Abstract
Coccidioidomycosis is a fungal infection caused by Coccidioides immitis and Coccidioides posadasii. The most severe disseminated coccidioidomycosis results in meningoencephalitis. Literature review reveals very rare reports of hyphal forms of coccidioides organisms detected in CSF. We present a case of a 58-year-old Hispanic male with a 10-year persistence of CM and many ventriculoperitoneal shunt reversions. The patient initially presented with a hydrocephalus of unknown source. The diagnosis of CM was made three years later based on a culture. On this presentation at the emergency department (ED), the patient reported one day of worsening left-sided weakness, altered mental status, and vomiting. We detected the hyphal forms of coccidioides in the cerebrospinal fluid (CSF) during cytopathology examination. Awareness of this reversion from spherule form to hyphal form is helpful in diagnosis and treatment. The hyphal forms of coccidioides may be associated with the increased risk of obstruction of the shunts. It is important to submit CSF for cytopathology examination for diagnosis, especially in patients with shunts.
Introduction
Coccidioidomycosis is a fungal infection caused by Coccidioides immitis and Coccidioides posadasii. Coccidioidomycosis mainly affects the lungs through inhalation of arthroconidia, although many other organs can be involved. The most severe disseminated coccidioidomycosis results in meningoencephalitis. We have reviewed the reports of hyphal forms of coccidioides detected in CSF in the published literature.
Case Report
Presentation
A 58-year-old Hispanic male with a history of type II diabetes mellitus, initially presented with an unknown source of hydrocephalus and was treated with right frontal ventriculoperitoneal (VP) shunt placement 10 years ago, followed by multiple revisions. Three years later, coccidioidal meningitis was diagnosed based on a positive culture of cerebral spinal fluid (CSF) of coccidioides. He was instructed to be on lifelong fluconazole therapy. The patient had many shunt revisions afterward. The most recent revision was two weeks ago, and he received a placement of a right temporal Miethke low-pressure (5 cm) miniNAV shunt system with a sprung reservoir. The patient had been doing well other than persistent left-sided weakness after discharge. On this presentation at the emergency department (ED), the patient reported one day of worsening left-sided weakness, altered mental status, and vomiting.
Assessment
The examination was notable for hemodynamic stability, weakness, fatigue, and nausea and vomiting. Laboratory data showed mild anemia and glucose of 116 mg/dL (normal: 70–99 mg/dL). Testing for human immunodeficiency virus (HIV) was negative. The CSF showed glucose of 107 mg/dL (normal: 50–80 mg/dL), WBC of 66/μL (normal: 0–5/μL), red blood cells (RBCs) of 59/μL (normal: 0/μL), protein of 150.7 mg/dL (normal: 15–45 mg/dL), neutrophil of 3% (normal: 0–6%), lymphocytes of 83% (normal: 40–80%), and monocyte of 9% (normal: 15–45%), plasma 1% (normal: 0%), and macrophages 4% (normal: 0%). The CSF coccidioides antibody (Ab) test (a complement fixation test that measures both IgM and IgG, performed by a reference lab) was positive with a titer of 1:2 seven years ago and a titer of 1:8 one year ago. No tests of coccidioides Ab for CSF or serology was done during the current admission. The CSF culture was positive for coccidioides in the patient’s third year of presentation. There was one CSF culture resulting as “Presumptively” positive coccidioides in the patient’s ninth year of presentation. Presumptively identification was usually based on pigmentation lacking during early culture and visible septate hyphae with barrel-shaped arthroconidia microscopically later. It was unclear and unable to reveal if the patient’s culture diagnosis as “Presumptively” positive coccidioides was based on “pigmentation lacking” in the past. The CSF cultures had grown “mixed molds,” which were interpreted as aspergillus and cladosporium in the past six months. The “mixed molds” was most likely a misinterpretation due to the findings of abundant hyphal forms of coccidioide organisms in CSF by cytopathology described in the “Management” section. The CSF culture showed coccidioides in the latest/current culture after the patient was discharged. Computed tomography (CT) of the head demonstrated persistent enlargement and cystic dilatation of the temporal horn of right lateral ventricle (Figure 1), which showed similar obstructive hydrocephalus as that observed by prior CT. Hydrocephaly and basilar meningeal enhancement were identified by previous magnetic resonance imaging (MRI). The differential diagnosis was most likely recurrence of shunt failure causing the vomiting and increasing left-sided weakness. Computed tomography of the head demonstrates persistent enlargement and cystic dilatation of the temporal horn of right lateral ventricle.
Management
The patient was given nothing by mouth (NPO) and started on intravenous (IV) hydration, esomeprazole, and insulin regular. A shunt tap in ED demonstrated a low pressure (5 cm H2O) and 25 mL of CSF removed. A sample of CSF was sent to the lab for analysis. The right temporal shunt was pumped 20 times, 3 times/day, and repeat CT head was performed. There was no significant change in the appearance of the right temporal horn dilatation; however, symptomatically, his left-sided weakness improved. The plan was to continue having the family pump the shunts regularly, with follow-up in the clinic in two weeks. Fluconazole used on this patient could not cover aspergillus, which grew in the CSF culture in the past six months before the current admission. During this hospital course, the more empiric voriconazole was used as lifelong therapy. Of note, the patient had compliance issues with medications and appointments in the past.
The CSF specimen was examined by cytopathology. The cytospins of CSF displayed abundant hyphal/mycelial form organisms with alternating arthroconidia with empty disjuncture pleomorphic cells by Diff-Quik, Papanicolaou, and Giemsa stains (Figure 2). The morphology was consistent with coccidioide organisms. The diagnosis was confirmed by CSF culture after the patient was discharged. The culture result was reported much later than the cytopathology observation of coccidioides. No PCR was performed to further classify Coccidioides immitis or Coccidioides posadasii. The culture was not sent for sensitivity. The cytospins of CSF show that abundant hyphal/mycelial forms of coccidioides can mature fully to form-alternating arthroconidia with empty disjuncture pleomorphic cells in (A) Diff-Quik stain, (B) Papanicolaou stain, and (C and D) Giemsa stain: Rectangular-1 and ovoid-2 (A) and square-3 and barrel-shaped-4 (C). Macrophages-5, and one red blood cell (RBC)-6 are present as internal controls of measurement (D). Arrows indicate the structures described.
Discussion
The diagnosis of coccidioidal meningitis (CM) in our patient is very challenging. The clinical symptoms of CM are varied. Early clinical consideration of CM as one of the broad differential diagnoses is very important. By MRI, there are approximately 50% of patients with coccidioidal CNS infection who may demonstrate hydrocephaly, basilar meningeal enhancement, and basilar vasculitic infarction. 1 This patient has obstructive hydrocephalus and basilar meningeal enhancement observed by several prior neuro-images.
The CSF of CM usually displays pleocytosis, predominantly with lymphocytes and elevated protein as that seen in our patient. In CM, the CSF glucose is usually low or normal. This patient’s elevated glucose is due to his diabetes. It is important to understand the CSF glucose level may not be reliable in diagnosis of CM in diabetic patients.
CSF culture is diagnostic for CM, but the sensitivity of the test is low, which is observed in this patient. Positive cultures are only found in approximately 25% of patients. 2 The presence of the lgG antibody in CSF by immunodiffusion or complement-fixing is almost considered as specific. However, they are not sensitive, and only 30–60% of tests are initially positive. Clinical manifestation, CSF parameters, and the IgG antibody detected by immunodiffusion or complement fixation in the serum are all helpful for the diagnosis of CM. 2 CSF coccidioidal antigen levels show a better sensitivity and have a specificity of 100%. 3 PCR testing has the advantage of being faster but has lower sensitivity and specificity than culture. 4
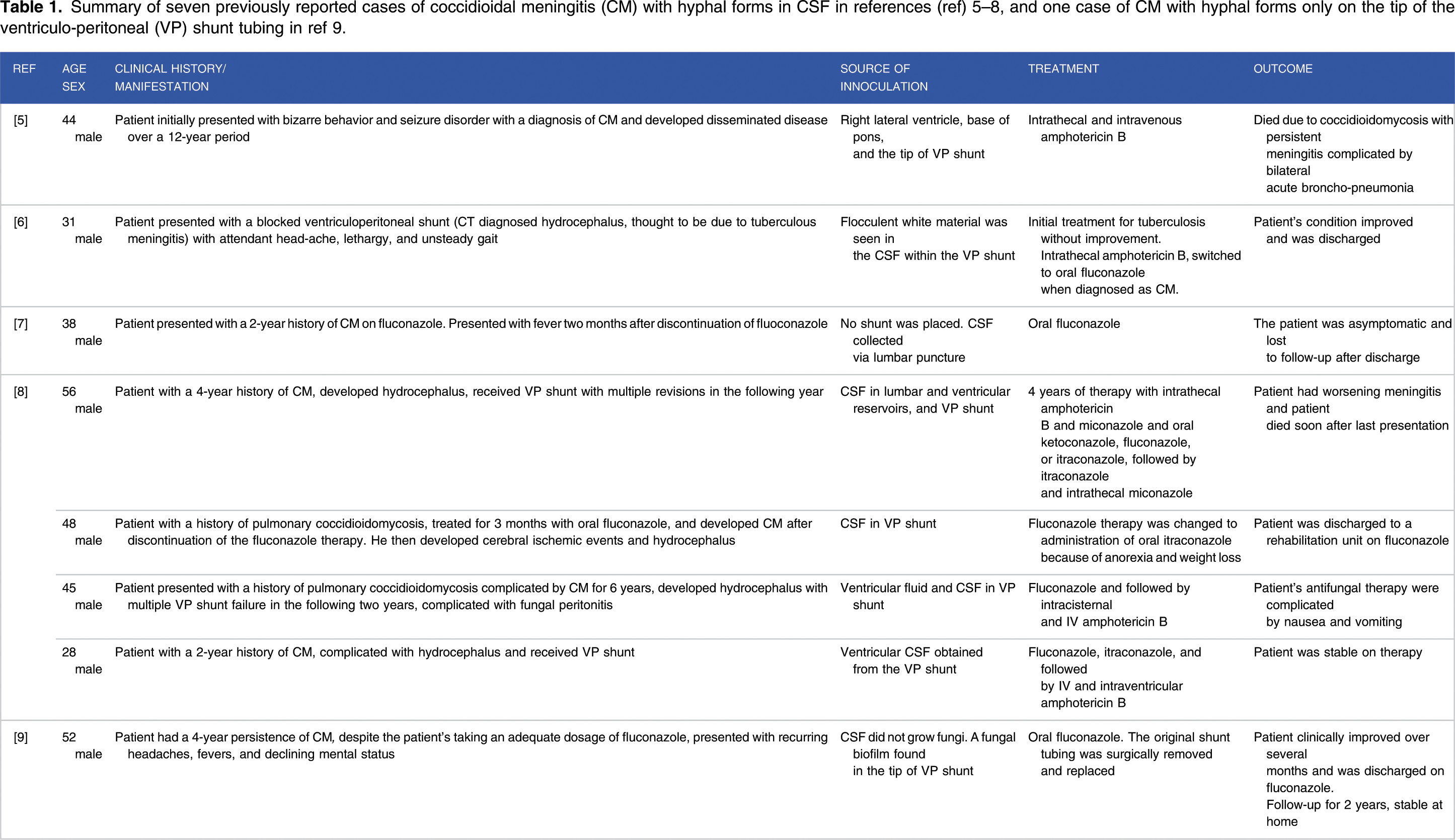
The CSF of our patient shows hyphal forms of coccidioides microscopically during cytopathology examination. Coccidioides is a dimorphic fungus, growing in a hyphal form in its natural environment of the soil and a spherule form in the human tissue. The hyphal forms are rarely seen in CSF. Only seven reported cases of CM with hyphal forms in CSF (Table 1) were found when searching PubMed.5-8 Among these seven cases, one case has no shunt/device. The plastic devices in central nervous system may be related to the presence of morphological reversion to the saprophytic form of coccidioides. Awareness of this reversion is helpful in diagnosis and treatment. Importantly, the hyphal forms of coccidioides may be associated with the increased risk of obstruction of the device(s). 8 This is subsequently confirmed by Davis et al 9 who have observed the hyphal forms of Coccidioides immitis dislodged from the tip of the VP catheter tip in a patient with 4-year persistence of CM despite the adequate treatment. A confluent biofilm formed on the tubing by hyphal elements, and coccoid cells of Coccidioides immitis has been identified under electron microscopy in the dehydration-condensed biofilm. Fungal biofilms should be considered as a cause for treatment failure in persistent CM, especially when artificial prostheses such as shunts/catheters are used. In our patient, there have been many shunt revisions performed because of shunt malfunction possibly due to fungal biofilms formed in the shunts.
For treatment of CM, fluconazole has been used as the primary oral medication since early 1990s. Patients with CM should receive lifelong medication with fluconazole or other azole monotherapy. 10 Voriconazole has been applied in CM treatment failure, although it is not used widely at present. Our patient has switched the medication from fluoconazole to voriconazole to cover a wider spectrum of fungal infection bases on multiple culture results of growing various fungi other than coccidioides (coccidioides grew in the last and different culture). Noncompliance in taking the fluconazole can be a cause of treatment failure as that may be the case in this patient. Intrathecal amphotericin B deoxycholate is usually used as rescue therapy when there is azole therapy failure and when the patient is pregnant due to the teratogenicity of azoles. 10
Note: Our patient had been followed up for four years until the pandemic started. The patient was on lifelong voriconazole and subsequently received two shunt revisions in the first two years, respectively, after his hospital discharge described in this report.
Conclusion
The diagnosis of CM can be difficult. Early clinical consideration of CM as one of the broad differential diagnoses is very important. Fungal biofilms should be considered as a cause for treatment failure in persistent CM, when shunts/catheters are used. The hyphal forms of coccidiodes can be visualized microscopically in CSF. In addition to regular CSF analysis, submitting CSF and the replaced malfunction shunts/catheters for cytopathology examination/diagnosis is recommended.
Summary of seven previously reported cases of coccidioidal meningitis (CM) with hyphal forms in CSF in references (ref) 5–8, and one case of CM with hyphal forms only on the tip of the ventriculo-peritoneal (VP) shunt tubing in ref 9.
Footnotes
Author contributions
YX conceptualized the study, developed the proposal, and coordinated the case study. JS, MW, and YX completed data collection and analysis, and wrote the report. All authors read and approved the final manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author note:
All authors had access to the data and a role in writing the manuscript.
Informed consent:
Written informed consent was obtained from the patient’s next of kin for publication of this article.
