Abstract
Studies have shown an association between continuous renal replacement therapy (CRRT) and thrombocytopenia. Patients on CRRT usually receive unfractionated heparin (UFH), and heparin-induced thrombocytopenia (HIT) is frequently suspected as a potential cause of thrombocytopenia. In the setting of HIT suspicion, changes in anticoagulation may put patients at risk of developing thromboembolic events or adverse medication effects.
Purpose:
The purpose of this study was to investigate current management of anticoagulation in patients with suspected HIT while on CRRT and identify complications associated with anticoagulation in this setting.
Methods:
This is a retrospective study of patients on CRRT with suspicion for HIT. Prevalence of HIT and anticoagulant use were collected for all patients. Outcomes included thromboembolic and bleeding events, final therapeutic doses of each anticoagulant, and time to reach therapeutic range. Thromboembolic and bleeding outcomes were compared between anticoagulants.
Results:
From January 2013 through December 2015, 74 patients were identified with suspicion of HIT while on CRRT. During the period of HIT suspicion, 20 patients received argatroban, 18 received bivalirudin, 1 received fondaparinux, 4 received UFH infusions, 10 received prophylactic dosing subcutaneous UFH, and 21 received no anticoagulation. The median final therapeutic doses of argatroban and bivalirudin were 1.00 μg/kg/min and 0.075 mg/kg/h, respectively, with no difference in time to therapeutic range (17.6 h vs 18.2 h, P = .485). Compared with bivalirudin, patients treated with argatroban had significantly more therapeutic activated partial thromboplastin time (aPTT) values (54.5% vs 42.8%, P = .008). Four patients (20.0%) treated with argatroban and 5 patients (27.8%) treated with bivalirudin experienced major bleeding (P = .709). Three patients (15.0%) treated with argatroban and 4 patients (22.2%) treated with bivalirudin experienced thromboembolic events (P = .687).
Conclusion:
Argatroban and bivalirudin were associated with similar rates of bleeding and thromboembolic events during the period of concurrent CRRT and thrombocytopenia, although patients treated with argatroban had more therapeutic aPTT values.
Keywords
Introduction
Approximately 35% to 50% of patients in the intensive care unit (ICU) develop thrombocytopenia, which is typically a multifactorial process with contributing factors including sepsis, bleeding, coagulopathies, and pharmacotherapy. 1 For critically ill ICU patients with severe acute kidney injury, it is often necessary to implement renal replacement therapy (RRT), commonly in the form of continuous renal replacement therapy (CRRT), which may further contribute to a decrease in platelet counts. Various studies have revealed an association between the initiation of CRRT and the development of thrombocytopenia; however, the reported incidence in the literature ranges widely from 18% to 70%, with usual declines in platelets of almost 50% from baseline within the first 3 to 5 days.1-3
To complicate the clinical picture further, it is common for patients on CRRT to receive either prophylactic or therapeutic unfractionated heparin (UFH), and therefore the diagnosis of heparin-induced thrombocytopenia (HIT) is often considered as a potential etiology of their thrombocytopenia. 1 While investigating the diagnosis of HIT in these patients, anticoagulation may be held, dose-adjusted, or changed to a nonheparin anticoagulant. Guidelines for treatment of HIT recommend changing heparin to a nonheparin anticoagulant, but there is currently limited evidence for optimally managing anticoagulation in these scenarios. 4 Intravenous direct thrombin inhibitors (DTIs) bivalirudin and argatroban have been studied for the treatment of HIT, but there is minimal literature regarding the use of these agents in patients on CRRT.5-7 CRRT with no anticoagulation has been shown to provide adequate filter life in some patients with high bleeding risk on CRRT, 8 but untreated HIT carries approximately 50% risk of thrombosis. 9
The purpose of this study was to investigate the current management of anticoagulation in patients with suspected HIT while on CRRT and to identify bleeding and thromboembolic complications associated with the management of anticoagulation in this setting.
Methods
Study design
This was a single-center retrospective study at a tertiary care academic hospital of patients on CRRT with suspicion of HIT from January 2013 to December 2015. Patients were identified based on dispensing reports from our electronic medical record of patients with orders for CRRT dialysate fluid along with a parenteral anticoagulant. Chart reviews were conducted to identify patients with a suspicion for HIT during the course of CRRT as evidenced by an order for the heparin-induced platelet antibody (HIPA) assay. Time of HIT suspicion was defined as the day on which a HIPA assay was ordered. Patients were excluded if HIPA was not ordered while on CRRT. Confirmed diagnosis of HIT was based on results of a serotonin release assay (SRA). At our institution, the SRA is only tested if the initial HIPA is positive.
Outcomes and definitions
For each patient with suspicion of HIT, a 4 T’s score 10 was calculated based on criteria at the time of HIPA order. Thrombocytopenia timing and magnitude was based on platelet counts on the day of HIT suspicion. Chart reviews identified that each patient was receiving at least 1 medication that could contribute to thrombocytopenia; therefore, 0 points were awarded to each patient for “other cause of thrombocytopenia,” because CRRT could also be a contributing factor.
Details of management of anticoagulation during the period of concurrent CRRT and thrombocytopenia, CRRT details including type and flow rates, and safety endpoints were collected via chart review. Choice of anticoagulation was identified, both prior to a HIPA assay order and during the period of HIT suspicion. Anticoagulant starting doses and final therapeutic doses were collected. Activated partial thromboplastin time (aPTT) values were collected for the duration of medication administration following suspicion of HIT while the patient remained on CRRT, for a maximum of 7 days. Goal therapeutic range was defined as 60 to 90 seconds, in accordance with our institutional guidelines. Therapeutic dose of the anticoagulant was defined as the first infusion rate to achieve 2 consecutive therapeutic aPTT values based on our institutional therapeutic range. Time to therapeutic dose was measured from medication start time to the first of 2 consecutive therapeutic aPTT values. Bleeding and thrombotic events were identified during the period of alternative anticoagulation. Bleeding outcomes were defined according to International Society for Thrombosis and Hemostasis criteria. 11 Thromboembolic events were identified by documentation of ultrasound imaging.
Outcomes were compared between patients who received argatroban or bivalirudin during the period of HIT suspicion. A separate analysis was conducted in patients who received argatroban or bivalirudin to compare patients stratified by initial anticoagulant dose. Patients were considered to have a “low” or “high” initial dose if their initial infusion rate was less than or equal to, or greater than the median initial rate, respectively.
Statistical analysis
Qualitative data were analyzed by chi-square or Fisher exact test. Nonparametric quantitative data were analyzed by Wilcoxon rank sum test. Parametric quantitative data were analyzed by Student t test. All analyses were performed using Statistical Package for Social Sciences (SPSS) version 23.0 (SPSS, Chicago, IL).
Results
Patient demographics
From January 2013 through December 2015, a total of 325 patients were identified with orders for parenteral anticoagulants while on CRRT. Among these, 102 had orders for HIPA assays. In 28 patients, HIPA was ordered while the patient was not on CRRT. The final cohort of patients with suspicion of HIT while on CRRT included 74 patients. Baseline characteristics are presented in Table 1.
Characteristics of the cohort (N = 74).

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferas; CRRT, continuous renal replacement therapy; CVVH, continuous venovenous hemofiltration; CVVHD, continuous venovenous hemodialysis; INR, international normalized ratio.
Data are presented as mean ± SD, median (IQR), or n (%).
Immediately prior to HIT suspicion (at the time of HIPA lab order), 1 patient was receiving argatroban, 25 UFH infusions, 37 prophylactic dosing subcutaneous UFH, and 11 no anticoagulation. Fifty-seven patients (77%) were changed to a new anticoagulant during the period of HIT suspicion. Twenty patients received argatroban, 18 received bivalirudin, 1 received fondaparinux, 4 received UFH infusions, 10 received prophylactic dosing subcutaneous UFH, and 21 received no anticoagulation. When calculating 4 T’s scores, 39 patients (53%) had low suspicion, 34 (46%) had intermediate suspicion, and 1 (1%) had high suspicion of HIT. SRA results were positive in 7 patients (9%). Among these patients with confirmed HIT, 4 were managed with argatroban, 2 with bivalirudin, and 1 with no anticoagulation (Figure 1).
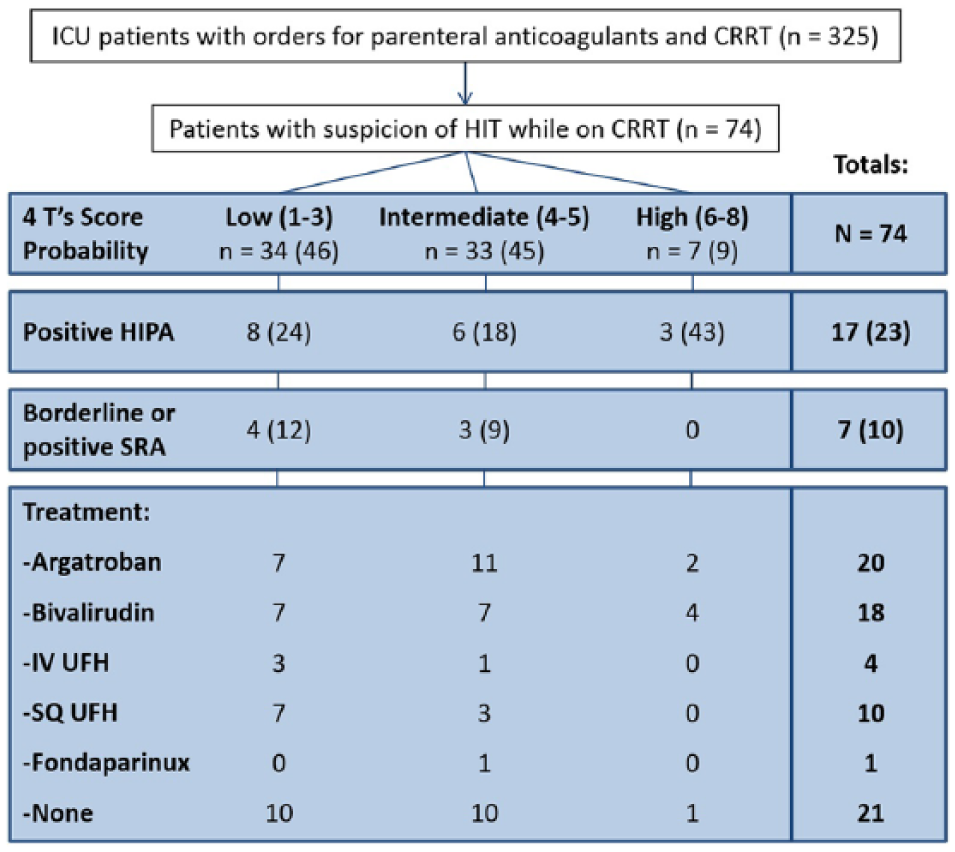
Patient selection flow diagram.
Argatroban vs bivalirudin
For patients on CRRT, our institutional guidelines for treatment of HIT recommend bivalirudin with an initial dose of 0.05 mg/kg/h. No recommendations are provided for argatroban in the setting of CRRT (1 μg/kg/min is recommended for critically ill patients and/or patients receiving intermittent hemodialysis). During the period of concomitant HIT suspicion and CRRT, initial infusion rates were significantly lower than therapeutic dosing rates among patients treated with argatroban or bivalirudin (0.50 μg/kg/min vs 1.0 μg/kg/min, P = .047; 0.050 mg/kg/h vs 0.075 mg/kg/h, P = .006). Seventeen patients treated with argatroban (85%) and 15 treated with bivalirudin (83%) achieved therapeutic anticoagulation, respectively. Patients treated with argatroban had significantly more therapeutic aPTT values than patients treated with bivalirudin (55% vs 43%, P = .008), but there was no significant difference in the time to reach a therapeutic dose (Figure 2).

Initial and therapeutic doses of argatroban and bivalirudin.
Therapeutic doses of argatroban in patients receiving continuous venovenous hemodialysis (CVVHD) and those receiving continuous venovenous hemofiltration (CVVH) were 3.62 μg/kg/min and 1.00 μg/kg/min, respectively (P = .371). Therapeutic doses of bivalirudin in patients receiving CVVHD and those receiving CVVH were 0.118 mg/kg/h and 0.070 mg/kg/h, respectively (P = .431). There was no significant correlation between dose and ultrafiltration rate for argatroban (R2 < 0.001) or bivalirudin (R2 = 0.082), but patients treated with bivalirudin had a trend toward requiring higher therapeutic doses with higher CRRT flow rates (Figure 3).

Correlation between dose and CRRT replacement fluid rate.
There were no differences in bleeding or thrombotic complications between patients treated with argatroban or bivalirudin (Table 2). Four patients treated with argatroban (20%) and 5 patients treated with bivalirudin (28%) met criteria for major bleeding (P = .573). Three patients treated with argatroban (15%) and 4 patients treated with bivalirudin (22%) developed new thrombosis (P = .566).
Argatroban vs bivalirudin during the period of HIT suspicion (N = 38).

Abbreviations: ALT, alanine aminotransferase; aPPT, activated partial thromboplastin time; AST, aspartate aminotransferas; HIT, heparin-induced thrombocytopenia.
Data are presented as median (IQR) or n (%).
Argatroban doses are reported in μg/kg/min; bivalirudin doses are reported in mg/kg/h.
High vs low initial dose
The median initial dose of argatroban was 0.5 μg/kg/min. No significant difference was found in time to therapeutic level between patients who received low or high initial doses (19.1 h vs 17.6 h, P = .558) (Table 3). The median initial dose of bivalirudin was 0.05 mg/kg/h. Compared with patients who received lower initial doses, patients who were started on doses greater than 0.05 mg/kg/h reached a therapeutic level significantly faster (18.6 h vs 8.3 h, P = .042). There were no significant differences in bleeding events between patients who received low or high initial doses of either argatroban or bivalirudin. Three patients (25%) of patients receiving a low initial dose of argatroban, and 4 patients (25%) of patients receiving a low initial dose of bivalirudin developed a new thrombosis. No patients receiving a high initial dose of either medication developed thrombosis, but these differences were nonsignificant.
Low vs high initial infusion rate during the period of HIT suspicion.

Abbreviations: aPPT, activated partial thromboplastin time; HIT, Heparin-induced thrombocytopenia.
Data are presented as mean ± SD, median (IQR), or n (%).
Table 4 describes types of new thrombotic events in patients during the period of HIT suspicion.
Thrombotic events during the period of HIT suspicion.

Abbreviations: DVT, deep venous thrombosis; HIT, heparin-induced thrombocytopenia; PE, pulmonary embolism.
Discussion
In this study, we investigated the management of anticoagulation in patients with suspected or confirmed HIT while on CRRT. Clinical practice guidelines recommend the use of nonheparin anticoagulants in patients with HIT. 4 More than half of patients with suspected HIT received appropriate guideline-recommended treatment.
Because patients on CRRT often develop thrombocytopenia, HIT may frequently be considered as a possible cause. Among ICU patients, the incidence of HIT has been reported to be <1%, much less common than thrombocytopenia among this population. 12 A recent study conducted in surgical ICU patients reported overdiagnosis and overtreatment of HIT, prompting a need for more optimal diagnostic criteria. 13 Unnecessary treatment with nonheparin agents may lead to increased cost and possible increased risk of bleeding, but because thrombocytopenia is so common in patients on CRRT, identifying patients at greatest risk of HIT may be difficult. 14 The “4 T’s Score” is commonly used by clinicians to estimate the likelihood of HIT and guide the need for confirmatory laboratory tests. 10 A systematic review of over 3000 patients found that a low probability 4 T’s score had a 0.998 negative predictive value, regardless of the study population. 15 In our study, 34 patients had low probability of HIT based on 4 T’s score, but still had HIT assays ordered. Interestingly, 4 of these patients had confirmed diagnosis of positive HIT with SRA. As our study was retrospective, it is possible that our 4 T’s score collected from manual chart review differed from the 4 T’s score calculated at the time of the encounter. A post hoc analysis of a study in medical-surgical ICU patients found disagreements between real-time 4 T’s scoring by research coordinators and scores calculated by central adjudicators. 16
In the setting of HIT, guidelines recommend switching to nonheparin anticoagulation, but there is minimal evidence to guide therapy in patients on CRRT. 4 Similar to comparative studies in patients with normal renal function, our study demonstrated similar efficacy and safety of argatroban and bivalirudin for treatment of HIT.5,17,18 Argatroban undergoes minimal renal elimination, so it may be preferred in patients with renal dysfunction.4,19,20 Institutional guidelines recommend argatroban first line for patients with renal insufficiency and normal or preserved hepatic function, with an initial dose of 2 μg/kg/min for normal hepatic function, 1 μg/kg/min in critically ill patients, and 0.5 μg/kg/min in patients with liver dysfunction; however, no dosing recommendations are made specifically for patients on CRRT. Favorable outcomes with argatroban have been reported in a small number of critically ill patients with HIT while on CRRT. In a small case series of patients receiving argatroban in the setting of RRT, a dose of 0.9 μg/kg/min achieved therapeutic aPTT in the single patient on CVVH. In this study, neither CRRT nor hemodialysis produced any clinically significant systemic clearance of argatroban. 21 In a previous study of argatroban in 30 patients on CRRT, a bolus of argatroban 100 μg/kg IV was administered to patients before starting a continuous infusion at 1 μg/kg/min and titrating to aPTT 1.5 to 3 times baseline. 22 Clinical trials have shown a decreased clearance necessitating a dose reduction in patients with hepatic failure and patients with higher Acute Physiology and Chronic Health Evaluation (APACHE) II scores.19,22-24 Based on the results of our study, we recommend an initial dose 1.0 μg/kg/min in patients on CRRT, and 0.5 μg/kg/min in patients with liver dysfunction on CRRT at our institution.
Unlike argatroban, bivalirudin is not Food and Drug Administration (FDA)-approved for HIT but is commonly used off-label with a recommended initial dose 0.15 to 0.2 mg/kg/h in patients with normal renal function.4,7,25 Current institutional guidelines suggest an initial dose of 0.05 mg/kg/h for patients on CRRT, but this dose was not high enough to reach therapeutic levels in the majority of patients in our study. Few studies have evaluated the use of bivalirudin for the management of HIT in patients with reduced renal function. A few studies reported that patients with renal dysfunction, with or without RRT, required lower doses of bivalirudin (mean dose 0.04-0.07 mg/kg/h) to achieve a therapeutic aPTT.26,27 But in each of these studies, patients receiving CRRT were not differentiated from other patients with renal dysfunction, making it difficult to guide our dosing in this population. Furthermore, prior studies have not consistently documented CRRT replacement fluid rates, and our institution often uses higher flow rates than previously reported. Another retrospective study found that patients on intermittent hemodialysis or CRRT required lower doses than patients with normal renal function but higher doses than patients with Clcr less than 30 mL/min not receiving dialysis. 26 In this study, average therapeutic dose of bivalirudin in the setting of CRRT was 0.07 mg/kg/h. Based on the results of our study, we have modified our institutional guidelines to recommend an initial dose 0.075 mg/kg/h for patients on CRRT.
Rates of bleeding and thrombosis in this study are comparable to those previously reported in similar patients. Prior studies in patients on RRT receiving argatroban have reported bleeding in 0% to 31%, and thrombosis in 3% to 11%.18,22,24 Studies in patients with HIT and renal dysfunction with or without RRT receiving bivalirudin have described major bleeding in 7% to 39% of patients, with rates of thrombosis 1% to 15%.18,26,27 In our study, argatroban and bivalirudin were associated with major bleeding in 20% and 28%, and thrombosis in 15% and 22%, respectively. Bleeding definitions may have differed between trials. Although not statistically significant, there were more thrombotic complications in patients who were started on a low initial dose compared with high initial dose of both argatroban and bivalirudin, with no difference in the incidence of bleeds. Previous literature has described various doses required to achieve therapeutic aPTT levels, some of which were lower than those found in our study.8,9,22,24,26 However, differences in CRRT characteristics, goal aPTT ranges, and other patient factors may make it difficult to compare doses between institutions.
There are few alternatives to these options for anticoagulation in the setting of HIT. Fondaparinux is FDA approved for treatment of HIT but contraindicated in renal dysfunction. 28 It is widely accepted that both UFH and low-molecular-weight heparins (LMWH) should be avoided in patients with HIT, and warfarin is unsafe to use in the management of HIT due to its early effects on protein C.29,30 While direct oral anticoagulants (DOAC) are not FDA approved for use in patients with HIT, they could be an attractive alternative due to their ease of administration and no requirement for monitoring. However, although case reports suggest safety and efficacy of rivaroxaban, dabigatran, and apixaban in the treatment of HIT in patients with normal renal function, there are no data on the use of these agents for patients on CRRT.31-33
This study had a number of limitations. The retrospective design makes it difficult to ensure accuracy and completeness in data collection. Manual chart review may have incorrectly identified and categorized bleeding events. The average weight of our population was more than 100 kg. It is unclear how obesity affected drug dosing and outcomes. The indication for anticoagulation in each patient was not collected, although this may have impacted patients’ risk of bleeding or thrombotic events and decisions regarding anticoagulation management. Clotting of the CRRT circuit was not tracked. We did not perform a power calculation to determine necessary sample size required to find significance in clinical outcomes. Finally, this was a small single-center study, so results may not be generalizable to other institutions.
Currently, there is a paucity of literature to guide management of anticoagulation in the setting of HIT in patients on CRRT. In this study, argatroban and bivalirudin demonstrated similar rates of bleeding and thromboembolism. Higher initial infusion rates of DTIs than what are currently being used at our institution in patients on CRRT may help improve target therapeutic anticoagulation levels and reduce the risk of thromboembolic events, with no increased risk of bleeding.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SP, HM, and AL conceived and designed the study. TC collected and analyzed data, SP, HM, and AL assisted with data analysis. TC wrote the manuscript. SP and AL helped with manuscript revisions.
