, time since stroke (subacute and chronic), level of spasticity, and the degree of impairment, ranging from “no movement possible” to “anti-gravity movement possible with restricted force” on the MRC scale. However, the study population was restricted to inpatient participants. Thus, the results cannot be generalized to patients in later chronic stages, living at home. Additionally, severe spasticity was defined as an exclusion criterion. Effects might therefore not apply to patients with a value higher than 3 on the Modified Ashworth Scale. Subgroup analyses allow the findings to be applied to patients with a severe hemiparesis and patients with a mild-moderate hemiparesis. However, the generalizability of subgroup analyses is limited due to small sample sizes, especially for the group of mild-moderately impaired patients.
Limitations
Activities of daily living-relevant hand functions were assessed by means of the ARAT. One limitation of this test is its bottom and ceiling effects, 51 which were also present in this trial. However, bottom effects might be present in any clinical test that requires object manipulation in a severely impaired population. Still, the ARAT is a valid test to investigate hand functions in ADL-relevant tasks with good clinical properties, such as good external validity and very high intra-rater reliability. 52 Another limitation of the ARAT is that there is not yet a minimal clinically important difference established for orthotic effects, which would be useful to better define and characterize the responders to FES assistance.
Investigating the orthotic effect in the design of a randomized cross-over study could induce some biases which we aimed to minimize. Since the target outcome was the online effect and not any therapeutic effect, we consider the study design as suitable. Carry-over effects, induced by familiarization and learning, were minimized by having 2 FES training sessions before testing and by randomization of the start condition. Due to the very good compliance of the patients and no drop-outs after the first testing, there was always a full dataset available for analysis. However, blinding of patients was not possible in this design.
The ramp-up time of the stimulation and therefore the resulting movement velocity was rather fast and could not be changed. Thus, good anticipation was required to place the fingers around the object at the right moment. Logically, FES support assisted mainly motor functions and less cognitive functions, including anticipation. However, cognitive deficits were at least considered when grouping patients into mild-moderately and severely impaired.
A general limitation of using electrical stimulation is its list of contraindications. Patients with cancer, with active or metal implants, with epilepsy or with wounds in the application area must not use the device. In the population which was screened, these contraindications were present in 16%, 9%, 5% and 1%, respectively. Thus, an overall proportion of 31% of patients would not be able to use the orthosis – at least not within the precautionary measures taken to conduct a clinical trial.
Outlook
One cause for the non-consistent orthotic effect across patients and objects might be the use of the current version of the stimulator with its limitations. These limitations include the challenge to control complex FES systems in an automated way, using various sensors (e.g., electromyography) to enable physiological functionality. 53 Designing a smooth, sophisticated and functional UL FES system for assistive applications remains a challenge and is to date still in developmental stages with literature consisting mainly of feasibility studies or technical reports.54-57 In addition, the great majority of patients needed proximal support to perform the test. This emphasizes the need for the combination of systems for distal and proximal support, such as augmenting the distal FES by a proximal exoskeleton.
Footnotes
Acknowledgements
The authors thank Priscila Bonina Silva, Clinical Specialist at Fesia Technology S.L., for providing technical support for the stimulator, and Katie Göttlinger for copy-editing the manuscript.
Author Contributions
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has received funding from the European Union’s Horizon 2020 research and innovation programme ReHyb under grant agreement no 871767.
Ethical Statement
Data Availability Statement
Patient data has to be anonymized and will therefore not be uploaded. Authors can be contacted for individual data requests.
