Abstract
Objective:
The objectives of this study are, first, to measure concordance between 5 different renal function estimates (methods) in terms of recommended drug doses, and, subsequently, to establish the potential for significant clinical differences between Cockroft–Gault (CG) and Modification of Diet in Renal Disease (MDRD) equations in dosing a specific medication, namely, meropenem.
Design and setting:
This study used a Monte Carlo simulation, and this is a computer–based study with no actual patient data.
Patients:
A total of 1200 and 8701 simulated cases to study the concordance for the 5 methods and the potential clinical significance of discordance between CG and MDRD, respectively, were chosen for the study.
Methods:
Simulated factors were age, sex, height, weight, serum creatinine, ethnicity, and albumin. We estimated the renal function using 5 formulas (ie, 10 combinations) including CG, MDRD, and Chronic Kidney Disease Epidemiology Collaboration (CKD–EPI). Next, the team evaluated concordance for each combination in dosing 22 drugs. Finally, our researchers reviewed and simulated data from the literature to show how CG versus MDRD use can result in clinically significant differences for meropenem.
Results:
Pairwise combinations yielded statistically significant differences (P < .0001) except for CG and MDRD (P < .5147). In addition, the highest concordance was for MDRD and CKD–EPI. Average discordance is in the range of 25% to 30% with the lowest being between CG and albumin–based estimates. Both CG and MDRD were largely discordant which can reach up to 40% with a drug like meropenem and may be associated with significant adverse outcomes.
Conclusions:
Both CG and MDRD in our simulation are statistically comparable. Clinically, nonetheless, they are significantly inconsistent in terms of recommended drug dosing. We encourage practical comparisons of outcomes for individual or groups of medications (eg, meropenem and antibiotics) empirically dosed in renal patients on the basis of equations used in distinct populations.
Keywords
Introduction
Chronic kidney disease (CKD) alters the concentrations and effects of various drugs and, as a result, may increase the risks of toxicity as well as treatment failure.1–6 In response, clinicians use multiple equations to estimate renal function and select doses of medications in such patients.2–4 Previous studies, however, show that up to 77% of medicines prescribed in this population were unadjusted.7–16 Almost two–thirds of these improper amounts potentially bear adverse consequences and about 3% may, in fact, result in severe outcomes and even death. 17 However, it is unclear which equation is best for estimation and assessment of the appropriateness of the doses selected.18–20 Moreover, the list of consequences associated with dosage regimen optimization in these patients is endless. 21 Clinicians need to see both, an explicit superiority example of one estimate over another and a model for evaluating this difference.
Variability in the pharmacokinetics and pharmacodynamics of drugs in patients with CKD, and on that account required dosing, is more prominent for certain drugs.1,3 For example, previous research indicates a vast baseline interindividual differences in the levels of meropenem in healthy subjects. 22 At the very outset, recommended doses of this antibiotic based on any estimate of kidney function would likely produce a broad range in blood concentrations. It is not surprising, then, that empiric dosing of this carbapenem is a considerable challenge with great adverse outcomes in various patient populations.23–36 In a pilot simulation, we identified a large discordance in its recommended doses by Cockroft–Gault (CG) and those given by Modification of Diet in Renal Disease (MDRD). Hence, meropenem seems to be a superb choice to investigate the potential superiority of one renal function estimate over another.
Consequently, our aim in this study is 2–fold: first, to simulate the drug–dosing mismatch of the different equations for commonly used medications in patients with CKD and, second, to demonstrate the potential clinical differences in empiric treatment with CG versus MDRD using meropenem. We would then qualify to propose a mechanism for evaluating the clinical outcomes associated with the use of the various methods with specific or groups of medicines.
Patients and Methods
We have completed 12 simulations of 100 patients each using Excel (Microsoft Office Professional Plus 2013). Tables 1 and 2 summarize the methods used in the simulation of patient variables and estimation of creatinine clearance (CrCl), respectively. For the drug dosing mismatch part, statistical assumptions are unnecessary because our aim is to detect the subpopulations where differences exist. On that account, we did not bind the data to any distributions. Concisely, our team produced a population of cases which contain all possible combinations. Categorical variables including age, sex, and ethnicity consisted of equal numbers in each group. In contrast, continuous variables spanned random ranges covering all patients of interest. For example, for serum creatinine, CKD stages 3, 4, and 5 are of primary importance. Therefore, our formula was a random fraction multiplied by 10 and then adding 1. This gives a range between 1 and 11 with progressively more cases in stages 3, 4, and 5. In terms of the normalized versions of renal function equations, we incorporated the adjusted MDRD as a representative. Be that as it may, normalization is unlikely to change the study findings and theme of variation of empiric dosing. Actual kidney function measurement with urine collection is also irrelevant as this is practically unused in most cases in practice. Excel sheets automatically generated each simulation by clicking a key. Once generated, all data were copied in a separate sheet for further analysis. Our study evaluated 22 medications commonly used in most hospitals in practice and which need a dose adjustment in patients with CKD. These medications were as follows: Acyclovir, allopurinol, apixaban, atenolol, amoxicillin/clavulanate, cefazolin, cefepime, digoxin, disopyramide, enoxaparin, eptifibatide, ranitidine, gabapentin, lamivudine, levofloxacin, meropenem, metformin, sitagliptin, sotalol, piperacillin/tazobactam, tenofovir, and tramadol. Well–known references such as Lexicomp and manufacturer inserts guided us for the dose tiers or recommendation cutoff values (Table 3). Next, data analyst matched the tiers with calculated CrCl or estimated glomerular filtration rate (eGFR) for each medication. Then, she generated binary codes with 0 denoting discordant and 1 meaning concordant for the 10 possible combinations. Conclusively, our team calculated the percentage concordance for each medication and couple of estimates. We have also evaluated the effect of sex, age, weight, CKD stage, and albumin on the concordance of the 3 most important pairs taking meropenem as a candidate. These were the CG with MDRD, Chronic Kidney Disease Epidemiology Collaboration (CKD–EPI), or albumin–based equations. The latter is usually used in patients with muscular atrophy or disease but they generally provide an alternative method of calculating CrCl. We used CKD–EPI estimates to determine the CKD stages.
Simulation methods.
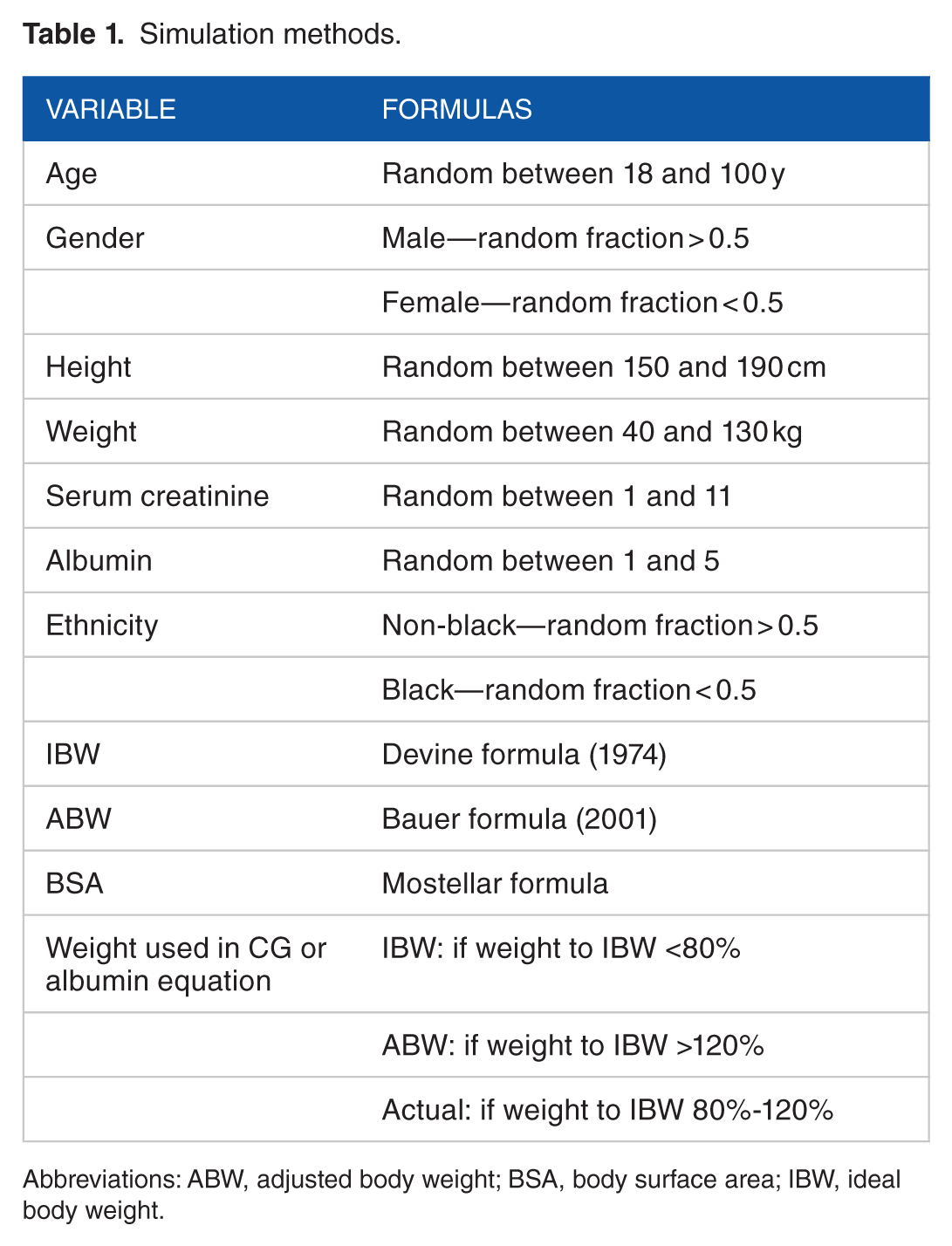
Abbreviations: ABW, adjusted body weight; BSA, body surface area; IBW, ideal body weight.
Five methods or equations for estimation of renal function.
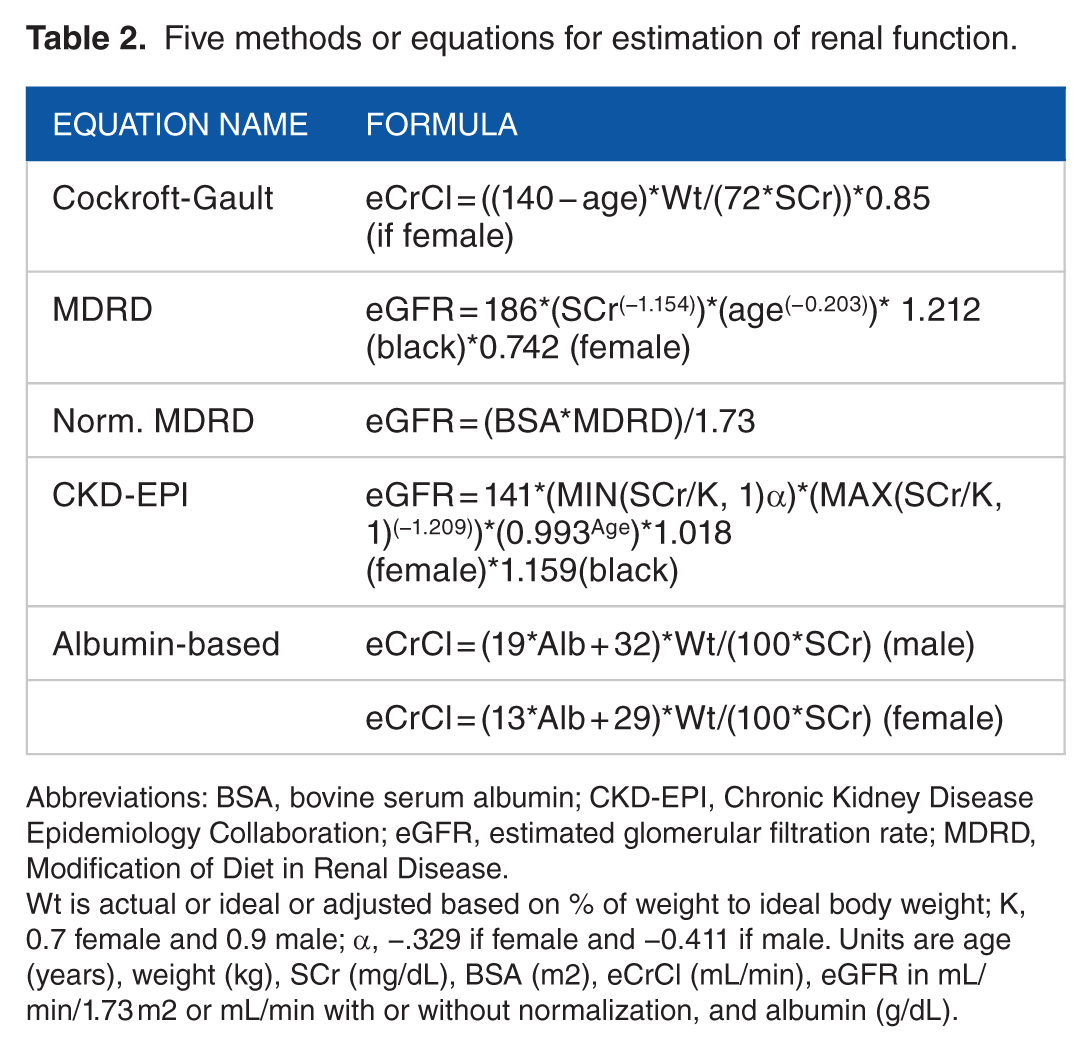
Abbreviations: BSA, bovine serum albumin; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; eGFR, estimated glomerular filtration rate; MDRD, Modification of Diet in Renal Disease.
Wt is actual or ideal or adjusted based on % of weight to ideal body weight; K, 0.7 female and 0.9 male; α, −.329 if female and −0.411 if male. Units are age (years), weight (kg), SCr (mg/dL), BSA (m2), eCrCl (mL/min), eGFR in mL/min/1.73 m2 or mL/min with or without normalization, and albumin (g/dL).
Tiers of renal dosing for 22 medications.
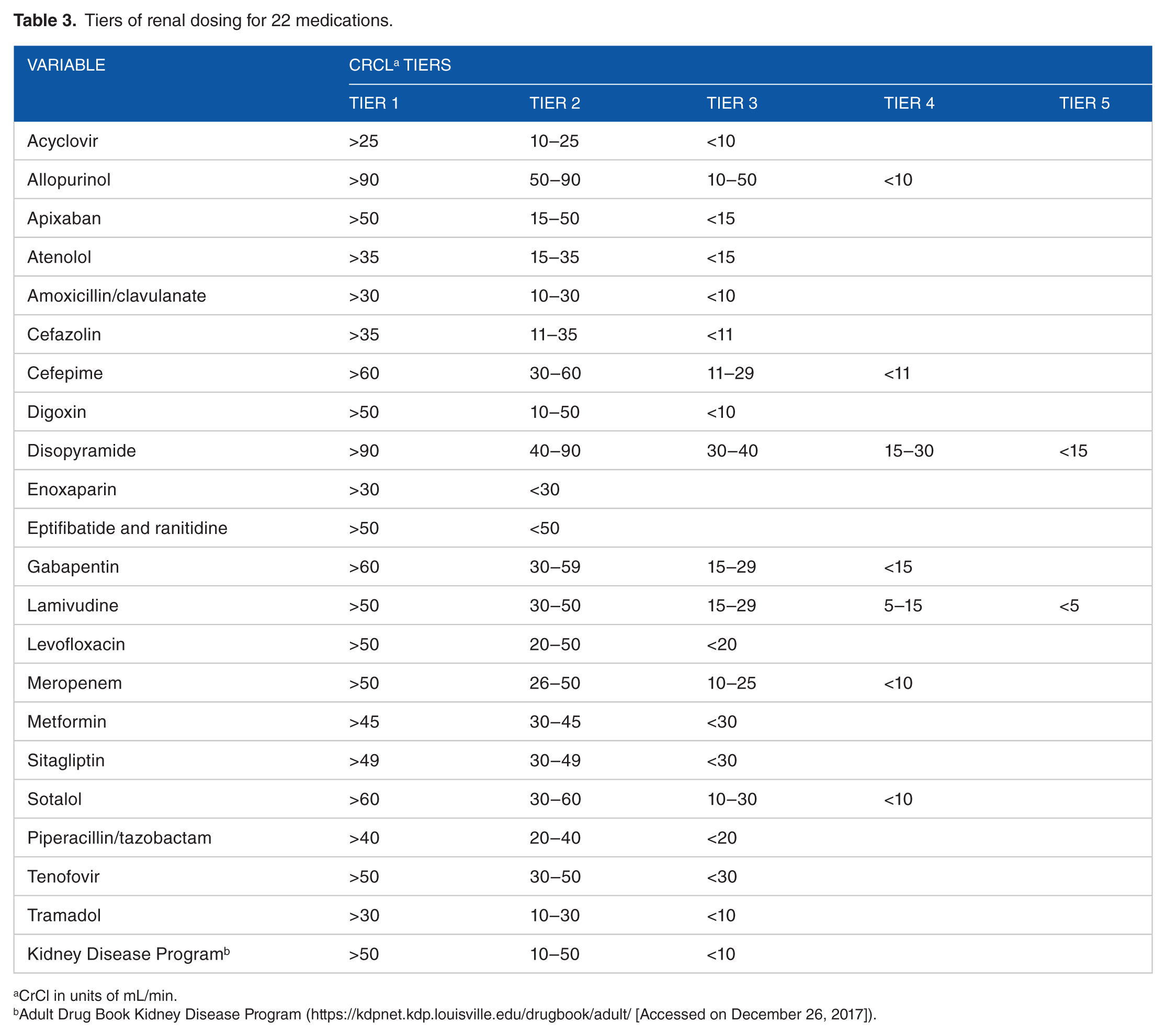
CrCl in units of mL/min.
Adult Drug Book Kidney Disease Program (https://kdpnet.kdp.louisville.edu/drugbook/adult/ [Accessed on December 26, 2017]).
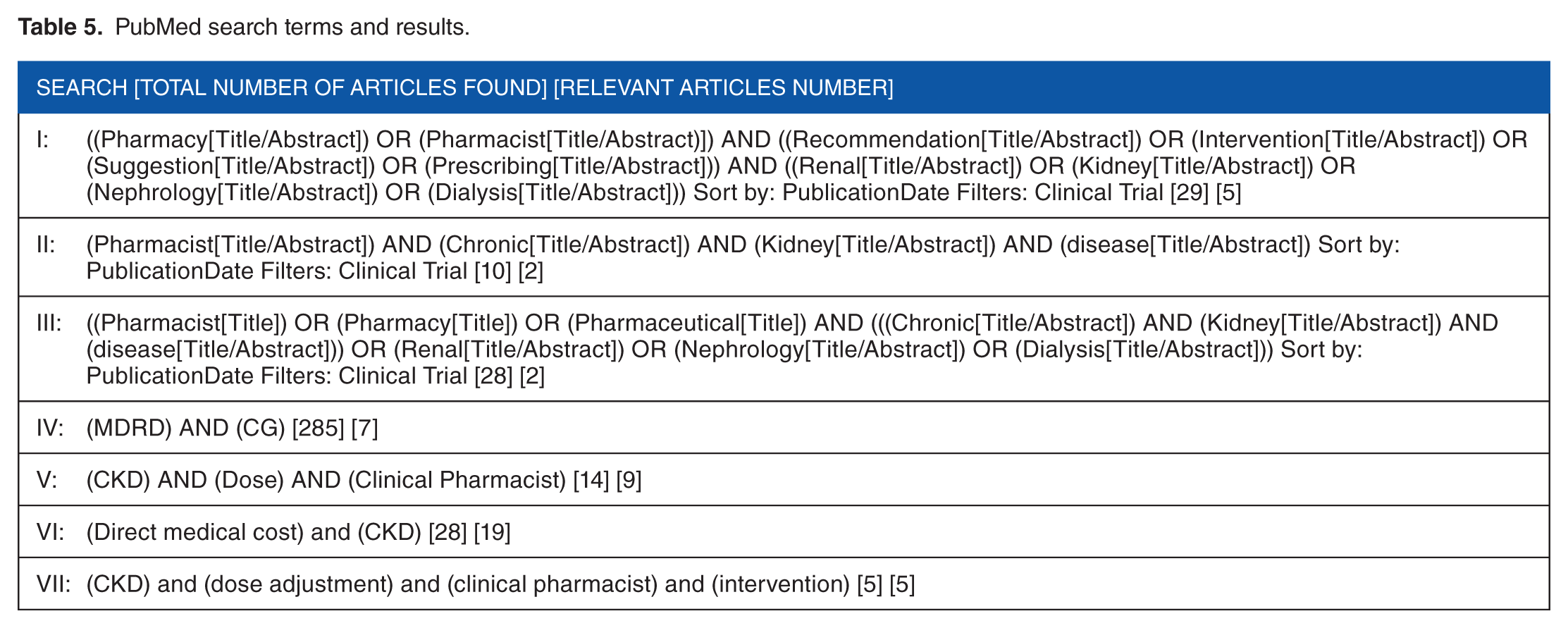
Finally, S.M.A.J. and L.M.S. performed a systematic review to retrieve all studies with renal function estimation and pharmacist interventions associated with specified clinical outcomes. In this part, the objective is to simulate data from the literature to quickly quantify the potential for measurable clinical differences between CG and MDRD. We used the secondary databases available, namely, Public/Publisher Medline (PubMed) and Elton B. Stevens Company (EBSCO). Key terms are summarized in Tables 4 and 5. In their first strategy, authors sought studies that had pharmacist interventions in patients with CKD and which reported outcomes in those 18 years old and above. They excluded all other studies. In the second strategy performed only on PubMed, the 2 used multiple combinations of key terms. The largest return was for the terms MDRD and CG with no limitations. Two independent investigators, L.M.S. and S.M.A.J., reviewed all abstracts of articles. They fully read the papers when both agreed on their inclusion. Differences in opinion were reconciled in a final meeting. Eventually, L.M.S. and S.M.A.J. simulated data only from articles that reported the clinical outcomes and the renal estimation methods. S.M.A.J. and L.M.S. used similar procedures as above for simulation with the exception that excel–generated data that resemble that found in each of the studies (ie, it complied with their statistical distributions). Study team conducted all parts in full accordance with the local and international ethical standards and the Declaration of Helsinki.
EBSCO search terms and results.

PubMed search terms and results.
Statistical analyses
We present continuous data as median (range) or mean ± standard deviation and categorical data as numbers and percentages. Our team used paired–data statistical tests as appropriate for comparisons of renal estimate groups for continuous and categorical data. They used StatTools, version 6.3.0 (Palisades Corp.) for performing all comparisons. Initially, authors performed simulations to merely assess concordance, and therefore, they made no sample size calculations. In effect, they simulated large sample sizes to make sure that they detect possible differences for all comparisons. Investigators also used multiple logistic regression to compare the impact of the different simulated factors on the discordance between CG and MDRD dosing.
Results
We summarize the overall data of the 1200 cases for the dosing concordance between equations in Table 6. The simulated population was diverse with a median age (range) of 58 (18–100) years. It was equally divided between sexes and ethnicities and largely uniformly distributed into the different body mass index categories. There were no significant differences in the demographics between men and women. For example, median height (range) was 171 (150–190) and 168 (150–190) for women and men, respectively. The majority were group 5 CKD with most of the remaining cases in categories 3 and 4. Figure 1 shows the distribution of the CrCl and GFR by CG, MDRD, and CKD–EPI. We observe that the CG distribution is more scattered than the MDRD and CKD–EPI. The CG estimates seem more likely to fall above or below the values for both the MDRD and CKD–EPI. Normalization of MDRD does not eliminate this difference in the scatter (data not shown). Figure 2 shows the concordance in dosing between the different renal function estimation methods for the 22 medications. Overall, it ranges from as low as 55% for lamivudine (CG vs albumin–based) to 99% for enoxaparin, eptifibatide, and ranitidine (MDRD vs CKD–EPI). Another observation is that discordance for combinations goes up and down together. Therefore, it is the cutoff values for dosing adjustments that increase or decrease the match between the different methods. For some medications such as eptifibatide and ranitidine, it can be as high as 98% regardless. In these cases, using any calculation is unexpected to change the clinical performance of the pharmacist who intervenes to make dosage adjustments. In contrast, differences in meropenem and cefepime doses can reach up to 37% and 42%, respectively. Unifying the tiers for all medications, as attempted in the Adult Drug Book Kidney Disease Program (Table 3), would still result in about 15% to 35% discordance (Figure 2). Pairwise CrCl or GFR comparisons showed that all formulas gave statistically different results (P < .0001) except for CG vs MDRD (P = .5147). There was a nonconsistent slight improvement in the concordance of CG with MDRD equation when normalized. For example, for meropenem, it increased from 63% to 66%.
Simulated cases overall summary.
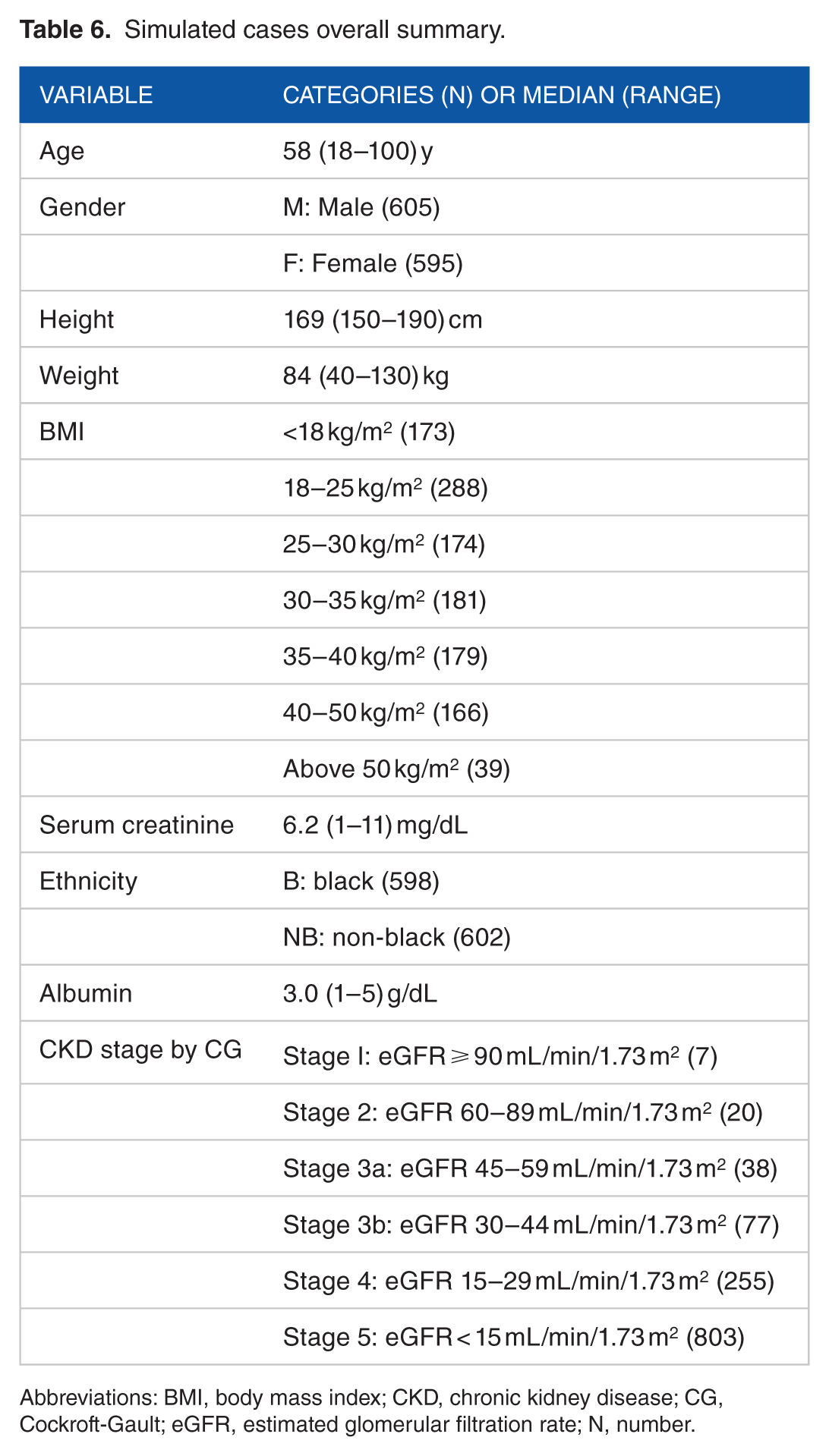
Abbreviations: BMI, body mass index; CKD, chronic kidney disease; CG, Cockroft-Gault; eGFR, estimated glomerular filtration rate; N, number.
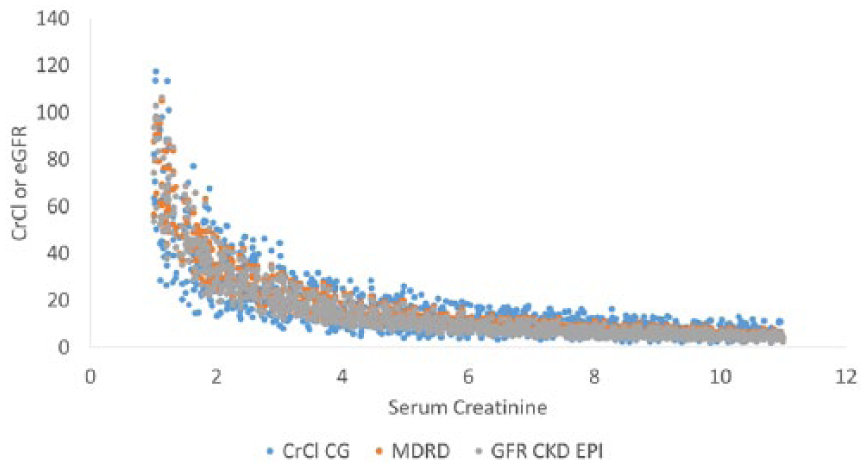
Scatter plot for all estimates of creatinine clearance or glomerular filtration rate for 1200 simulated cases using CG, MDRD, and CKD-EPI. CKD-EPI indicates Chronic Kidney Disease Epidemiology Collaboration; CG, Cockroft-Gault; CrCl, creatinine clearance; GFR, glomerular filtration rate; MDRD, Modification of Diet in Renal Disease.
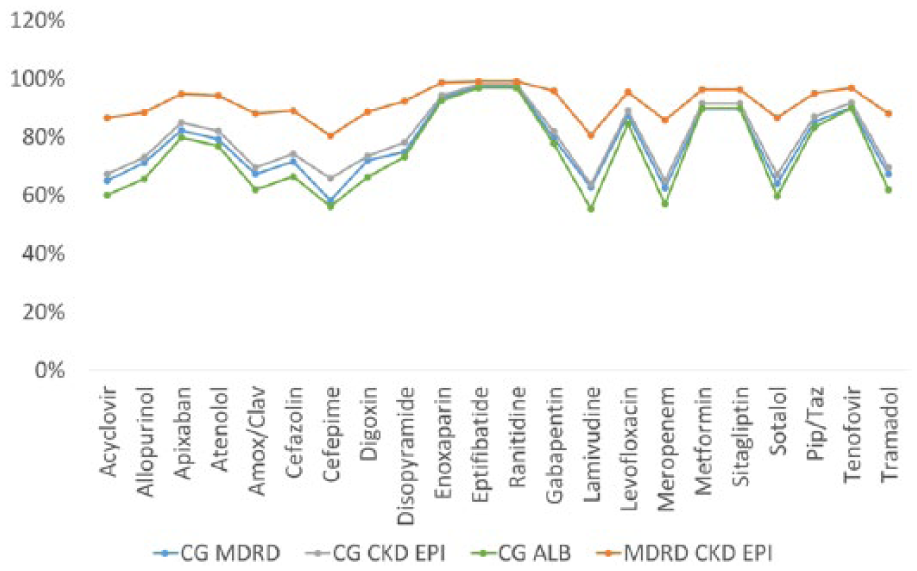
Concordance in dosing of different drugs for the different equations of renal function. CKD-EPI indicates Chronic Kidney Disease Epidemiology Collaboration; CG, Cockroft-Gault; CrCl, creatinine clearance; GFR, glomerular filtration rate; MDRD, Modification of Diet in Renal Disease.
Sex may slightly affect the discordance in meropenem dose of CG with CKD–EPI, but not that between CG and MDRD (Figure 3). One can also note that CG is always more concordant with the other equations in elderly patients (ie, those above 65 years old) (Figure 4). Comparatively, weight has a variable effect on the concordance (Figure 5). Notably, when actual to ideal falls below 80%, the concordance of CG with albumin–based estimates increases. Discordance between the two increases with an albumin level below 3 (Figure 6). Considering CKD stages, there is a consistent order of descending concordance for CG with both MDRD and CKD–EPI as follows: I > II > IIIb > V > IV > IIIa (Figure 7). Albumin–based equations, nonetheless, have totally different and messy concordance values with CG. Multiple models of logistic regression failed to converge. Only one model did so to null hypothesis meaning that no factor would emerge as statistically more predictive of discordance between CG and MDRD.
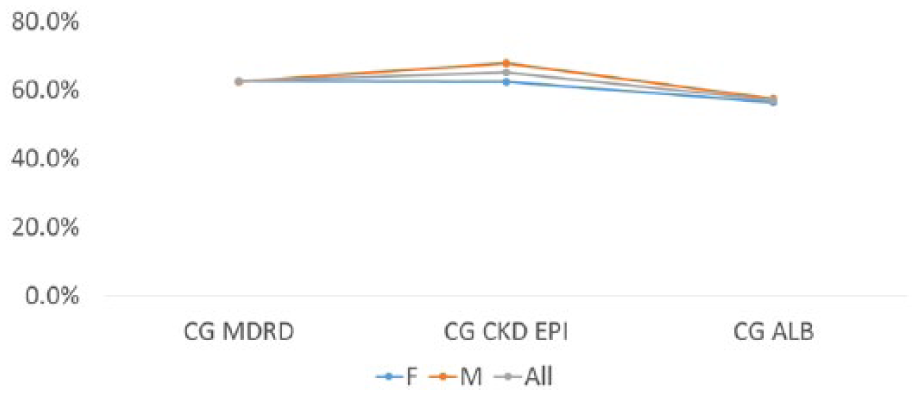
Concordance of the CG equation with different equations for meropenem dosing using sex as a stratification factor (M for male and F for female). CKD-EPI indicates Chronic Kidney Disease Epidemiology Collaboration; CG, Cockroft-Gault; MDRD, Modification of Diet in Renal Disease.
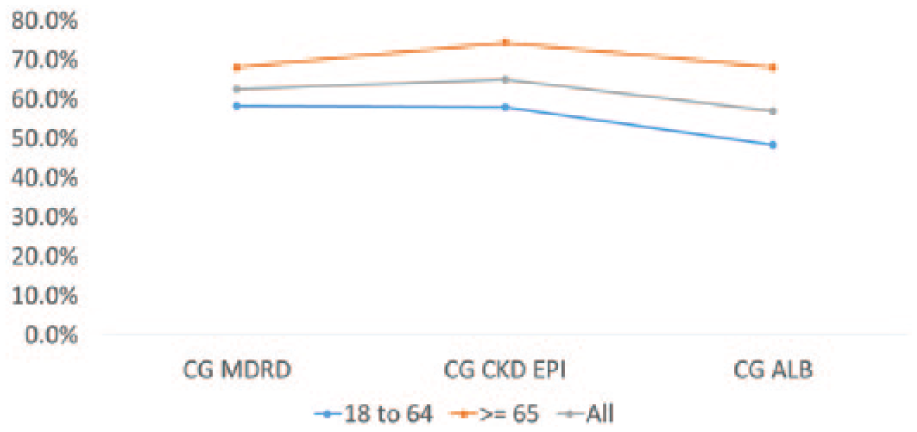
Concordance of the CG equation with different equations for meropenem dosing using age as a stratification factor (age in years). CKD-EPI indicates Chronic Kidney Disease Epidemiology Collaboration; CG, Cockroft-Gault; MDRD, Modification of Diet in Renal Disease.
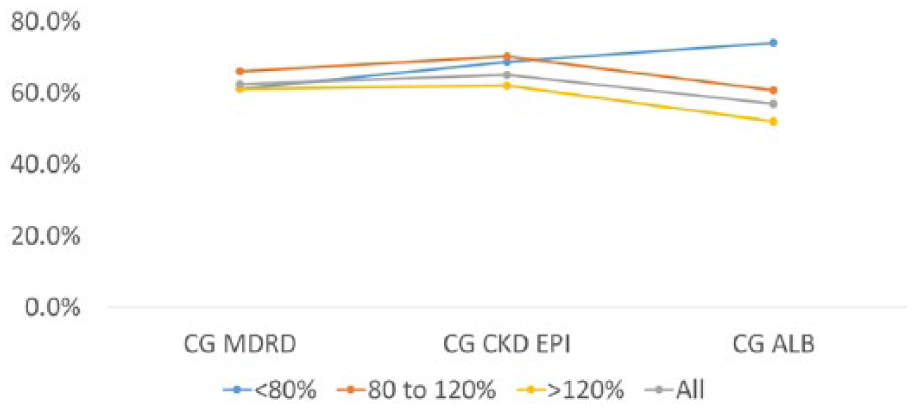
Concordance of the CG equation with different equations for meropenem dosing using weight to ideal body weight as a stratification factor. CKD-EPI indicates Chronic Kidney Disease Epidemiology Collaboration; CG, Cockroft-Gault; MDRD, Modification of Diet in Renal Disease.
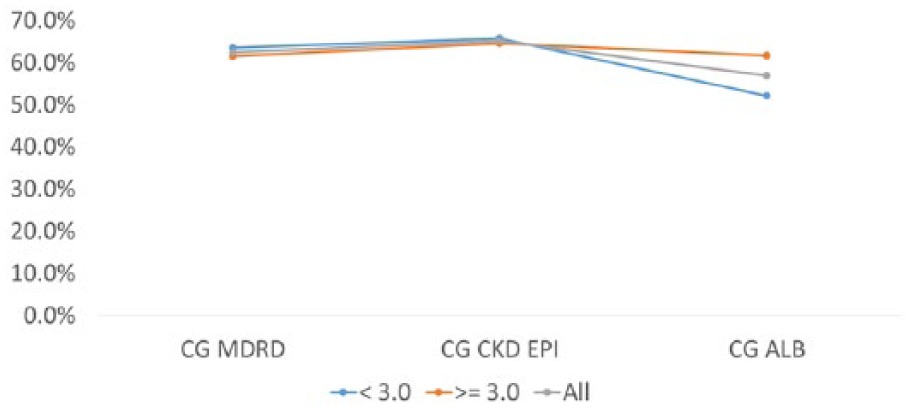
Concordance of the CG equation with different equations for meropenem dosing using albumin level as a stratification factor (albumin level units in g/dL). CKD-EPI indicates Chronic Kidney Disease Epidemiology Collaboration; CG, Cockroft-Gault; MDRD, Modification of Diet in Renal Disease.
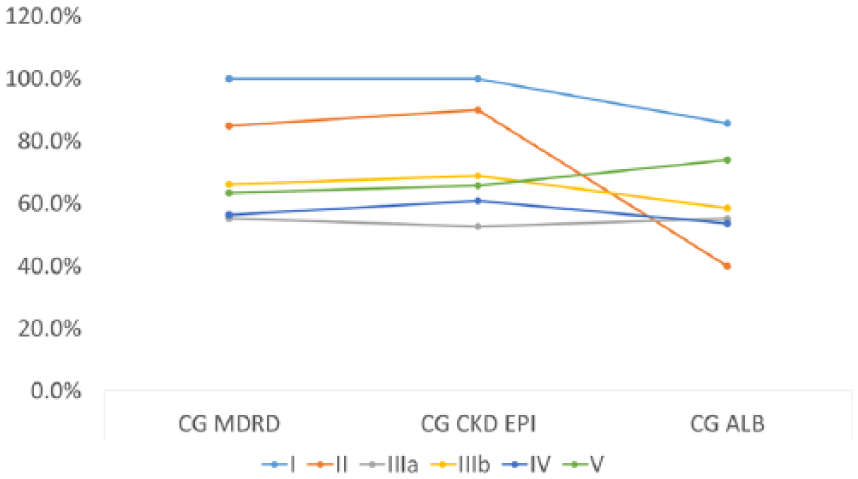
Concordance of the CG equation with different equations for meropenem dosing using CKD stage as a stratification factor. CKD-EPI indicates Chronic Kidney Disease Epidemiology Collaboration; CG, Cockroft-Gault; MDRD, Modification of Diet in Renal Disease.
For the second part, we found 467 articles using our first search strategy. In all, 65 of them met our inclusion guide, whereas our second round yielded 332 and 40 of these met the criteria. After removing all duplicates, only 62 remained as relevant papers. Among these, one recent systematic review performed on the prevalence of inappropriate prescribing in CKD, associated clinical outcomes, and the impact of pharmacist interventions served as an index. 37 Eight of the cited references had an assessment of clinical outcomes. These were also the same retrieved with our search strategy. Our team could get the full text of 7 with the eighth publication (in French) being deemed unretrievable even after a formal request from our librarian. Five based decisions on CG and 2 preferred MDRD. After review, we excluded 6 of the 7 as they failed to provide enough information for simulation. We simulated the data from this one study which used the MDRD equation for estimation of the renal function. The total number of patients here was 8701. Summary statistics for the 2 simulations we performed are presented in Table 7. Readers can see that 2 are pretty similar to the original. In these, our group has defined a new variable, namely, mortality switch. Here, we kept mortality outcome the same if the CG and MDRD equations had similar tier suggestion and switched it when they differed. Table 8 summarizes the simulated dosing for CG compared with MDRD on the basis of Breton et al. 38 There were 2610 and 2631 different recommendations in simulations 1 and 2, respectively. Of these, 577 and 607 would survive with the change, and the remaining would expire. Consequently, there is a significant margin of about 16% for a change in mortality if CG is used versus MDRD for dosing in these patients.
Summary statistics of 2 simulations used to compare the clinical outcomes of using CG versus MDRD equations.
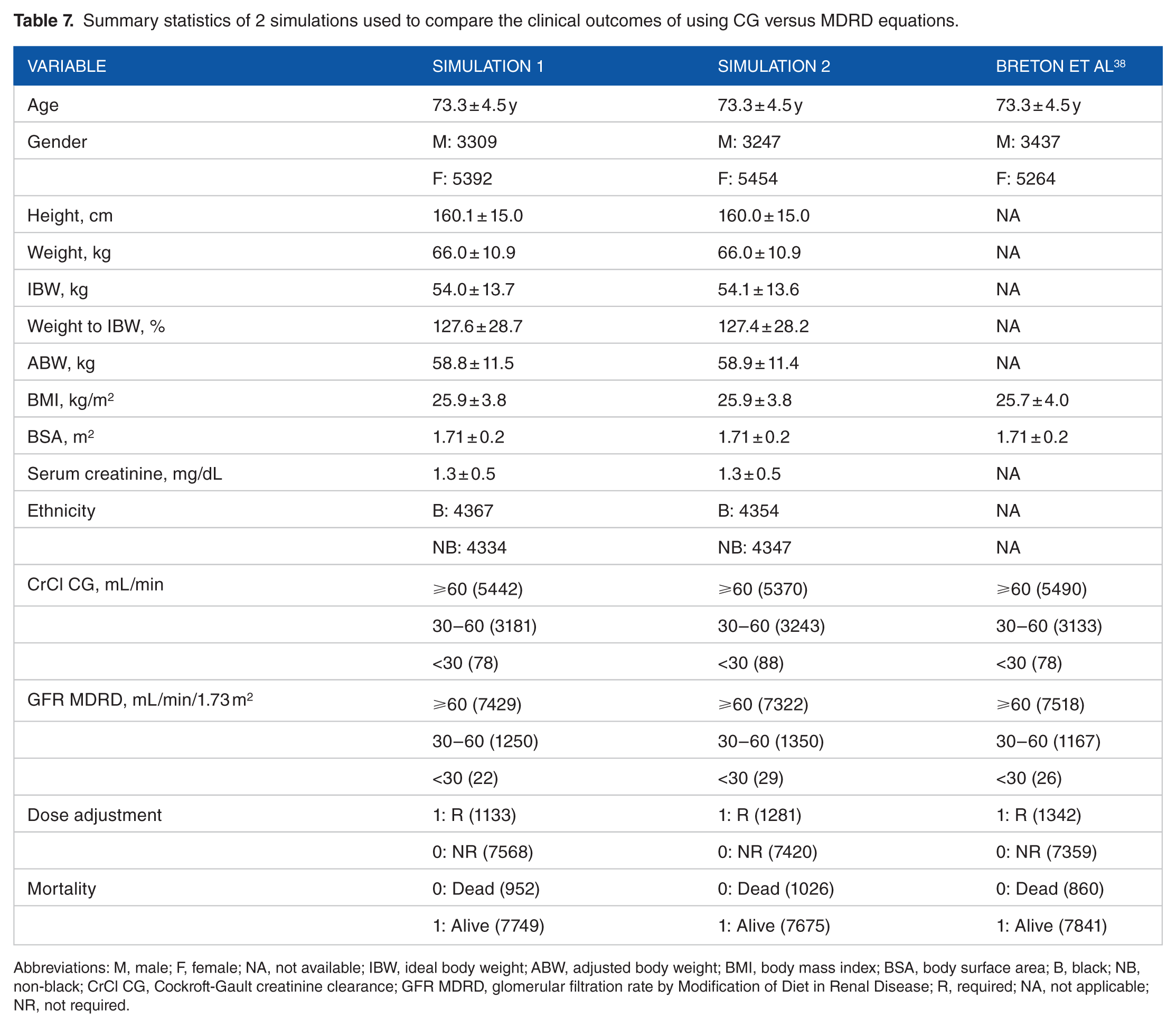
Abbreviations: M, male; F, female; NA, not available; IBW, ideal body weight; ABW, adjusted body weight; BMI, body mass index; BSA, body surface area; B, black; NB, non-black; CrCl CG, Cockroft-Gault creatinine clearance; GFR MDRD, glomerular filtration rate by Modification of Diet in Renal Disease; R, required; NA, not applicable; NR, not required.
Dosing recommendation of CG compared with MDRD.
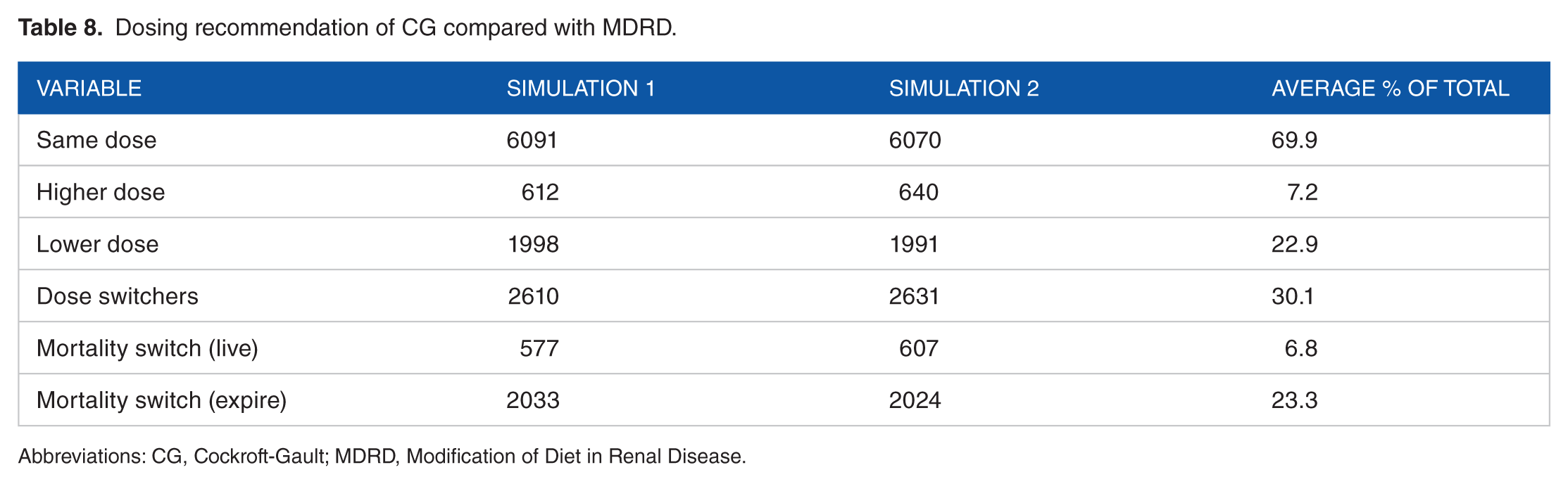
Abbreviations: CG, Cockroft-Gault; MDRD, Modification of Diet in Renal Disease.
Discussion
Our study is the first to characterize the effect of changing the dosing tiers on the concordance between renal function estimates for a vast number of medications. The choice of the equation forms probably an insignificant factor in dictating the clinical outcomes for some drugs such as enoxaparin, eptifibatide, and ranitidine. For example, Melloni et al reported enoxaparin dose adjustments in 19.0% and 9.6% with CG and MDRD, respectively. 39 However, the authors of the paper reported using actual weight in calculating CrCl in the CG equation. Adjusting for those above 120% or below 80% of the ideal body figure offsets the difference. So the discordance reported by Melloni et al is almost identical with our study. Hence, it seems that adjusting for this variable in CG would eliminate 10% of the mismatch irrespective of the drug. In the same study by Melloni et al, we find that eptifibatide dosing varied by almost 18%. In this case, variation again disappears if we both adjust the weight in CG and match the ages to our study. Note that those of Melloni et al were largely below 65 years of age. Therefore, knowing the sources of variability would clarify the differences observed and permit a more judicious use of renal function estimates.
Antimicrobials present considerable differences in doses between CG and MDRD in patients with CKD. In our simulated population, antibiotics were associated with consistently more deviation between CG and MDRD approximately in the range of 20% to 40%. It was minimally affected by normalization and is comparable with that seen in reality which ranges from 17% to 36%.40,41 For example, Golick and Lawrence 40 had about a 32.4% different meropenem regimens between CG and MDRD. Stevens et al 41 showed 10% to 19% discrepancy for 2– to 5–tier medications, respectively. These findings emphasize our point that populations in practice have pretty similar ranges of variability which can be broad enough to alter clinical outcomes. This is especially noteworthy with the fact that, in most settings, urine collections are unperformed. Clinical significance seems to be huge for meropenem. Suffice it to disclose that unpublished data from our antimicrobial stewardship program showed that 100% of our multidrug–resistant Klebsiella pneumonia is, in fact, carbapenem–resistant enterobacteriaceae as well. Obviously, dosing may be a prime contributing suspect. Clinical pharmacy use of CG to adjust antibiotic doses may promote the emergence of these resistant pathogens in our institution. Readers should note that our facility has a large population of both CKD and malnourished patients. In addition, sex and albumin had little effect on the discordance between CG and MDRD for meropenem. Other factors, however, such as age, CKD stage, and weight clearly affected concordance. For example, CKD stage IIIa has minimal dosage similarity for CG with MDRD and CKD–EPI. Hence, the urgency of establishing which method should be preferably used in this patient group is highest. Another critical message is that in malnourished patients, albumin–based equations may emerge as a better estimate of renal function for drug dosing. It goes in line with the findings from an old study by Sanaka et al. 42 Albumin–based equations provided more accurate and less biased CrCl estimates than CG in patients with muscular atrophy, renal insufficiency, and albumin levels below 2.8 g/dL. Overall, no one factor emerged in multiple logistic regression as predictive of discordance. Therefore, real–life studies can assist in finding the best renal estimates for empiric dosing. The gold standard, however, lies in optimal clinical judgment based on patient responses.
Pharmacotherapy Assessment in Chronic Renal Disease (PAIR) criteria is a validated tool to assess medication safety and use issues in patients with CKD. It is noteworthy that inappropriate dosage comprised more than one–third of the drug–related problems (DRPs) in the PAIR criteria. 43 Actual reported DRPs were also mostly dose–related in another study conducted in residents of aged care facilities. 44 Some articles, albeit few in number, have shown that CG and MDRD perform better with varied scenarios.44–47 However, the one on meropenem found no difference between CG and MDRD. 48 It screened samples from 61 patients which are a very modest size. Our simulations showed that for an outcome such as mortality, having a sufficient number of cases may help elucidate the actual difference between these methods in dosing medications. Both of our simulations demonstrated that a significant proportion of patients (about 16%) were likely to switch their outcome based on the method used. Interestingly, if we take the discordance of about 38% among CG and MDRD for meropenem, one can calculate a 5% anticipated difference in mortality. Accordingly, it would be very likely to detect superiority in clinical studies using quasi–experimental designs. Readers should note our key finding compared with that of Breton et al. 38 They simply showed that use of CG is associated with more dose adjustments overall in the population of elderly patients with renal impairment. Our study, in contrast, shows that for a specific drug meropenem, the potential clinical impact of using CG versus MDRD is large. Therefore, more research is needed to find out in this specific area of practice which equation would perform optimally in selecting initial antibiotic doses.
Future research designed to elucidate the ideal equation to use for empiric dosing of antibiotics (eg, meropenem) should take the predictors of a larger discordance between estimates into consideration. According to the current findings, these risk factors are male sex, 18 to 65 years old, obesity, hypoalbuminemia, and CKD stages III, IV, V, and sometimes II. Some of these sources of variability were well established and studied as summarized by Lascano and Poggio. 49 However, multiple other factors may be involved as discussed by Cockroft and Gault.50–52 Dersch and McCormack 53 correctly concluded that dosage selection in renal patients must be a clinical activity which ought to be individualized. Nevertheless, real–life studies can help determine whether using CG or MDRD is superior empirically. Individual patient response and sound clinical judgment would then pave the way for careful escalation or de–escalation of therapy. Therefore, one suggested primary outcome would be dose changes due to suboptimal response while maintaining the estimate of CrCl or GFR and/or adverse effects. The latter includes things such as seizures and death. Finally, taking meropenem as a role model, our team will recruit patients with predictors of a larger discordance in future studies. Our team will clinically test the hypothesis that either CG or MDRD is superior in empiric dosing of meropenem. Our primary outcome will incorporate treatment escalation or de–escalation while maintaining a stable CrCl or GFR.
Conclusions
Proper drug dosing is a clinical activity which should be based on more than mere calculations. It should take the patient characteristics and clinical response into consideration. Statistically similar CrCl and eGFR with the CG and MDRD formulas, respectively, seem to be, in fact, clinically largely discordant in terms of drug dosing. Therefore, this study supports practical comparisons of outcomes for individual medications, especially antibiotics, dosed in patients with CKD using different renal function estimates.
Footnotes
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
SMAJ performed the literature search, analyzed the data, and prepared the manuscript. AHK analyzed the data, reviewed the manuscript, and guided the team during the publication process. LMS performed literature search and gave conceptual guidance, analyzed the data, and prepared the manuscript. SASS analyzed data and review manuscript. DAK reviewed manuscript and assisted the team during publication.
Availability of data and materials
The data sets used and/or analyzed during this study are available from the corresponding author on reasonable request.
