Abstract
Purpose:
Abiraterone acetate is an androgen synthesis inhibitor used to treat metastatic castration-resistant prostate cancer. The therapeutic equivalence, pharmacokinetics, and safety of abiraterone acetate fine particle (AAFP) versus originator abiraterone acetate (OAA) were examined in a Phase-2 study; sufficient similarity was observed between the 2 treatments. This 1-year, open-label, extension study evaluated the long-term safety and efficacy of AAFP.
Patients and methods:
This was an open-label, single-arm extension study. Patients who completed the 84-day, Phase-2, run-in study could enroll to receive 500 mg AAFP once daily and 4 mg methylprednisolone twice daily for up to 1 year. Safety was the primary endpoint, as assessed by adverse event monitoring throughout the study. Secondary efficacy endpoints included change from run-in study baseline levels of serum testosterone and prostate-specific antigen measured at baseline, 6 months, and 1 year.
Results:
Twenty patients enrolled, of whom 9 and 11 received prior AAFP and OAA in the run-in study, respectively. Both prior treatment groups maintained a significant decrease in serum testosterone from run-in baseline at all timepoints; mean change from run-in baseline ± standard deviation at 1 year of −6.24 ± 2.02 (P = .0023) and −5.87 ± 3.64 (P = .0026) ng/dL for the prior AAFP and OAA group, respectively. Change from run-in baseline in serum prostate-specific antigen was not significant. Most (84.0%) adverse events were Grade 1 or 2.
Conclusion:
No new safety signals were identified, and patients were able to successfully switch from OAA to AAFP with no impact.
Introduction
Androgen receptor activation has long been recognized as the driver of prostate cancer growth, even in the setting of metastatic castration-resistant prostate cancer (mCRPC).1,2 Androgen deprivation therapy serves as the foundation of treatment for progressive and advanced prostate cancer. 3 Castration, either medical or surgical, markedly reduces circulating androgen levels, achieving a remission in select prostate cancer phenotypes. However, in certain subtypes, the disease progresses to castration-resistant prostate cancer (CRPC) which will ultimately result in death.3-5 In the CRPC setting, malignant cells can survive, proliferate, and migrate despite castrate levels of testosterone through a variety of escape mechanisms, including production of testosterone from extra gonadal sources or de novo androgen synthesis by the prostate cancer cells themselves.6-8 Treatment of these patients requires targeting these alternative pathways.
Abiraterone is an irreversible inhibitor of cytochrome P450 17-alpha hydroxylase/17,20 lyase (CYP17), a key enzymatic step in androgen biosynthesis. 9 Originator abiraterone acetate (OAA; Zytiga®; Janssen Biotech, Inc; Horsham, PA) is a formulation of abiraterone that, in combination with prednisone, has been found to improve overall survival in patients with mCRPC previously treated with docetaxel and progression-free survival in patients with mCRPC, both with and without prior docetaxel treatment.10-12 Treatment with monotherapy OAA, in combination with prednisone, 5 mg twice daily, is recommended by the National Comprehensive Cancer Network, American Urologic Association/Society for Urologic Oncology, and American Society of Clinical Oncology (ASCO) for patients with mCRPC.13-15
Abiraterone acetate fine particle (AAFP; Yonsa®; Sun Pharmaceutical Industries, Inc; Cranbury, NJ) is a novel formulation of abiraterone designed to provide comparable absorption to OAA at a lower dose using SoluMatrix Fine Particle Technology™ that in combination with methylprednisolone is indicated for the treatment of mCRPC. 16 A Phase-2, randomized, open-label, active-controlled study established therapeutic equivalence between 500 mg AAFP once daily to 1000 mg OAA once daily in patients with mCRPC and reported no new safety concerns associated with 84 days of AAFP treatment. 17 This study evaluated the safety and continued efficacy of an additional 1 year of treatment with AAFP in patients who had received either OAA or AAFP in the run-in study.
Methods
Study design
This 1-year, multicenter, open-label, single-arm extension study (NCT02962284) adhered to the tenets of the Declaration of Helsinki and the International Council for Harmonization guidelines for Good Clinical Practice. The study protocol and all relevant supporting information were approved by an institutional review board prior to study initiation, and all enrolled patients provided informed consent.
Patients who successfully completed the 84-day Phase-2 therapeutic equivalence study 17 and could benefit from 1 year of treatment with AAFP as judged by the investigator were eligible to enroll in the extension study if they were receiving ongoing therapy with gonadotropin-releasing hormone agonist or antagonist, and had a serum testosterone level <50 ng/dL and a life expectancy of at least 9 months at screening. Patients were excluded from the study if they were unable to swallow tablets whole or had a serious concurrent illness that could interfere with study participation; hypersensitivity to abiraterone acetate, methylprednisolone, or any component of the study drugs; or had moderate-to-severe hepatic impairment. Results from the Day 84 (last day) visit of the Phase-2 therapeutic equivalence study were used to determine eligibility for this study.
In the run-in study, patients were randomized in a 1:1 ratio to either 500 mg AAFP once daily with 4 mg methylprednisolone twice daily, or 1000 mg OAA once daily with 5 mg prednisone twice daily for 84 days. In the extension study, all patients received 500 mg AAFP once daily and 4 mg methylprednisolone twice daily for 360 days. Key prohibited concomitant medications and therapies during the extension study included medications known to modulate hepatic drug metabolism, substrates of cytochrome P450 2D6 with a narrow therapeutic index, and antiandrogens and estrogens.
Assessments
Efficacy assessments
Change from run-in study baseline in serum testosterone (T) and prostate-specific antigen (PSA) levels at 6 months and 1 year or early termination were secondary endpoints in the study. Serum T was measured using an ultra-sensitive assay (Covance Inc, LabCorp, Burlington, NC) with a lower limit of quantification (LLOQ) of 0.1 ng/dL. An assay with LLOQ of 0.02 ng/mL (Esoterix Laboratory Services, Calabasas, CA) was used to measure serum PSA. Assessments performed at Days 0 and 84 of the run-in study were used as prior baseline and baseline measurements, respectively, for this study.
Safety assessments
The primary endpoint was safety of AAFP 500 mg once daily with 4 mg methylprednisolone twice daily in patients with mCRPC more than 1 year, following an initial treatment of either AAFP 500 mg once daily with 4 mg methylprednisolone twice daily or OAA 1000 mg once daily with 5 mg prednisone twice daily. Key safety assessments included adverse event (AE) monitoring, liver function tests, serum hematology, and vital signs.
Adverse events were monitored throughout the study and reported by system organ class and preferred term using Medical Dictionary for Regulatory Activities, versions 18.1 and 19.1, for AEs and serious AEs, respectively. Assessments of intensity grade were made according to the National Cancer Institute Common Terminology Criteria for AEs, version 4.03. Liver function tests, including serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), and total bilirubin, were performed monthly starting at 1 month until 1 year or early termination. Serum hematology was assessed at baseline (Day 84 of the run-in study), 6 months, and 1 year or early termination. Vital signs were measured at baseline and every 3 months until 1 year or early termination.
Statistical analyses
The intent-to-treat and safety populations were identical and included all patients who were enrolled and received at least 1 dose of study drug in the extension study. For statistical analyses, patients were divided into 2 groups according to the patient’s treatment in the run-in study (herein referred to as prior AAFP and prior OAA groups).
Categorical variables were summarized using frequencies and percentages, whereas continuous variables were summarized using descriptive statistics (n, mean, standard deviation [SD], median, minimum, and maximum). Hypothesis testing was performed at the 5% significance level using Student’s t-test for serum T and PSA levels and an analysis of covariance model on change from baseline with treatment and baseline (covariate) included for liver function tests.
Results
Patient disposition and demographics
A total of 20 patients enrolled; 9 (45.0%) received prior AAFP and 11 (55.0%) received prior OAA. Twelve (60.0%) patients completed the study (Figure 1); 4 (44.4%) in the prior AAFP group and 8 (72.7%) in the prior OAA group. Reasons for discontinuation included death (2 [22.2%] patients in the prior AAFP group), progressive disease (2 [22.2%] and 3 [27.3%] patients in prior AAFP and OAA groups, respectively), and withdrawal of consent (1 [11.1%] patient in the prior AAFP group). Enrolled patients were predominantly White (75.0%) with a median ± SD age of 76.5 ± 8.8 years (Table 1). Overall, 13 (65.0%) patients had a Gleason grade >7, 3 (15.0%) had a Gleason grade of 7, and 3 (15.0%) had a Gleason grade <7.
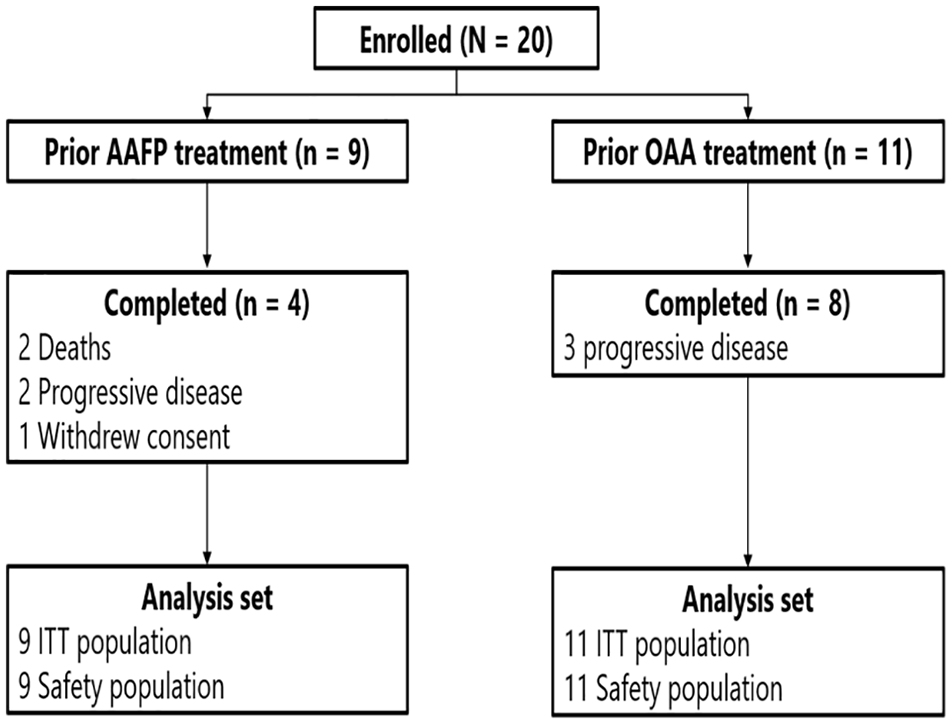
Patient disposition.
Demographic and baseline disease characteristics.
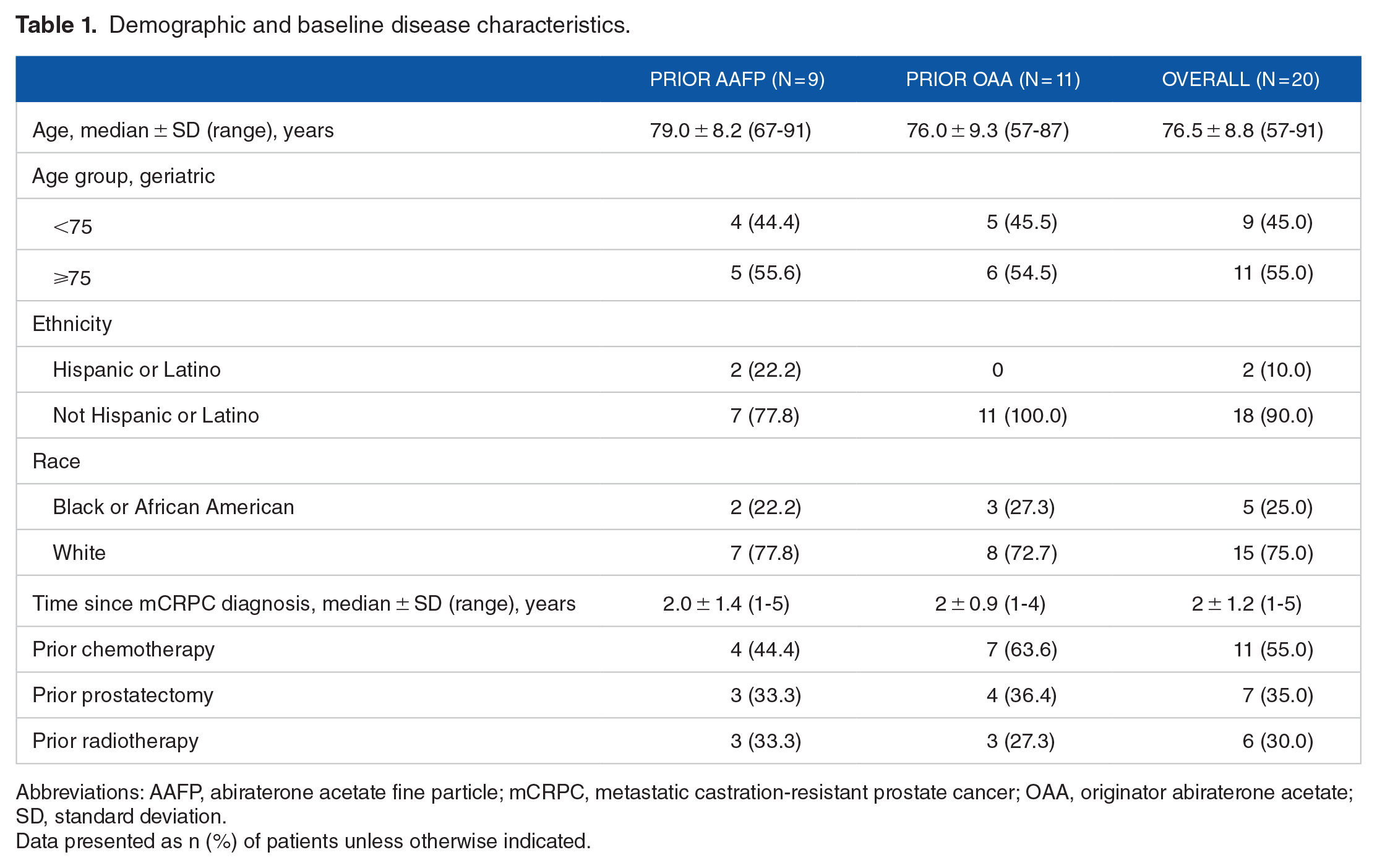
Abbreviations: AAFP, abiraterone acetate fine particle; mCRPC, metastatic castration-resistant prostate cancer; OAA, originator abiraterone acetate; SD, standard deviation.
Data presented as n (%) of patients unless otherwise indicated.
Efficacy
Serum testosterone levels
Serum T levels were significantly decreased from run-in study baseline at all timepoints in both prior treatment groups (Figure 2). For the prior AAFP group, the mean run-in study baseline serum T ± SD was 6.53 ± 1.68 ng/dL. Serum T mean change from baseline ± SD was −6.27 ± 1.72 (P < .0001), −4.93 ± 0.86 (P = .0100), and −6.24 ± 2.02 ng/dL (P = .0023) at extension study baseline, 6 months, and 1 year, respectively. For the prior OAA group, run-in study baseline mean serum T ± SD was 7.39 ± 4.42 ng/dL. Serum T mean change from baseline ± SD was −7.20 ± 4.45 (P = .0003), −7.26 ± 5.80 (P = .0280), and −5.87 ± 3.64 ng/dL (P = .0026) at extension study baseline, 6 months, and 1 year, respectively.
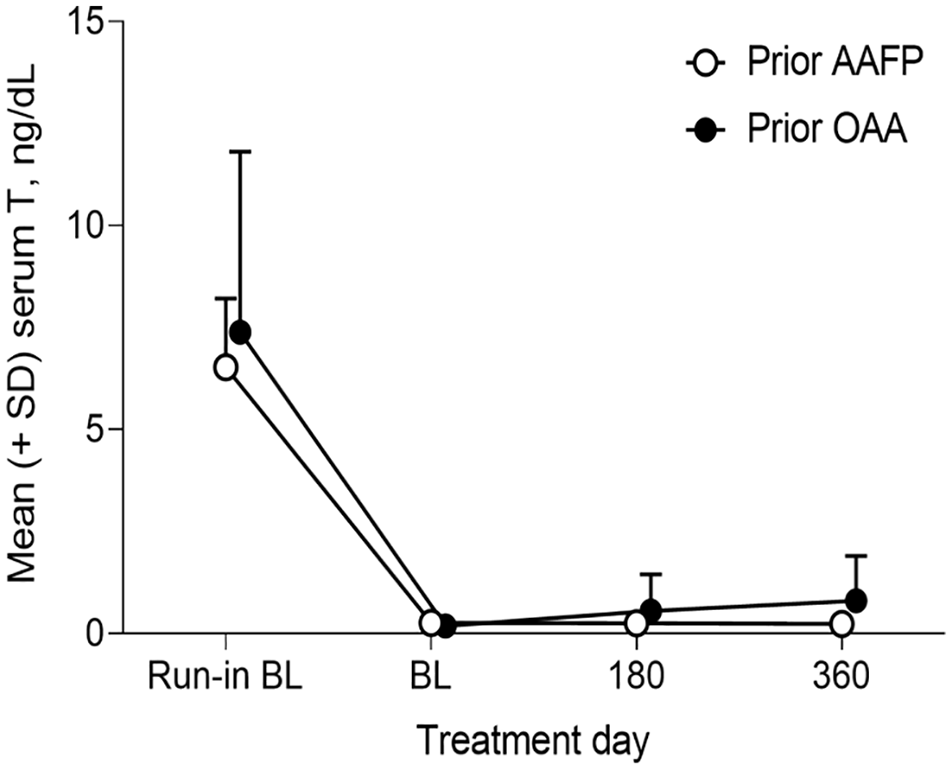
Mean serum testosterone levels ± standard deviation over time.
Serum PSA levels
The mean run-in study baseline serum PSA ± SD for prior AAFP group was 55.72 ± 84.55 ng/mL versus 133.32 ± 296.04 ng/mL for prior OAA group (Figure 3). The change in serum PSA from run-in study baseline was not statistically significant at any timepoint of this study for either prior treatment group. The mean change from run-in study baseline ± SD for prior AAFP group was −48.66 ± 79.45 (P = .1035), −56.18 ± 103.90 (P = .2426), and −60.12 ± 115.63 ng/mL (P = .3096) at baseline, 6 months, and 1 year, respectively, versus −94.46 ± 192.80 (P = .1352), −106.38 ± 214.42 (P = .1511), and −112.76 ± 239.85 ng/mL (P = .1961) at baseline, 6 months, and 1 year, respectively, for prior OAA group.
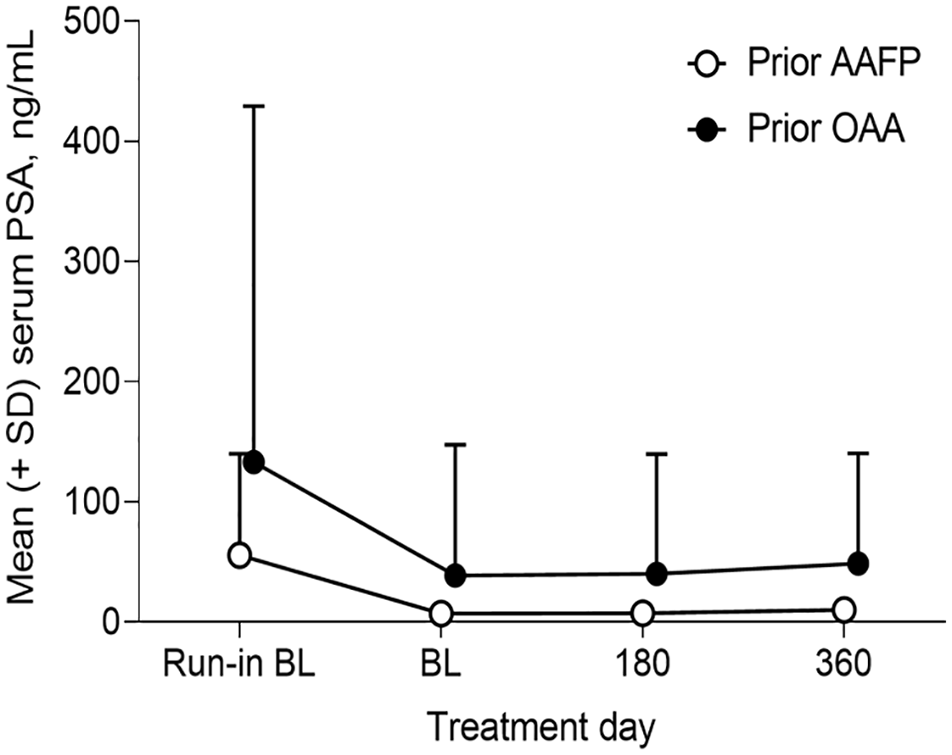
Mean serum prostate-specific antigen level ± standard deviation over time.
Safety
Liver function tests and other clinical evaluations
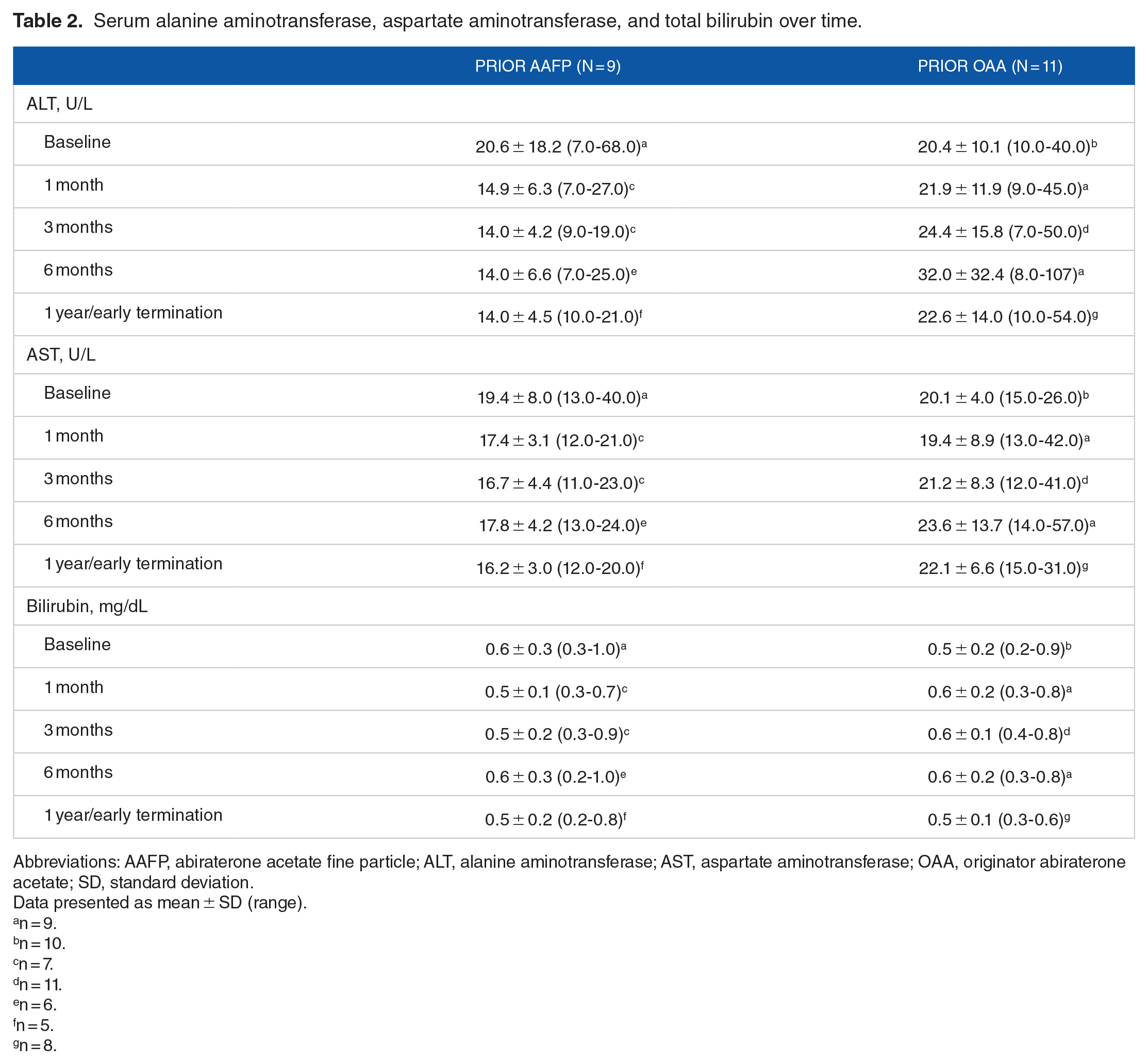
Changes from baseline in serum ALT, AST, and total bilirubin were small with no consistent significant changes from baseline or significant differences between prior treatment groups (Table 2). No individual patient exhibited major changes from baseline throughout the study.
Serum alanine aminotransferase, aspartate aminotransferase, and total bilirubin over time.
Abbreviations: AAFP, abiraterone acetate fine particle; ALT, alanine aminotransferase; AST, aspartate aminotransferase; OAA, originator abiraterone acetate; SD, standard deviation.
Data presented as mean ± SD (range).
n = 9.
n = 10.
n = 7.
n = 11.
n = 6.
n = 5.
n = 8.
Mean serum ALT ± SD was 20.56 ± 18.20 versus 20.40 ± 10.06 U/L at baseline for prior AAFP versus prior OAA group, respectively; and 14.00 ± 4.53 versus 22.63 ± 13.96 U/L at 1 year/early termination for the prior AAFP versus prior OAA group, respectively. Mean change from baseline in serum ALT ± SD was significant at 1 month (−7.43 ± 14.89 vs 1.67 ± 5.15 U/L for prior AAFP vs prior OAA group, respectively; P = .0345) but not significant at any subsequent timepoints.
Baseline serum AST had a mean ± SD of 19.44 ± 8.03 and 20.10 ± 4.04 U/L for prior AAFP and prior OAA group; at 1 year, mean serum AST ± SD was 16.20 ± 3.03 and 22.13 ± 6.58 U/L for the prior AAFP and prior OAA group, respectively. Mean change from baseline in serum AST ± SD was significant at 10 months (3.20 ± 5.63 vs −4.14 ± 3.24 for prior AAFP vs prior OAA group; P = .0446) and 11 months (4.20 ± 2.95 vs −3.71 ± 1.80 for prior AAFP vs prior OAA group, respectively; P = .0008) but not significant at all other timepoints.
Serum total bilirubin mean ± SD at baseline was 0.57 ± 0.25 and 0.50 ± 0.19 mg/dL for the prior AAFP and prior OAA group, respectively; at 1 year, mean ± SD was 0.52 ± 0.24 and 0.45 ± 0.12 mg/dL for the prior AAFP and prior OAA group, respectively. There was a significant mean change from baseline in serum total bilirubin ± SD at 5 months (−0.10 ± 0.15 vs 0.13 ± 0.12 for prior AAFP vs prior OAA group, respectively; P = .0096) and 7 months (−0.18 ± 0.13 vs 0.13 ± 0.17 for prior AAFP vs prior OAA group, respectively; P = .0125) but not significant at all other timepoints.
Serum hematology and vital sign assessments throughout the study did not exhibit major changes from baseline or significant differences between prior treatment groups.
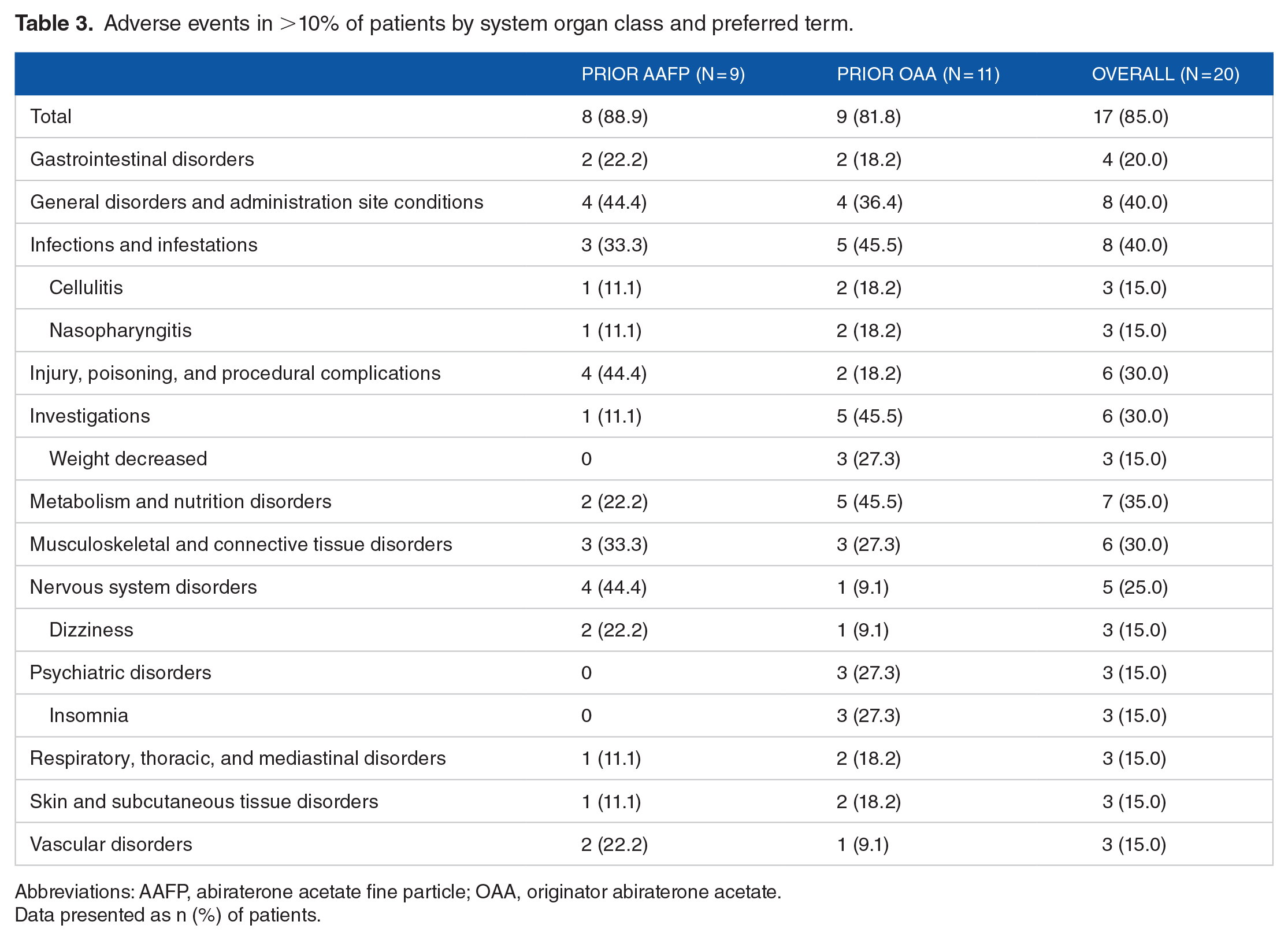
Adverse events
A total of 8 (88.9%) and 9 (81.8%) patients in the prior AAFP and OAA groups, respectively, reported AEs (Table 3), which were mostly mild or moderate; no severe AEs were considered related or possibly related to study drugs. Most (84.0%) AEs were Grade 1 or 2 in toxicity. One patient in the prior AAFP group reported a Grade 3 AE (syncope) that was considered related or possibly related to study drugs; all other grade ⩾2 AEs were considered unrelated or unlikely to be related to study drugs. Two deaths occurred, both in the prior AAFP group; 1 due to septic shock and 1 due to head injury, and neither was considered related to study treatment. The 2 deaths were the only discontinuations due to AEs. Serious AEs were reported in 2 additional patients—1 patient from the prior OAA group experienced cellulitis and 1 patient from the prior AAFP group experienced 4 serious AEs (abasia [inability to walk due to impaired coordination], asthenia, dehydration, and noncardiac chest pain)—all considered unrelated to treatment.
Adverse events in >10% of patients by system organ class and preferred term.
Abbreviations: AAFP, abiraterone acetate fine particle; OAA, originator abiraterone acetate.
Data presented as n (%) of patients.
Most common AEs by system organ class included general disorders and administration site conditions in 4 (44.4%) and 4 (36.4%) patients in the prior AAFP versus prior OAA groups (Table 3). Hepatic AEs included ALT increased in 1 (9.1%) patient from the prior OAA group. Fluid retention AEs included face edema (1 [11.1%] patient in the prior AAFP group), peripheral swelling (1 [11.1%] patient in the prior AAFP group and 1 [9.1%] patient in the OAA group), hyperkalemia (2 [18.2%] patients in the prior OAA group), and hypokalemia (1 [9.1%] patient in the prior OAA group).
Discussion
This study found no impact on safety or efficacy when switching patients from treatment with OAA to treatment with AAFP. A statistically significant decrease in serum T levels from the run-in study baseline was successfully maintained through 1 year of treatment with AAFP in patients who received either prior AAFP or prior OAA. The mean change from run-in baseline in serum PSA levels was not statistically significant for either prior treatment group. AAFP with methylprednisolone was well tolerated throughout the study, and no new safety concerns were raised.
National Comprehensive Cancer Network recommends OAA or AAFP with a corticosteroid (either prednisone or methylprednisolone) for patients with mCRPC, along with monthly monitoring of liver function, potassium and phosphate levels, and blood pressure. 14 The official ASCO and Cancer Care Ontario guidelines include OAA plus prednisone among the treatments that have demonstrated survival and quality-of-life benefits and should be offered to all patients with mCRPC. 13 American Urologic Association recommends abiraterone plus prednisone—among other therapies—be offered to patients with mCRPC and good Eastern Cooperative Oncology Group (ECOG) performance status (abiraterone may still be offered to patients with poor ECOG performance status). 15
At present, there is no cure for mCRPC, and treatment needs to be continued indefinitely until decision to discontinue or death. 13 A real-world retrospective observational study found a mean treatment duration ± SD of 195.0 ± 168.9 days with OAA. 18 Therefore, treatment with abiraterone can potentially be long-lasting; clinical evidence of sustained efficacy and safety of abiraterone over the long term is needed to establish real-world benefit for patients with CRPC, and the 1-year duration of this study suggests its conclusions are relevant to a real-world setting.
Limitations of the study include the small sample size of 20 patients, which could potentially contribute to the observed lack of significant change in PSA levels from prior study baseline. The study also used surrogate measurements (serum T and PSA levels) to evaluate efficacy, although this limitation is somewhat mitigated by safety being the primary endpoint of the study. Assessment of serum dehydroepiandrosterone (DHEA) and sulfonated DHEA levels would provide more direct evidence of longitudinal CYP17 inhibition than serum T. Further studies with greater sample size and number of assessments would strengthen the observations of AAFP efficacy and safety established here.
In summary, AAFP with methylprednisolone showed a favorable benefit–risk profile over 1 year of treatment in patients with mCRPC. Switching patients from OAA to AAFP did not impact safety or efficacy of AAFP.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored and funded by Churchill Pharmaceutical, LLC, King of Prussia, PA. Writing and editorial support for manuscript preparation were provided by Ginny Feltzin, PhD, of AlphaBioCom, LLC, King of Prussia, PA, and funded by Sun Pharmaceutical Industries, Inc, Princeton, NJ, USA. All authors met the International Council of Medical Journal Editors criteria and received neither honoraria nor payment for authorship.
Declaration of Conflicting Interest:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.C-.R. is an employee of Adastra Pharmaceuticals. C.S. receives honoraria from Astellas, Pfizer, and Sanofi; consulting fees from Pfizer and Sun Pharmaceutical Industries, Inc; research funding from Roche; and travel compensation from Sanofi. N.S. is an employee of Sun Pharmaceutical Industries, Inc and receives honoraria from Galderma, Leo, Novartis, Pierre Fabre, and Roche; consulting fees from Galderma, Leo, Novartis, Pierre Fabre, and Roche; patents, royalties, or other intellectual property from Genentech/F. Hoffmann-La Roche, Ltd.; and travel, accommodations, or expenses from Galderma, Leo, and Roche. R.C. serves as a consultant for Cellay; Clovis; Dendreon; Integra Connect; and Sun Pharmaceutical Industries, Inc; and serves on the speaker’s bureau for Amgen, Astellas, and Pfizer.
Author Contributions
All authors have contributed to the conception, development, writing, and final approval of the manuscript.
