Abstract
Epilepsy is a chronic neurological disorder manifested by recurring unprovoked seizures resulting from an imbalance in the inhibitory and excitatory neurotransmitters in the brain. The process of epileptogenesis involves a complex interplay between the reduction of inhibitory gamma-aminobutyric acid (GABA) and the enhancement of excitatory glutamate. Pro-BDNF/p75NTR expression is augmented in both glial cells and neurons following epileptic seizures and status epileptics (SE). Over-expression of p75NTR is linked with the pathogenesis of epilepsy, and augmentation of pro-BDNF/p75NTR is implicated in the pathogenesis of epilepsy. However, the precise mechanistic function of p75NTR in epilepsy has not been completely elucidated. Therefore, this review aimed to revise the mechanistic pathway of p75NTR in epilepsy.
Plain Language Summary
Roles of p75 neurotrophin receptor (p75NTR) in epilepsy: Epilepsy is a chronic neurological disorder manifested by recurring unprovoked seizures resulting from an imbalance in the inhibitory and excitatory neurotransmitters in the brain. The process of epileptogenesis involves a complex interplay between the reduction of inhibitory gamma-aminobutyric acid (GABA) and the enhancement of excitatory glutamate. Pro-BDNF/p75NTR expression is augmented in both glial cells and neurons following epileptic seizures and status epileptics (SE). Over-expression of p75NTR is linked with the pathogenesis of epilepsy, and augmentation of pro-BDNF/p75NTR is implicated in the pathogenesis of epilepsy. However, the precise mechanistic function of p75NTR in epilepsy has not been completely elucidated.
Keywords
Introduction
The p75 neurotrophin receptor (p75NTR) was initially identified in 1973 as a low-affinity nerve growth factor receptor (LNGFR).
1
The p75NTR is considered to be a functional receptor for various growth factors, such as pro-brain derived neurotrophic factor (pro-BDNF), neurotrophin 3 (NT-3), and neurotrophin 4 (NT-4).
2
Noteworthy, p75NTR is a member of the superfamily of tumor necrosis factor receptors.
3
It consists of 3 domains, intracellular, transmembrane and extracellular.
2
The tendency of p75NTR is toward dimerization rather than trimerization, owing to the activation of the tyrosine kinase co-receptor.
4
The p75NTR promotes neuronal maturation, differentiation, and survival.
4
The induction of apoptosis by neurotrophins through p75NTR involves the activation of p53, caspase, and c-Jun N-terminal kinase signaling.
5
The engagement of neurotrophins with p75NTR enhances the viability of neurons through the initiation of the nuclear factor kappa B (NF-κB) signaling pathway.
6
p75NTR also involves in interactions with various receptors, such as interaction with the sortilin receptor, which serves as a co-receptor-for-the-functional effects of multiple neurotrophins, including BDNF.
7
Additionally, the pro-BDNF exhibits a more specific binding affinity towards p75NTR in the presence of sortilin receptors.
8
Moreover, the interaction between p75NTR and tropomyosin receptor kinase A (TrkA), resulted in the reduction of p75NTR-induced apoptosis.
9
Also, p75NTR with the help of the nogo-66 receptor (NgR1) induces apoptosis and inhibits neuronal myelin sheath.
10
The effects of diverse signaling pathways, such as NF-κB, which plays a crucial role in the advancement and progression of neurodegenerative disorders, are mediated by p75NTR
11
(Figure 1). Pathway of p75NTR.
It has been shown that p75NTR is highly expressed during neuronal development, though these receptors are re-expressed in different pathological conditions including neurodegenerative diseases and epilepsy. For example, toxic peptides and proteins like amyloid beta (Aβ) which accumulated in AD are regarded as a ligand for p75NTR that induces axonal degeneration and synaptic dysfunction. 12 It has been demonstrated that the signaling of p75NTR plays a role in epilepsy, though the precise mechanism was not fully elucidated. Thus, this review tries to show how p75NTR signaling is intricate in epilepsy neuropathology.
Role of p75NTR in epilepsy
Epilepsy is a chronic neurological disorder manifested by recurring unprovoked seizures resulting from an imbalance in the inhibitory and excitatory neurotransmitters in the brain. 13 Epilepsy is classified as a primary (idiopathic) epilepsy when the underlying causes are unidentified. 14 However, secondary (symptomatic) epilepsy is caused by different neurological disorders such as brain injury, brain tumors, neurodegenerative diseases, and infection. Epilepsy is regarded as the fourth common neurological disease affecting more than 50 million in the world. The incidence of epilepsy is 2%–5% in the general population.15,16 Notably, many signaling pathways are implicated in the pathogenesis of epileptogenesis and epilepsy. 16
It has been reported that pro-BDNF and mature BDNF (mBDNF) induce divergent physiological responses by activating p75NTR and TrkB respectively. 17 Of note, pro-BDNF/p75NTR is essential for cognitive function, and impairment of this signaling is associated with the development of neurodegenerative and neurodevelopmental disorders. 18 Neuronal cell death in the hippocampus following status epilepticus (SE) is mainly mediated by pro-BDNF/p75NTR signaling since administration of antibodies against pro-BDNF prevents hippocampal cell death following pilocarpine-induced SE. 19 Of interest, proBDNF is localized to mossy fibers and microglia following SE. in addition, p75NTR is momentarily induced primarily in axons and axon terminals following SE, as well as in neuron and astrocyte cell bodies. Furthermore, proBDNF and p75NTR increased independently of cell death and their localization was different depending on the severity of SE. The expression of pro neurotrophin co-receptors, sortilin and sorCS2. Following severe SE, sorCS2, but not sortilin, was elevated in neurons and astrocytes. 20 These data indicate that important differences exist between rat and mouse in the pro-neurotrophin response following SE. Moreover, the proBDNF and p75NTR increase after seizures in the absence of significant cell death suggesting that proneurotrophin signaling may play other roles following SE. However, Pro-BDNF/p75NTR expression is augmented in both glial cells and neurons following seizure and SE in mice regardless of neuronal cell death. 20 This finding signifies that pro-BDNF/p75NTR is linked with epileptogenesis by inhibiting GABA neurotransmission. 20 It has been observed that pro-BDNF/p75NTR signaling inhibits GABAA receptor and synaptic plasticity though BDNF/TrkB signaling activates GABA long-term potentiation.21,22 In addition, pro-BDNF/p75NTR via the Rho-ROCK pathway increases the internalization of the GABAA receptor and induces degradation of these receptors by the lysosomes leading to disruption of GABAergic neurotransmission in the cortical neurons. 23 In SE, cleavage of BDNF to mBDNF by plasminogen and plasminogen activator is impaired leading to increased pro-BDNF which via the activation of p75NTR induces recurrent seizure by inhibiting GABAergic neurotransmission.24,25 Mutation of the plasminogen gene in mice results in recurrent seizure and SE. 26 Therefore, a deficiency of plasminogen induces severe epilepsy through augmentation of augmentation of pro-BDNF/p75NTR. 26 Consequently, dysregulation of neuronal plasminogen is involved in the pathogenesis of epilepsy. 27 However, many studies confirmed that neuronal plasminogen has little or no role in the pathogenesis of epilepsy.28,29 Experimental and clinical studies showed that endogenous tissue plasminogen activator (t-PA) was increased following stroke but not intricate in the pathogenesis of epileptogenesis. 28 Likewise, in animal model epilepsy, t-PA plays a little role in the pathogenesis of epileptogenesis. 29 Thus, t-PA is not involved in the pathogenesis of post-stroke epilepsy.
Furthermore, KCC2 which regulates GABAergic neurotransmission and balance between inhibitory/excitatory neural circuits, inhibits epileptogenesis. Down-regulation of KCC2 leads to epileptogenesis in both preclinical and clinical studies.30,31 Neurons from patients with temporal lobe epilepsy (TLE) had low expression of KCC2.
30
Interestingly, down-regulation of KCC2 following SE persists for weeks leading to the reduction of inhibitory GABAergic neurotransmission and induction of excitatory neurotransmission with subsequent recurrent seizures.
31
Of note, pro-BDNF/p75NTR inhibits the expression of neuronal KCC2 in the TLE animal model.
32
However, BDNF/TrkB promotes the expression of KCC2 in the GABAergic neurons.
33
Indeed, pro-BDNF triggers the expression of p75NTR,
23
leading to epileptogenesis by inhibiting GABAergic neurotransmission. Notoriously, GABA-mediated depolarization provokes the expression of p75NTR.
34
These findings illustrated that pro-BDNF/p75NTR is involved in the pathogenesis of epilepsy. Conversely, inhibition of p75NTR before or at the onset of SE did not prevent subsequent seizures in pilocarpine-induced SE in mice.
35
In addition, p75NTR null mice experience higher excitability in the cortical neurons.
36
Thus, pro-BDNF/p75NTR signaling seems to have a neuroprotective against epileptogenesis.
36
A postmortem study involved 18 hippocampal specimens from patients with TLE showed that p75NTR expression was increased in hippocampal neurons contributing to epileptogenesis and neuronal cell deaths.
37
Furthermore, the increased expression of p75NTR in hippocampal neurons of TLE may critically influence neuronal survival during the epileptogenic process.
38
These observations suggest that over-expression of p75NTR is linked with the pathogenesis of severe epilepsy and TLE and SE (Figure 2). However, the mechanistic role of p75NTR and related ligands in epilepsy is not fully elucidated. Role of p75NTR in epilepsy.
Mechanistic pathways of p75NTR in epilepsy
PI3K/Aκt
It has been shown that phosphoinositide 3 kinase (PI3K) and protein kinase B also known as the Aκt signaling pathway regulate several neurological processes that are critical for the development of the central nervous system, including neurogenesis, synaptic plasticity, neuronal differentiation, and autophagy. 39 Exaggerated PI3K/Aκt is linked with hippocampal neuronal injury in rat epilepsy model, and inhibition of this pathway prevents hippocampal neurodegeneration. 40 Upregulation of neuronal PI3K is linked with epileptogenesis and induction of epilepsy. 41 Therefore, PI3K inhibitor LY294002 prevents pentylenetetrazole (PTZ)-induced epileptic seizure in zebrafish model. 41 Besides, the Aκt signaling pathway promotes epileptic seizure by enhancing the expression of the mechanistic target of rapamycin (mTOR) in the rat TLE model. 42 In this state, Aκt inhibitor perifosine attenuates kainic acid (KA)-induced TLE in rats. 42 It has been shown that the PI3K/Aκt signaling pathway promotes mTOR-induced epileptogenesis in hippocampal cultured neurons of the post-traumatic epilepsy model. 43 Interestingly, a dual inhibitor of mTOR and PI3K NVP-BEZ235 attenuates the development and progression of epileptic seizures. 43
On the other hand, p75NTR activation is linked with stimulation of the PI3K/Aκt/mTOR signaling pathway. It has been reported that p75NTR signaling increases the activation of the PI3K/Aκt signaling pathway. 44 Remarkably, pro-BDNF/p75NTR regulates the excitability of cortical neurons by modulating the PI3K/Aκt signaling pathway. 36 However, the preclinical study demonstrated that p75NTR activates autophagy by inhibiting the PI3K/Aκt/mTOR signaling pathway. 45 Of note, over-expression of p75NTR during aging promotes the expression of mTOR-mediated synaptic dysfunction in mice. 46 In addition, mTOR inhibitor rapamycine promotes autophagy through a p75NTR-dependent pathway. 47 Different studies confirmed that mTOR signaling is highly upregulated in epilepsy.48,49 It has been observed that the BDNF/p75NTR axis promotes hippocampal glutamatergic excitotoxicity by enhancing the mTOR signaling pathway. 50
Likewise, The PI3K/Aκt/mTOR signaling pathway is modulated by p75NTR-mediated enhancement of phosphatase and tensin homolog (PTEN) expression. 47 Of note, PTEN overexpression is associated with glutamate excitotoxicity, neuronal cell death, and epileptogenesis. 51 An experimental study revealed that the administration of PTEN inhibitor picolinate attenuates KA-induced TLE in mice. 51 Interestingly, PTEN variants are associated with drug resistance in patients with focal epilepsy. 52
Therefore, p75NTR activation is implicated in epileptogenesis by increasing PTEN expression and dysregulation of the PI3K/Aκt/mTOR signaling pathway (Figure 3). p75NTR activation and epileptogenesis.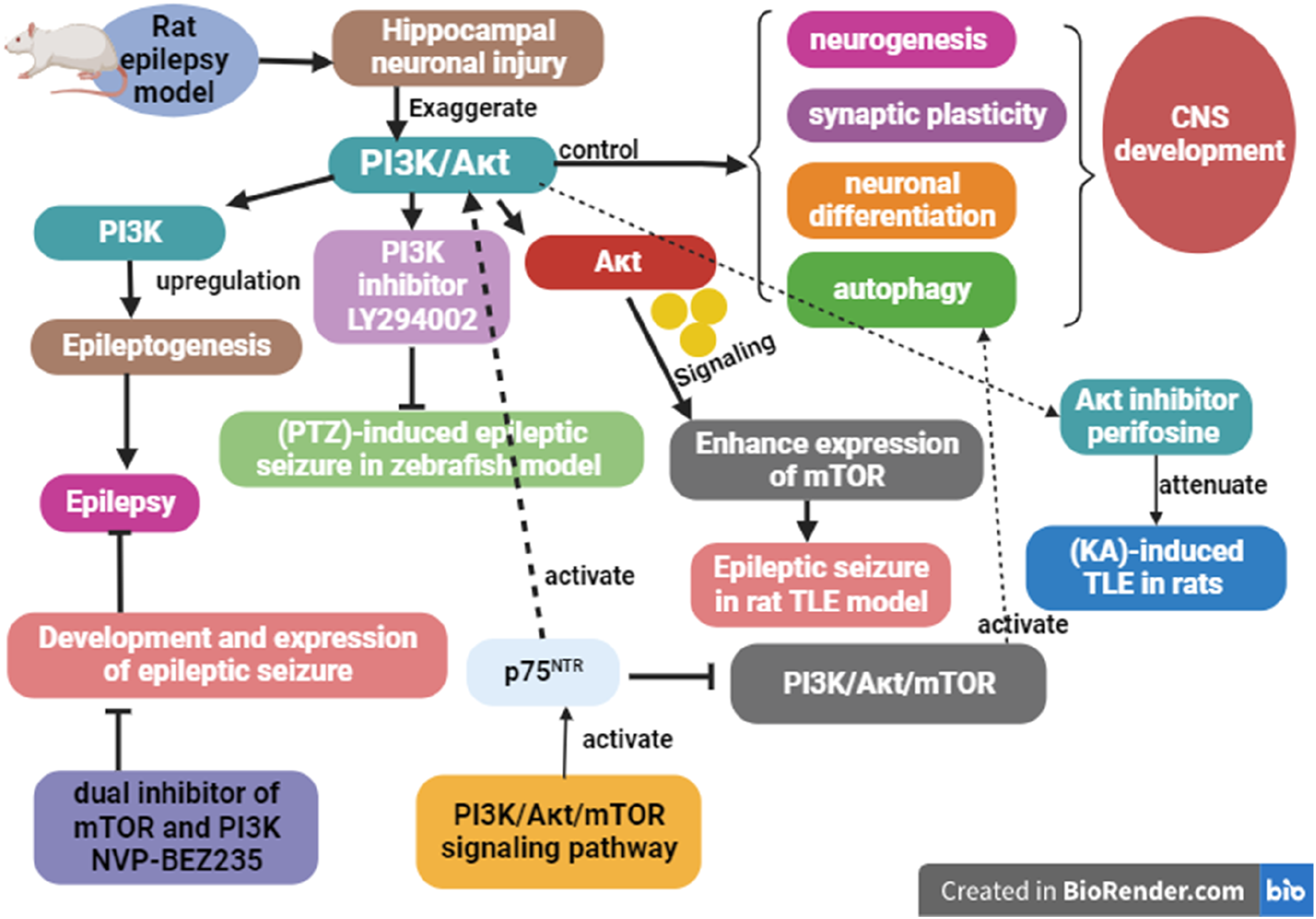
Caspase 3 and p75NTR
Cysteine proteases, or caspases, play a key role in apoptosis. Caspases are produced as dormant proenzymes and can be classified as effectors (caspase 3, 6, 7), or initiators (caspase 2, 8, 9, 10). The primary mediator of neuronal apoptosis is caspase 3, which also performs non-apoptotic tasks. The physiological functions of caspases are control memory and synaptic functions. 53 The p75NTR activates caspase 3 leading to progressive neuronal injury. 54 Song and coworkers 55 found that PTEN is necessary for shifting the switch between apoptotic and survival pathways in rat brain neurons. Regardless of whether TrkB is activated by BDNF or not, apoptosis is triggered instantly by pro-BDNF and p75NTR. The inhibition of PTEN blocks the apoptotic pathway and augments the BDNF/TrkB signaling pathway. This evidence is corroborated by the confirmation that inhibiting PTEN decreases p75NTR-mediated neuronal damage and cell death in rats with pilocarpine-induced epilepsy. 55
Caspase 3 is extremely activated in patients with TLE
56
signifying that caspase 3 is involved in epileptogenesis, and inhibition of this pathway could be a novel pathway in the management of epilepsy. A previous study indicated that over-expression of caspase 3 is present in resected neocortex from patients with TLE.57,58 Thus, targeting of caspase 3 may be effective against epileptogenesis. Besides, caspase 3 inhibitors attenuate neuronal loss in traumatic brain injury which is intricate in the development of epilepsy.
59
Different studies reported that pharmacological suppression of caspase 3 was effective in TLE.
60
In this state, pro-BDNF/p75NTR through activation of caspase 3 may lead to neuronal injury, apoptosis, and induction of epileptogenesis.
61
Therefore, direct inhibition of the pro-BDNF/p75NTR signaling pathway or indirect suppression of caspase 3 could be an effective therapeutic strategy in epilepsy (Figure 4). Caspase 3 and p75NTR in epileptogenesis.
NF-κB and p75NTR
NF-κB, a collective of transcription factors, plays a crucial role in diverse physiological and pathological processes.
62
Two principal pathways of NF-κB have been described, namely the canonical and non-canonical pathways. The canonical NF-κB pathway is initiated in reaction to various external stimuli involved in immune response, inflammation, cell proliferation, and survival.
63
This pathway results in the rapid release of NF-κB and the subsequent inhibition of IκB, which triggers IκB kinase.
64
Higher levels of NF-κB expression trigger the expression of negative regulators like IκBα, which limit the conical NF-κB pathway.
62
However, tumor necrosis factor-alpha (TNF-α) stimulates the non-conical NF-κB pathway by activating NF-κB inducing kinase (NIK).
63
The non-conical NF-κB pathway plays a complex role in the thymus’s T cell maturation and immune response regulation.
64
Moreover, an in vitro study found that p75NTR activates NF-κB during the apoptosis pathway.
65
It’s interesting to note that p75NTR causes cognitive impairment in an animal model study by triggering NF-κB.
6
Furthermore, NF-κB is intricate in the epileptogenesis process by increasing the expression of genes involved in the release of pro-inflammatory cytokines, chemokines, and the development of inflammation.
66
NF-κB is highly expressed in glial cells and neurons, and upregulated of NF-κB is associated with BBB dysfunction and GABA inhibition.
66
NF-κB is highly upregulated in neurons and astrocytes following epileptic seizure within 4 hr in animal epilepsy models.
67
Wang et al.
68
observed that neuronal expression of NF-κB following SE persist for 14 days. Pro-inflammatory cytokines which released from astrocytes and glial cells during epileptic seizure promote the expression of NF-κB and induction of further recurrent epileptic seizure.
69
Therefore, p75NTR via activation of NF-κB may induce the development and progression of epilepsy (Figure 5). NF-κB and p75NTR in epileptogenesis.
Ras homology family member A/Rho/ROCK and p75NTR
RhoA, a member of the Ras homology family, is a small GPTase that plays various cellular roles. It is extensively expressed in neurons and in glial cells.
70
RhoA overexpression is linked to the onset and progression of neurodegenerative diseases.
71
It has been shown that pro-BDNF-induced p75NTR activation increases RhoA signaling and the expression of apoptosis-mediated proteins like synaptophysin, which causes neuronal apoptosis.
70
Additionally, the p75NTR/RhoA signaling pathway is triggered in response to isoflurane, which causes neurotoxicity.
72
Moreover, fingolimod enhances the synaptic plasticity of the hippocampus by suppressing the expression of p75NTR and lowering RhoA signaling in the Huntington disease (HD) mouse model.
73
Identical to Ras, Rho is balanced by regulatory proteins such as GTPase activating protein. ROCK which is also known as Rho kinase is a serine/threonine protein kinase with a molecular weight of 160 kDa. Two types of ROCK are present, ROCK1 which is expressed in non-neuronal tissues and ROCK2 is mainly expressed in neuronal tissues including the brain and spinal cord.
74
Rho-ROCK signaling pathway is implicated in different pathophysiological processes, for example, it inhibits axonal growth during brain injury, and Rho-ROCK inhibitors can improve axonal growth.
75
In addition, over-expression of the Rho-ROCK signaling pathway is associated with the development of Alzheimer’s disease (AD).
76
Rho-ROCK is highly expressed in neurons and astrocytes that are intricate in synaptic plasticity.
77
Expression of the Rho-ROCK signaling pathway in the hippocampus is augmented following KA-induced SE.
78
It has been shown that Rho-ROCK inhibitor fasudil attenuates KA-induced neuronal injury.
79
Likewise, Rho-ROCK inhibitor Y-27632 decreased the expression of ROCK2 in experimental epileptic seizure in mice.
80
Different studies revealed that p75NTR signaling pathways activate neuronal expression of Rho-ROCK and linked neuronal injury.81-83 These findings that p75NTR signaling pathway via activation of RhoA and Rho-ROCK signaling increases the risk of epileptic seizure (Figure 6). Rho/ROCK and p75NTR in epileptogenesis.
Taken together, p75NTR signaling pathway is implicated in epileptogenesis and the pathogenesis of epilepsy by inducing various molecular pathways. Targeting of p75NTR signaling pathway could be a novel pathway in the management of epilepsy.
Conclusions
Epilepsy is a chronic neurological disorder manifested by recurring seizures resulting from an imbalance in the inhibitory and excitatory neurotransmitters in the brain. Many signaling pathways are implicated in the pathogenesis of epileptogenesis and epilepsy. It has been observed that p75NTR a function receptor of proBDNF is dysregulated in epilepsy and implicated in the development and progression of SE and TLE. However, the mechanistic role of p75NTR in the pathogenesis of epilepsy is not completely clarified. Therefore, this review was suggested to review the mechanistic pathway of p75NTR in epilepsy. Interestingly, p75NTR activation is implicated in epileptogenesis by increasing the expression of different signaling pathways. For example, p75NTR increases PTEN expression and dysregulation of the PI3K/Aκt/mTOR signaling pathway that induces epileptogenesis and the induction of epileptic seizure. As well, p75NTR enhances the activity of caspase 3 with subsequent progressive neuronal injury and epileptogenesis. In addition, p75NTR promotes the expression of NF-κB, RhoA and Rho-ROCK signaling which induces the progression of epileptic seizure and the development of TLE. Taken together, the p75NTR signaling pathway is associated with epileptogenesis and the pathogenesis of epilepsy by inducing various molecular pathways. Hence, targeting of p75NTR signaling pathway and associated pathways could be an innovative therapeutic strategy in the management of epilepsy. This review suggests doing preclinical and clinical studies to confirm this scientific claim.
Footnotes
Author contributions
AT, HMA-K, AIA and AkA conceptualized the manuscript, wrote, edited and reviewed the main text and approved the final edition of the manuscript. OE, AAF, FA, HMS and GE-SB prepared the figures, wrote, corrected, amended and approved the final edition of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
