Abstract
Cardiorenal syndrome is a group of disorders resulting from the pathological interaction between the heart and the kidneys. The most accepted classification of cardiorenal syndrome differentiates the types based on the temporal sequence of organ involvement. Although many mechanisms seem to be involved in the pathogenesis, cardiac hemodynamic changes are still considered one of the major parameters that affect the cross talk between these organs. As heart failure and chronic kidney disease reach endemic proportions, the necessity to understand the pathophysiology, hemodynamics, and effective therapy of cardiorenal syndrome becomes fundamental. The aim of this review is to identify and explain the hemodynamic changes behind the syndrome and evaluate potential noninvasive methods that can help develop a practical tool for managing the cardiorenal patient.
Introduction
Heart failure (HF) and chronic kidney disease (CKD) have become an endemic problem during the past decade; both entities share risk factors where the progression and/or decompensation of one can significantly affect the other. According to the Centers for Disease Control and Prevention, more than 10% of adults in the United States have CKD with varying stages of disease progression and about 5.7 million adults have HF, 1 costing the nation several billion dollars each year. 2 This is evidenced in the increase in mortality and length of hospitalization in patients with HF and worsening renal function.3–10
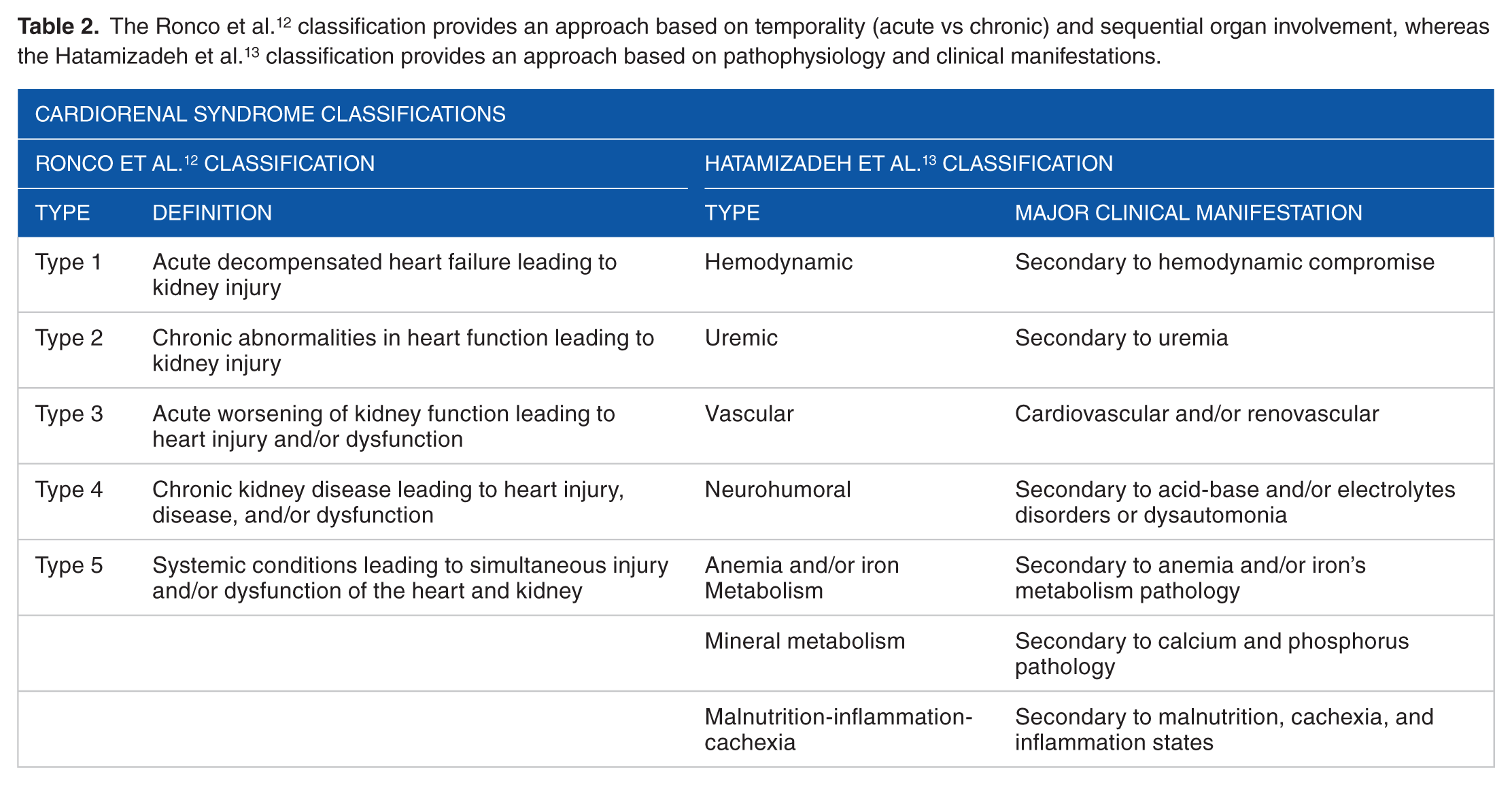
The heart and the kidneys interact closely to form an intricate network to maintain hemodynamic stability and organ perfusion. Cardiorenal syndrome (CRS) is a group of disorders resulting from the pathological interaction between the heart and the kidneys whereby acute or chronic dysfunction in one organ may induce acute or chronic dysfunction in the other. 11 Several definitions and classifications have been developed to help simplify, better understand, and adequately evaluate the multifaceted interaction between the heart and the kidneys in CRS. This is done with the goal of providing better management and appropriate monitoring of these patients. Currently, the most accepted classification is the one proposed by Ronco et al, 12 which distinguishes between 5 distinct types of CRS based on the presumed initial trigger and pathogenesis, temporal sequence of organ injury and/or failure, and whether the injury is acute or chronic in nature. Other authors have proposed different CRS classifications based on the necessity of a better clinical approach. Hatamizadeh et al 13 divided CRS into 7 types from a pathophysiologic standpoint, aiming for a more clinically relevant approach.
The pathophysiology of CRS is complex and involves multiple factors. Renal congestion, secondary to increased central venous pressure (CVP), and decreased cardiac output (CO) have been frequent findings in CRS.14,15 The late Arthur Guyton developed a model of the heart-kidney interaction with respect to extracellular fluid volume, CO, and mean arterial pressure (MAP). 16 This can be used to understand the systemic hemodynamic effects in combined renal and cardiac failure; however, it cannot explain all aspects implicated in the pathophysiology of this syndrome. 17 The pathophysiology of CRS also includes many other components which could potentially be used as targets for management. 13 Neurohormonal mechanisms are essential concepts in understanding CRS and developing diagnostic strategies. Activation of the renin-angiotensin-aldosterone system has a negative effect on both the heart and the kidneys. 18 Angiotensin II and aldosterone lead to sodium and water reabsorption, which in turn leads to renal and systemic vasoconstriction. Subsequently, there is an increase in venous return, end-diastolic ventricle pressure, oxidative stress, and production of cytokines which eventually leads to cardiac and renal fibrosis and degeneration.19,20
Understanding the hemodynamics of CRS is complex and challenging especially when relying only on clinical parameters. Implementing routine noninvasive testing into the evaluation of the patient with CRS may serve as an invaluable tool that can elucidate the pathophysiology of the syndrome. In addition, it may aid in the categorization, evaluation, management, and treatment of these patients. Point-of-care ultrasound, a noninvasive modality, can be used in routine patient assessments performed at bedside. Such intervention, which with adequate training can be of great yield, comes at a low cost, has high reproducibility, and increases patient satisfaction. 21
The objective of our review is to identify and assess the hemodynamics of CRS through noninvasive diagnostic tests from the evidence provided in the literature to provide clinicians with a tool to evaluate the patient with CRS.
Hemodynamic Parameters on Cardiorenal Subtypes
Cardiorenal syndrome’s hemodynamic parameters and its noninvasive evaluation will be surveyed and divided based on the Ronco classification, which categorizes CRS into 5 subtypes. 12 It is relevant to highlight that a structural or functional abnormality must exist in both organs for CRS to be diagnosed, and organ cross talk is involved in the global acute and chronic pathogenesis. 22
CRS type 1
Cardiorenal syndrome type 1 occurs in the setting of acute decompensated HF (ADHF) leading to kidney injury and/or acute kidney dysfunction. It is estimated that up to 40% of patients hospitalized with ADHF develop acute kidney injury (AKI).5,8 In CRS type 1, it is believed that both renal congestion and decreased renal perfusion contribute to the pathophysiology of this phenomenon. 23 Several studies have shown that increased venous pressure is a key player in the development of AKI in ADHF. 24 In many patients, venous congestion, more so than arterial perfusion pressure, affects renal blood flow and therefore worsens renal function. Furthermore, renal vasoconstriction also plays a hemodynamic role in CRS type 1; this has been demonstrated by Pesce et al 25 through a mouse model where renal dysfunction was induced by coronary artery ligation.
Hemodynamic parameters
Renal perfusion pressure depends on arterial pressure as well as on the transrenal perfusion pressure gradient, which equals MAP minus CVP. 26 Therefore, a decrease in MAP and/or an increase in CVP will cause a decrease in transrenal pressure. Studies have shown that increases in CVP or jugular venous pressure (JVP) on examination are associated with impaired renal function.15,27 Firth et al 28 demonstrated that increasing CVP above 19 cmH2O produced significant reduction in glomerular filtration rate (GFR) and sodium excretion in an isolated rat kidney, which was reversible when CVP normalized. Similarly, Winton et al 29 observed that by increasing the renal venous pressure above 20 mm Hg, the diuresis of an isolated canine kidney was significantly and progressively reduced. In these cases, it can be deduced that the main culprit is renal congestion which occurs in the parenchymal region. Renal congestion has been associated with increased renal interstitial pressure which may directly play a role by compressing vessels in the renal parenchymal regions or by reducing vessel compliance leading to a mechanical obstruction in the renal tubules subsequently decreasing urine output. 30 Right ventricular failure leading to high filling pressures—ie, elevated CVP/pulmonary capillary wedge pressure (PCWP)—is associated with a lower renal perfusion pressure, which subsequently leads to impaired renal function. Evidence suggests that the main hemodynamic determinant for the development of worsening renal failure is CVP and not CO. 14
Noninvasive diagnostic evaluation
Noninvasive modalities can be useful in evaluating the parameters previously mentioned. Certain symptoms and clinical signs can reliably predict the presence of venous congestion. A systematic review of 12 studies suggested that an increase in JVP was very helpful for detecting increases in filling pressures. 31 Echocardiography may help in diagnosing the congestive state by considering right atrial (RA) and ventricular pressures and the diameter of the inferior vena cava (IVC). Estimation of CVP using bedside echocardiography is a quick and reliable method to do so. Beigel et al described how point-of-care echocardiography can be used as a guide for treatment by measuring hemodynamic parameters including CVP, systolic pulmonary arterial pressure, PCWP/left atrial pressure, and CO. The association between high venous pressures and worsening renal function seems to be superior to the effect of arterial blood pressure, cardiac index (CI), and PCWP as predictors of CRS type 1.14,15 Assessment of systemic venous congestion is possible by measuring the size and collapsibility of the IVC. 32 According to Rudski et al, 32 dilated IVC is defined as a diameter greater than 2.1 cm. If the IVC is not dilated with 50% collapsibility, the estimated RA pressure is 3 mm Hg. A dilated IVC with less than 50% collapsibility has an estimated RA pressure of 15 mm Hg. When the IVC diameter and collapsibility do not match, the RA pressure is estimated to be 8 mm Hg. IVC dilation identifies patients who are at risk for worsening renal function during hospitalization and is associated with an increased risk of adverse events during the 2-year follow-up. 33 Furthermore, rapid assessment of IVC physiology could be performed at the bedside by a noncardiologist34,35 (Table 1).
Estimation of RA pressure based on IVC parameters. 32

Abbreviations: IVC, inferior vena cava; RA, right atrial.
In scenarios in which IVC diameter is >2.1 and collapse is >50%, an intermediate value of 8 mm Hg (range: 5-10 mm Hg) may be used. 36
Besides CVP, other useful echocardiocardiographic measurements include lateral and septal wall longitudinal motion (E′) in relation to the mitral inflow velocity (E). E/E′ ratio directly correlates with PCWP. An E/E′ above 15 correlates with a PCWP of ≥18 mm Hg37,38 This finding is independent of the systolic function, as 50% of patients with congestion have preserved ejection fraction—ie, diastolic dysfunction. CVP has been shown to be an independent predictor of worsening renal function, especially in the setting of low CO.14,39 Venous pressure depends on volume and venous tone, where venous tone is primarily mediated by neurohormonal activation. This explains why CVP is not a reliable marker for fluid overload. 40 The high compliance of the venous system predisposes for a relative pressure-volume disconnection. 41 A good example is how unrestrained sympathetic activation with concomitant vasoconstriction may result in impaired capacitance function and volume shifts from the splanchnic circulation toward the systemic circulation. 42 From the intrarenal perspective, Iida et al 30 found that intrarenal venous flow patterns measured by intrarenal Doppler ultrasound were associated with RA pressure and correlated strongly with clinical outcomes. Intrarenal venous flow can be noninvasively measured by Doppler ultrasonography and is mainly affected by both central venous hemodynamics and renal parenchymal compliance as related to renal congestion. 30 Conversely, the resistive index (RI) of the renal artery, despite correlating with CVP, MAP, and CI, 43 is not independently associated with clinical outcomes. 30 Preexisting conditions such as atherosclerosis and nephropathy may lead to RI being less specific in assessing renal congestion.44,45 Renal arterial RI depends on parenchymal abnormalities, such as glomerulosclerosis, tubular fibrosis, tubule-interstitial lesions, and vascular abnormalities, such as renal artery atherosclerosis and renal venous congestion.46,47 Pesce et al 25 propose to measure renal vasoconstriction by the pulsatility index (PI) through echo-Doppler as a noninvasive marker of CRS type 1 in mice, thus introducing new perspectives for the evaluation and management of this CRS subtype. In this study, renal interlobar arteries were identified using color Doppler to measure the arterial flow in the renal cortex. This was used to measure peak velocity (Vmax), end-diastolic velocity (Vmin), and mean velocity (VM), as well as calculate the PI where PI = (Vmax − Vmin)/Vmean. 25 Pulsatility index has been found to correlate with renal vascular resistance, filtration fraction, and effective renal plasma flow in chronic renal failure. 12
CRS type 2
CRS type 2 involves chronic abnormalities in heart function leading to kidney injury or dysfunction. 11 Approximately 63% of patients with HF meet the definition of CKD stages III-V with an estimated GFR <60 mL/min/1.73 m. 14 CRS type 2 occurs when HF incites a sequence of processes involving the endothelium, glycocalyx, tubules, nephrons, and fibroblasts in the skeleton of the kidney that result in progressive loss of functioning nephrons and tissue fibrosis. 23
Hemodynamics parameters
Vascular damage is thought to be one of the main factors involved in the development and progression of CRS of type 2. 48 Multiple methods have been proposed to assess vascular damage, which are conveyed by endothelial dysfunction, increased arterial stiffness, and abnormal pressure wave reflection. 48 Vascular damage has been linked with the development and progression of HF and a decline in renal function. 24 Increased arterial stiffness is associated with an increase in cardiac afterload, impaired coronary blood flow, and abnormal arterial-ventricular coupling leading to myocardial damage.48–50 In addition, worsening renal function from pulsatile nephropathy and changes in central hemodynamics are contributing variables.48–51 Assessing vascular damage through noninvasive methods is indispensable in the evaluation and management of these patients. Efforts to evaluate renal perfusion parameters have been attempted to identify the cardiorenal interaction early on. Measurement of renal artery RI by noninvasive Doppler assessment has been studied and established as a marker of organ damage in cardiovascular disease.12,52 Nevertheless, RI specificity decreases with the progression of renal disease. Similarly, RI has poor sensitivity identifying the direction of cardiorenal damage; however, it is mainly influenced by CO rather than by renal function.52,53
Noninvasive diagnostic evaluation
In these patients, CRS is insidious and patients are rarely treated in specialized units but rather in general wards or in the outpatient setting. Due to the coexistence of several comorbidities, it is not easy to clearly differentiate between the subtypes, often encountering difficulty reaching a definitive diagnosis. 54 Ultrasonographic evaluation can provide information regarding the chronicity of kidney disease, volume status, and hemodynamics of the heart. This is established by assessing ejection fraction, right ventricular volume, and estimated pulmonary arterial pressure. Furthermore, this type of CRS can be assessed with Doppler using pulse wave velocity sampling. 54 The assessment of central arterial stiffness through carotid-femoral pulse wave velocity, which reflects segmental arterial elasticity, is the preferred method to evaluate arterial stiffness as a component of vascular damage.55,56 Carotid-femoral pulse wave velocity is defined as the velocity of a pulse traveling from the heart to the carotid and the femoral artery. 24 Increases in arterial stiffness increase the pulse wave velocity. 57 This can be measured using a central arterial pressure waveform analysis system and subtracting the carotid suprasternal notch distance from femoral suprasternal notch distance, which correlates better with invasive measurements. 58 The European Society of Hypertension/European Society of Cardiology guidelines consider measuring aortic pulse wave velocity as a tool for the assessment of subclinical target organ damage, where the threshold for diagnosis is a value of 12 m/s.56,59 Further studies are needed to confirm whether the reduction in pulse wave velocity by itself is related to a reduction in cardiovascular events. 60
Hemodynamic monitoring has always been the foundation of HF management. One of the newer devices to assess pulmonary pressures is CardioMEMS HF System, 61 which is a pressure sensor that is percutaneously placed in the pulmonary artery branch and interrogated via a wireless detection system which can then be remotely reviewed by clinicians. 62 It allows for outpatient measurement of right-sided pressures and can be extrapolated to predict volume status in a format similar to invasive right heart catheterization. This device’s landmark trial excluded patients with CKD IV or V due to the premise that these patients may be difficult to diurese. This could be very promising and will add to clinicians’ objective assessment of a patient’s volume status and pulmonary pressures.
CRS type 3
Cardiorenal syndrome type 3 is defined as an acute worsening of kidney function leading to heart injury and/or dysfunction. 11 CRS type 3 is a frequent event in the nephrological emergency setting. In this clinical pattern, volume overload, due to acute renal failure, leads to acute HF. Acute salt and water retention, volume overload, and the immediate effects of uremia on the myocardium are all postulated mechanisms precipitating ADHF in CRS type 3. 23
Kidney size and echogenicity provide primary features to discern between acute and chronic nephropathies. Physicians should know that kidney volume and longitudinal diameters correlate with patients’ height and body surface area and that a normal ultrasound does not exclude CKD. However, a hyperechogenic renal cortex with medullary hypoechogenicity and low corticomedullary ratio is predictive of chronic nephropathy.63,64 Enlarged renal parenchyma may suggest edema in the acute setting, human immunodeficiency virus–related nephropathy, early stages of diabetic nephropathy, amyloid nephropathy, polycystic kidney disease, and interstitial nephritis. Likewise, renal ultrasound evaluation is crucial in the differential diagnosis of obstructive nephropathies. 54 In the acute setting, calculating the RI from the interlobar or arcuate artery of the kidney using pulse wave Doppler can be a helpful in determining the diagnosis and prognosis of this CRS subtype. 65 An RI greater than 0.75 can be used to differentiate between acute tubular necrosis and prerenal AKI. 66 In addition, an RI greater than 0.795 predicts persistent AKI in critically ill patients. 65
CRS type 4
Cardiorenal syndrome type 4 is CKD leading to heart injury, disease and/or dysfunction. CKD is an independent risk factor for the progression of HF to hospitalization and death. 67 In a recent meta-analysis, an exponential relation between the severity of renal dysfunction and the risk for all-cause mortality was described. 68 Hemodialysis in end-stage renal disease (ESRD) is associated with myocardial stunning, tissue swelling, and marked increases in cell signaling with enzymatic activation of matrix metalloproteinase-2 which increases the burden of insults to the myocardium. 69 These changes cause an activation of T cells and cardiac macrophages, thereby influencing the cycle of cardiac myocyte apoptosis and replacement fibrosis. This fibrosis has been implicated in the major mechanisms of death in ESRD, HF, and sudden cardiac death. 70
ESRD patients with arteriovenous fistula (AVF) represent a particular group of interest. In 2003, the Centers for Medicare & Medicaid Services and ESRD networks formed a National Vascular Access Improvement Initiative called Fistula First Initiative. This initiative focused on promoting the use of AVF in all suitable dialysis patients; there has since been a significant increase in AVF creation. 67 AVF is the preferred dialysis access because of higher flow rates, superior patency, and low incidence of infections; however, they are not exempt of complications.
Since the early development of this surgical technique, it has been known that radio-cephalic fistulae lead to a hyperdynamic circulation because of an increase in the CO and a decrease in the peripheral vascular resistance. 71 Typically, an arteriovenous dialysis access requires about 20% of the CO—similar to the native kidneys in the absence of renal failure. The normal access flow (Qa) to CO ratio Qa/CO ranges between 17% and 23%. When the Qa/CO ratio increases beyond this range, the risk for cardiac failure increases. 72
Even when there is no formal high flow access definition, the term has been used when the access has a flow of 1 to 1.5 L/min or more than 20% of the CO. Usually reserved for upper arm fistulae, the access flow in the forearm (600-900 mL/min) tends to be half of those in the upper arm (900-1500 mL/min). High-output HF and pulmonary hypertension are clinical complications associated with high-output fistulae. High-output HF can be defined as signs or symptoms of HF in the presence of a CO greater than 8 L or a CI greater than 3.9 L/min/m2. The ejection fraction of these patients tends to be preserved. It is usually seen with Qa greater than 2 L/min or Qa/CO greater than 20%.67,71,72
Noninvasive diagnostic evaluation
In these cases, ultrasonography can be used to detect pulmonary congestion in CKD, identify hemodynamic changes caused by AVF, and subsequently guide management of dry weight and vascular access.57,72–74 In CRS type 4, ultrasonographic examination can verify similar indices as previously mentioned in CRS type 2.
CRS type 5
Cardiorenal syndrome type 5 consists of systemic conditions leading to simultaneous injury and/or dysfunction of the heart and kidneys. It may develop in the normal setting or in previously impaired organ function. 75 Although this subtype does not have a primary and secondary organ dysfunction, situations do arise where both organs simultaneously are targeted by systemic illnesses—the prototype being sepsis. The 11th Consensus Conference of the Acute Dialysis Quality Initiative considers septic shock as the model disorder to elucidate the pathophysiology involved in CRS type 5. 75
Septic shock is a disease of the microcirculation, and its degree of compromise is directly proportional to the degree of organ dysfunction and mortality. These microvascular alterations are caused by inflammation and massive cytokine release. Fluid resuscitation improves microcirculation by optimizing stroke volume and CO, thus increasing the total flow to the organs and tissues. 76 Evidence has shown that “sepsis-related myocardial dysfunction” is volume responsive, thereby increasing stroke volume after a fluid challenge. 77
Noninvasive diagnostic evaluation
Focused cardiac ultrasound is an invaluable tool for the assessment of the patient in cardiogenic shock; it can confirm the correct diagnosis, detect the cause, guide the hemodynamic management, and monitor the response. Various shock assessment protocols have incorporated point-of-care ultrasound, allowing the assessment of the IVC, aorta, and lungs. 78 Severe hypovolemia, biventricular hyperkinesis, small IVC, and IVC respiratory collapse are all typical echocardiographic findings of septic shock. 77 Once the afterload is restored by vasopressors and fluid therapy, left ventricular (LV) myocardial dysfunction improves.77,79,80 At this stage, the IVC diameter recovers and the respiratory collapsibility fades, similar to the profile in cardiogenic shock. Recognition of these sonographic findings can help clinicians tailor appropriate treatments to the different stages of septic shock. 78
Other Classifications
The Ronco classification is based on the presumed initial trigger of CRS pathogenesis, but it is not usually possible to specify whether the kidney or the heart initiated the events. 13 Thus, it can be challenging to clinically approach and manage patients with CRS based on this classification. Despite the contribution of this classification to the understating of the complexity of CRS, it is not as easily applied in clinical practice. 41 Due to lack of clinical and therapeutic guidelines, Hatamizadeh et al 13 introduced a new classification of CRS. It classifies CRS into 7 categories including the following: hemodynamic, uremic, vascular, neurohormonal, anemia/iron metabolism related, mineral metabolism related, and protein-energy wasting related (Table 2). This classification focuses on neurohormonal pathways as an approach to the treatment of CRS.
Future Direction
With the widespread use of dialysis for renal replacement therapy and the recent development of LV-assisted devices (LVADs) as bridge to transplant, and more importantly as destination therapy, a newer CRS classification is needed as these artificial organs will alter the pathophysiology and possibly the interaction between the 2 organs.
LVAD and its implication in the cardiorenal paradigm
Heart transplantation remains the gold standard for the treatment of advanced HF. The demand for heart transplantation exceeds the organ supply; and certain comorbidities preclude patients with HF from being candidates for heart transplant. This has lead to an increase in popularity of LVAD either as a bridge to transplant or as destination therapy in the management of advanced HF. 81 Nevertheless, patients with CKD are sometimes poor candidates for this therapy, as there is a paucity of data in long-term mortality and morbidity. For example, in patients with renal failure, for each gram of increased creatinine, the risk of LVAD infection increases by 70%. 82 Several studies have shown improved or stable renal function after implantation of LVADs regardless of whether the devices are continuous or pulsatile. The patients included in these studies had preexisting renal dysfunction, which ranged from mild to moderate.
In different studies, a significant improvement in kidney function within 1 month after LVAD was noted.83–85 Although some report a gradual decline in GFR during the first 6 to 12 months, most had an improvement in GFR at 2 years. The same results have been reported in animal models, where long-term continuous-flow devices maintained normal end-organ function.86,87 Lack of improvement in renal function after LVAD implant may be due to other causes of kidney damage such as intrinsic renal disease, persistent right HF, and/or a new renal insult, such as hemoglobinuria secondary to LVAD thrombosis leading to hemolysis. In a recent publication, the presence of hemolysis in patients with short-term LVAD did not directly translate to the need of renal replacement therapy, although an upward trend in the creatinine level was noted. 88
Following LVAD implantation, CRS is often reversed. Initially, there is a recovery of kidney function which seems to be followed by worsening in GFR at 1 to 2 months after implantation. The more the GFR increases after LVAD transplantation, the more it decreases chronically, although GFR generally remains above preimplantation levels throughout the duration of follow-up. 89
Conclusions
There is a need to understand acute and chronic hemodynamic mechanisms that regulate ventricular function, coronary flow, renal, and myocardial perfusion at different levels. 11 The assessment of hemodynamic parameters of the subtypes of CRS is fundamental in guiding management, but not all of them are well understood or rather not all CRS pathophysiology is hemodynamically dependent. Regarding CRS types 2 and 4, it is useful to determine whether guiding management from the evaluation of vascular dysfunction through noninvasive methods is a useful strategy in the future. Physical examination, together with ultrasonography, is a cost-effective, noninvasive method to assess CRS. After reviewing the 5 subtypes of Ronco and other classifications, we propose that there is a need for a better categorization of CRS; a classification that has better clinical usefulness and applicability for an entity that involves a multidisciplinary approach.
Footnotes
Acknowledgements
The authors would like to acknowledge Mai Sedki, MD, for her contributions in editing this manuscript.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
CC and MI contributed to literature review search and analysis and wrote the first draft of the manuscript. CC, MI, FCR, and GAH contributed to the writing of the manuscript. CC, MI, FCR, and GAH agree with manuscript results and conclusions; jointly developed the structure and arguments for the paper; and made critical revisions and approved final version. All authors reviewed and approved the final manuscript.
Disclosures and Ethics
The authors have confirmed that this article is unique and not under consideration or published in any other publication.