Abstract
Cadmium is a non-essential metal which because of its extensive use in industry and agriculture presents a worldwide health hazard. Long-term exposure to cadmium results in nephropathy characterised by interstitial nephritis affecting the renal tubules. In addition to occupational exposure, cadmium can contaminate food and water increasing the number of people at risk of developing chronic renal disease. It is now known that smoking is an independent risk factor for renal disease. The current knowledge of the molecular mechanisms initiating and progressing cadmium nephrotoxicity is now partly understood and research on the molecular effects of nicotine on the kidney is ongoing. The exacerbation of renal disease by smoking is an increasing problem. It is concluded that where there is potential exposure to cadmium, either occupationally or environmentally, individuals should be encouraged to cease smoking.
Introduction
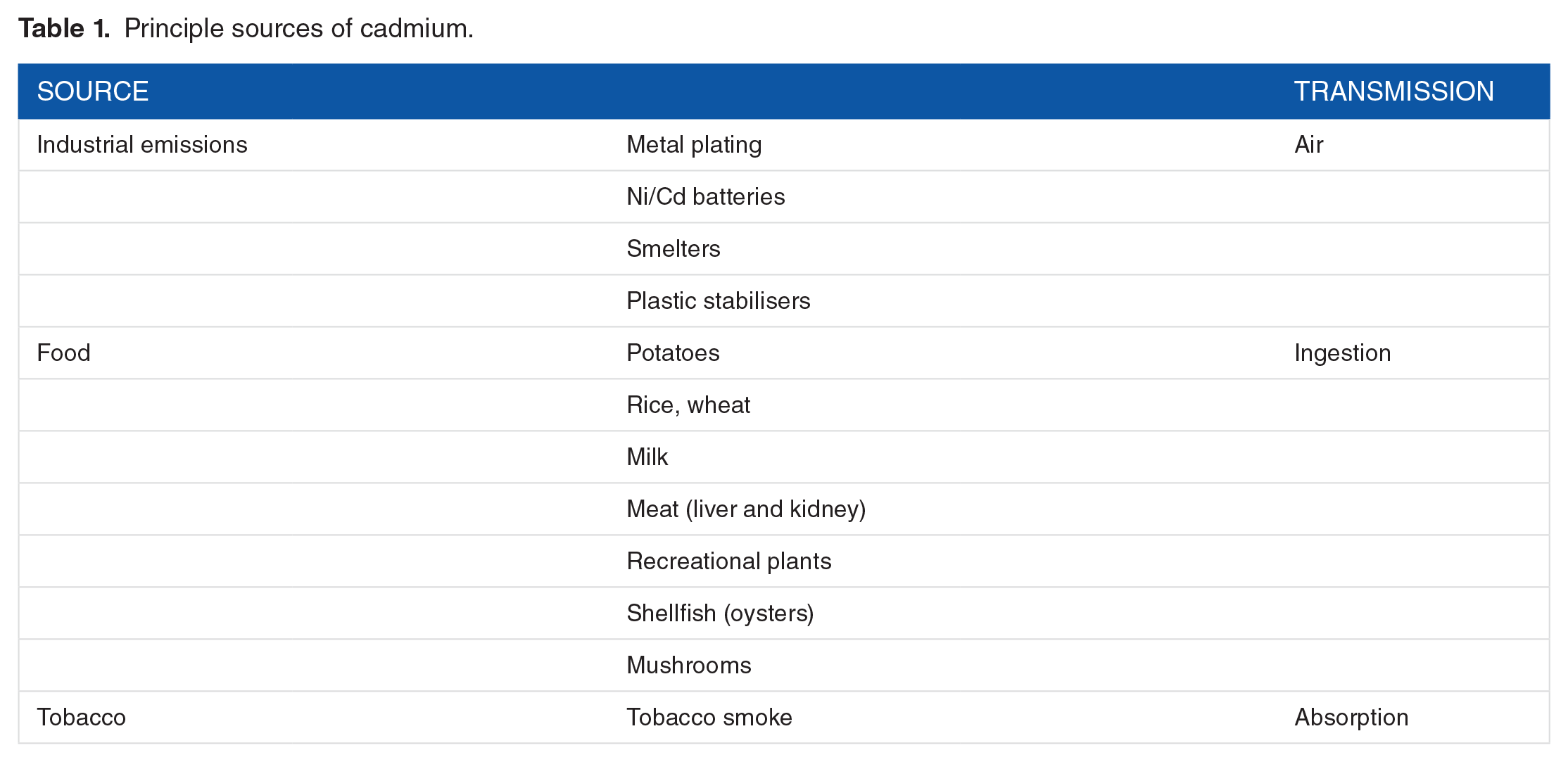
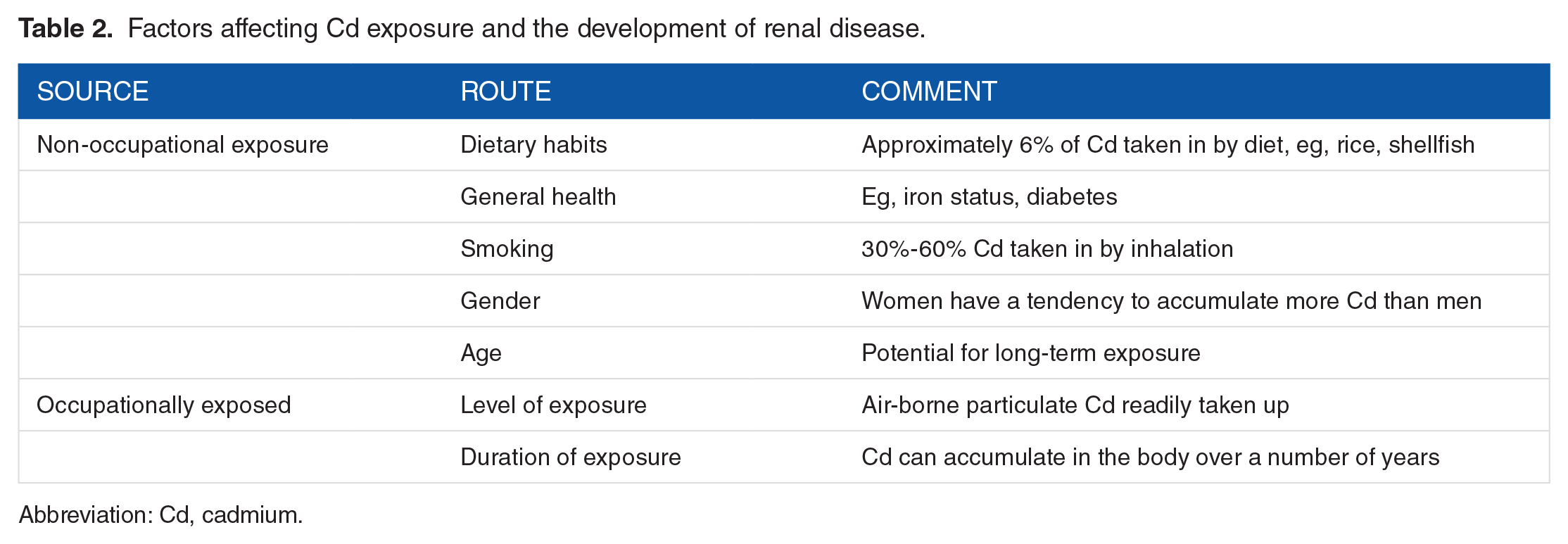
Cadmium (Cd) is found naturally in ores together with other heavy metals, it has been used extensively in alloys, some plastics, and nickel-Cd batteries. Cadmium poses a great health risk even at low quantities. The toxic effect of Cd on the kidney and bone was first reported in the 1950s in Sweden 1 and Japan. 2 Chronic low levels of Cd exposure have also been linked to cardiovascular disease and cancer. 3 The body has limited capacity to respond to Cd, as it is unable to undergo metabolic degradation to a less toxic form and it is poorly excreted. 4 Cadmium enters the body either by ingestion in foods that contain high levels of Cd, such as shellfish, liver and kidney meat, some grain cereal products, potatoes, and some leafy vegetables, or via inhalation (Table 1). Only about 2% to 6% of Cd ingested is taken up into the body and the amount depends on a number of factors including the form of Cd in food. In contrast, 30% to 60% of inhaled Cd is taken up. 5 Cadmium causes heavy metal nephropathy which is characterised by interstitial nephritis and low-molecular-weight proteinuria. It can be distinguished from other types of nephropathies, eg, diabetic and IgA nephropathies by the mechanism of cellular damage involved and the excretion of urinary protein. Cadmium-induced nephrotoxicity is characterised by renal glycosuria, aminoaciduria, hypophosphaturia, hypercalciuria, polyuria, and decreased ability to buffer acid load. 6 Jarup 6 concluded that the correlation of low levels of exposure to Cd with albuminuria partly explained the observed increase in cases of chronic renal disease (CRD). The multiple toxic effects of Cd at the cellular and organ levels have been recently reviewed.7,8 Its toxic impact appears to be proportional to the body burden of Cd; detoxification with EDTA and other chelating agents is possible and has been shown to be therapeutically beneficial in humans and animals. 9 Environmental contamination arises from industrial emissions, soil, and contaminated food as well as smoking10,11 (Table 2). Urine Cd levels are higher in people living in contaminated areas, and smokers have about twice the concentration in their urine as non-smokers. 12 The use of Cd in Europe has decreased due mainly to the gradual phasing out of Ni-Cd batteries; however, Cd consumption and production have increased worldwide. 12 Cadmium retention is generally higher in women than men, and the severe Cd–induced itai-itai disease is mainly a women’s disease. 13 Sex differences are more pronounced at higher levels of exposure and there is evidence that Cd has an oestrogenic effect. 14 Julin et al 15 studied the dietary intake of premenopausal non-smoking women and concluded that dietary Cd intake can be used as a predictor of short-term and long-term exposure to Cd. Risk assessment of the effect of Cd in a population should include older women as they generally have higher blood Cd (B-Cd) and kidney Cd concentrations than men as a result of their higher gastrointestinal absorption.13,15 Julin et al 15 concluded that estimates of dietary Cd predict biomarkers in both short-term and long-term Cd exposure; this prediction was improved when the iron status of the women was taken into account. Major determinants of intestinal Cd uptake are dietary composition in particular crude fibres and trace elements, especially iron. 16
Principle sources of cadmium.
Factors affecting Cd exposure and the development of renal disease.
Abbreviation: Cd, cadmium.
Cadmium can be released into the atmosphere by smelting or other processes and travel long distances. Once deposited onto the ground it is taken up into the food chain by cereals and root crops. Cadmium is taken up more rapidly by plants than other metals such as lead and mercury. Cadmium can also enter the food chain via water (for further details, see Agency for Toxic Substances and Disease Registry [ATSDR] publications).
The kidney is the principal organ targeted by chronic exposure to Cd. 17 Cadmium nephrotoxicity may result from inhalation or ingestion. Human studies suggest a latency period of about 10 years before the clinical onset of clinical damage. 17 The nephrotoxic effect of Cd and the resulting renal disease has severe human and economic consequences. 18 Although Cd nephropathy has been observed in workers exposed to high levels of Cd, it is now understood that people living in polluted industrial areas who are exposed to relatively low levels of Cd are also at risk of developing renal dysfunction. 10 An excellent review describes the Cd-induced renal effects and the results of environmental exposure in Sweden, Japan, and Belgium. 19 Ferraro et al 20 confirmed that there is a risk of CRD as a result of low-level exposure to Cd by analysing a large population in the US National Health and Nutritional Examination Survey (NHANES) (1999-2006) study. Chaumont et al 21 studied the lifetime trend of U-Cd in 6 cohorts of the general public in Belgium. They found that at low Cd exposure levels, the relationship between U-Cd and kidney Cd levels seen in industrial workers does not hold as it is influenced by recent Cd intake and renal handling of proteins, particularly low-molecular-weight proteins. Tobacco is a major source of Cd, and in 2015, more than 1.1 billion people smoked tobacco with more men than women smoking. Although the number of people smoking is generally falling, tobacco smoking is increasing in the Eastern Mediterranean. 22 Smoking was initially linked to kidney disease when it was demonstrated that heavy smokers had proteinuria more frequently than light smokers. This finding was consolidated by the data from the Multiple Risk Factor Intervention Trial (1982) which indicated that smoking is one of the principle risk factors that can lead to CRD. 23 Smoking is also the most important remedial cardiovascular risk factor, as well as being an independent risk factor for the progression of renal disease. The amount of Cd taken up by the roots of Nicotiana (tobacco) plants depends on the species and the availability of Cd in the soil. 24 As a result, the level of Cd varies in different brands of cigarettes from 1.30 to 2.78 μg g−1,1,25 whereas cigars contain less Cd (0.75-1.69 μg g−1). 26 The nephrotoxic components of cigarette smoke are Cd and nicotine. It has been estimated that for a smoker, 50% of total Cd intake comes from smoking. 27 It has been calculated that a cigarette contains 1 to 2 µg of Cd, of which about 10% is inhaled when smoking. 28 Cigarette smoking is therefore a major source of Cd exposure and can significantly increase B-Cd levels. 29 The concentration B-Cd in smokers is on average 4 to 5 times higher than those of non-smokers, and smoking is therefore the principle non-occupational source of Cd.
Biomarkers of Cd Nephropathy
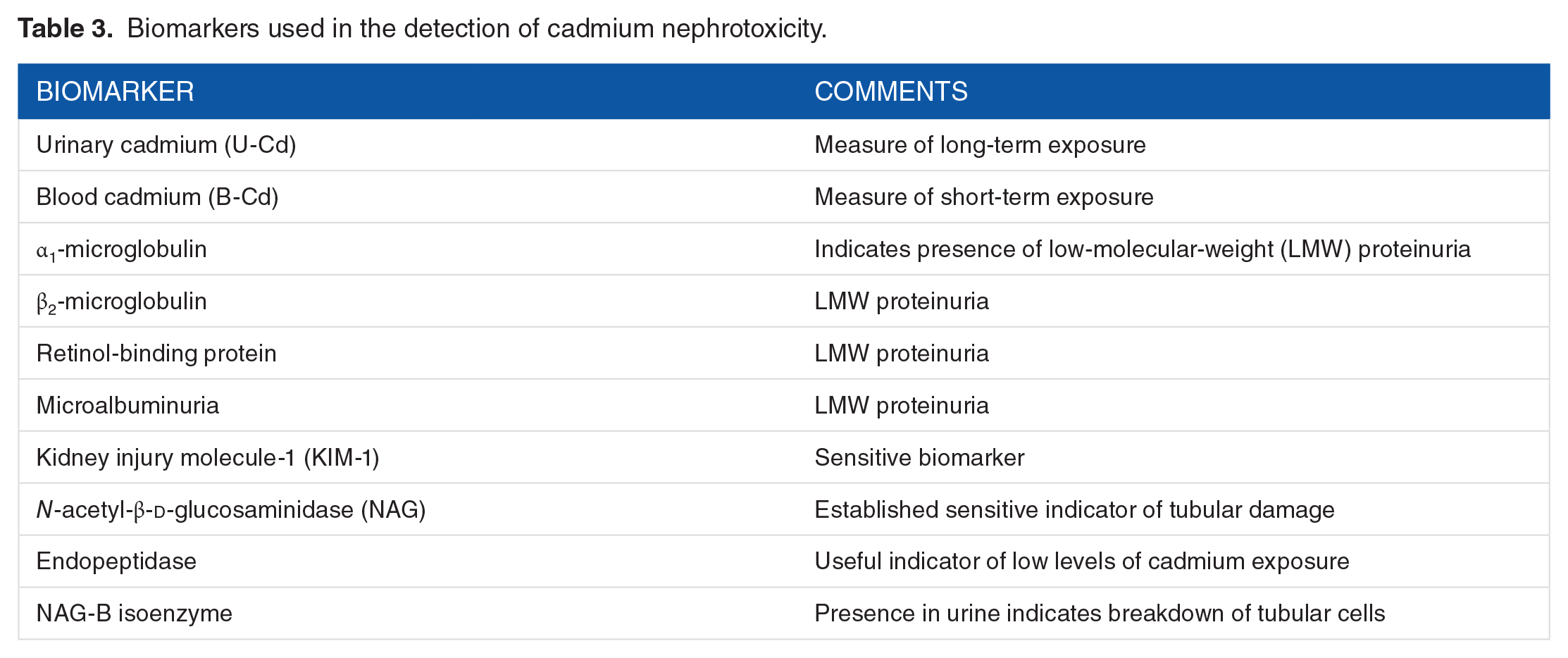
It is now possible to draw a more accurate picture of early kidney damage by nephrotoxic substances such as metals. The body burden can be measured and related to various levels of different biomarkers (Table 3). It is advisable to use small combinations of tests or panel of biomarkers that are characteristic of certain patterns of damage. The advantage of using sensitive biomarkers of renal damage is that susceptible people can be identified early when damage to their kidneys can be halted or delayed. 18 Roels et al 30 demonstrated clear differences in urinary biomarker excretion between a control group and groups exposed to low levels and high levels of Cd; they concluded that when screening for the effects of Cd proximal tubular markers, eg, α1-microglobulin and urinary enzymes should be measured in addition to glomerular markers. A threshold of 2 µg/g creatinine is mainly associated with biochemical alterations, whereas at 4 µg/g creatinine, the glomerular barrier progressively leaks and cytotoxic effects occur in the proximal tubule, and a third threshold of 10 µg/g creatinine corresponds to the onset of proximal tubular dysfunction demonstrated by an increase in the excretion of low-molecular-weight proteins.
Biomarkers used in the detection of cadmium nephrotoxicity.
N-acetyl-β-
Cd Nephrotoxicity
Cadmium can be absorbed either by the lungs or gastrointestinal tract (Figure 1) and it then binds to serum albumin in the blood. On entering the liver, it forms a complex with MT-1.
36
The Cd-metalloprotein-1 gene (MT) complex is absorbed into the renal tubule where it is stored in the lysosomes. Because Cd is a non-essential toxic heavy metal, there are no specific Cd transporter systems in the kidney.
37
However, there is considerable evidence that it can compete with other metals for transport-mediated entry into the proximal tubule. Most prominent of these are zinc and calcium transporters. Evidence is growing that rather than the accumulation of Cd in the proximal tubular cells causing non-specific toxic damage, the early stages involve more specific reactions.
38
Cadmium may interfere with cell signalling via the ROS, ERK, and AKT signalling pathways and it also interacts with receptors, second messengers, and transcription factors.
39
Cell-cell adhesion, modification of the cell signalling pathways, and autophagic responses occur well before necrosis or apoptosis.
38
These cellular changes lead ultimately to apoptosis and cell death.
40
It is known that that Cd toxicity of the kidney varies with different individuals exposed to the same levels Cd. One explanation comes from a cross-sectional study of people with a history of Cd exposure through contaminated rice in South-Eastern China.
41
Polymorphism of the MT genes, MT1A and MT2A, influenced Cd-related kidney damage.
41
Increases in B-Cd and U-Cd as well as increases in urine N-acetyl-β-
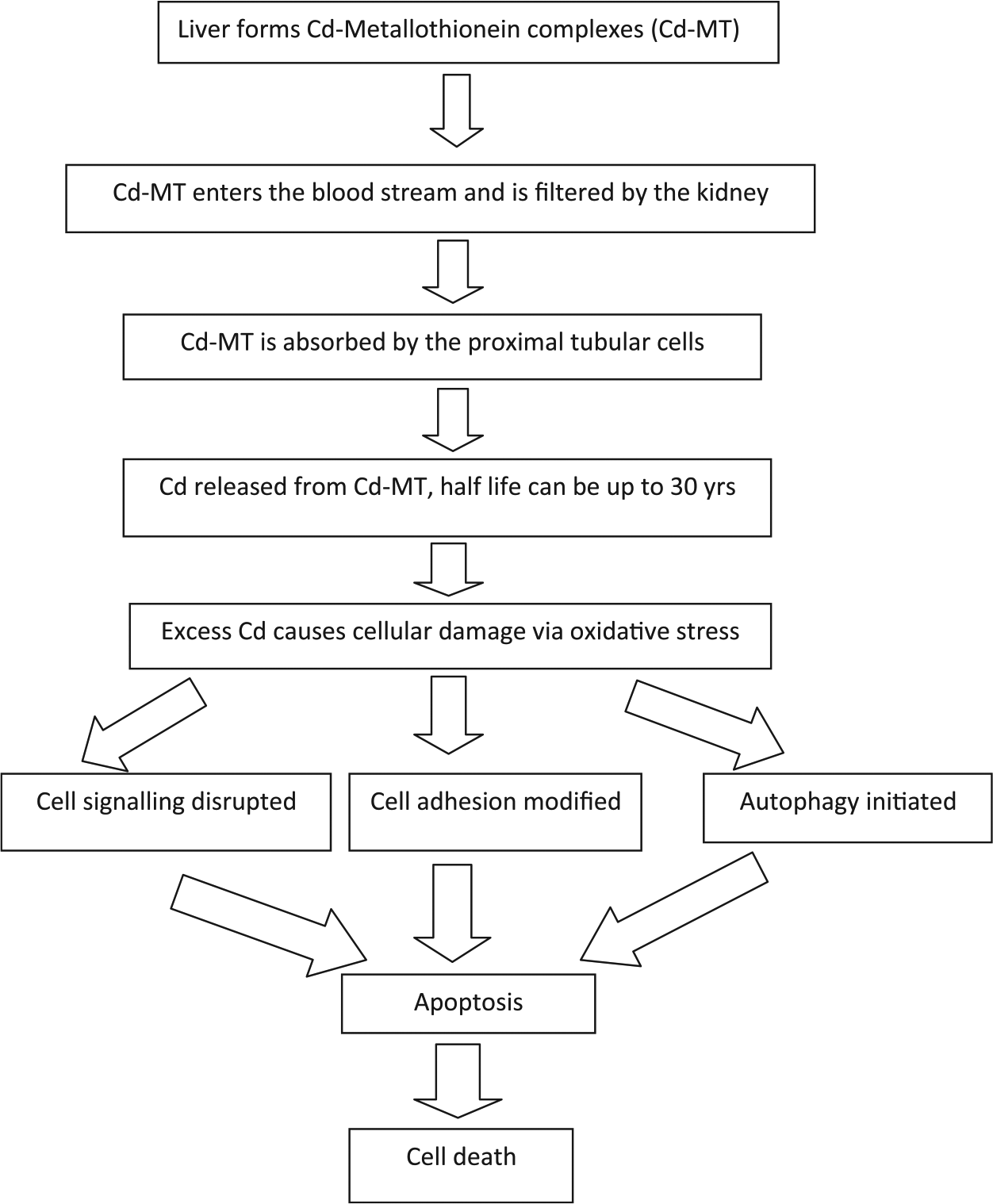
Suggested mechanisms of renal toxicity of cadmium (Cd).
Even low-level chronic Cd exposure from food intake and smoking can produce adverse health effects. 42 Early reports from Japan demonstrated that long-term exposure to Cd-contaminated rice can cause itai-itai (ouch-ouch) disease which is characterised by osteomalacia and osteoporosis. 2 Satarug and Moore 43 had provided evidence linking Cd-related kidney dysfunction and decreases in bone mineral metabolism in non-occupationally exposed populations showing no signs of nutritional deficiency. This finding challenges the previous view that kidney and bone damage in itai-itai disease was the result of Cd toxicity in combinations with nutritional deficiency. Satarug and Moore 43 present evidence that the then current provisional tolerable weekly intake for Cd (provisional tolerable weekly intake) should be lower than 1 µg/kg body weight. Cadmium-linked toxicities are found more frequently in women than in men, and reduction in exposure is pivotal because at present there are no therapeutically effective chelating agents to reduce the body burden. Nishjo et al 44 established a benchmark dose (BMD) for renal effects for health risk assessments of residents living in Cd-polluted areas in Thailand. They measured B-Cd and Cd-U, β2-microglobulin, retinol-binding protein, and NAG as renal tubular markers in control and exposed groups consisting of women and men more than 40 years of age. They concluded that the BMD limits of urinary Cd and B-Cd for renal effects were 4.4 to 8.1 µg/g creatinine and 5.0 to 6.2 µg/L, respectively. On the basis of these values, they calculated that more than 40% of the residents were at high risk of renal effects due to Cd exposure. A long-term study conducted in the polluted Jinzu River basin in Japan concluded that inhabitants with renal effects caused by Cd exposure had poor life expectancy for both sexes and it may also increase mortality in women from cancer, ischaemic heart disease, and renal disease. 45 However, Ikeda et al 46 found no increase in urinary biomarkers of tubular function in a population with up to 3 µg Cd/g creatinine in their urine. Investigations 47 into a population living near a zinc smelter in Avonmouth (UK) demonstrated that concentrations were close to the levels where kidney and bone effects have been found. The presence of early renal changes was confirmed by the dose-response relationship between U-Cd and NAG-U, and the authors concluded that there was a need for measures to reduce the concentration of environmental exposure to Cd. 47 Occupational exposure to Cd has been well-documented in the manufacture of nickel-Cd batteries, metal plating, zinc and lead refining, and smelting of lead and Cd. Cadmium exposure can also occur in more specialist occupations, and, eg, a 28-year-old female non-smoker was found to have a significant body burden of Cd after working with jewellery manufacturing for 3 years, and as she had B-Cd level >10 µg/L, she therefore met the US Occupational Safety and Health Administration (OSHA) screening guidelines for mandatory removal. 48
The first signs of renal abnormalities occur when the U-Cd level reaches 2 to 4 nmol/mmol creatinine. Tubular dysfunction manifests itself as low-molecular-weight proteinuria including β2-microglobulin, α1-microglobulin, retinol-binding protein, and NAG.10,49 Initial tubular damage may progress to more severe damage including decreased glomerular filtration rate (GFR). 10 An association between low levels of Cd exposure and CRD requiring renal replacement was demonstrated in both environmentally and occupationally exposed Swedish populations. 50 These authors concluded that exposure to occupational or relatively low levels of Cd is a determinant for end-stage renal disease. A later study of 1797 adults participating in a cross-sectional survey in Korea (KNHANES [Korean National Health and Nutrition Examination Survey]) 51 confirmed that low-level Cd exposure was associated with CRD especially in adults with hypertension or diabetes. Long-term exposure to Cd can also cause skeletal damage. 2 Bioaccessibility of Cd has not been well studied. Ju et al 52 compared the health risk with Cd through inhalation and seafood consumption and found that fish had higher bioaccessibility than that of shellfish. A cross-sectional study 53 assessed whether exposure to Cd is associated with renal dysfunction in 1699 Belgium subjects. Five urine biomarkers were found to be significantly related to Cd excretion which exceeded 2 to 4 μg/24 hours. A similar study 46 of residents living in an area contaminated with heavy metals from a zinc smelter demonstrated a similar change in the urinary biomarkers NAG and alanine aminopeptidase (AAP) at low levels of U-Cd again indicating early renal dysfunction. A study of a small community in Pennsylvania who were exposed to heavy metals originating from a zinc smelter 54 found that NAG and AAP activity significantly increased in adults more than 18 years of age when the U-Cd was greater than 1.0 μg/g creatinine. The results from these studies indicate that renal changes occur below the 2.0 μg/g level.
Smoking and Nephrotoxicity
The major source of Cd apart from occupational exposure and diet is smoking. Cigarettes contain nicotine as well as Cd which also has nephrotoxic effects. Nicotine is a psychoactive substance which is responsible for tobacco dependence. Nicotine is metabolised to the inactive metabolite cotinine by the enzyme cytochrome P450 2A6 (CYP2A6). 55 The additive and pharmacologic effects of smoking are primarily mediated by the alkaloid nicotine. 56 Elevated serum and Cd-U in smokers result in glomerular dysfunction, and nephropathies are accelerated by nicotine. 57 Satarug et al 57 concluded that elevated CYP2A6 activity occurs in subjects who show signs of Cd-linked renal toxicity. Nicotine exposure causes a worsening of nephropathy in diabetic mice. 58 Nicotine leads to vascular endothelial cell dysfunction, which is also one of the earliest consequences of hyperglycaemia. Nicotine upregulates the expression of transforming growth factor β (TGF-β) which is involved in the pathogenesis of diabetic nephropathy. 59 Transforming growth factor β promotes extracellular matrix accumulation which results in expansion of the mesangial matrix and thickening of the basement membranes of the microvessels both of which are prominent features of nephropathy.60,61 Cytochrome P450 2A6 coumarin 7-hydroxylase is the principle human nicotine C-oxidase, so the inter-individual and inter-ethnic variation in the CYP2A6 levels can be attributed to polymorphism in the CYP2A6 gene. 62 This would help to partly explain the varying degrees of effect that smoking has on renal function. Where coexisting illness is present, eg, diabetes, polycystic kidney disease, or hypertension, then smoking is a risk factor for the development of CRD. Nicotine activates non-neuronal nicotinic acetylcholine receptors, some of which are located in the kidney. 63 The renal effects of nicotine are linked to the increased generation of reactive oxygen species and activation of pro-fibrotic pathways. 63 Nicotine also increases blood pressure which is accompanied by reductions in GFR. Cessation of smoking may offer cardiovascular as well as renal benefits. 64 Smoking increases the renal risk in both diabetic and non-diabetic renal diseases. 65 Satarug et al 57 investigated the interrelationships between phenotype of hepatic CYP2A6 and the link to nephropathy in 118 non-smoking healthy Thai men and women who were exposed to lead and Cd. Mild nephropathy was detectable at urine concentrations of 1 to 2 µg/g creatinine corresponding to renal Cd concentrations ≤50 µg/g kidney cortex. Cadmium was an inducer of CYP2A6, and CYP2A6 is known to metabolise 90% of nicotine. 62
Smoking is a modifiable renal risk factor and it can influence progression to CRD via a number of different routes. 66 Smoking can result in hypoxia and vascular damage together with matrix changes which result in glomerulosclerosis ending in chronic kidney disease. Data from the Multiple Risk Factor Intervention Trial indicated that in men, smoking increased the risk of progression to end-stage renal failure. 66 This was particularly apparent in older men and may be independent of any underlying renal disease; however, renal graft survival was also reduced. Cessation of smoking reduced the rate of progression. A striking result was the poor survival of patients with diabetes on dialysis who continued to smoke. A study with patients with type 1 diabetes investigated 158 male patients with type 2 diabetes and compared them with a similar number of men who had never smoked. 67 They concluded that the risk of low GFR (<60 mL/min per 1.73 m2) in male patients with type 2 diabetes was enhanced by smoking and mediated by metabolic syndrome. Smokers had a significantly lower probability to attain renal transplantation if they continued to smoke, indicating that transplantation provides a strong incentive to stop smoking. 68 An in-depth review considered whether smoking is an independent progression factor in renal disease and summarised the evidence from a number of large prospective, population-based studies. 69 They considered smoking as a risk factor in patients with primary hypertension or primary renal disease, patients with diabetes, and patients with renal transplants. The evidence that smoking is an important risk factor in kidney disease was overwhelming and more effort should be made to induce patients to stop smoking. In a later review, the same authors investigated the possibility of patients developing renal failure in the future in a large population-based sample. 70 They considered sex-specific effects, dose-response relationships, and whether cessation reduced the risk due to smoking. They confirmed that smoking is indeed a significant risk factor for future renal failure and reported that smoking cessation decreased the risk of renal failure in men, although the data are not clear for women.
About one-third of the world’s tobacco is produced and consumed in China with 350 million smokers and 740 million passive smokers. 71 The death toll which can be directly attributed to tobacco use is expected to rise to 2 million per year. A major 15-year study of Chinese men and women and their smoking habits demonstrated that Chinese men now smoke a third of the world’s cigarettes as a result of a surge in urban and rural usage. In contrast, smoking has declined in Chinese women. Although the overall adult mortality rate is falling in China, the proportion of male deaths attributed to smoking is increasing. 72 Jin et al 73 studied a large Chinese cohort living in Singapore to determine the effect of smoking and found a strong predictive association of smoking with CRD. The risk reduces with non-smokers, but only after a long period of time. They noted that the number of people with CRD in the region was increasing and strongly advocated encouraging people to cease smoking. The effect of age, smoking habits, and occupational exposure to Cd were considered when calculating the BMD for Cd-induced renal tubular disease. 74 These authors studied a group of 490 randomly selected non-smoking women aged 35 to 54 years from non-occupationally exposed areas and measured U-Cd, β2-microglobulin, and NAG. They calculated that the BMD reference point is 1 µg/g creatinine a similar value to that suggested by the European Food Safety Authority confirming the need to reduce Cd exposure to improve human health. 75 In an earlier study, 76 they found that when the Cd level in blood was 0.38 µg/L (median) and urine 0.67 µg/g creatinine, there were significant effects on the renal tubules demonstrated by increases in urinary NAG. These authors commented that subjects with diabetes appeared to be at increased risk, and although the changes at this level of exposure were small, these adverse effects may affect large sections of the population. They emphasised that in areas where exposure to Cd is mainly through the diet, the long half-life of Cd in the soil will be slow which emphasised the need to aggressive reduction in smoking among the population.
A recent large cross-sectional study compared traditional foods and cigarette smoking as a potential source of Cd among aboriginal groups in Quebec, Canada, and the data obtained indicated that traditional foods do not increase Cd exposure, but smoking did. 77 Abd-El-Ghany et al 78 compared Egyptian male smokers and non-smokers and found that increased smoking was related to increased U-Cd level and there was a positive correlation with U-Cd levels and β2-microglobulin activities but a negative correlation with creatinine clearance suggesting early Cd nephropathy. 78 A recent study demonstrated the additive effect of arsenic and Cd exposure on kidney dysfunction. 79 The values of arsenic and Cd levels in a local lake were above the level recommended by World Health Organization and their values were 3 to 4 times higher in the local tobacco than in branded cigarettes. The biomarkers measured, particularly NAG, were found to be elevated in the exposed groups compared with the controls groups confirming renal dysfunction. A similar study in Pakistan demonstrated the additive effect of the combined exposure to aluminium and Cd. 80 They found that exposed patients with renal disorders had higher levels of these elements in their blood than exposed control groups, and lower values were found in the control groups.
Diabetes and adverse effects of smoking
Cigarette smoking in diabetes has repeatedly been confirmed as an independent risk factor for the onset and progression of diabetic nephropathy. 81 Chakkawar 81 has reviewed the probable biochemical changes in the kidney which manifest as the functional and structural features of diabetic nephropathy. In particular, nicotine exposure due to chronic cigarette smoking can cause the acceleration of microvascular complications in diabetes. Smoking mediates high oxidative stress resulting in the generation and low availability of endothelial NOS (nitric oxide synthetase) and may be the initial step in the pathogenesis of glomerulosclerosis in diabetic nephropathy. A total of 3613 patients with type 1 diabetes in the Finnish Diabetic Nephropathy Study were assessed over a 12-year period for the development of CRD. 82 Progression was followed by measuring microalbuminuria and macroalbuminuria and cumulative smoking (packs/year). Their data confirmed that smoking is a risk factor for progression of diabetic nephropathy and is dose related. Surprisingly they found that ex-smokers carry the same risk of progression of diabetic nephropathy. Barregard et al 83 considered whether patients with diabetes are more sensitive to nephrotoxicity from Cd compared with non-diabetics. These authors studied 590 women in their 60s and measured estimated GFR, albumin, and retinol-binding protein in 12-hour urine samples. Blood and urine Cd levels were determined by mass spectrometry. The results of this study provide support for the hypothesis that women with diabetes have higher risk of renal damage from Cd exposure than women without diabetes. The presence of MT antibody (MT-Ab) in the circulation of patients with both type 1 and type 2 diabetes has been reported in a study of 262 Chinese patients with diabetes. 84 This confirmed a previous report 85 that various autoantibodies were present in similar patient groups. Their data proved that the presence of MT-Ab can potentiate tubular dysfunction among patients with diabetes and that patients with high MT-Ab levels are more prone to the development of tubular damage. Interestingly, they were the first to report example of a sex difference in the distribution of MT-Ab and showed that the level of MT-Ab was higher in the female subjects than in the male subjects. Their results were similar to a previous study with occupationally exposed groups, in that there was no significant relationship between the level of this antibody and smoking habits, diabetes management, age, urinary Cd, or B-Cd. 86
Conclusions
There is now overwhelming evidence that smoking is an independent risk factor for renal disease due to the nephrotoxic effects of nicotine and Cd. 87 It also has an additive effect on existing renal disease due to hypertension, diabetes, or occupational or environmental exposure to Cd. Renewed efforts should be made to encourage anyone with existing renal disease or living in an area where there is a risk of Cd exposure to cease smoking. There is mounting evidence that 1 µg/Kg body weight should be adopted as the upper limit of normal exposure to Cd.
Footnotes
Peer review:
Five peer reviewers contributed to the peer review report. Reviewers’ reports totalled 2911 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
RGP wrote and researched this article.
