Abstract
Critically ill children require nutritional support that will give them nutritional and non-nutritional support to successfully deal with their disease. In the past few years, we have been able to better understand the pathophysiology of critical illness, which has made possible the establishment of nutritional strategies resulting in an improved nutritional status, thus optimizing the pediatric intensive care unit (PICU) stay and decreasing morbidity and mortality. Critical illness is associated with significant metabolic stress. It is crucial to understand the physiological response to stress to create nutritional recommendations for critically ill pediatric patients in the PICU.
Introduction
Children have small macronutrient reserves and high energetic requirements, a situation which worsens in those who are critically ill due to the intense stress on the body. The critically ill child undergoes pathophysiological changes which, added to the treatment carried out in the intensive care unit (ICU), put him or her at nutritional risk due to the using up of energy reserves, favoring malnutrition, worsening of the underlying illness, delayed recovery, greater complications, infections, and increased morbidity, leading to a prolonged ICU stay. 1 For this reason, nutritional metabolic support is part of the therapeutic strategy, beginning at admission, of any critically ill child.2–5 This article reviews the physiological foundations of nutritional support, the concept of nutritional risk, nutritional goals, enteral nutrition (EN) and parenteral nutrition (PN) controversies, as well as new strategies in the nutritional support of critically ill children.
Physiological Foundations of Nutritional Support
The following phases may be defined during critical illness:
Acute phase: It occurs after an event that requires vital organ support. It is characterized by the activation of the inflammatory cytokine cascade and the central nervous system to survive. This phase may last hours or days and is made up of 2 kinds of response (Figure 1):
(a) Neuroendocrine response: activation of the hypothalamic-pituitary axis, increasing adrenocorticotropic hormone and thyroid-stimulating hormone (TSH) levels; inactivation of peripheral hormones (triiodothyronine [T3] and insulin-like growth factor); and increased cortisol.
(b) Immune and metabolic response: catabolism secondary to the great mobilization of amino acids and fatty acids from muscle to guarantee nutrients. In addition, neutrophils, monocytes, and lymphocytes are activated. 6

Phases of neuroendocrine response to critical illness. 6 EGP indicates endogenous glucose production; GH, growth hormone; IGF-1, insulin-like growth factor 1; MPS, muscle protein synthesis; PT, protein turnover; rT3, reverse triiodothyronine; T3: triiodothyronine.
Stable phase: This phase is characterized by the stabilization or the beginning of the withdrawal of support to vital organs, without having resolved the response generated by the stress of the first phase. This phase may last for days or weeks.
Recovery phase: This phase involves normalization of the neuroendocrine, immune, and metabolic alterations and minimal or no support to vital organs. It may last for weeks or months. 6
Nutrition During the Phases
The amount of nutrients administered during the acute phase is determined by a close relationship between autophagy and nutritional therapy. 7 Autophagy, described in 1970, is a lysosomal process capable of eliminating damaged organelles, unfolded proteins, microorganisms, and excessive fat or carbohydrate stores. It is a quality control system that plays a role in immunity, inflammation, and infection and is regulated through stimulating factors (hunger, oxidative stress, glucagon, and glutamine) and inhibiting factors (feeding, insulin secretion, hyperglycemia, and excessive nutrients).8,9
This autophagy, which presents during the acute phase, may be inhibited by early nutritional therapy, theoretically reducing mitochondrial function and oxidative stress tolerance, which would explain the adverse effects of early total EN, such as a greater risk of multiple organ failure and mortality.10,11
Today, we know that nutrient restriction in the acute phase decreases even further the levels of T3, thyroxine, and TSH hormones, as well as the T3/reverse T3 ratio, which is associated with a better result in critically ill patients. The administration of complete nutrition during this phase has not been shown to decrease catabolism or muscular atrophy. 12
In the stabilization phase, characterized by a decrease in C-reactive protein and normalization of T3, 6 the administration of medications and prolonged immobilization deteriorate the nutritional status and increase the presentation and severity of muscle weakness. It has been proven that the administration of nutrients during the acute phase does not diminish its incidence; on the contrary, it increases its severity. 13
The objective of these phases (stabilization and recovery) is to recover lean mass and ensure mobility and reconditioning of the patient, with the objective of reaching the growth percentile. 6
Nutritional Goals
Critically ill children should receive a precise initial evaluation and planning of their nutritional requirements: caloric and energy needs, with follow-up in the event of energy and protein imbalance to avoid under- or over-feeding.14–16
Energy Requirements
Energy requirements in critically ill children should be individualized because they change constantly as the condition evolves. Indirect calorimetry is the measurement that provides a precise calculation of energy expenditure.6,14
The American Society of Parenteral and Enteral Nutrition (ASPEN) recommends indirect calorimetry for estimating energy needs and determining the achievement of the basal nutritional status on admission to the pediatric intensive care unit (PICU), which may also help identify patients at risk of refeeding syndrome.6,17 However, if indirect calorimetry is not available, the initial energy calculation may be based on the formulas of Schofield and the World Health Organization (WHO), which provide estimates of caloric needs (Table 1).6,16-18
Equation for predicting basal metabolic rate.

Abbreviations: H, height; W, weight.
Adapted with permission from Skillman and Mehta. 14
Macronutrient Requirements
Proteins, fats, and carbohydrates are the macronutrients which provide energy for carrying out all bodily functions. The optimal amount and composition needed of each one of these elements is a difficult goal to achieve and may change throughout the course of the disease. The goal of nutritional therapy is to ensure an adequate intake of proteins and energy for maintaining a positive nitrogen balance, in the interest of improving the loss of lean body mass and facilitating the healing of wounds and the inflammatory response.16,17
Proteins supply 4 kcal/g of energy, and pediatric requirements are related to age, being higher in critically ill children because catabolism reaches its maximum level 8 to 14 days after the initial lesion. 18
The protein requirements of seriously ill children according to the ASPEN guides, by age groups, are described in Table 2. In situations such as kidney or liver disease, it may be necessary to decrease the supply, and in certain states of severe stress, additional protein supplements may be necessary to satisfy the metabolic demands. 17
Protein requirements of critically ill children. 17

Lipids are the first source of energy used in critically ill children, and their metabolism is increased in cases of stress. 17 In PN, lipid requirements start at 1 g/kg/day, increasing every 24 hours up to 2-4 g/kg/day, with a maximum intake of 3 to 4 g/kg/day in infants and 2 to 3 g/kg/day in older children (with a maximum infusion rate of <2.5 mg/kg/min). Lipids may be administered as long-chain triglycerides (soy oil) and medium-chain triglycerides (fish oil) and are a source of essential fatty acids, omega-6 fatty acids (linoleic acid), and omega-3 fatty acids (linolenic).17,19
Carbohydrates are the main source of energy and should supply 40% to 60% of the total needs, with glucose being the essential substrate used by the brain, the erythrocytes, and the renal medulla, with a contribution of 4 kcal/g. The upper limit of dextrose is determined by its oxidation rate (Table 3; the maximum oxidation rate tolerated by small children is 12-15 mg/kg/min). If glucose is administered within this limit, its oxidation produces energy and glycogen; if the limit were exceeded, negative effects would be generated (such as lipogenesis) and the appearance of steatosis would be facilitated.18,19
Recommendations regarding glucose infusion. 17
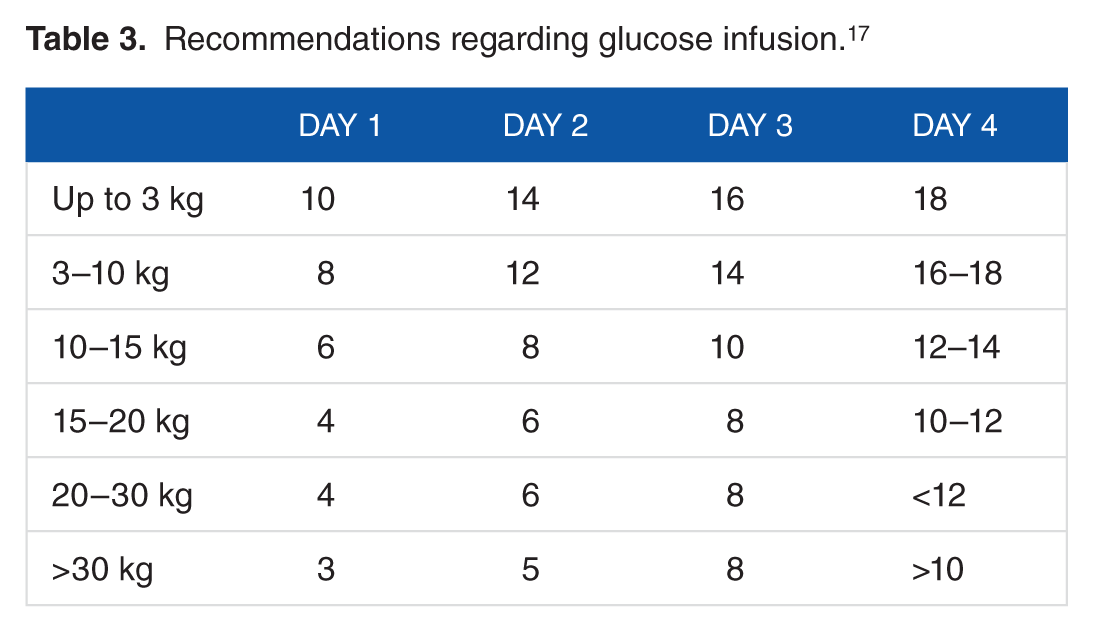
Table 4 describes the suggested nutritional input in the different phases.
Suggested energy and macronutrient contribution during the different phases of critical illness. 6

Abbreviation: REE, resting energy expenditure.
Enteral Nutrition
Enteral nutrition continues to be the most used strategy. It should be implemented early, once the gastrointestinal tract allows for it. Enteral nutrition has many benefits: it favors trophism of the intestinal mucosa, stimulates neuroendocrine mechanisms, decreases the possibility of bacterial translocation, has a lower risk of complications, does not require complicated asepsis techniques, and is economical. Parenteral nutrition should only be considered if EN is contraindicated or if there is intolerance.
Pre- vs Postpyloric EN
Enteral nutrition may be delivered to the stomach, duodenum, or jejunum through a nasogastric or orogastric tube, percutaneous endoscopic gastrostomy, or surgical gastrostomy tubes. When it is delivered to the duodenum or jejunum, it is considered to be postpyloric. Among the advantages are a greater ease of access with fewer comorbidities, and the disadvantages include an increased risk of bronchoaspiration and pneumonia. Strategies for minimizing aspiration include postpyloric delivery, administration of motility agents, and elevation of the head of the bed. Matheny et al found a significant relationship between the route of administration and the risk of pulmonary complications.20,21
Early vs. Late EN
Early EN is defined as feeding begun 24 to 48 hours after initiation of ventilation. It reduces inflammation, preserves enterocyte function, decreases infectious complications, serves as a vehicle for providing immunomodulating agents, and acts as an effective means of prophylaxis for stress ulcers.
The retrospective analysis of 4049 adult patients showed that ICU stay, and hospital mortality in general, was lower in critically ill patients who received early EN (18.1% vs 21.4%, P = .01, and 28.7% vs 33.5%, P = .001, respectively). 22 In the meta-analysis by Lewis et al, surgical patients who received early postoperative EN had fewer infections (relative risk [RR]: 0.72; 95% confidence interval [CI], 0.54-0.98; P = .03), a reduction in hospital stay (mean, 0.84 days; range, 0.36-1.33 days; P = .01), and a tendency toward a reduction in dehiscence of the anastomosis (RR, 0.53; 95% CI, 0.26-1.08; P = .08), compared with those who did not receive postoperative nutritional support. The meta-analysis by Dhaliwal et al of 8 studies of patients in critical condition reported a lower mortality with early vs late EN (RR, 0.52; 95% CI, 0.25-1.08; P = .08), concluding that EN should be preferred in the critically ill child.
Parenteral Nutrition
Parenteral nutrition represents one of the most advanced techniques within the field of nutrition and constitutes a salvage modality for many critically ill patients who are unable to tolerate EN.2,23,24
Children who require PN cannot be classified in a single group as they differ in multiple factors, such as the type of illness, the phase they are in, the physiological characteristics, and the energy requirement for fulfilling their needs for growth and development; this makes the composition and prescription of PN vary in the PICU.6,19
Routes of Administration
Parenteral nutrition may be administered peripherally or centrally, each having advantages and disadvantages that should be considered to achieve an adequate prescription. The peripheral route has the advantage of being economical, having a less complex vascular access, and having a lower risk of complications, mainly related to infection and thrombosis. A disadvantage is that the allowed osmolarity is less than 900 mOsm/L with less than 12% glucose, which does not permit the administration of complete PN and necessitates frequent peripheral access change due to the risk of thrombophlebitis. 25
Central line access has the advantage of allowing the use of solutions with an osmolarity greater than 900 mOsm/L, supplying complete nutrition and, if necessary, administering nutrition over a long period of time (>3 months). Its disadvantage is a greater risk of infections and vascular complications. 26
Composition
The composition of PN in pediatric patients should be prescribed according to age, weight, clinical condition, underlying pathology, and, especially, the disease phase, as previously mentioned. 6
Complications
The main complications of PN are related to the venous catheter, initially with the insertion technique, such as pneumothorax, vascular laceration, arrhythmias, cardiac perforation with tamponade, air embolism, nerve plexus lesion, or anomalous placement of the catheter. Following insertion, there can be rupture, accidental displacement, occlusion, venous thrombosis, and infection.19,26
In addition, patients with long periods of PN present metabolic complications, such as metabolic bone disease, refeeding syndrome, and hepatobiliary syndrome. These pathologies are grouped together in the acronym PNALD (Parenteral Nutrition–Associated Liver Disease).19,25
Parenteral nutrition cholestasis is one of the most difficult problems related to its prolonged use, especially when it progresses to hepatic disease. It is a multifactorial complication, having both modifiable (sepsis, PN duration) and nonmodifiable (prematurity, birth weight) risk factors.
Controversies in PN
There are great controversies in the nutritional support of the critically ill child, the supplementation of EN with PN, and early vs late PN.
There are great doubts regarding the supplementation of EN with PN, understood as adding PN to patients who receive insufficient or hypocaloric EN (trophic enteral input less than 60% of the basal caloric input), and the available evidence is not conclusive. The exact time to add supplementary PN to a hypocaloric EN continues to be controversial. If a patient is at high nutritional risk while receiving hypocaloric EN, the supplementary addition of PN after 7 days could be considered reasonable according to expert opinion, keeping in mind that this combination may be suspended when the patient tolerates more than 80% of proteins and calories through the gastrointestinal tract.27,28
Regarding the controversy of early (the first 48 hours after admission to the ICU) vs late (8 days after admission) PN, starting from the fact that EN is preferred over PN, and based on the evidence, we could conclude that in those patients with contraindications for EN, initiation of PN may be delayed 7 to 10 days, which has shown an impact on the reduction of infectious events in the ICU and the number of days of mechanical ventilation, without a major impact on mortality. 26
Nutritional Strategies in the Critically Ill Child
Once stabilization has been achieved, nutritional support should be started. The energy input and time of initiation are critical because underfeeding or overfeeding the child, or beginning early or late, may have negative consequences, causing comorbidities and repercussions which could lengthen the PICU stay.6,23
An appropriate evaluation of the nutritional state (identifying nutritional risk) is very important, adjusting the energy input to the resting caloric expenditure, to avoid an unbalanced supply of nutrients. Follow-up is carried out by monitoring weight gain, evidence of overfeeding, and altered hepatic function with the development of steatosis/cholestasis and hyperlipidemia.6,23
Currently, there is greater knowledge regarding input adjustment and methods for administering PN in critically ill children, with permissive underfeeding and strategies for the administration of lipids as examples.
Permissive underfeeding is defined as the restriction of nonprotein calories, with a complete supply of the protein requirements. With permissive underfeeding, the emphasis is on providing a sufficient level of protein intake during the first phase of the disease to satisfy the total protein requirements and fulfill the metabolic demand. Moderate calorie restriction with preserved protein intake has been shown to positively influence the results.29,30
Table 5 describes the results of studies where permissive underfeeding was used as a treatment strategy in critically ill patients.
Studies of permissive underfeeding.
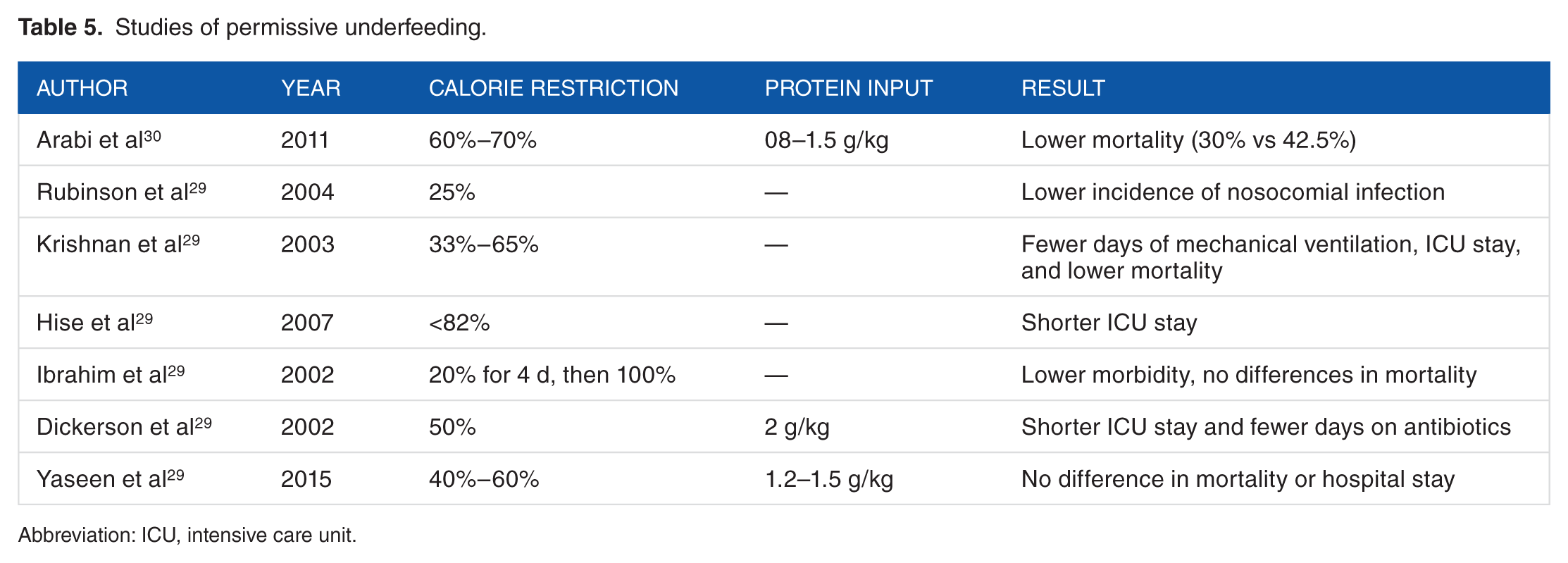
Abbreviation: ICU, intensive care unit.
According to what was stated above, a greater caloric intake does not attenuate protein catabolism in critically ill patients and its impact in the stable phase. However, its administration during the acute phase may inhibit the autophagy and oxidative stress regulation processes, which would explain the adverse effects of early nutritional therapy.
Finally, the administration of lipids could have problems such as macrophage stimulation and an increased risk of cholestasis in a patient receiving PN. Lipids are causal factors of mitochondrial oxidative stress and hepatic steatotosis, with similar pathological characteristics to nonalcoholic fatty liver. 31
Macrovesicular steatosis is the presence of small to large lipid drops in the cytoplasm of hepatocytes, with peripheral displacement of the cell’s nucleus. This type of hepatic lesion is generally reversible; however, in time, it may evolve to steatohepatitis and cirrhosis.31–34
Traditional lipid emulsions supply energy in the form of long-chain triglycerides derived from soy or olives, medium-chain triglycerides, fish oils, or a mix of these ingredients. 35 The current bibliography suggests that naturally occurring phytosterols with steroid alcohols derived from plant cellular membranes are used as part of the intravenous lipid components of PN, and the omega-6 fatty acids derived from soy have been implicated in the damage of hepatocytes through a proinflammatory mechanism fostered by the production of proinflammatory prostanoids and leukotrienes, resulting in increased oxidative stress and systemic inflammation.35–37
Recent studies have shown that PN cholestasis may be prevented or treated by restricting lipids, soy-based formulas, mixed sources, or lipid emulsions of fish oil. During the past decades, different fish oil–based alternatives have been developed, which could have fewer proinflammatory effects and fewer antioxidant effects than the standard emulsions. 38
These strategies entail different formulations of soy oil combined with medium-chain triglycerides, olive oil containing omega-9 monounsaturated fatty acids, oleic acid and fish oil containing omega-3 fatty acids, eicosapentaenoic acid, and docosahexaenoic acid, but in a 2.5:1 ratio of omega-3 and omega-6 fatty acids, respectively: Omegaven (Fresenius Kabi, Germany), Lipoplus/Lipidem (B. Braun, Germany), and SMOFlipid (Fresenius Kabi, Germany). Lipoplus has a mix of 50% medium-chain triglycerides, 40% soy oil, and 10% fish oil, and SMOFlipid has a mix of 4 oils, namely, 30% soy oil, 30% medium-chain triglycerides, 25% olive oil, and 15% fish oil. 38
Omegaven 10% is a fish oil emulsion which does not contain phytosterols and has an omega 6/3 ratio of 1:7. This product has been used to prevent and treat hepatic disease related to PN; it is not currently approved by the Food and Drug Administration. Another strategy is the weekly limiting of the amount of soy-based lipid emulsions to 1 g/kg 2-3 times per week, showing a reduction in total bilirubin and the risk of cholestasis.1,38–40
Conclusions
Critical illness is associated with significant metabolic stress. Understanding the response to this stress and the characteristics of its 3 phases is essential for understanding and implementing the nutritional recommendations for critically ill pediatric patients.
After reviewing the literature, we consider that the assessment of energy requirements in the critically ill pediatric patient should be individualized, and nutritional risk should be determined to optimize nutritional support. If indirect calorimetry is not available, the recommended formulas are Schofield and the global health organization, always taking into account the physiological response to stress and phases of the disease (acute, stable, and recovery) for adjusting nutritional inputs. Permissible underfeeding with 60% to 70% of the estimated standard calorie requirement with nonprotein calorie restriction and supplying complete protein needs during the first phase of critical illness satisfy the requirements of total protein and meet metabolic demand, improve catabolism, and stimulate autophagy during the acute phase and have been shown to positively influence outcomes in these patients in the short and long term. In critical children with intact bowel, EN is the route of choice as it is associated with lower morbidity and mortality, and in patients who are definitely not able to initiate EN, PN should be initiated and it may even delay up to 7 to 10 days. In addition, it should be taken into account that when initiating NTP, all strategies should be optimized to reduce the complications associated with it.
Footnotes
Peer review:
Five peer reviewers contributed to the peer review report. Reviewers’ reports totaled 516 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceived and designed the experiments: IJAG, CBG, PAMP, ETMS, JDTB, JPCH. Analyzed the data: IJAG,CBG, PAMP, ETMS, JDTB, JPCH. Wrote the first draft of the manuscript: IJAG,CBG, PAMP, ETMS, JDTB, JPCH. Contributed to the writing of the manuscript: CBG, JFS. Agree with manuscript results and conclusions: All authors. Jointly developed the structure and arguments for the paper: IJAG,CBG, PAMP, ETMS, JDTB, JPCH, JFS. Made critical revisions and approved final version: IJAG,CBG, JFS. All authors reviewed and approved of the final manuscript.
