Abstract
Peptic ulcer bleeding remains an important medical emergency. Important recent advances are reviewed. These include further support for a more restrictive transfusion strategy aiming for a target haemoglobin of 70-90 g/L. The Glasgow-Blatchford score remains the most useful assessment score for identifying the lowest risk patients suitable for outpatient management and predicting the need for intervention. Newer scores such as the AIMS65 and Progetto Nazionale Emorragia Digestive score (PNED) may be more accurate in predicting mortality. Pre-endoscopy erythromycin improves outcomes and is underused. A new disposable Doppler probe appears to provide more accurate determination of both rebleeding risk and the success of endoscopic therapy than purely visual guidance. Over-the-scope clips and haemostatic powders appear to have some role as endoscopic salvage therapies. Non-
Keywords
Introduction
Acute upper gastrointestinal haemorrhage continues to be an important clinical problem. The incidence of non-variceal acute upper gastrointestinal bleeding is about 85/100 000 per year in the United Kingdom. 1 Although the specific mortality associated with portal-hypertensive variceal bleeding is higher, 2 peptic ulcer bleeding (PUB) is still the commonest cause of both acute gastro-intestinal (GI) bleeding overall and significant bleeding requiring a blood transfusion.2,3 There have been considerable advances in many aspects of the management of PUB, but the overall mortality remains significant (around 10%): the advances in therapy being offset by the increasing age and comorbidity of the patients.
There are several comprehensive, evidence-based guidelines supporting the management of PUB,4,5 although comprehensive audits and observational studies have shown that all aspects of management do not always reliably follow guidelines.3,6,7 New data are continually becoming available in all aspects of PUB, and in many cases, these have had insufficient time to directly inform published guidelines. In addition, guidelines by their very nature are broad and overarching and are not optimally positioned for discussion of new and evolving techniques and management strategies of developments in specific, niche but important areas.
This narrative review covers important and/or controversial recent developments in all aspects of PUB. These will be placed in clinical context to further enhance the management of patients. The aim is to concisely summarise important evolutions in therapy for generalist physicians and surgeons as well as highlight important new literature to those with more focused interests in the area, in particular where this adds to, or even contradicts, standard guidelines.
The management of ulcer bleeding can be divided for convenience into several overlapping areas: recognition, risk assessment, resuscitation, endoscopic and salvage therapies and drug therapies, both in the acute presentation and primary and secondary preventive strategies. There are important new developments in all of these areas (see Figure 1), areas that will be highlighted include the application of novel prognostic scoring systems, the peri-procedural pharmacological management of the patient and co-morbidity, novel endoscopic techniques (Doppler ultrasound probe, over the scope clips, haemostatic powders), reassessment of classical endoscopic stigmata and secondary prevention strategies focused on the management of polypharmacy and co-morbidity.
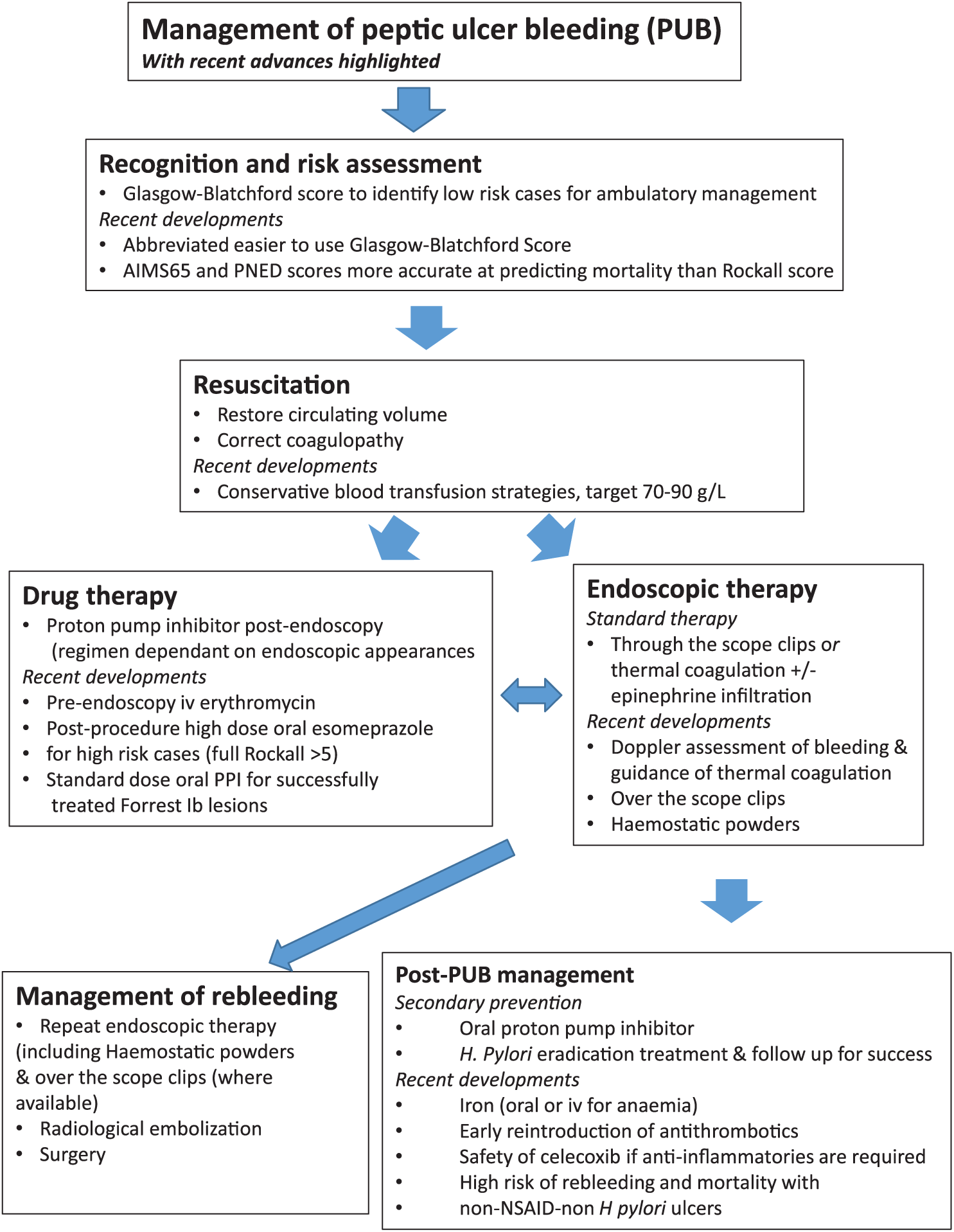
Management of peptic ulcer bleeding with recent advances in therapy highlighted.
Recognition
The presence of blood in the vomitus or melaena passed rectally is usually sufficient to alert a clinician to the presentation of acute upper GI bleeding and enough to instigate appropriate management. Sometimes rapid fresh rectal bleeding presents a dilemma: could this be colonic source or very rapid transit from an upper GI source? This has implications for the management. In the United Kingdom, National Confidential Enquiry into Patient Outcome and Death (NCEPOD) audit of severe GI bleeding, actual site of bleeding correlated poorly with the presenting general clinical features and clinical suspicion of the admitting team. 3 The presence of a pulse rate greater than the systolic blood pressure was associated with an upper GI source for fresh rectal bleeding. 3 Further prospective studies examining this index are required, but it certainly seems reasonable to perform a gastroscopy initially, before lower GI endoscopy in these patients showing that degree of circulatory compromise after they have received appropriate resuscitation.
Resuscitation
Despite the high prevalence of PUB, there are few data on any specifics of fluid resuscitation and general principles on restoring circulating fluid volume and adequacy of organ perfusion are usually employed, although it seems inevitable that there will be individual choice in terms of fluids used and rate given. There is an increasing body of data concerning transfusion strategies, although they are somewhat difficult to interpret applied to a dynamic situation, such as significant PUB when blood forms but one part of an overall fluid resuscitation strategy. Randomised studies in this area are difficult to do, and hence, the key trial examining blood transfusion strategies was extremely welcome. 8 In common with an increasing body of data from other critically ill patients, across the spectrum of medicine including major sepsis9,10 a restrictive blood transfusion strategy seems at least as good as a more traditional liberal strategy. In the trial, 8 a single unit, and then repeated as required, strategy with a transfusion trigger of 70 g/L to maintain the haemoglobin at 70-90 g/L was as safe and effective as a more traditional haemoglobin target of 90-110 g/L. The trial recruited all comers with upper GI bleeding and was not specifically designed to look at subgroups by aetiology of bleeding. Mortality was lower in the restrictive transfusion group (5% vs 9%). Those with variceal bleeding or Child’s A or B cirrhosis particularly seemed to benefit from the conservative transfusion strategy with improved mortality and rebleeding rates. In the subgroup with PUB, there appeared to be less difference between the two strategies. The restrictive strategy produced favourable clinical outcomes: mortality 3% versus 5%, rebleeding 10% versus 17% and surgery 2% versus 6%, although given the small numbers these were not statistically significant. Recruitment to the trial was, by necessity, selective excluding those with very severe bleeding and significant circulatory diseases, which may limit generalisation to other populations.
There are data showing contrary effects: one study published only in abstract form 11 examined real-world outcomes after transfusion for PUB, and showed an association between with more units transfused and lower death rates. A further uncontrolled study also from Los Angeles reported that initial blood transfusion during resuscitation was associated with a lower risk of death from PUB (odds ratio [OR]: 0.84 (95% confidence interval [CI]: 0.75-0.94), 12 although it is possible that blood transfusion in this context is more of marker for the adequacy and aggressiveness of initial fluid resuscitation.
A meta-analysis reported the results of pooling the four available studies of transfusion strategies in acute upper GI bleeding, individually these all have different methodologies and inclusion criteria. 13 Not all of the included studies specifically examined only acutely bleeding peptic ulceration. Overall, the results favoured a restrictive transfusion strategy: there were significant reductions in death and length of stay with the restrictive strategy. Rebleeding rates were also non-significantly lower in the restrictive group (OR: 0.26; 95% CIs: 0.03-2.10). A subsequent further meta-analysis including only data on acute gastrointestinal bleeding from five randomised controlled trials showed that a restrictive transfusion strategy was associated with lower all-cause mortality (relative risk: 0.65; 95% CI: 0.44-0.97) and rebleeding (relative risk: 0.58; 95% CI: 0.40-0.84) without any effect on ischaemic events. 14 The exact optimal resuscitation strategy is unclear and always needs to be individualised to the specific patient. However, the consistency and biological plausibility of the results, combined with the costs and potential harm of blood transfusion, mean that it would seem appropriate to employ a conservative transfusion strategy for most patients with PUB, while maintaining adequate circulating fluid volumes.
It is important to emphasise that blood-transfusion strategies are but only one component of fluid resuscitation in PUB and that in the acutely bleeding patient haemoglobin levels only form one part of the assessment of circulatory instability. Any choices concerning fluid and blood replacement must be governed by the need to restore adequate organ perfusion. It seems that crystalloid fluid resuscitation followed by blood to maintain the haemoglobin level at 70-90 g/L is most appropriate for most patients, further data are required for those with severe or critical vascular and circulatory diseases.
Risk Stratification
Many systems that have been used to stratify risks in upper GI bleeding (see Table 1). Probably the two most widely used and studied are the Rockall scores (both pre- and post-endoscopy) and the Glasgow-Blatchford Score (GBS). 20 Additional scores have been proposed. The AIMS65 score has been advocated as an even simpler score requiring scoring only on a 5-point score for each of albumin <30 g/L, INR (>1.5), Glasgow Coma score <14. Systolic blood pressure < 90 mmHg and age > 65. Although the AIMS65 can reliably predict mortality, it appears less accurate than the GBS in determining the need for interventions such as blood transfusion or admission to critical care.15,17
Principle risk scores used in acute upper GI bleeding. Showing the components of each score and the main clinical uses. References as cited in main text.
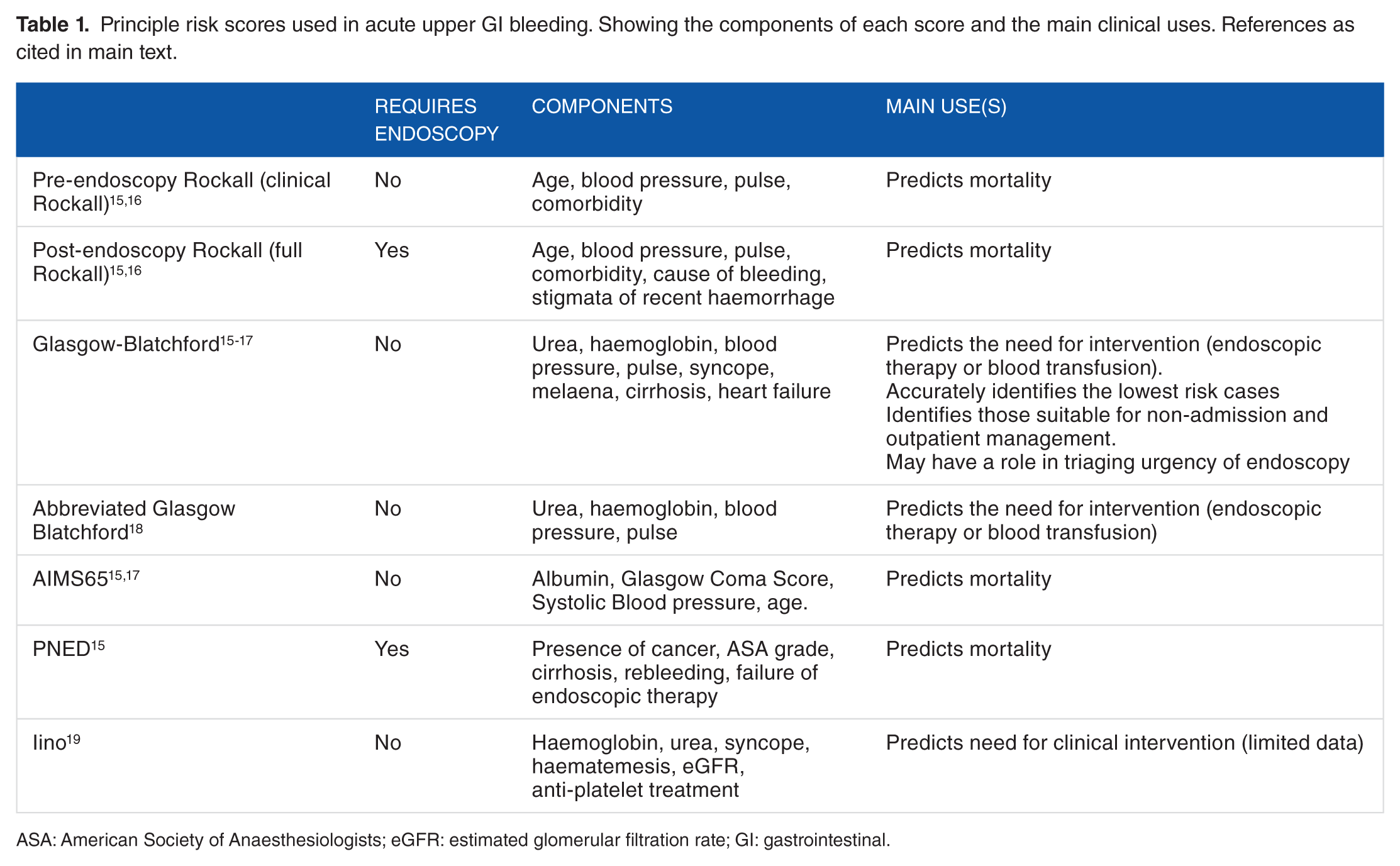
ASA: American Society of Anaesthesiologists; eGFR: estimated glomerular filtration rate; GI: gastrointestinal.
A further score, the Progetto Nazionale Emorragia Digestive score (PNED) system which relies on a rather cumbersome multipart scoring utilising age, the presence of cancer, renal failure, American Society of Anaesthesiologists (ASA) grade, cirrhosis, rebleeding and failure of endoscopic therapy has been proposed. A large prospective study of over 3000 patients confirmed that the GBS clearly performed best in identifying the lowest risk patients and also in predicting important interventions such as endoscopic therapy or blood transfusion. Although the PNED and AIMS65 scores were best at predicting mortality, none of the scores apart from the GBS appeared to be clinically useful in determining either the safety of outpatient management or the need for endoscopic therapy. 15 A GBS score of 7 or more was best at predicting the need for endoscopic treatment. 15
Despite the fact that the GBS and Rockall scores have always been designed to assess somewhat different aspects, there continues to be studies comparing the clinical utility of these studies. It should be highlighted that the Rockall scores assess the mortality risk and were never designed directly as decision tools (accepting that any risk assessment of the patient obviously does inform clinical-decision making). In contrast, the GBS was explicitly designed and validated to detect those cases not needing intervention (therapeutic endoscopy or blood transfusion). Thus, not surprisingly, the GBS consistently performs better in identifying lower risk cases, especially those suitable for safe direct discharge and outpatient management.16,21 A recent international validation of the GBS confirmed that a score of 0 or 1 is associated with a very low risk of intervention and hospital admission and emergency endoscopy is not required. 15
Despite the plethora of existing scores, a further model has recently been proposed based on seven factors: systolic blood pressure <100 mmHg, syncope, haematemesis, haemoglobin < 100 g/L, blood urea 22.4 mg/dL, estimated glomerular filtration rate <60 mL/min/1.73 m2, and the use of anti-platelet medications. 19 In a cohort of Japanese patients, this score was superior to the pre-endoscopy Rockall and AIMS65 scores in predicting clinical intervention. 19 However, it is requires further study and in particular comparison against the GBS before the exact role in management can be established.
Use of anti-thrombotic agents at the time of upper GI bleeding does not seem to be associated with adverse outcomes. A prospective study of 619 patients from North America, Europe and Asia that required endoscopic intervention confirmed that patients taking anti-thrombotics were generally older with more co-morbidity and hence higher risk scores (AIMS65, Rockall and Glasgow-Blatchford) but actually had lower mortality (4% vs 11% and shorter hospital stay (mean of 6.9 days vs 7.9 days) when compared to non-users. 22
Cirrhosis is an important independent risk factor for mortality in PUB. A prospective analysis of consecutive cases of all-cause upper GI bleeding in cirrhotic patients showed that 45-day mortality from PUB (17%) was similar to acute variceal bleeding (19%), although deaths in both groups were due to liver failure and not controlled bleeding. 23
The timing of emergency endoscopy in acute upper GI bleeding remains controversial and although immediate endoscopy (as early as possible) seems theoretically attractive, this approach has not been supported by evidence. Some studies have demonstrated that very early endoscopy is not associated with better outcomes, and in some case worse outcomes have been reported (although this latter effect could have been an artefact of the design of the observational studies).24,25 Perhaps more usefully, those patients with a GBS of 12 or more were shown (again in an observational study) to have lower mortality with a presentation to endoscopy time of <13 hours, whereas in those with lower GBS scores there was no association between early such endoscopy and clinical benefit. 26 These findings have not been university reproduced, although the retrospective nature of the all the studies conspires against determining the exact nature of the relationships between timing of endoscopy and outcomes. Ahn et al reported that urgent (<8 hours from presentation) endoscopy was not associated with better clinical outcomes than early (8-24 hours) endoscopy. Although those having early endoscopy tended to have higher risk scores and higher risk lesions seen at endoscopy, when analysis was confined to those patients with a higher risk (here defined as a pre-endoscopy Rockall score of 3 or more), urgent endoscopy was not associated with any positive or negative clinical outcomes. 27 In contrast, another retrospective study showed a clear association between urgent (<12 hours) and adverse outcomes compared to early (12-24 hours). Those undergoing urgent endoscopy were 5 times more likely to reach a composite end-point of death, inpatient rebleeding, endoscopic, surgical or radiological intervention. 28 This effect may have been influenced by causation bias (those having urgent endoscopy had higher AIMS65 and GBS) but analysis of the results according to GBSs is very informative. Interestingly in this study, urgent timing of endoscopy was not associated with either better or worse outcomes in higher risk cases (defined as GBS of 12 or more). However, in the lower risk group (GBS of <12), urgent endoscopy was clearly associated with worse outcomes (composite outcome 29% vs 5%, rebleeding 10% vs 3%). The reasons for this perhaps counter-intuitive association are unclear. The authors postulate, quite reasonably, this could be due to inadequate resuscitation of these patients considered low risk. One further recent retrospective study from Korea 29 showed that in higher risk patients (defined as GBS of > 7) urgent endoscopy (<6 hours) was associated with lower 30-day mortality (2.5%) than later (6-48 hours) endoscopy (3.8%). However, this was not associated with any association with rebleeding, transfusion requirements, intensive care admission or hospital stay, all or which are usually associated with mortality. Although more endoscopic therapy was applied in the urgent group, this was mainly driven by increased treatment of oozing Forrest Ib lesions (that are probably not as high risk as previously believed, as will be discussed subsequently).
The conclusion is that GBS scores can be used effectively to triage patients not requiring admission. It may also be possible to detect those that may benefit most relatively early endoscopy and equally detect those in who very early endoscopy may actually be associated with harm.
Further modifications of the GBS have also been reported, omitting the most subjective of the criteria and relying merely on objective measurable haemodynamics and laboratory values, omitting the scoring for chronic disease/major co-morbidities, melaena and syncope, Interestingly, the abbreviated score seemed to perform as well as the full GBS score and was also superior to the Rockall scores for predicting the need for clinical intervention. 18 Further validation studies are required, but this may prove to be a useful simple modification to enhance clinical practice.
In addition to these predominantly clinical scores, the endoscopic appearances can provide additional prognostic information. Reviewing a large cohort of patients with PUB from Los Angeles, Camus et al
12
showed that ulcer size was an independent predictor of 30-day rebleeding and death. Increasing ulcer size above 10 mm was associated with an increased risk of death of 1.08 per 10% increase in ulcer size in patients presenting
Although this is an interesting finding, at present, it does not seem that ulcer size alone will be sufficient to influence decisions about interventions or second-look endoscopies, but it would be wise to consider reports of ulcer size when considering any further descriptions of scoring systems or treatments that have a bearing on prognosis in PUB.
Endoscopy and Endoscopic Therapy
Endoscopy obviously remains the essential haemostatic modality in PUB. Dual therapy, with adrenaline/epinephrine infiltration combined with either thermal coagulation with a bipolar probe or mechanical haemostasis with endoclips remains the optimal endoscopic therapy advocated in the major guidelines.4,5 The thermal or mechanical aspects are the most important and although adrenaline is often used to clear the endoscopic field it probably adds little to the haemostasis as secured by other means. 15 There is a variety of through-the-scope endoscopic clips available from different manufacturers, but there are no data showing clear superiority of any one clip.
Within endoscopy, there are four important recent developments: Doppler probe-guided lesion assessment and treatment, the reassessment of the bleeding risk associated with oozing (Forrest Ib) ulcers, the availability of large over-the-scope clips and the new haemostatic powders. The exact place of these new haemostatic methods within the management pathway requires further assessment but all seem to offer some advantages in certain circumstances, although arguably the Doppler system offers the greatest change to overall strategies.
Doppler probe assessment of ulcers to detect significant arterial signals in the ulcer base is not a new technique, having been reported many years previously. 30 However, the lack of availability of the equipment and lack of convincing evidence of efficacy at the time, with available endoscopic modalities rather precluded wider adoption. More recently, there has been an increase in interest, stimulated by the availability of an easier to use Doppler unit and disposable relatively low-cost endoscopic probes (Vascular Technologies Inc, Nashua, New Hampshire, USA). Two studies from the same group have shown initially how Doppler probe assessment is more accurate than classical endoscopic scoring in predicting rebleeding risks 31 and second in a randomised-trial that Doppler probe-guided management reduces rebleeding and further intervention compared to standard treatment. 32 Doppler assessment demonstrated that many oozing ulcers (Forrest Ib) are actually not associated with significant arterial flow into the ulcer (only 46.7% showed a positive Doppler signal) and that these ulcers are associated with a lower rebleeding rate than typically assumed. The rate of Doppler-positive arterial flow in oozing ulcers is actually significantly lower than the prevalence of positive Doppler signal for active arterial bleeding (not surprisingly 100%), non-bleeding visible vessel (Forrest IIa, 90.7%) and those with adherent clot (Forrest IIb, 68.4%). Interestingly, 40.5% of ulcers with flat haem spots alone (Forrest IIc) which are classically associated with a low risk of rebleeding had a positive arterial Doppler signal, while in clean-based ulcers (Forrest III) only 8.3% had a positive Doppler signal. Repeating Doppler assessment post-standard endoscopic treatment showed a considerable reduction in arterial flow and that persistent arterial inflow was associated with an increased risk of rebleeding. 31
It is interesting to compare these results with those from the original endoscopic Doppler studies. 33 In those Forrest Ia and Ib were not assessed, as they were actively bleeding, it was assumed they must have a significant feeder artery, but arterial inflow was detected in 61% of Forrest IIa, 70% Forrest IIb, 50% of Forrest IIc and 21% of Forrest III. These are very similar to those with the new disposable technology, although further studies particularly on low-risk Forrest III lesions are required.
A subsequent randomised trial compared use of the Doppler probe to both guide the decision to use endoscopic therapy and the success of that therapy against standard haemostatic treatment based purely on endoscopic visualisation. 32 Endoscopic therapy was applied based on the presence of a Doppler signal rather than endoscopic appearance and after endoscopic therapy, the ulcer was reinterrogated and retreatment applied if an arterial signal was still present. Doppler use in this manner was associated with a significant reduction in rebleeding. Rebleeding at 30 days was 8/72 (11.1%) in the Doppler-treated arm compared to 20/76 (26.3%) in the standard care group. Residual arterial signal despite maximal endoscopic therapy (adrenaline, bipolar probe, through-the-scope clips) was strongly associated with rebleeding (8/9 cases, 88.9%) compared to 0/8 (0%) of those that had continued endoscopic retreatment until the Doppler signal was obliterated.
This technique looks extremely promising. The application of the Doppler probe allows more accurate definition of the rebleeding risk of ulcers (it is superior to standard endoscopic stigmata) and facilitates tracing of the underlying artery for direction of haemostatic methods and allows post-treatment reanalysis to define the efficacy of endoscopic therapy. Further studies in other populations with less experienced and committed operators are required before widespread adoption and further data on the efficacy of this advance are awaited with interest. Certainly, Doppler-guided therapy has the potential to dramatically reform the endoscopic approach to PUB. Here, it is important to note that bipolar coagulation is probably the optimal method of primary endoscopic haemostasis with the Doppler probe, clips may be difficult to place over a hidden feeder artery and may also obstruct further Doppler investigation because of their position.
These data with the Doppler probe showing that oozing ulcers (Forrest Ib) are associated with significantly lower risks of rebleeding post-endoscopic therapy31,32 are in keeping with a retrospective analysis of data from one of the large studies of proton-pump inhibitor therapy after endoscopic therapy in PUB 34 and should lead us to reappraise the risks associated with oozing ulcers. After optimal standard endoscopic treatment, in the placebo-treated arm, rebleeding was much lower in those with oozing ulcers (4.9%) than in spurting (Forrest Ia, 22.5%), adherent clot (Forrest IIb, 17.6%) and visible vessel (Forrest IIa, 11.3%). Again supporting the hypothesis that oozing ulcers tend to have smaller feeding vessels and rebleeding rates are lower than previously believed on the basis that this was ‘active bleeding.’ Interestingly, this study also showed that intravenous esomeprazole did not reduce this already low post-endoscopic rebleeding rate in the oozing ulcers (compared to the beneficial effect in the other high-risk stigmata). This suggests that parenteral acid suppression may be withheld from this group after successful haemostasis and standard oral therapy used instead. 34 This reappraisal of the rebleeding rates associated with classical endoscopic stigmata of recent haemorrhage has major implications for the interpretation of existing studies and the design of future studies. Grouping all active bleeding groups (Forrest Ia and Ib) together now seems inappropriate given the clearly divergent rebleeding risks. Certainly, all future studies of drug or endoscopic techniques should stratify effects according to this subdivision of endoscopic stigmata.
The main limitations of the standard endoscopic clips are the relatively small size and the limited pressure the jaw can apply to close tissue and provide mechanical haemostasis. The much larger and stronger over the scope endoscopic clip (OTSC; Ovesco Endoscopy,Tubingen, Germany) overcomes many of these drawbacks. It is able to grasp larger and more fibrotic areas than standard clips and is able to apply more pressure onto feeding arteries. The obvious drawbacks of this are having to preload the clip on the endoscope before intubation, the much greater unit cost and sometimes the difficulty passing the clip through the upper oesophageal sphincter. The OTSC clip is Food and Drug Administration (FDA) approved and available in many areas, being utilised to close fistulas and perforations in addition to acute PUB. Several case-series have reported successful haemostasis with this device when other endoscopic methods have failed. Honegger et al 35 reported 85% success in treating PUB (28/35), although in a smaller case series haemostatic success was only reported in 4/7 cases of refractory bleeding peptic ulcer. 36 The OTSC clip has also been used successfully as primary treatment for PUB: Manno et al 37 reported 100% success in 21 cases. A retrospective review of experience in the quaternary referral practice of the Mayo Clinic, 38 showed an overall success rate of 70.1% (47/67 patients) when used as primary or secondary treatment. Schmidt et al performed an important, albeit small-sized (66 subjects), randomised trial comparing the OTSC with ‘standard’ endoscopic methods for recurrent PUB after initial successful haemostasis. These results looked somewhat more impressive in that persistent bleeding at the time of endoscopy after per-protocol second line haemostasis was significantly lower in the OTSC arm than standard treatment (6.0% vs 14%), although there was no difference in subsequent rebleeding after successful second-line haemostasis, blood transfusion requirements or hospital stay. 39 It is important to note that essentially all the ‘standard’ endoscopic therapy used in this particular study was throughout the scope clips plus epinephrine, with very minimal use of thermal coagulation. Slightly surprisingly for a study focusing on failed initial haemostasis (given the data from the Doppler studies discussed previously) approximately 50% of the study cohort compromised oozing (Forrest Ib) ulcers, where rebleeding is generally much lower. When the OTSC was applied as rescue therapy after failed per-protocol endoclips, haemostasis was secured in 100% of cases. It is also worth noting that no problems with passing the OTSC (even the larger 12 mm OTSC) into position were reported. Once again, this does seem a very promising technique, although the studies have not adequately stratified for the underlying rebleeding rates of the different ulcer phenotypes. However, at present, this does seem to be useful second-line endoscopic technique, although a degree of training is required to use the equipment effectively in the emergency.
Haemostatic powders are in a similar position. These seem a promising technology, but not yet supported by comprehensive randomised trial data. There are now several powders available in various geographical locations, the first available was Hemospray (Cook Medical) but others are now available, including Endoclot (Premier Endoscopy Limited), Purastat (Diagmed Healthcare) and Ankaferd (Ankaferd Drug Inc), although availability of the different products varies in different countries. For example, Hemospray has only recently been approved for use in the United States (7 May 2018). These are proprietary mineral, amino acid, herbal or polysaccharide preparations that when sprayed onto a bleeding area through a cannula inserted through the channel of an endoscope provoke rapid haemostasis. The powders appear acts both as a physical barrier by polymerising upon contact with moisture and a powerful procoagulant by concentrating clotting factors at the site of application. There are no randomised trials, but several case-series showing successful haemostasis after failure of first-line endoscopic therapies showing that several of these powders may be usefully employed in those most difficult refractory bleeding ulcers.
In a comprehensive literature review of reported cases, Hemospray was successful in 88% success in 81 cases of bleeding peptic ulcers. 40 A further review showed Hemospray to be effective in 88% of a total of 234 GI bleeding cases of all causes (the majority of use as primary [83%] rather than rescue therapy (17%)). However, the overall rebleeding rate was relatively high at 16.2% within 72 hours, and there are a paucity of data specifically referring to PUB. 41 Even within the 81 cases of PUB reported, the majority were at the lower-risk end of the spectrum (62% Forrest Ib) and the true clinical efficacy of Hemospray still needs to be defined.
Obviously, this method provides no obliteration of the underlying artery (as clips or bipolar probe do) and the rate of rebleeding and the subsequent natural history of PUB bleeding in this context are unknown. Haemostatic powders do not influence the underlying arterial inflow and by obscuring the view and access conceivably may reduce definitive haemostasis. At present, it is unclear if rebleeding rates with highest-risk stigmata (spurting arteries or those with significant positive Doppler traces) treated by Hemospray are clinically problematical. The powder application invariably obscures the endoscopic view, and perhaps repeat second-look endoscopy will be required to perform more secure haemostasis. The FDA release accompanying the approval of Hemosray in the United States includes data on the success (95% initial haemostasis) but also warnings about the risk of rebleeding (quoting a figure of 20% at 30 days). 42 At present, this cannot be regarded as a routine first-line therapy but in some cases can be extremely useful when other methods have failed. The technique is relatively easy, although care must be taken to avoid premature exposure to the powder to liquid, which activates the powder. However, because the spray does not need to be as precisely targeted as clips or bipolar probing, it may offer some additional advantages when access is limited, such as posterior duodenal ulcers or proximal lesser curve ulcers where is it difficult to obtain direct approach to the lesion. In the author’s experience, applying this in a duodenal cap with a rapidly bleeding ulcer is often quite difficult, but it can be applied for bleeding lesions when other methods are technically impossible or have failed and may provide rescue haemostasis in those cases. It is important to note that the requirement for blood or liquid for effective activation often reduces the effective use of haemostatic powders on non-bleeding but protuberant arteries (Forest IIa lesions) which do merit some form of endoscopic therapy.
Most of the published data about powders concerns Hemospray and given the very different properties of the different preparations, it would be incorrect to extrapolate between them. Data are even more preliminary for the other forms of haemostatic powders. Ankaferd was successful in stopping bleeding from seven cases of upper GI cancer 43 and was reported to be effective in 26/30 cases of upper GI bleeding of various causes, although the report does not provide sufficient data about the risk stratification of the bleeding peptic ulcers included. 44 A further small study examined the use of Ankaferd as the primary method of endoscopic haemostasis for bleeding Forrest Ia and Ib lesions (although the relative distributions of the two was not given) and initial success was seen in 19/26 (73%) with a rebleeding rate of 15.8%. 45 Interestingly, other more standard endoscopic methods could be immediately applied after failed Ankaferd application and these secured haemostasis in all cases.
Purastat has been mainly used this far as an adjunct to prevent rebleeding after endoscopic resections such as endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD) 46 and as it has not been reported to control active arterial haemorrhage it cannot be recommended at present as either a primary or rescue therapy for actively bleeding peptic ulcer. Further studies examining its role as an adjunct after other forms of endoscopic therapies to prevent rebleeding are awaited with interest, Endoclot is perhaps more promising. A preliminary study of its use as rescue therapy 21 cases of upper GI bleeding refractory to treatment with bipolar probe or standard through-the scope clips reported immediate haemostatic success in all cases (100%) and a rebleeding rate of 4.8%. It should be noted that the majority of ulcers (76%) treated were oozing (Forrest Ib) and the efficacy in higher-risk contexts remains to be explored. 47 Further studies reporting the different haemostatic powders in relation to more standard haemostatic methods and in ulcers with different bleeding stigmata will help refine the place of the powders in management (Table 2).
Haemostatic powders available. Not all preparations are available in all territories. References as cited in main text.

Although endoscopic therapy is effective, rebleeding remains an important problem is a small subset of patients. The potential role of interventional radiology as an adjunct to endoscopic therapy continues to be explored. Lau et al performed a well-designed randomised trial comparing radiological embolisation with standard high-quality management in patients after successful endoscopic haemostasis. Patients with high-risk ulcers based on clinical criteria (size >20 mm, shock on presentation, spurting ulcer or haemoglobin <90 g/L on presentation). Overall, there was no significant effect of adjunctive embolisation (in the intention to treat analysis, rebleeding occurred in 10.2% of the embolisation group and 11.4% of the control group) and although transfusion requirements were lower in the embolisation group, other clinical outcomes were equivalent. While this study shows that routine post-endoscopy embolisation is not indicated after successful haemostasis, post hoc analysis of the study does show some interesting effects that should be followed up by more focused studies. Embolisation did seem to reduce rebleeding in ulcers >15 mm and half of the rebleeding in the embolisation group occurred in the 18% of the group randomised to active intervention arm that did not receive effective embolisation. With per protocol analysis the hazard ratio for rebleeding without embolisation was 1.89 (95% CI: 0.73-4.92). 48
Thus, there are an exciting array of tools becoming available to assist the assessment and endoscopic treatment of PUB. Further studies are required to define how best we should use these together.
Drug Therapy
Pre-endoscopy proton pump inhibitor (PPI) infusion is recommended by some guidelines but not universally. 15 Although this treatment seems to downstage the endoscopic appearance of bleeding ulcers, the effect on objective clinical end-points such as rebleeding or hospital stay is debateable. In contrast, post-endoscopy PPI treatment after endoscopic therapy to high-risk ulcers has been shown repeatedly to be better than placebo in reducing rebleeding and surgery. 49 However, despite the multitude of studies in this area, the optimal PPI regimen is unclear. Many clinicians use the original ‘Hong-Kong’ regimen (bolus followed by continuous infusion of omeprazole, pantoprazole or esomeprazole) for 72 hours. Other dose regimens have been shown to be effective, including intermittent parenteral dosing and even high-dose oral PPI, and it is not clear what the optimal regimen is50,51 (for full details of the many different regimens studied please refer to the meta-analyses cited). As previously discussed, the rebleeding rate after successful endoscopic haemostasis in oozing (Forrest Ib) ulcers is low and does not seem to be reduced by parenteral high-dose acid suppression and hence treatment may be rationalised in those patients to standard oral PPI therapy. 34
The Rockall score can be used to focus the use of post-procedure PPI. Cheng et al 52 showed that 11 days high dose oral esomeprazole (40 mg twice daily), started after endoscopic therapy and 72 hours iv PPI was superior to once daily esomeprazole 40 mg (with 40 mg once daily subsequently for both groups) in preventing rebleeding (10.8% vs 28.7% in the 4-28 days post-index bleed) in patients with a full Rockall score of 6 or more. There was no significance difference in mortality, hospital stay or blood transfused. Thus, there is rationale for treating the higher risk patients (Rockall 6 or more) with higher dose PPI for the period after initial stabilisation.
There is sound plausibility and evidence for using prokinetics before endoscopy in upper GI bleeding to clear the stomach and both improve the endoscopic views and probably improve safety. While individual underpowered trials have shown inconsistent results, several recent meta-analyses, including up to 8 trials (598 subjects), showed that intravenous erythromycin before endoscopy was associated with meaningful clinical benefit in terms of reduction in need for repeat endoscopy (by about 50%), improved mucosal visualisation, as well as length of stay, while metoclopramide seemed to be less effective.53-56 No effect on mortality or utilisation of endoscopic therapy was reported and the effect of blood transfusion was inconsistent and not significant. 56 Erythromycin is probably underused and seems to be a simple intervention that would improve outcomes. The optimal erythromycin regimen is unclear, trials have used doses ranging from 125 mg to 4 mg/kg, given over 5-30 minutes and starting the gastroscopy 20-90 minutes after completing the erythromycin. 56
Tranexamic acid, used to prevent fibrinolysis of a clot overlying an artery is an attractive, simple pre-endoscopy treatment. Tavakoli et al 57 randomised 410 patients presenting with signs suggestive of PUB to pre-endoscopy treatment with either intravenous tranexamic acid, intravenous plus nasogastric tranexamic acid or saline placebos. There was no significant difference in any clinically meaningful end-point (rebleeding, mortality, and need for endoscopic intervention). However, there were no thromboembolic complications reported in the first week after treatment. The study population was of generally low-risk PUB (80% clean-based ulcers) and was underpowered and inappropriately designed to show a clinical effect of tranexamic acid on the more significant clinical outcomes. Results of the much larger worldwide HALT-IT study should shed more light on the role of tranexamic acid. 58
The management of concurrent anticoagulation is an increasing problem for all of those involved in the care of acute PUB. The use of prothrombin complex concentrate to reverse the anticoagulation effects of vitamin K antagonists (VKAs) such as warfarin is well-established. 15 The directly acting anticoagulants (DOACs), the thrombin antagonist dabigatran and the factor X inhibitors apixaban, rivaroxaban and eduxoban present more of a conundrum. Due to renal clearance of these drugs, the anticoagulation effect declines relatively rapidly. However, in some patients, life-threatening bleeding will require anticoagulation reversal. The first specific reversal agent for dabigatran (idarucizumab) has just been licenced and available for clinical use. Although it is expensive, it should be available to treat significant dabigatran-associated PUB. Idarucizumab is a monoclonal antibody against dabigatran and will not reverse the other DOACs.59,60 There are relatively few data concerning the treatment of upper GI bleeding associated with these agents and although some guidelines have been produced, 61 these have not been informed by comprehensive studies. Widespread use of tranexamic acid is not routinely indicated in PUB 62 (although the results of the large worldwide HALT-IT trial are awaited with interest 58 ), but in this particular situation of DOAC-induced PUB, the use of tranexamic acid seems reasonable, albeit not supported by convincing evidence. 63 Prothrombin complex concentrate seems to reverse the anticoagulation effect of factor X inhibitors in healthy volunteers and should probably be considered in severe life-threatening bleeding. Although there are really no data specifically showing an effect in PUB.61,64 A specific antidote to factor Xa inhibitors has been developed and has recently been shown to rapidly reverse the anticoagulant effect and hopefully will be available for clinical use soon. 65 Further studies are needed to better define the management strategy for anticoagulated patients, especially those taking DOACs in terms of timing of endoscopy, application of pharmacological reversal strategies and the specifics of endoscopic therapies. At present, it would be wise to resuscitate and support such patients and risk stratify as per standard policies. It will be important to establish whether different forms of endoscopic therapies, which cause different degrees of tissue damage (thermal methods versus clips) are preferable in this situation.
Acute upper gastrointestinal bleeding is a significant drain on the body’s iron stores and many patients are anaemic after completing initial management. A randomised trial compared subsequent management strategies in this group with non-variceal upper GI bleeding (mostly peptic ulcer related). Iron therapy, either a one-off dose of intravenous ferric carboxymaltose or oral ferrous sulphate 200 mg daily, was more effective than placebo at restoring haemoglobin levels to normal. After 12 weeks, 17% of iron-treated patients were still anaemic compared to 70% of placebo-treated. 66 There was no difference in the rates of improvement in anaemia between parenteral and enteral iron groups, although higher ferritin levels were seen in the parenteral group. 66 There was no significant toxicity, and it seems logical supplemental iron therapy should be used in those patients with anaemia at the cessation of the peptic ulcer bleed.
Management of Refractory Bleeding
Despite advances in endoscopic and pharmacological therapies, a significant minority of patients experience significant rebleeding. Surgery has traditionally been regarded as the most appropriate approach. Increasingly interventional radiology is been seen and often regarded as the initial therapeutic approach before surgery. There are no randomised trials to guide therapy, and it seems unlikely any will be performed. Although not supported by trial data, endoclips can be placed to mark the site of bleeding, in those deemed at highest risk of needing embolisation (most obviously in those with failed endoscopic haemostasis, but in also in those with technically difficult but successful haemostasis) to facilitate subsequent radiological localisation and embolisation. If this is available locally.
A number of case series have reported high technical success and acceptable complication rates with radiological embolisation for acute PUB,67-69 and it is generally believed that the overall safety of interventional radiological is significantly better than surgery. Hence, most guidelines now advocate radiological embolisation as the rescue therapy of choice. However, not all studies are concordant with this: single centre observational studies and a meta-analysis have suggested that rates of rebleeding are higher following embolisation than surgery and mortality rates equivalent.70,71 All such studies are somewhat difficult to interpret because of case mix: as patients undergoing embolisation tended to be older with more co-morbidities. Where the appropriate skills in interventional radiology are available, embolisation does seem to provide an effective rescue tool, but as with many other decisions in these patients, clinical management should be individualised.
Follow-up and Prevention
An understanding of the major causes of PUB naturally leads into developing strategies for both primary and secondary prevention. The major and easily ameliorable causes of are PUB are
Strategies to deal with
For many years, it has been documented that the sensitivity of all endoscopy-based
Although aspirin and other antiplatelet agents are clearly associated with an increased risk of PUB, in many cases these agents are indicated because of the underlying vascular disease, and it is now accepted that where indicated aspirin should be continued (or interrupted for a minimal interval of < 3 days) in acute PUB. 73 A small risk in early rebleeding is more than compensated by a significantly reduced risk of vascular events and death. This approach is supported by data from both a relatively small clinical trial and observational follow-up data.77,78
A further non-randomised case-control study from Japan 79 compared outcomes from patients with PUB that were taking antithrombotic medications, which were continued throughout with those from patients without any anti-thrombotic drug exposure. Overall, in 346 patients, continuing anti-thrombotic treatment, which was mainly anti-platelet therapies, was associated with a significantly increased risk of rebleeding (13.9% vs 5.8% within 30 days), but there was no difference in mortality or other outcomes. In this study, collection of data on post-bleed thrombotic complications was not rigorous. The endoscopic therapies applied, the majority either argon plasma coagulation or injection therapies alone, were probably suboptimal compared to other studies. The exact proportion of Forrest Ib ulcers, which as discussed previously have a relatively low rebleeding rate, were not provided. Thus, generalisation of these finding is slightly difficult, but overall, these data are consistent with the previous studies suggesting that continuation of anti-platelet therapies is usually indicated in PUB.
The most appropriate treatment after an aspirin-induced bleed is aspirin plus a PPI, 80 this is superior to the P2Y12 antagonist clopidogrel alone as secondary treatment. There are less data on the newer P2Y12 antagonists ticagrelor and prasugrel, but these are more potent anti-platelet agents and not surprisingly the risk of GI bleeding seems to be higher than aspirin or clopidogrel.81,82 Follow-up of a large non-randomised cohort (11 955 patients) after myocardial infarction treated with percutaneous intervention, where PPI-use (about 18% overall) was at the physicians’ discretion, showed that while those treated with a PPI had a higher rate of adverse cardiovascular outcomes (hazard ratio 1.38 (95% CI: 1.21-1.58)), this was similar between clopidogrel- and prasugrel-treated patients 83 and suggests that this association is due to confounding by indication with higher risk patients receiving gastroprotection and not due to a significant effect of PPIs on clopidogrel pharmacokinetics. 73 The rates of clinically significant bleeding were not significantly different between PPI-treated clopidogrel and prasugrel patients, although interestingly were numerically lower in the prasugrel group (1.9% vs 4.6%), and this may reflect underlying confounding in this non-randomised study
Patients with drug-eluting coronary artery stents do need to continue dual antiplatelet therapy for a year, PPI co-treatment reduces bleeding in those taking aspirin plus clopidogrel. 84 Further subgroup analysis of the COGENT trial confirmed that omeprazole co-treatment with aspirin-clopidogrel therapy is similarly effective at reducing GI bleeding in the two main groups receiving dual-therapy: post-acute myocardial infarction (upper GI bleeding at 180 days, placebo-treated 1.21%, omeprazole-treated 0.24%) and after elective percutaneous intervention (0.73% and 0.23% respectively). 7 Importantly, omeprazole-treatment was not associated with increased adverse cardiovascular outcomes in any subgroup. Although the COGENT study was prematurely terminated due to funding difficulties, these data are very reassuring as to the efficacy and safety of omeprazole in this situation.
These most recent data do again confirm the clinical safety and efficacy of PPI co-treatment with clopidogrel-based anti-thrombotic regimens. 85 Although there is undoubtedly a pharmacokinetic interaction, in that several PPIs, most noticeably omeprazole, do inhibit the CYPP450 mediated biotransformation and activation of clopidogrel, this is not translated into a reduction in clinical efficacy. 73 A further detailed study on these interactions showed that four different PPIs (omeprazole, esomeprazole, lansoprazole and rabeprazole (pantoprazole was not tested), all had the potential to reduce the anti-platelet activity of clopidogrel; however, no effect was seen if rabeprazole was taken 4 hours after the clopidogrel. 86
The relative benefits and risks of aspirin plus PPI versus clopidogrel plus PPI after an aspirin-induced PUB are not fully defined. However, observational data suggest that aspirin is safer, more effective and preferable.87,88 An interesting effect of the increasing use of gastro-protection with anti-platelet agents is that the incidence of upper GI bleeding has decreased, but as might be expected, the incidence of lower GI bleeding has remained stable, so that in patients taking dual antiplatelet agents with PPI cover the risk of lower GI bleeding is approximately three times higher than upper GI bleeding. 89
In general, PPI co-treatment has been advocated with aspirin for primary and secondary prevention. This has been outlined in existing guidelines from the American Heart Association and the American College of Gastroenterology, although these are over 10 years old now, they are still generally applicable. 90 While all cases of previous PUB are high-risk and warrant PPI-secondary prevention. Primary prevention needs can be stratified according to the number other risk factors (including age, other antithrombotics, selective serotonin reuptake inhibitors) and primary prevention focused on those are highest risk. 73 A recent study looking at secondary prevention in a Chinese population showed that famotidine was equivalent to rabeprazole. 91 Previous data suggested that PPI treatment was better, 92 and until more data in wider populations are available, PPI treatment continues to remain the treatment of choice.
The importance of primary prevention of PUB was highlighted in a population-based study from the United Kingdom. Follow-up of over 3000 patients starting treatment with an anti-platelet agent (96% aspirin) without any specific gastroprotection protocol over 10 years (it is worth noting that choice of this was at the prescriber’s discretion and only about 25% received gastroprotection) confirmed that the risk of acute upper GI bleeding increased substantially with age (age > 75, hazard ratio [HR]: 4.13 (95% CI: 2.60-6.57) but importantly demonstrated that these bleeding events in this most elderly group were far from benign in outcome. The majority of PUBs (62%) in the over 75 age-group were fatal or associated with major subsequent disability 6 Estimates from these data suggest that in those patients over 85, the number-needed-to-treat with a PPI to prevent an aspirin-induced PUB is as low as 25. In comparison, it may be as high as 338 in subjects under 65. 6
Similar to the case with antiplatelet agents, it is now becoming apparent that where indicated early resumption of anticoagulation for atrial fibrillation after a PUB is beneficial. The increased risk of rebleeding is compensated by reductions in stroke and death. The exact optimal time for reintroduction of anticoagulation after a PUB is unclear. It appears that leaving reintroduction for 3 months imposes an excessive risk of thrombotic events,93,94 while very early reintroduction does increase the rebleeding risk and a compromise of reintroduction after 7-15 days probably seems to provide the optimal reduction in thromboembolic events compared to rebleeding.95,96 Data in support of this come from Qureshi et al, 97 who reported on outcomes in 1329 patients with atrial fibrillation and a warfarin-associated significant gastrointestinal haemorrhage (about 50% were from an upper GI source). Restarting warfarin was clearly associated with better outcomes: reduced thromboembolism (HR: 0.71; 95% CI: 0.54-0.93) and mortality (HR: 0.67; 95% CI: 0.54-0.93) but no effect on recurrent bleeding (HR: 1.120; 95% CI: 0.78-1.86). An analysis by time of reintroduction of warfarin showed that rebleeding was higher if restarted less than 7 days after index bleeding and thromboembolism was higher if restarted after 30 days. Reintroduction between 7 and 15 days seemed to provide optimal efficacy. More studies specifically examining this effect are required, particularly focused on PUB.
Previous studies have confirmed that selective COX-2 inhibitors are safer than traditional non-selective NSAIDs in terms of gastrointestinal complications. 73 The combination of celecoxib plus a PPI is associated with the lowest risk of rebleeding after an NSAID-induced PUB, when reintroduction of anti-inflammatory therapy is required. 73 However, concerns about the cardiovascular safety of COX-2 inhibitors lead to many clinicians being reluctant to prescribe these. The publication of recent data has allowed an important reappraisal of the role and risk associated with COX-2 inhibitors, particularly celecoxib. 98 A large observational study of real-world practice showed that the risk of myocardial infarction appeared to be increased with all cyclo-oxygenase inhibitors and the risk associated with naproxen (generally thought to be the safest cardiovascular NSAID) was equivalent to other NSAID and that the increase in risk with celecoxib was also comparable other agents. 99 The risk with the now withdrawn COX-2 inhibitor rofecoxib was higher. Subsequently, a updated meta-analysis of outcomes from randomised trials and observational studies of cyclo-oxygenase inhibitors showed that rofecoxib appeared to be an outlier with higher risks of myocardial infarction, stroke and combined cardiovascular outcomes that placebo, other NSAIDs or other COX-2 inhibitors. 100 Celecoxib was not associated with any increased risk of any cardiovascular endpoint compared separately to placebo, NSAIDs or COX-2 inhibitors and in fact seemed to be associated with a lower risk of stroke compared to other non-specific NSAIDs. There seemed to be no correlation between the COX-2 selectivity of an agent and cardiovascular risks. 100
Subsequently, a recent, large randomised study powered specifically to look at cardiovascular rather than gastrointestinal outcomes showed no inferiority of celecoxib compared to either naproxen or ibuprofen (all combined with esomeprazole) and cardioprotective doses of aspirin as indicated. The PRECISON trial included over 24 000 subjects with osteoarthritis or rheumatoid arthritis and increased cardiovascular risk, with mean follow-up of 20 months. Overall, cardiovascular adverse outcome rates were comparable (celecoxib 2.3%, naproxen 2.5% and ibuprofen 2.7%), but celecoxib was associated with a significantly lower incidence of GI side effects (composite GI end-point celecoxib 1.1%, naproxen 1.5%, ibuprofen 1.6%). 101 Subsequently, further detailed analysis of the gastrointestinal effects of PRECISON have confirmed the overall superior safety of celecoxib, with significantly lower total clinically significant gastrointestinal events even in those also treated with aspirin and corticosteroids. The number of the individual complications such symptomatic ulcer or ulcer bleeding were low throughout the study and although numerically in favour of celecoxib, failed to show statistical significance. 102 This suggests that when clinically indicated celecoxib (in a dose up to 200 mg daily) plus a full-dose PPI should not be withheld from patients requiring treatment after a NSAID-induced bleed. 98
Although these data on the relative cardiovascular safety of commonly used COX-inhibitors are very reassuring, the missing piece of the puzzle has always been which is the most appropriate anti-inflammatory agent to use with low-dose aspirin to prevent GI bleeding and yet maintain cardiovascular safety. 73 Initial studies showed that as might be expected adding aspirin to a COX-2 inhibitor ameliorated much of the GI safety of COX-2 inhibitor compared to a non-specific NSAID, 103 however that end point was based predominantly on endoscopically detected ulcers in average-risk patients and not complications such as bleeding.
An important trial (CONCERN) has now demonstrated that in the highest risk patients, those with a combined aspirin-NSAID induced peptic ulcer bleed, who need to continue both therapies, celecoxib plus aspirin plus a PPI is the optimal strategy. 104 The Hong Kong group randomised 514 subjects to aspirin plus esomeprazole plus either celecoxib or naproxen. After 18 months follow-up, recurrent PUB was significantly lower in the celecoxib group (5.6%) than the naproxen group (12.3%). There was no difference in cardiovascular outcomes, although these were numerically less common in the naproxen arm.
PPI co-treatment would be usually recommended after a PUB and primary prevention of PUB in higher risk patients taking anticoagulants is usually advocated by some guidelines but not all.73,105–108 Surprisingly there are little data specifically to support this practice. A recent study showed that omeprazole co-treatment did reduce rebleeding in warfarin-treated patients, although this effect was only apparent in those also taking anti-platelet drugs or NSAIDs. 109 A further observational study showed that concurrent use of PPIs or H2-receptor antagonists with anticoagulants were both associated with a reduced risk of acute upper GI bleeding, and this effect was most marked in those with a history of peptic ulcer disease. 110 Therefore, despite the relative lack of evidence, co-prescription of gastroprotection with anticoagulant therapy would seem to be indicated in secondary prevention, for primary prevention, a case- and risk-based approach seems sensible pending further data. Those with highest risk of bleeding are most likely to gain from use of acid suppression.
The rate of GI bleeding with DOACs compared to VKAs remains controversial. While overall bleeding rates associated with DOAC use at all sites combined do seem to be lower, this is mainly driven by a reduction in cerebral bleeding and the rate of GI bleeding may actually be increased but certainly does not seem convincingly lower. Several studies have shown either a higher rate of GI bleeding with DOACs or equivalent rates to VKAs.111–114 The increased GI bleeding risk of DOACS seems especially marked in the most elderly (aged over 76).
This effect on GI bleeding may not be unexpected, as most of the DOACs are taken as active drugs (in contrasts to VKAs) and relatively higher levels and local anticoagulant effects may be seen at the level of the gastrointestinal mucosa. The choice of which, if any, anticoagulant to reintroduce after a PUB currently needs to be made on an individual basis taking into account underlying risks and comorbidity, availability of antidotes and patient preferences. The scoring systems for thrombotic risks in atrial fibrillation (CHADS2-Vasc) and bleeding risk (HAS-BLED) can give important guidance on the relative risks of these important outcomes; 115 however, there are no data specifically related to using these scores prospectively to manage patients influences outcomes.
An increasing number of studies from disparate geographical regions have shown an important increase in apparently idiopathic peptic ulcers as the cause of upper GI bleeding. This was initially in the Far East, but further studies from the United Kingdom and Europe have confirmed this. These ‘ideopathic’ ulcers can contribute up to 40% of ulcers in some series and an even higher proportion of the bleeding peptic ulcers occurring in hospital inpatients.116–119 Although labelled idiopathic, they are typically seen in the more elderly population with significant comorbidity and these ulcers may represent a marker of systemic vascular pathology. The importance of recognising this group lies not only in determining secondary preventive management of PUB by cause (
Conclusions
Acute PUB remains an important clinical problem, but the management continues to be refined. Important recent developments that can be incorporated into practice include the confirmation of the usefulness of the GBS in determining the lowest risk patients, who may be safely managed as outpatients. The Glasgow-Blatchford is most useful for predicting the need for intervention but another score either the Rockall Score or AIMS65 should also be assessed as a predictor of mortality to aid assessment of outcomes. The abbreviated GBS may prove easier to use clinically, and just as accurate, if supported by further data.
A restrictive transfusion threshold with a trigger of 70 g/L and a target of 70-90 g/L is appropriate for most patients. Pre-endoscopy erythromycin is probably under used, and recent evidence supports wider use to improve endoscopic views. The addition of high dose oral esomeprazole after initial proton pump therapy in high-risk cases (Rockall scores of 6 and above) seems to have a useful benefit in reducing rebleeding. For endoscopic treatment, the haemostatic powders and over the scope clips are useful tools when standard modalities are ineffective or impractical. The new Doppler equipment and disposable endoscopic Doppler probe appear to be very promising in more accurately predicting ulcer rebleeding and the success of endoscopic therapy and are likely to be widely used if further studies are confirmatory.
Oral iron should be prescribed to anaemic patients after a PUB episode to enhance normalisation of the haemoglobin level. Strategies to manage the bleeding episode and prevent rebleeding, need to include the management of comorbidities and risks and in at present must include the management of bleeding associated with all antithrombotic agents, but particularly the DOACs. Specific antidotes are being developed and are just entering the clinical arena. Recent data suggest that the cardiovascular risk of celecoxib is not excessive compared to standard NSAIDs and when used appropriately celecoxib should once again be a useful tool in the primary and secondary prevention of PUB. Non-
Footnotes
Declaration of conflicting interest:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
ILPB conceived the concept of the manuscript, performed the literature search and literature review, wrote all drafts of the manuscript and is the guarantor of the paper.
