Abstract
Background
Depression is one of the most common symptoms experienced by multiple sclerosis patients and may be secondary to the disease itself as well as other variables such as age, disease severity and side effects of treatment.
Objective
To determine if there is an association between disease modifying therapies and depression rates based on PHQ9 scores in multiple sclerosis.
Methods
This was a retrospective chart review. Patients followed at the University of Texas Southwestern Multiple Sclerosis and Neuroimmunology Clinic from 2017 to 2020 were included in this study. Patients’ most recent PHQ-9 scores were used. The following data was extracted from patient charts: disease modifying therapy, age, disease duration, gender, antidepressant use and ambulatory status.
Results
Data from our study included 2611 individual PHQ-9 scores. The majority of our patients were female and the mean age across all treatment groups was 50.37 years old. The median disease duration across all treatment groups was 12.74 years. Most patients in this cohort required no ambulatory assistance. 43.86% of patients were on antidepressants and use was correlated with a higher PHQ9 score. The median PHQ 9 score across all treatment groups was 4 (Interquartile range = 7). Across treatment groups, patients on interferon therapy had the lowest PHQ 9 scores with a median of 2.
Conclusions
Our study demonstrated that there were lower PHQ-9 scores among interferon treatment group as compared to other disease modifying therapies and non-treatment groups
Introduction
Multiple sclerosis (MS) is a chronic condition characterized by immune mediated attacks on the central nervous system. 1 It impacts multiple domains of patients’ lives with numerous associated symptoms including mobility difficulties, sensory loss, pain, fatigue, depression, spasticity, genitourinary dysfunction and bowel symptoms. 2 Of these symptoms, depression is one of the most common with a prevalence of approximately 30%.2-4 Depression can lead to worsening disability and poorer quality of life for MS patients, and because of this, depression screening is now considered as a quality measure by the American Academy of Neurology.5,6
Multiple factors have been cited as influencing the occurrence of depression in MS patients. These include severity of disease, younger age at diagnosis, less social support and female gender. There is conflicting literature relating to disease duration and the negative or positive correlation with depression.7-11
We sought to determine if there was an association between disease modifying therapy use and depressive symptoms based on PHQ9 scores. Interferon therapy has previously been shown to induce depressive symptoms when used in treatment of hepatitis and malignancy with prevalence ranging from 30-70%. Postulated mechanisms for this revolve around immunotherapy cytokine network modulation.12-14 While interferons have been associated with depression, fingolimod has previously been reported to lower the risk of depression. Potential mechanisms thought to contribute to this include inhibition of histone deacetylases and increased production of brain derived neurotrophic factor in the central nervous system. 15 Based on this, we hypothesized that PHQ9 scores in MS patients would be influenced by disease modifying therapy based on both efficacy and mechanism of action with those higher efficacious, immunomodulating drugs having lower total PHQ9 scores compared to other groups.
Methods
Participants
This was a retrospective chart review (study number STU-2019-1604). Given the retrospective nature of the study, University of Texas Southwestern IRB approved the study to be conducted with an exemption from a formal ethical approval application process, and waived the need for patient consent. Multiple sclerosis patients followed at the University of Texas Southwestern Multiple Sclerosis and Neuroimmunology Clinic from 2017 to 2020 were included in this study. All MS variants were included. PHQ-9 scores that were completed at office visits during this time period were reviewed. Data was retrieved from our electronic data warehouse. Individual patient charts were pulled based on ICD-9 and ICD-10 codes. Charts were reviewed with respect to disease modifying therapy, demographics and ambulatory status at the time of PHQ completion.
Inclusion and Exclusion Criteria
Inclusion criteria included the following: 1) a diagnosis of MS, 2) PHQ9 data availability, and 3) patients evaluated by a practitioner at our center. We excluded patients with incomplete data, those that did not have a diagnosis of MS, and those on non-traditional disease modifying therapies.
Clinical Data
Cohort Characteristics.

a: Fully ambulatory, b: Walks with assistance of cane or walker, c: Wheelchair.
We used the most recent available PHQ-9 score in gathering data for the purpose of this study. Additionally, for all subjects either the date of diagnosis or the date of symptom onset was known up to the month and year. Disease duration was computed by taking the difference between the date of PHQ-9 administration and the date of symptom onset, if available, or the date of diagnosis otherwise. Given that the date of diagnosis and date of symptom onset were known only up to the month and year, the day was assumed to be the 15th. Similarly, if only the year was known, the month was assumed to be June and if only the decade was known, the fifth year within that decade was specified. For instance, if the date of diagnosis or symptom onset was only known to have been in the 80s, the date of diagnosis (or symptom onset) was specified to be June 15th, 1985.
Treatment at the time of testing was classified into 6 subtypes: 1) No treatment, 2) Interferons, 3) Low-efficacy, immunomodulatory (not including interferons), 4) Low-efficacy cell depleters, 5) High-efficacy, cell depleters, and 6) High-efficacy, cell restrictors. Interferons consisted of interferons (beta-1a/1b and peg-interferon). Low-efficacy, immunomodulatory agents (not including interferons) included glatiramer acetate. Low-efficacy, cell depleters included teriflunomide and fumarates (dimethyl fumarate and diroximel fumarate). High-efficacy, cell depleters included alemtuzumab, rituximab, ocrelizumab, and cladribine. Lastly, high-efficacy, cell restrictors included natalizumab, fingolimod, and siponimod.
Statistical Analysis
Based on the PHQ-9 total score (the summation of the 9 questions constituting the PHQ-9), subjects were inferred to be depressed if the total score was greater than or equal to 10 and not depressed otherwise. This score was chosen based on a prior study conducted by SB Patten and JM Burton et al evaluating the validity of PHQ9 scores in a MS specific population that found that a score of 10 or greater correlated with a sensitivity of 95.0% and specificity of 85.9% (95% CI 78.7-91.4) when used as a cut-off for depression. 16
We analyzed the effect of age, gender, disease duration, anti-depressant usage, ambulatory status, and disease modifying therapy on the binary diagnosis of depressed vs not using logistic regression. Then, we analyzed the effect of the same covariates on the PHQ-9 total score using ordinal regression assuming a logit link. The proportional odds assumption of the ordinal regression model was verified before reporting results.
Lastly, a Tukey’s all-pairs comparison was performed to compare the effect of each disease modifying therapy grouping independently for the logistic and ordinal regression models discussed above. In order to account for multiple comparisons, a Bonferonni’s adjustment was applied to all P-values independently for the logistic and ordinal regression model.
All analyses were performed in R (version 3.6.1). Ordinal regression was performed using the clm package in R, as well as testing the validity of the logit link and accompanying assumptions.
Results
Depression based on PHQ-9 Total Score (including all 9 Questions)
Logistic regression modeling results with the no disease modifying therapy cohort serving as the reference population.

a: Centered and scaled age at the time of testing; mean = 50.37, standard deviation = 12.90, b: Walks with assistance of cane or walker, c: Wheelchair bound, d: Interferons (beta-1a/1b and peg-interferon), e: Glatiramer acetate, f: Teriflunomide and fumarates (dimethyl fumarate and diroximel fumarate), g: Alemtuzumab, rituximab, ocrelizumab, and cladribine, h: Natalizumab, fingolimod, and Siponimod.
Logistic regression comparison among the 6 treatment groups.
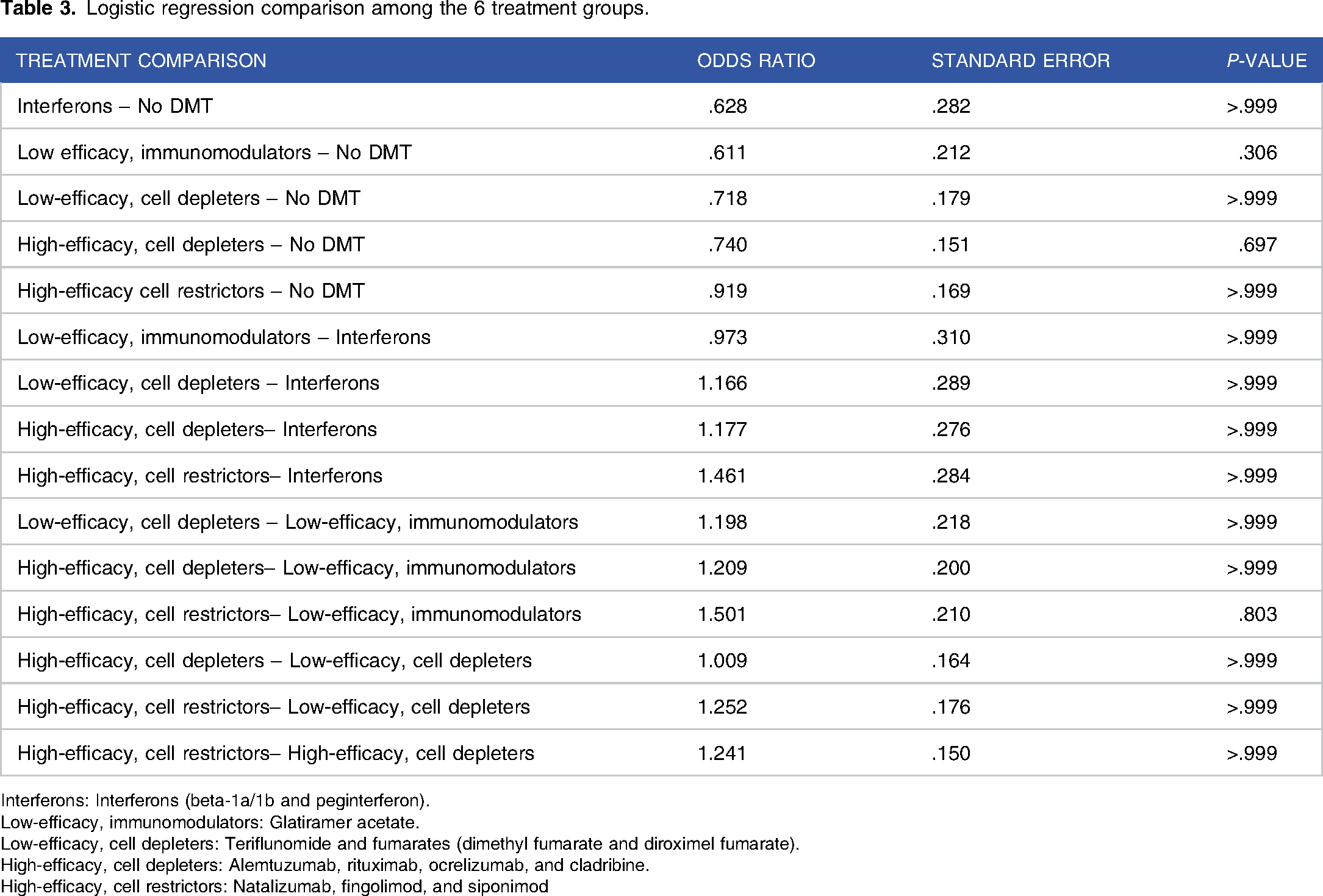
Interferons: Interferons (beta-1a/1b and peginterferon).
Low-efficacy, immunomodulators: Glatiramer acetate.
Low-efficacy, cell depleters: Teriflunomide and fumarates (dimethyl fumarate and diroximel fumarate).
High-efficacy, cell depleters: Alemtuzumab, rituximab, ocrelizumab, and cladribine.
High-efficacy, cell restrictors: Natalizumab, fingolimod, and siponimod
Ordinal regression modeling demonstrating effect of age, disease duration, antidepressant use, ambulatory status and treatment type on PHQ9 scores.

a: Centered and scaled age at the time of testing; mean = 50.37, standard deviation = 12.90, b: Walks with assistance of cane or walker, c: Wheelchair bound, d: Interferons (beta-1a/1b and peg-interferon), e: Glatiramer acetate, f: Teriflunomide and fumarates (dimethyl fumarate and diroximel fumarate), g: Alemtuzumab, rituximab, ocrelizumab, and cladribine, h: Natalizumab, fingolimod, and Siponimod
Ordinal regression comparison between individual treatment effects compared to no treatment and other treatment groups and effect on PHQ9 scores.

Interferons: Interferons (beta-1a/1b and peginterferon).
Low-efficacy, immunomodulators: Glatiramer acetate.
Low-efficacy, cell depleters: Teriflunomide and fumarates (dimethyl fumarate and diroximel fumarate).
High-efficacy, cell depleters: Alemtuzumab, rituximab, ocrelizumab, and cladribine.
High-efficacy, cell restrictors: Natalizumab, fingolimod, and Siponimo
Discussions
In this study we sought to determine if there was an association between higher PHQ-9 scores and disease modifying therapies used to treat multiple sclerosis. In contrast to prior studies, we determined that PHQ-9 total scores were lowest in the interferon treatment group when compared to all other treatment groups and the non-treatment group. This was unexpected given the previously reported association of depressive symptoms and interferon use. However, it should be noted that our study was in line with smaller studies focused on this topic.10,17 One possible explanation for our findings could focus on disease severity. Those patients with more severe disease may have been on higher efficacious treatments, and as shown previously, disease severity has been correlated with higher rates of depression.
Several other factors were found to have an association with depression apart from disease modifying therapy class. Consistent with several prior studies, we found that shorter disease duration correlated with higher PHQ9 scores. One potential reason for this could be patient reaction to initial diagnosis and coming to terms with the impact this may have on his/her life. We also found that those patients who required an assistance device such as a walker or cane for ambulation were more likely to have higher PHQ9 scores compared to those without ambulatory restrictions and also those patients who were wheelchair dependent. In our study, ambulatory status was used as a surrogate for disability as EDSS scores were not readily available.
The conclusions of this study are limited by several factors. This was a retrospective, cross-sectional study that did not evaluate longitudinal data, specifically disease modifying therapy transitions. It is possible that those patients who experienced depressive symptoms on interferon therapy had already transitioned to another class of medication at the time of PHQ9 completion. This factor may have led to selection bias and could account for why the interferon group had lower scores. We were also not able to control for the impact of concurrent medical illnesses or other extraneous factors such as social support that may have positively or negatively influenced PHQ9 scores.
We also found that nearly half of our patient population was on antidepressants and use of these medications correlated with higher PHQ9 scores in our cohort. These medications have numerous indications and are commonly used for neuropathic pain as well as depression in MS patients. It was not possible to track the specific indications for the use of these agents, and therefore this factor should not be used as reliable marker of depression in our patient population. One could argue that neuropathic pain could be an independent driver of depression given it is a common symptom experienced by MS patients. 18 It is also possible we created a selection bias by selecting depressed patient, and antidepressant use could be considered an indicator of depression in this scenario.
In this study, depression was evaluated with the PHQ-9. Question 3 and 4 specifically focus on fatigue and sleep which are significant symptoms in the MS patient population. It is possible that these 2 questions drive higher PHQ9 scores and lead to inappropriate diagnosis of depression. There is no available normative data excluding question 3 and 4, and this brings to question if the PHQ9 is biased towards over-scoring in MS patients due to the high prevalence of fatigue and insomnia in this patient population. For the purpose of this study, we did a secondary analysis removing questions 3 and 4 and no meaningful change was noted on the results.
Finally, this study was completed at a single site which could have also introduced selection bias.
Future prospective, longitudinal studies are needed to confirm our findings.
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by Lauren Tardo, MD with assistance from Harris Majeed. Statistical analysis were performed by Morgan McCreary Ph.D. The first draft of the manuscript was written by Lauren Tardo, MD, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Conceptualization and supervision was provided by Benjamin Greenberg, MD, MHS.
Declaration of conflicting interest:
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Benjamin Greenberg has received consulting fees from Alexion, Novartis, EMD Serono, Viela Bio, Genentech/Roche, Greenwhich Biosciences, Axon Advisors, Rubin Anders, ABCAM, and PRIME Education. He has received grant funding from PCORI, NIH, NMSS, The Siegel Rare Neuroimmune Association, Clene Nanomedicine and the Guthy Jackson Charitable Foundation for NMO. He serves as an unpaid member of the board of the Siegel Rare Neuroimmune Association.
Funding:
The authors received no financial support for the research, authorship, and/or publication of this article.
