Abstract
Background:
Clinicians may choose to administer antiplatelet medications to patients with cerebral aneurysms following endovascular coiling to prevent thrombus formation and vascular occlusion, if they fear a thrombus will form on the platinum wire where it diverges into the vessel from the aneurysm sac. However, the mechanism by which vascular endothelial cells repair a vessel in the living body in the event of a coil deviation and the effects of antiplatelet drugs on these cells have not been fully elucidated. We aimed to investigate the association between endothelial progenitor cells (EPCs) and endothelium formation at the surface of the platinum coils deployed in the carotid artery of rats, and to determine the effects of different antiplatelet drugs on this process.
Subjects and Methods:
We established an experimental model using normal and diabetic rats at 12 months of age. The diabetic rats were assigned to 4 different diet groups, distinguished by whether they were fed plain rat feed, or the same feed supplemented by 1 of 3 antiplatelet drugs (cilostazol, aspirin, or clopidogrel: all 0.1%) for 2 weeks, and the carotid artery was perforated by an embolization coil (“carotid coil model”). We monitored the process by which vascular endothelial cells formed the new endothelium on the surface of the coil by sampling and evaluating the region at 1, 2, and 4 weeks after placement. This repair process was also compared among 3 groups treated with different antiplatelet drugs (i.e. aspirin, clopidogrel, and cilostazol). One-way analysis of variance tests were performed to evaluate the differences in vascular thickness between groups, and P < .05 was considered statistically significant.
Results:
The diabetic rats showed delayed neoendothelialization and marked intimal hyperplasia. Cilostazol and clopidogrel effectively counteracted this delayed endothelial repair process. Flk1 immunostaining revealed greater expression in the diabetic rats administered cilostazol, second only to normal rats, suggesting that this agent acted to recruit EPCs.
Conclusion:
Neoendothelialization is delayed when vascular endothelial cells fail to function normally, which consequently leads to the formation of hyperplastic tissue. Cilostazol may remedy this dysfunction by recruiting EPCs to the site of injury.
Background
Endovascular coiling using a detachable platinum wire is an increasingly popular treatment for cerebral aneurysms worldwide, often achieving the best outcomes for certain patients and aneurysm characteristics. 1 However, during the perioperative period of an embolization surgery, clinicians may administer antiplatelet drugs if they fear that a thrombus will form on the embolized wire deviating into the vessel from the aneurysm sac. Although the usefulness of antiplatelet drugs has been reported, no study has identified the process supporting their use. 2 We hypothesized that antiplatelet drugs are involved in vascular endothelial repair by acting on endothelial progenitor cells (EPCs).
Thus, we aimed to investigate the association between EPCs and endothelium formation at the surface of the platinum coils deployed in the carotid artery of rats, and to determine the effects of different antiplatelet drugs on this process.
Subjects and Methods
This study was conducted with the approval from the ethics association of our hospital (Research at Kindai University Faculty of Medicine Ethics Committee, the approval number 31-003).
Our experimental model employed Otsuka Long-Evans Tokushima Fatty (OLETF) rats (a strain that spontaneously develops diabetes), based on their susceptibility to vascular endothelial damage, which is enhanced under hyperglycemic conditions. 3 At 12 months of age, these animals were assigned to 4 different diet groups, distinguished by whether they were fed plain rat feed (CE-2: CLEA Japan, Inc.), or the same feed supplemented by 1 of 3 antiplatelet drugs (cilostazol, aspirin, or clopidogrel: all 0.1%) for 2 weeks. For comparison, a control group was also established, consisting of normal Long-Evans Tokushima Otsuka (LETO) rats of the same age that were fed the plain diet (Table 1) for 2 weeks. We established our novel “carotid coil model” after 2 weeks of feeding by surgically exposing the common carotid artery, and perforating it transversely with a platinum coil (Guglielmi detachable coil: GDC™, manufactured by Boston Scientific for Stryker Neurovascular Portage, MI, USA), such that the coil was suspended across the center of the intravascular space (Figure 1). The study of the effect of various drug combinations was not carried out, as this was considered to render it difficult to identify causal relationships.
Summary of the targeted rats and feed.
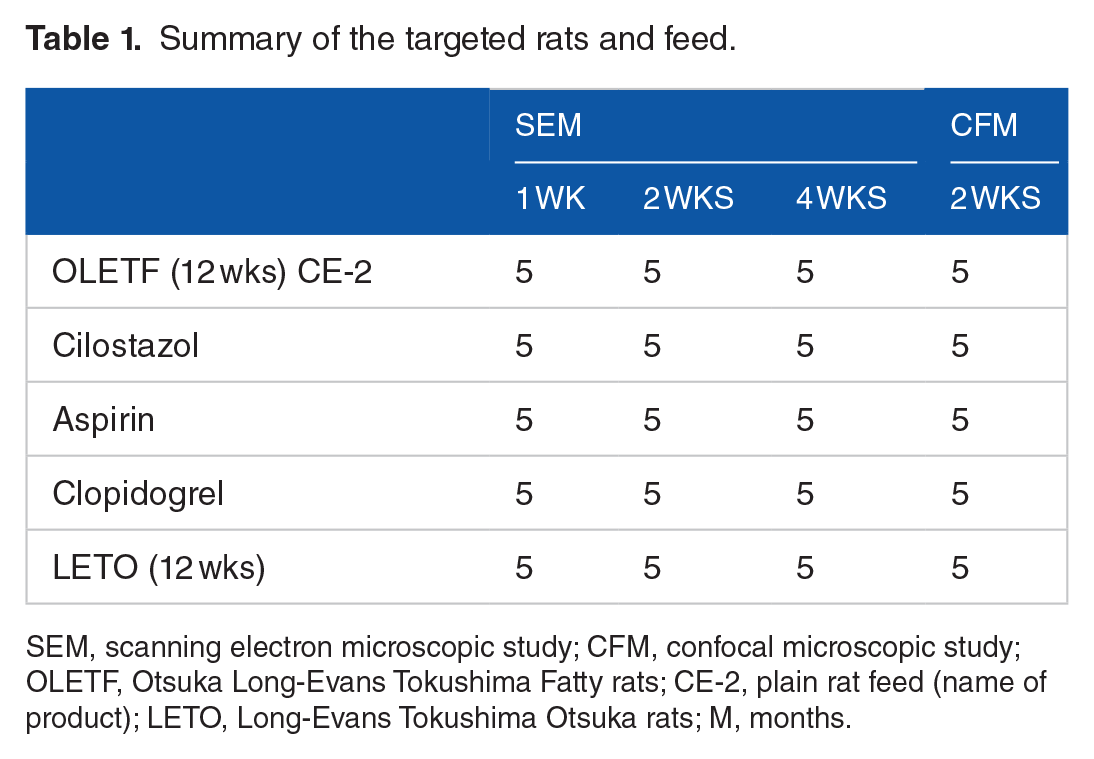
SEM, scanning electron microscopic study; CFM, confocal microscopic study; OLETF, Otsuka Long-Evans Tokushima Fatty rats; CE-2, plain rat feed (name of product); LETO, Long-Evans Tokushima Otsuka rats; M, months.
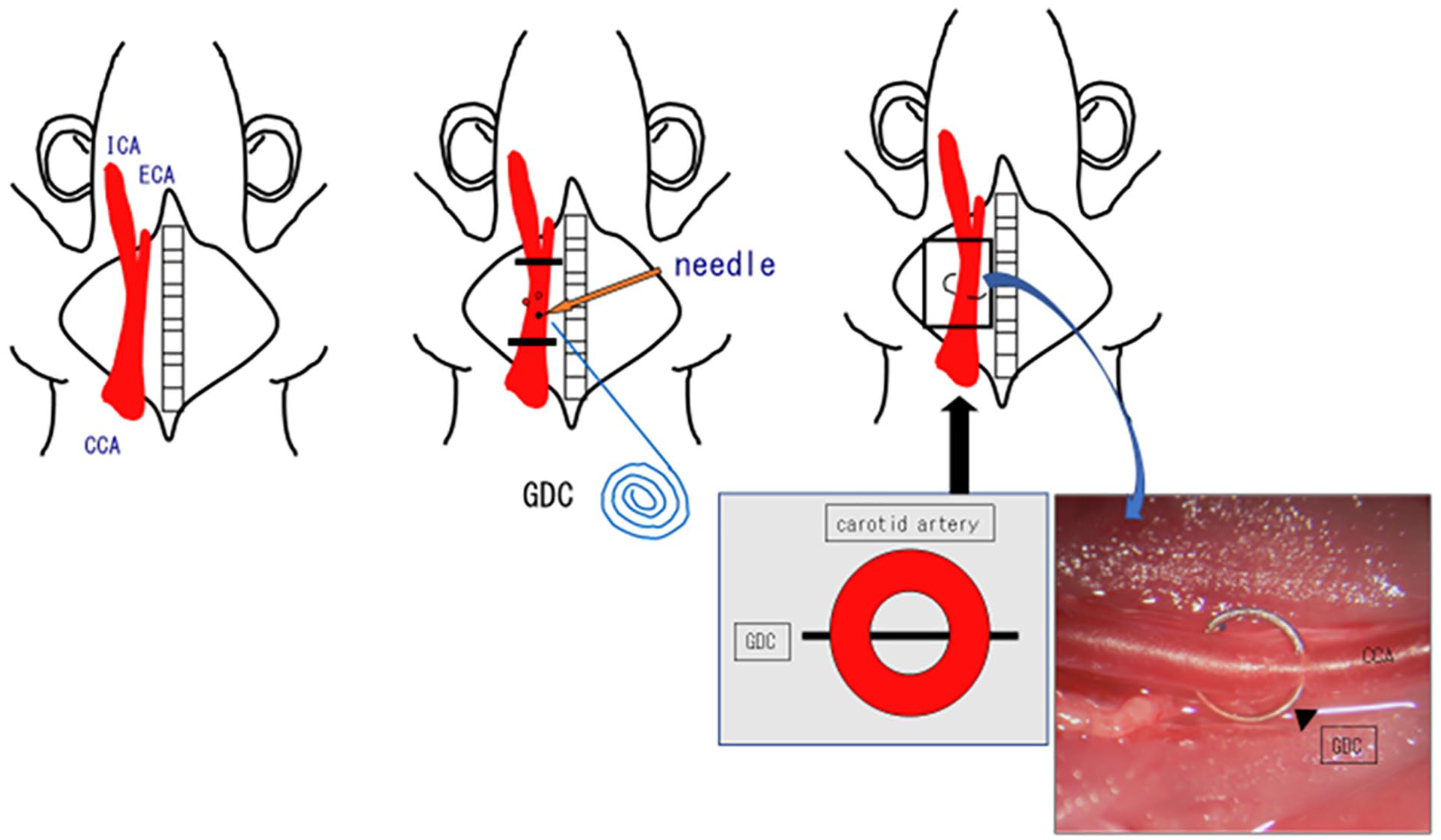
Model of a jailed coil in the carotid artery. The carotid artery is perforated transversely with a platinum coil, suspending it across the center of the intravascular space.
Rats were sacrificed at 1, 2, or 4 weeks after coil placement to excise the region for observation, and the specimens were evaluated by scanning electron microscopy (SEM) and confocal microscopy (CFM) after fluorescent immunostaining for Flk1.
The endothelial coverage on the coil surface, observed using by SEM and CFM, was graded by using the Fukawa staging (F-stage: Figure 2), a novel framework developed for this model, which was defined as follows:
□ F-Stage I: Endothelialization has proceeded along the distal side of the coil (i.e. the side facing the brain, away from the heart), spanning its long axis.
□ F-Stage II: The new endothelium has spread to cover the entire circumference of the coil where it meets the vessel wall, while expanding toward its center; coverage spans <50% of the coil width (short axis).
□ F-Stage III: The new endothelium covers >50% of the coil width.
□ F-Stage IV: The coil surface is completely covered by vascular endothelial cells.

Fukawa-stage (F-stage). The F-stage reflects the classification of the formation of endothelial progenitor cells around the coil in the carotid artery. Stage I: endothelialization proceeds (arrow) along the distal side of the coil (i.e. the side facing the brain, away from the heart), spanning its long axis. Stage II: the new endothelium spreads to cover the entire circumference of the coil where it meets the vessel wall, while expanding toward its center; the coverage spans <50% of the coil width (short axis). Stage III: the new endothelium covers >50% of the coil width. Stage IV: the coil surface is completely covered by vascular endothelial cells.
Endothelialization was graded in each group at 1, 2, and 4 weeks after coil placement. Group averages were additionally calculated, and the endothelial diameter was compared among the groups (i.e. thickness = endothelialized coil diameter – coil diameter).
ANOVA (analysis of variance) tests were performed to evaluate differences in vascular thickness between groups, and P < .05 was considered statistically significant. All statistical analyses were performed with EZR4 (Saitama, Japan), which is a modified version of the R commander designed to add statistical functions that are frequently used in biostatics.
Result
Figure 3 shows a summary of the number of rats in each group according to the F-stage, based on the degree of endothelialization on the coil surface. In most rats in the LETO (control) group, the coil had become completely covered by the vascular endothelium by 1 week after placement. In contrast, none of the OLETF rats (with one exception) had reached F-stage IV by 1 week; the majority of rats took 4 weeks to reach F-stage IV, a consequence of the strain’s hyperglycemic inhibition of endothelialization. Furthermore, after 1 week, a higher number of rates were rated as F-stage III in the cilostazol group than in the aspirin and clopidogrel groups. Figure 4 shows a comparison of the average diameter of the new endothelium formed on the coil surface among the 5 groups. The neoendothelium was significantly thicker in the OLETF rats fed a normal, unadulterated diet than in the LETO rats fed the same diet (one-way ANOVA; P = .002). Similarly, the neoendothelium was significantly thicker in OLETF rats administered aspirin than in the control group (P = 0.000, respectively), but not in those administered cilostazol (P = 0.026). In comparison, the neoendothelium was significantly thinner in OLETF rats in the cilostazol and clopidogrel groups than in OLETF rats fed a normal, unadulterated diet (P = 0.026 and P = 0.000, respectively). In addition, the neoendothelium was significantly thicker in the aspirin group than in the cilostazol group (P = 0.000; Figure 4–2).

Summary of the F-stage classification of the vascular endothelial cells in the 5 study groups. In normal rats fed a plain diet (LETO), the endothelium had formed over the entire coil surface within the first week after coil placement. In diabetic rats (OLETF) administered different antiplatelet drugs (cilostazol, aspirin, or clopidogrel) or those with no drug administered (CE-2, control), complete coverage was delayed until the fourth week. The cilostazol group had a higher number of rats at F-stage III at 1 week after coil placement than the other groups.
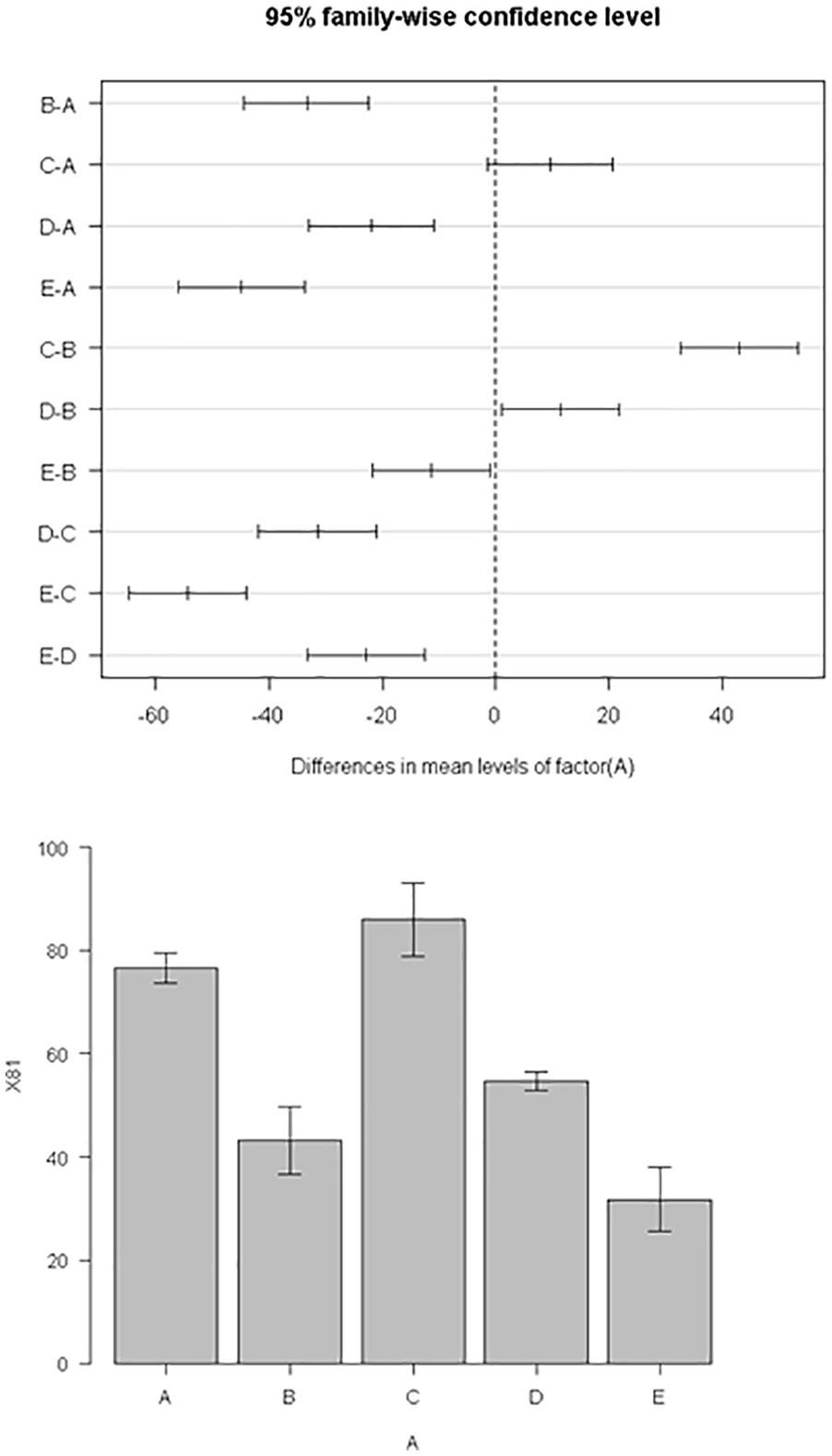
Vascular endothelial diameter: comparisons to that in normal rats (ANOVA). The neoendothelium was significantly thicker in the OLETF rats fed a normal, unadulterated diet than in the LETO rats fed the same diet (one-way ANOVA; P = .002). the neoendothelium was significantly thicker in OLETF rats administered aspirin than in the control group (P = .000, respectively), but not in those administered cilostazol (P = .026). The neoendothelium was significantly thinner in OLETF rats in the cilostazol and clopidogrel groups than in OLETF rats fed a normal, unadulterated diet (P = .026 and P = .000, respectively). the neoendothelium was significantly thicker in the aspirin group than in the cilostazol group (P = .000).
Discussion
The present study data seem to indicate that neoendothelialization is delayed when the vascular endothelial cells cannot function properly, resulting in intimal hyperplasia. In addition, cilostazol and clopidogrel inhibited this hyperplastic response in diabetic rats in our carotid coil model.
In the present study, we established a novel experimental model, in which the carotid artery of diabetic (OLETF) rats was perforated with a platinum embolization coil,4,5 with the assumption that the damage to the vascular endothelium would be conspicuously sustained under hyperglycemic conditions. 3 Flk1, also known as vascular endothelial growth factor receptor 2 (VEGFR-2), is typically expressed by EPCs and plays an important role in angiogenesis.6,7 Thus, the components observed at the coil surface that stained positive for Flk1 were most likely cells that differentiated from EPCs. There is already an abundance of evidence that EPCs are a subpopulation of CD34-positive somatic stem cells, which are present in the peripheral blood and direct vasculogenesis under the control of VEGF and other factors.8,9
Vascular endothelial cells were visible around the entire circumference of the coil at 1 week after placement in normal rats, but this coverage was not as complete as in diabetic rats. Thus, our experimental model strongly supports the idea that endothelial damage occurs in diabetic rats. We continued to monitor the endothelialization process in the carotid artery at the surface of the coil over time, rating it in terms of the F-stage, a classification scheme that we created especially for our carotid coil model, dividing the repair process into 4 stages (I–IV: Figure 2). The new endothelium started to form from the distal side of the coil surface, that is, opposite to the incoming blood (Figure 5, New Endothelialization Step 1). Next, the cells started to wrap around its circumference at the vessel wall, while simultaneously spreading inward toward its center, until ultimately the tissue covered the entire coil surface (Figure 5, New Endothelialization Step 2). Using the endothelial thickness of this F-stage classification, we compared the thickness of the new endothelium between normal and diabetic rats fed a normal diet, and found it to be significantly thicker in the latter (one-way ANOVA; P = .002). The cells that formed at the coil surface were most likely EPCs, as evidenced by the positive Flk1 staining on the right side of Figure 6, suggesting that, in diabetic rats, the intimal hyperplasia of the new endothelium proliferates unabated.

The mechanism of endothelialization on the surface of the coils.
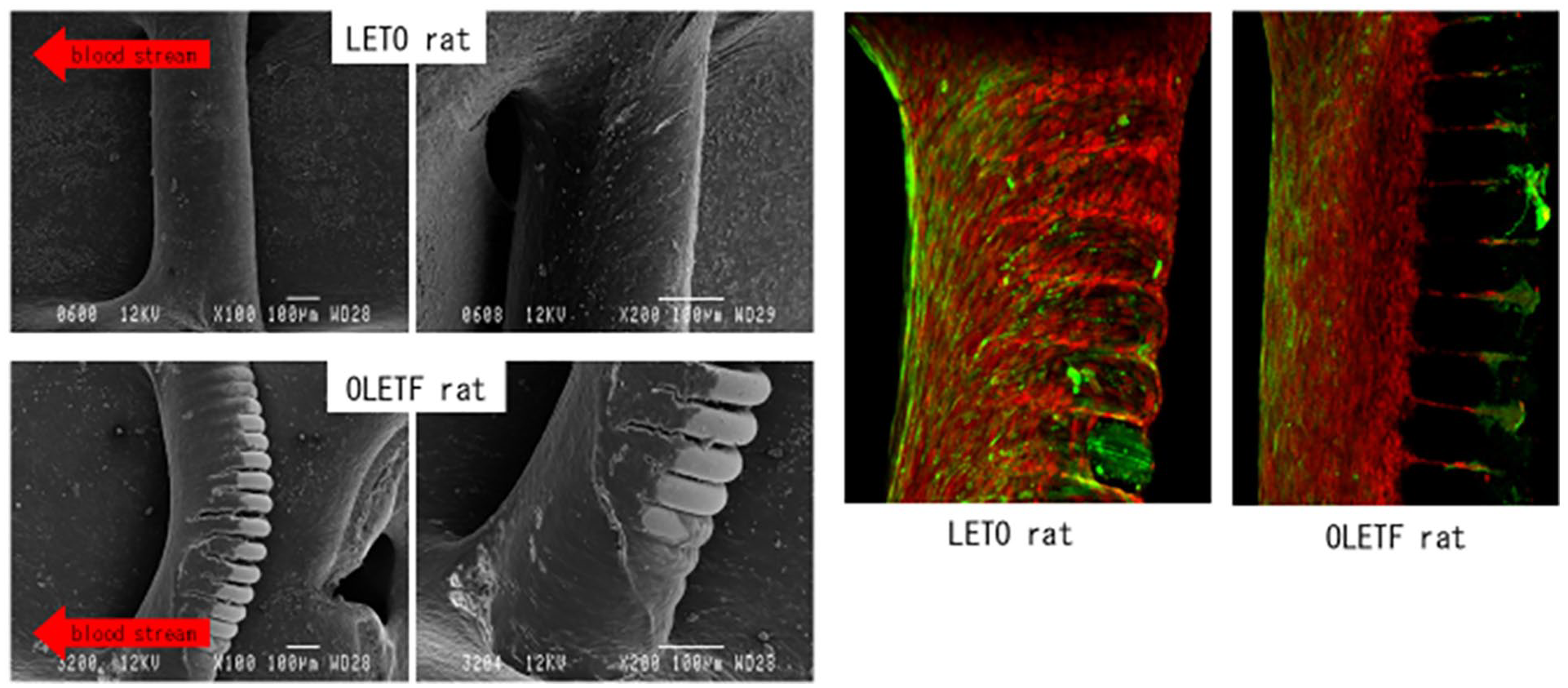
Scanning electron microscopy (SEM) of the jailed coil model after 1 week (left) and the confocal microscopic study by Flk1 staining (right). Left: SEM images taken at 1 week after coil insertion. The upper part shows images of normal rats (LETO); the endothelial cells are observed in the entire circumference of the coil within 1 week. The lower part shows images of diabetic rats (OLETF); during the same time period, the coil circumference is not entirely covered. Upper: in normal rats (LETO), endothelial cells have already covered the entire circumference of the coil at 1 week after placement. Lower: in diabetic rats (OLETF), endothelial coverage does not span the entire circumference of the coil at 1 week after placement. Right: CFM images of specimens stained for Flk1 (2 weeks after coil insertion). LETO rats are well-dyed with Flk1, but the OLETF rats are less stained.
The present study also determined the effects of antiplatelet drugs on angiogenesis. We found that, while intimal proliferation was not inhibited in diabetic rats administered aspirin, it was suppressed in those administered cilostazol or clopidogrel (P = .00 and P = 0.00, respectively). These 2 drugs seem to counteract the negative effects of the hyperglycemic environment on vascular endothelium repair, thereby acting to suppress the intimal hyperplasia. Cilostazol appeared to be superior to clopidogrel in this regard; the neoendothelium in the cilostazol group was significantly thinner than that of the clopidogrel group (P = 0.026), nor was it significantly thicker than that of the normal, non-diabetic rats. Furthermore, Flk1 staining in the neoendothelium was more intense in rats administered cilostazol than in rats administered clopidogrel (Figure 7).
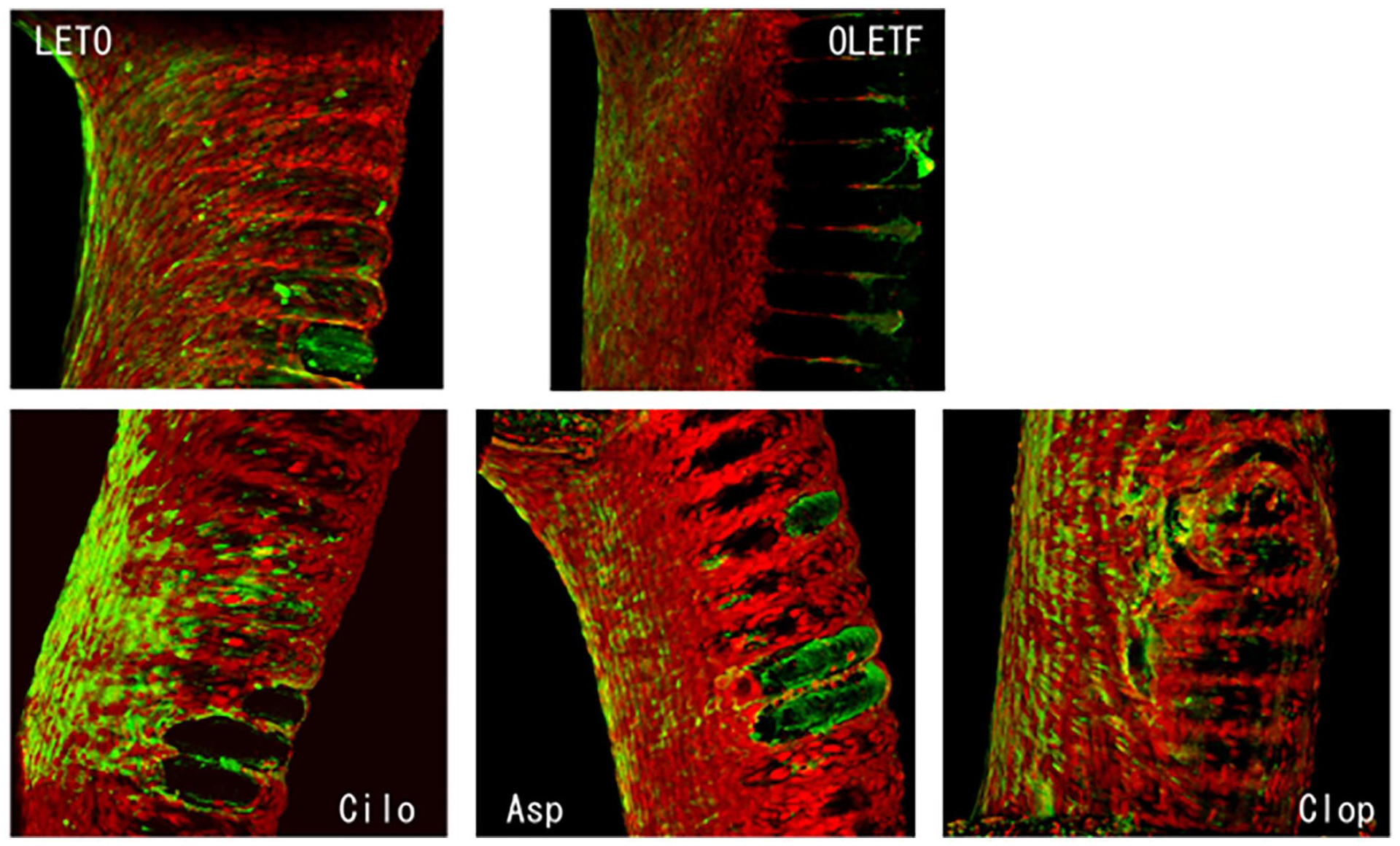
Confocal microscopic study at 2 weeks after placement. Green spots are abundant in both LETO and OLETF specimens. Specimens from rats administered with cilostazol contained the highest number of green spots among the antiplatelet groups, comparable in density with that of the LETO (control) group.
Several factors could be responsible for the different patterns of intimal hyperplasia observed among the treatment groups. Aspirin suppresses the activity of cyclooxygenase-1 (COX-1), which acts to inhibit the synthesis of thromboxane A2, thereby resulting in platelet aggregation. 10 Although aspirin inhibits platelet aggregation only through this pathway, both cilostazol and clopidogrel do so by elevating the levels of cAMP.11,12 Cilostazol is an antiplatelet drug often used for secondary prevention in patients with high-risk ischemic stroke in Japan. Cilostazol was found to better suppress intimal hyperplasia in the carotid artery (i.e. the neoendothelium showed a significantly smaller diameter) than did aspirin in a randomized control trial, 13 with the authors offering EPC recruitment due to elevated cAMP levels as a potential mechanism. Furthermore, Asai et al 14 have reported that cAMP promotes EPC accumulation during the repair of vascular damage. EPCs were likely recruited to the coil surface in our model via these mechanisms as well.
Discrepancies in cardiac outcomes have been observed among patients taking clopidogrel, corresponding to genetic polymorphisms. 15 It is possible that our observations of weaker Flk1 staining and less-inhibited intimal proliferation among diabetic rats in the clopidogrel group, in comparison with those in the cilostazol group, were related to the similar genetic differences. Thus, in the future, we need to verify whether there are differences in the mechanisms of action between these 2 drugs, as suggested, for example, by cilostazol’s apparently superior ability to recruit EPCs to form the vascular endothelium.
The available evidence, thus, suggests that the new endothelium forms at a slower rate when EPCs cannot function properly, and the tissue that forms is thicker than that under normal conditions. Accordingly, cilostazol’s ability to suppress intimal proliferation could be a key to improving the function of vascular endothelial cells. Clinical evidence is lacking regarding the relative efficacy of different antiplatelet drugs in managing deviations following coil embolization to treat cerebral aneurysms and in maintaining penetrating branch patency during stent placement to treat stenotic arteriosclerotic lesions. Our findings bolster the idea of cilostazol’s efficacy in such clinical settings. Other reports have implicated neovascularization in the formation of unstable plaques, which are a cause of cerebral infarction. 16 Our work suggests that cilostazol could additionally act to prevent such events by suppressing the abnormal proliferation of new blood vessels. One study directly implicated EPCs in the reduction of infarct volume and improvement of neurological symptoms after stroke, 17 suggesting that their abnormal proliferation contributes to ischemia. We demonstrated that cilostazol can suppress this abnormal proliferation.
It makes sense that the new endothelium would initially form at the distal side of the platinum coil in our model, because that is where the shearing stress due to blood flow is the weakest; from there, it spreads against the flow to cover the coil, moving up in the F-stage classification. EPCs are hypothesized to be involved in this process (Figure 5). Although the so-called tip, stalk, and phalanx are believed to be involved in angiogenesis, the details of their contributions are still unclear. 18 Hereafter, it will be necessary to examine the involvement of EPCs with these antigenic factors.
There is increasing evidence supporting cilostazol’s clinical utility. 19 A systematic review found that cilostazol effectively reduces diabetes-associated microvascular damage, 20 whereas a meta-analysis found that it effectively decreases amputation risk and helps in maintaining patency in cases of arterial lesions of the lower limbs following an interventional surgery. 21 We find it ethically difficult to clinically investigate the vascular endothelial repair effects of each antiplatelet drug. We eagerly await further reports on the drug’s benefits and applications.
Conclusion
Cilostazol appears to remedy endothelial cell dysfunction by recruiting EPCs to the site of vascular injury.
Footnotes
Acknowledgements
Funding:
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contribution
NF, TU, YK, JT were involved in study design and data interpretation. TO and TU were involved in the data analysis. All authors critically revised the report, commented on drafts of the manuscript, and approved the final report.
