Abstract
Background:
This study compared urinary and serum nerve growth factor levels and serum C-reactive protein levels in patients with overactive bladder, before and after treatment with antimuscarinic agents, to evaluate their diagnostic and therapeutic importance.
Methods:
This was a prospective case-control study conducted between February 2014 and December 2014 which included patients with a confirmed diagnosis of overactive bladder (overactive bladder group) and healthy controls (control group). All patients visiting urology out-patient department with urgency, frequency, and urge incontinence were assessed for overactive bladder and, if eligible, were enrolled in the study. Biomarkers were evaluated before in both groups and after antimuscarinic treatment in the overactive bladder group.
Results:
Overall, 80 participates were enrolled (overactive bladder, n = 40; control, n = 40). The mean age was not different between the two groups (overactive bladder, 48.6 years; control, 46.9 years [P = 1.01]) and females were more in both the groups (60% and 55%, respectively). The majority (70%) of patients presented with frequency and urgency. After 3 months of treatment with antimuscarinics, the mean overactive bladder symptom score decreased significantly from 8.4 to 4.85 (P < .001). The mean urinary and serum nerve growth factor levels and serum C-reactive protein levels also decreased significantly from baseline after 3 months of treatment with antimuscarinics (24.78 pg/mL, 22.46 pg/mL, 0.89 mg/L, respectively; P < .0001).
Conclusions:
Measurement of urinary and serum nerve growth factor levels has diagnostic and therapeutic potential in patients with overactive bladder.
Introduction
Overactive bladder (OAB) is a common and chronic disorder with an increasing overall prevalence with age, around 10% in adults and may exceed >40% in elderly population. 1 It has a considerable impact on the quality of life and is a significant burden on health care system. International Continence Society (ICS) has defined OAB as the presence of urinary urgency, with or without urge incontinence, and usually with an increased daytime frequency and nocturia in the absence of proven infection or other obvious pathology. 2 According to NOBEL Study, the overall prevalence of OAB among community-dwelling individuals was 16.0% and 16.9% in men and women, respectively. 3 In India, the prevalence of OAB in men is reported around 13.6%. 4
The diagnosis of OAB is based on clinical history, physical examination, urinalysis, uroflowmetry, and urodynamic study. Although evaluation of detrusor overactivity (DO), occurrence of involuntary detrusor contractions of variable amplitude and duration during urodynamic testing, could be helpful during the diagnosis of OAB, DO can only be evaluated by urodynamic assessment, an invasive and expensive test. In a study, it was demonstrated that DO was present in 45% and 60% of dry-OAB and wet-OAB patients. 5
Currently, several biomarkers are being evaluated for diagnosis of OAB including urinary and serum nerve growth factor (NGF), prostaglandin E2 (PGE2), and urinary and serum C-reactive protein (CRP). Nerve growth factor is a small secretory protein produced by detrusor muscle cells and urothelium, and it is found to be present in OAB patients. Urinary NGF has been shown to be elevated in OAB patients, and it is considered as a potential biomarker.6–9 Prostaglandin E2 produced in the bladder by detrusor muscle stretch, stimulation of nerves, and following mucosal injury, is also considered as an OAB biomarker. 10 Serum CRP were also found to be higher in patients with OAB. 11 However, there is limited evidence to consider these biomarkers as reliable tests to diagnose OAB.
The first-line pharmacological treatment of OAB includes antimuscarinic drugs.12,13 These drugs offer significant improvement in OAB symptoms and improve quality of life. In the present study, we compared urinary and serum NGF levels and serum CRP levels in patients with OAB, before and after treatment with antimuscarinic agents, to evaluate their diagnostic and therapeutic importance. In addition, we explored possible correlation between these markers and imaging and urodynamic findings.
Methods
Study population
Study population included patients aged more than 15 years of either sex with a confirmed diagnosis of OAB and DO. Patients with history of malignancy, pelvic radiotherapy, neurologic disease, and any systemic or inflammatory conditions were excluded from the study. Patients with bladder outlet obstruction, renal impairment, post-void residual urine (PVRU) >50 mL, or taking any medication within 14 days (before sample collection) that could affect bladder function were also excluded. In comparator group, equal number of healthy controls (asymptomatic without any OAB symptoms and free of lower urinary tract symptoms [LUTS]) were enrolled.
Study design
This was a prospective case-control study conducted between February 2014 and December 2014 at the Institute of Medical Sciences, Banaras Hindu University, Varanasi, Uttar Pradesh, India. All patients visiting urology out-patient department with urgency, frequency, and urge incontinence were assessed for OAB and, if eligible, were enrolled in the study after obtaining a written informed consent. Patients underwent thorough history taking and clinical examination, routine blood investigations, urine analysis and culture, uroflowmetry, and urodynamic study. The diagnosis of OAB was also confirmed using OAB symptom score (OABSS) 14 and the modified Indevus Urgency Severity Score (IUSS) 15 questionnaires. Patients were also asked to record a 3-day voiding diary to verify urgency, urgency urinary incontinence, and functional bladder capacity. Patients were subcategorized as OAB wet (at least one urgency urinary incontinence per 3 days) and OAB dry (urgency at least once per day without urgency urinary incontinence). Each enrolled study patient was treated with antimuscarinic agents as their routine treatment (and not as study treatment) for at least for 3 months and followed up at 3 months.
The study protocol was reviewed and approved by the institutional review board. The study was conducted in accordance with the International Conference on Harmonization Good Clinical Practice guidelines, the approved protocol and the ethical principles that have their origin in the Declaration of Helsinki. The written informed consent (assent where applicable) was obtained from each study participant before participation in the study.
Evaluation
Before initiation of antimuscarinic agents, all patients were evaluated by transabdominal ultrasound for PVRU and detrusor wall thickness (DWT), serum and urinary NGF, and serum CRP. All participants from control group were evaluated for DWT, serum and urinary NGF, and serum CRP. After 3 months of treatment with antimuscarinic agents, patients were again followed up and evaluated for transabdominal ultrasound for PVRU and DWT, serum and urinary NGF, and serum CRP. Patients were considered as responders or non-responders based on OAB questionnaire responses.
Sample collection and biochemical analysis
Blood samples (5 mL) were collected from each study participant in plain vial and after 1 hour, centrifuged at 3000 r/min for 10 minutes and serum was collected in the Eppendorf tube and preserved at −20°C. Urine samples were collected when participants felt full bladder sensation, centrifuged at 5000 r/min for 10 minutes, and the supernatant was separated in aliquot tubes and preserved at −20°C. Both serum and urinary NGF levels were measured by enzyme-linked immunosorbent assay (ELISA; using beta NGF human simple step ELISATM Kit). The serum CRP was measured using Cobas Integra 400 autoanalyzer.
Statistical analyses
All statistical analyses were performed using SPSS (version 11.0). The Student t-test was used to test the statistical significance between compared groups. The Pearson correlation coefficient was used to measure the strength of linear regression between 2 variables. Statistical significance was considered if P-value was less than .05. Demographics and baseline characteristics were presented using descriptive statistics.
Results
Overall, 80 participates were enrolled (OAB group, n = 40; control group, n = 40) in the study and all patients completed the study. The mean age was not different between the 2 groups (OAB group, 48.6 years; control group, 46.9 years [P = 1.01]). Overall, females were more in both the groups (Table 1). The majority (70%) of patients presented with frequency and urgency, followed by frequency, urgency, and urge incontinence (22.5%). Incontinence was reported by a total of 11 (27.5%) patients (female, n = 8; male, n = 3). In total, 8 patients had dry OAB and 3 patients had wet OAB. Urodynamic evaluation showed that DO was present in 82.5% of patients (Table 1).
Demographics and baseline characteristics.
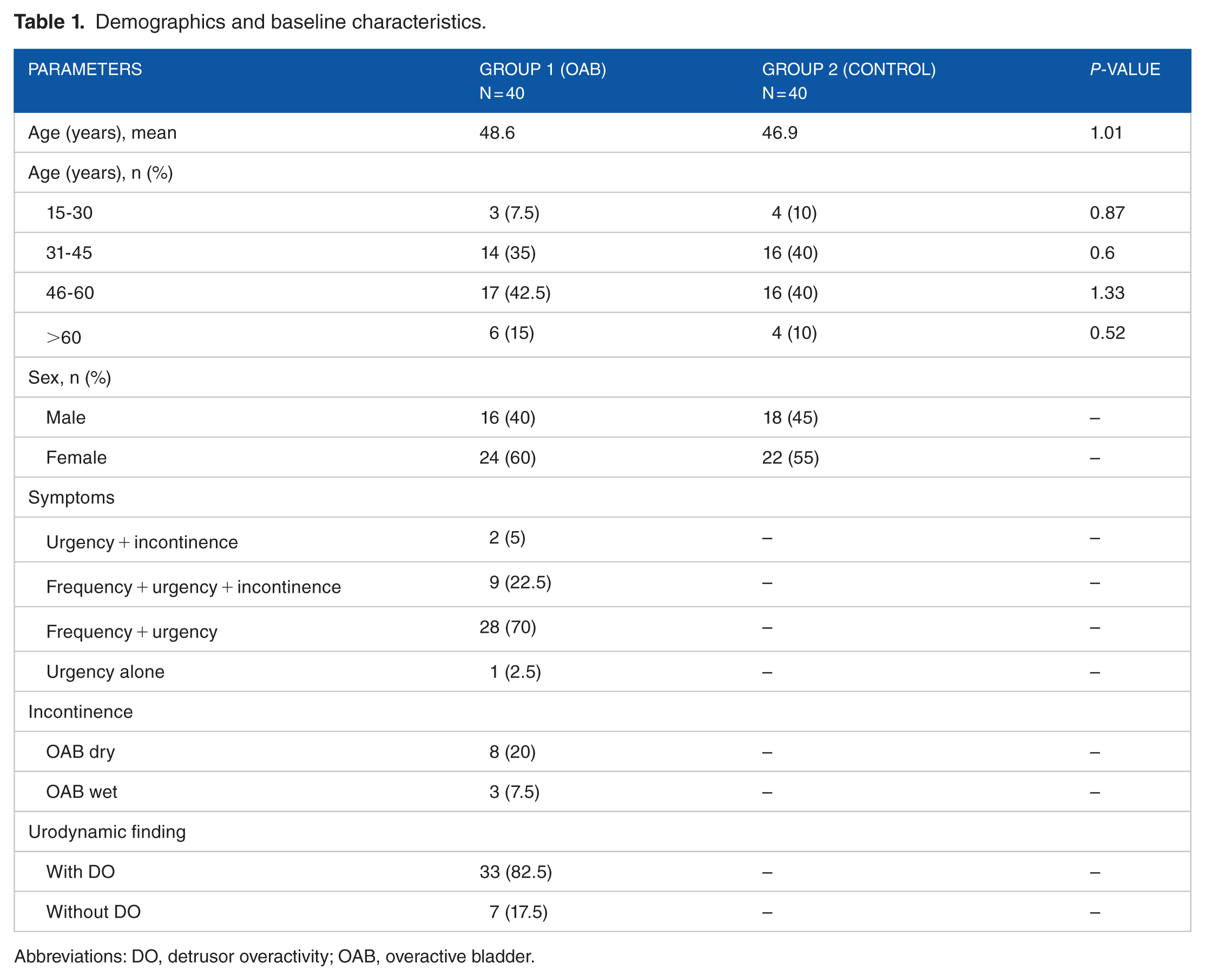
Abbreviations: DO, detrusor overactivity; OAB, overactive bladder.
The mean OABSS in patients with OAB at baseline was 8.4. The mean DO was 5.2 mm in patients with OAB, almost similar to controls (4.7 mm; P = .65). At baseline, the mean urinary NGF, serum NGF, and serum CRP levels in patients with OAB were 37.8 pg/mL, 34.58 pg/mL, and 1.36 mg/L respectively, significantly higher than control group (4.34 pg/mL, 4.82 pg/mL, 0.50 mg/L, respectively; P < .0001) (Table 2).
Summary of study parameters (pre- and post-treatment).
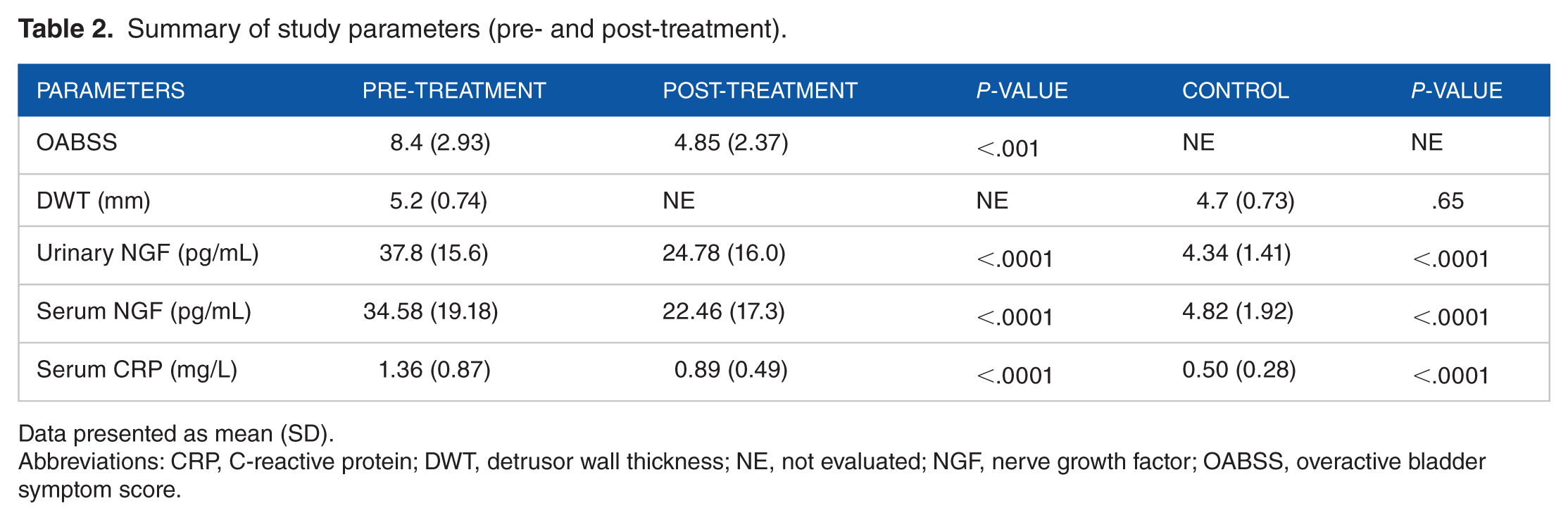
Data presented as mean (SD).
Abbreviations: CRP, C-reactive protein; DWT, detrusor wall thickness; NE, not evaluated; NGF, nerve growth factor; OABSS, overactive bladder symptom score.
After 3 months of treatment with antimuscarinics, the mean OABSS decreased significantly from 8.4 to 4.85 (P < .001). The mean urinary NGF, serum NGF, and serum CRP levels also decreased significantly from baseline after 3 months of treatment with antimuscarinics (24.78 pg/mL, 22.46 pg/mL, and 0.89 mg/L, respectively; P < .0001) (Table 2). When OABSS and biomarkers were compared based on patients who responded or did not respond to treatment, the difference between pre- and post-treatment was significant (P < .05) for OABSS and all the 3 biomarkers (Table 3). Among patients who responded to treatment, the urinary NGF decreased from 27.5 to 14.4 pg/mL, serum NGF decreased from 22.97 to 11.8 pg/mL, and the serum CRP decreased from 1.21 to 0.78 mg/L (Table 3). The relationship between OABSS and NGF (both urinary and serum) showed positive correlation (Figure 1A and B). Similarly, a positive correlation was observed between urinary and serum NGF (Figure 1C).
Pre- and post-treatment comparison of biomarkers in OAB patients.
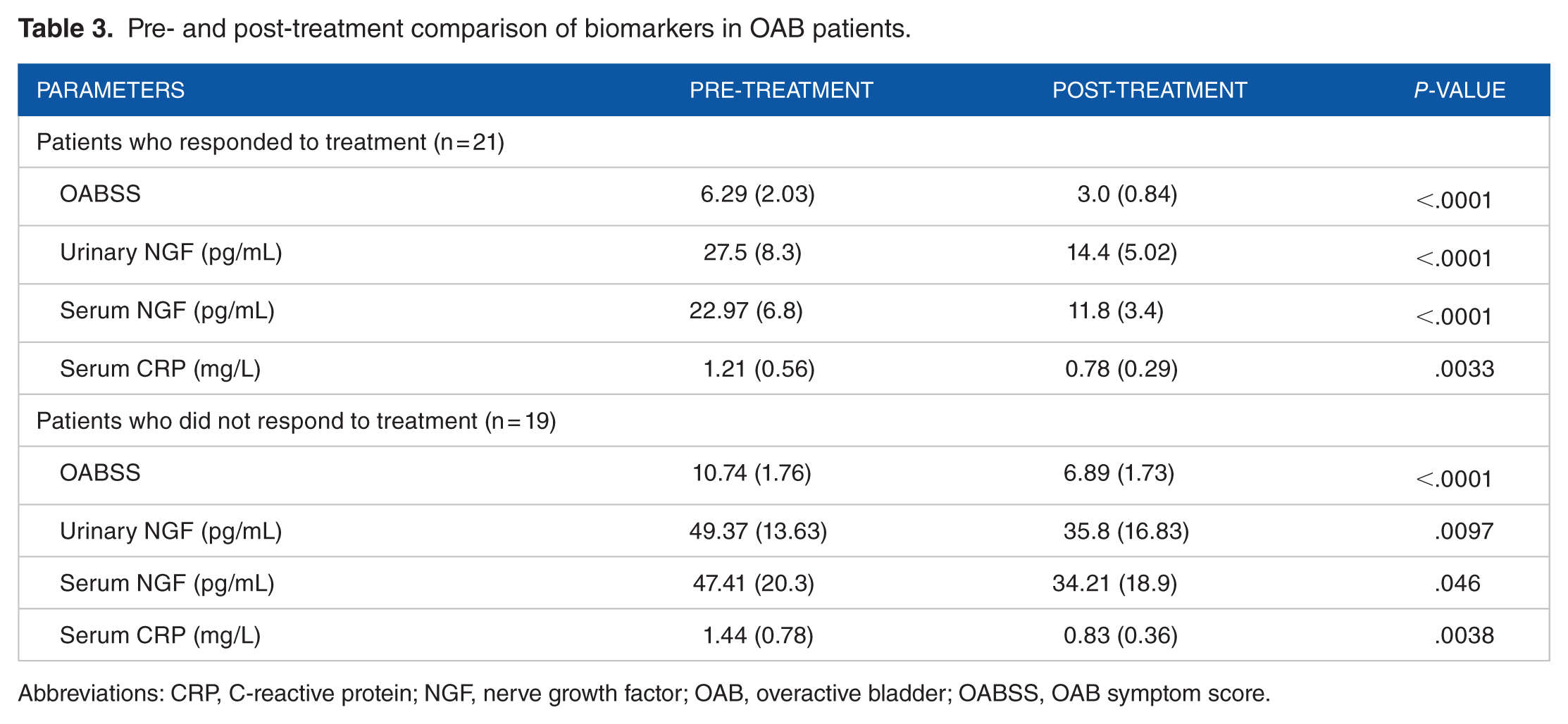
Abbreviations: CRP, C-reactive protein; NGF, nerve growth factor; OAB, overactive bladder; OABSS, OAB symptom score.
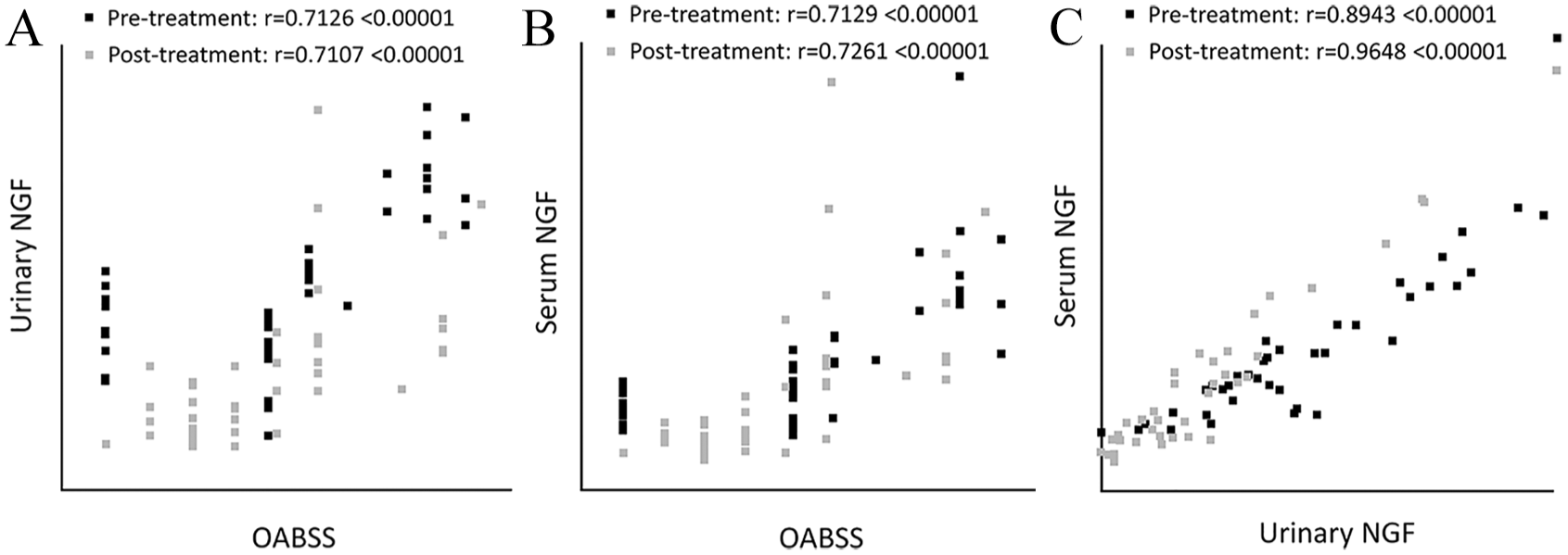
(A, B) Correlation between OABSS and NGF levels and (C) between urinary and serum NGF levels. The r-value was derived using the Pearson correlation coefficient test. NGF indicates nerve growth factor; OABSS, overactive bladder symptom score.
Discussion
In this prospective case-control study, we aimed to compare the urinary NGF, serum NGF, and serum CRP levels before and after antimuscarinic treatment in patients with OAB to evaluate their diagnostic and therapeutic utility. Results from this study showed that urinary and serum NGF levels have diagnostic potential and could also be used in monitoring treatment effect in patients with OAB.
In real-world clinical practice, the diagnosis of OAB is primarily based on patient-reported symptoms and sometimes on objective assessments. The choice of treatment is mainly limited to pharmacotherapy which may reduce micturition frequency and increase blabber capacity; however, there could be limited benefit to health-related quality of life. Multi-item questionnaires have been introduced to evaluate the OAB and its treatment outcomes. The OABSS is one of the few questionnaires that actually measures the severity of symptoms from the patient’s perspective without placing the obligation on the patient to complete the voiding diary. 14 It is also a useful tool to evaluate the OAB symptom severity after medical treatment. The OABSS is also easy for physicians to interpret the patient’s situation and for patients to understand the questions. Hence, in the present study, OABSS was used as one of the major tools for the diagnosis and treatment of OAB patients.
In the present study, DWT was similar in both control and OAB groups. These results were similar to a previous study by Chung et al, who found no difference in DWT between OAB patients and individuals with no OAB. 11 These results demonstrate that DWT may not be useful for the assessment OAB. However, in a previous study, a trend of increasing DWT was associated with the severity of urgency. 16 In another study, authors evaluated the total DWT in female patients with idiopathic DO and found that around 59% of patients had a mean DWT greater than 5 mm, of which the majority (94%) had DO. They concluded that, with a cut-off of 5 mm, the measurement of DWT by transvaginal ultrasound could be a potential screening method for OAB. 17 Few studies have reported reduction in DWT after antimuscarinic treatment.18,19 In other studies, a positive correlation had been found between presence of OAB and high DWT.20,21
In the present study, urinary and serum NGF levels were significantly higher in OAB group compared with control group. Likewise, urinary and serum NGF levels were significantly reduced after 3 months of antimuscarinic treatment. The urinary NGF levels also correlated with OABSS, and patients with higher OABSS had higher NGF levels. Since the NGF levels differ significantly among normal controls and OAB patients, urinary and serum NGF levels could be used as a biomarker for the diagnosis of OAB. These results are consistent with previous studies, where increase in urinary NGF7,8,18,22–26 and serum NGF levels 27 was seen in OAB patients. A recent meta-analysis, which included 17 studies, concluded that urine NGF/Cr (NGF normalized to urine creatinine) could be a useful biomarker for OAB in future. 28 However, the authors specify that NGF/Cr could not be used at present due to lack of specificity; but consider that the specificity in the diagnosis of OAB may improve when NGF is combined with other biological indicators. In addition, similarity in the urinary and serum NGF levels in the present study is not in consonance with many of the previously published reports. However, the serum and urinary NGF levels in the body are believed to be derived from multiple sources and the normal ranges of these have not been defined. In the present study, we observed a positive correlation between pre- and post-treatment urinary and serum NGF levels, which means there was a tendency for high serum NGF levels with high urinary NGF levels and vice versa. This correlation demonstrates that either urinary or serum NGF levels may be used for the assessment of OAB.
The CRP is a well-known biomarker of acute and chronic inflammation. However, it has also been demonstrated that patients with OAB may have higher levels of CRP, particularly in wet OAB. 11 The results from our study were consistent (0.50 mg/L in control group and 1.36 mg/L in OAB group) with the previous study. 11 However, after antimuscarinic treatment, a significant reduction in serum CRP levels was seen even in patients who did not respond to treatment. These results demonstrate that serum CRP may have a diagnostic potential, but however, it has limited therapeutic potential.
Authors acknowledge following limitations of the study. The major limitations of the study include relatively small sample size and study conducted at single center, which could limit the generalizability of our results to wider population. Multicenter studies with larger sample size are warranted to provide more meaningful conclusion on the use of these biomarkers to aid diagnosis and treatment. In addition, we did not evaluate patients with urodynamic study to compare post-treatment outcomes, which could have provided more information regarding refractory nature of disease and its correlation with NGF levels.
In conclusion, measurement of urinary and serum NGF levels has diagnostic and therapeutic potential in patients with OAB. Multicenter studies with larger sample size are warranted to confirm these results.
Footnotes
Acknowledgements
The authors acknowledge Dr Kiran Lavand for his assistance in writing this manuscript.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ST conceived and designed the study, wrote the manuscript, overall supervision and formatting of the study; PP collected the data – selection of cases and controls and follow up record keeping and contributed data analysis tools; YR collected the data – selection of cases and controls and follow up record keeping; FAK collected the data – selection of cases and controls and follow up record keeping; RS performed the analysis - sample collection, storage and biochemical analysis of serum and urinary samples for NGF and CRP; and USD supervised the study.
