Abstract
Brexpiprazole, a novel atypical antipsychotic agent, has recently been approved as an adjunctive treatment for major depressive disorder (MDD) when monotherapy only provides a partial response. The mechanism of action is likely related to its partial agonist activity at D2 and 5-HT1A receptors, as well as potent 5-HT2A antagonist effects. The purpose of this systematic review is to provide a detailed overview available evidence on its role in treating MDD, based on all clinical publications in the English language between January 1, 2014 and April 30, 2017 identified from PubMed, Google Scholar, Scopus, Web of Science, PsycINFO, and ClinicalTrials.gov. Two primary pivotal trials are reviewed in detail and a further 10 supporting reviews and open-label studies are discussed. Brexpiprazole is compared with aripiprazole according to pharmacologic and clinical activities. Overall, this appears to be a useful antidepressant adjunctive therapy with a favorable side effect profile and comparable efficacy with existing agents.
Keywords
Introduction
Major depressive disorder (MDD) imparts a considerable burden on social relationships, work productivity, and overall quality of life. 1 In 2017, depressive disorders were ranked as the leading cause of disability worldwide. 2 This highlights the importance of identifying better targeted, rapidly acting treatments with higher rates of response and remission.3,4
Despite advances in the pharmacologic treatment of MDD, only 50% to 60% of depressed patients respond to first-line antidepressant treatment (ADT). 5 Several strategies have been implemented following inadequate response to first-line ADT, including dose escalation, switching to another antidepressant, or augmenting with another medication. 6 Among these different approaches, the evidence is strongest for augmentation with atypical antipsychotics, specifically aripiprazole, quetiapine, and risperidone.6–8 However, early discontinuation of treatment due to adverse effects and to concern about long-term safety have restricted their use. 9 Therefore, the emergence of a novel antipsychotic agent with at least comparable efficacy and potentially a more favorable tolerability and safety profile would help to reduce this current unmet need. Here, we review current evidence that brexpiprazole is a potential alternative for adjunctive antidepressant therapy.
Brexpiprazole received its first approval in July 2015 by the US Food and Drug Administration as monotherapy for schizophrenia and as an adjunctive therapy for MDD in an adult population. 10 In 2017, brexpiprazole was also approved in Canada and Australia as a treatment for schizophrenia in adults. The efficacy of brexpiprazole for the treatment of schizophrenia was supported by 2 randomized, double-blind, placebo-controlled, phase 3 trials,11,12 which are beyond the scope of this review. Here, we summarize the pharmacologic properties of brexpiprazole and examine the evidence to support its efficacy and tolerability as an adjunctive therapy to antidepressants in MDD.
Method
We conducted a systematic literature review focusing on English-language, peer-reviewed publications in PubMed, Google Scholar, Scopus, Web of Science, PsycINFO, and ClinicalTrials.gov from January 1, 2014 to April 30, 2017. All searches contained the terms major depressive disorder, inadequate treatment response to antidepressant treatment, brexpiprazole, major depressive disorder-related comorbidities, comparison of brexpiprazole and aripiprazole. Randomized controlled trials, open-label trials, and post hoc analyses were included. The primary focus in most trials was the mean change in total Montgomery-Åsberg Depression Rating Scale (MADRS). Bibliographies from these publications were searched to identify additional articles of interest.
Pharmacodynamics
The precise mechanism of action of brexpiprazole for clinical use in MDD is currently unknown but is likely related to its activity at D2, 5-HT1A, and 5-HT2A receptors. 13 Brexpiprazole has demonstrated high affinity (binding affinity [Ki] <1 nM) to D2, 5-HT1A, 5-HT2A, α1B, and α2C receptors; partial agonist activity at D2 and 5-HT1A receptors; and potent antagonist activity at 5-HT2A, α1B, and α2C receptors. 13 Brexpiprazole also displays affinity (Ki < 5 nM) for 5-HT2B, 5-HT7, α1A, α1D (antagonist), and D3 (partial agonist) receptors, as well as moderate and very low affinities for histamine H1 and muscarinic M1 receptors, receptively. 13
Comparison with aripiprazole
Similar to aripiprazole, brexpiprazole’s preclinical profile is consistent with its low potential to induce extrapyramidal symptoms (EPS) based on the catalepsy test. 14
According to proposed Neuroscience Based Nomenclature, both drugs are classified as dopamine multifunctional agents. 15 However, as a serotonin-dopamine activity modulator, brexpiprazole has higher affinity for 5-HT1A than D2 receptors, whereas the reverse is true for aripiprazole. 13 Regarding the D2 receptor, brexpiprazole has about 3 times the affinity and approximately three-quarters of the intrinsic activity of aripiprazole at this receptor, 13 suggesting a better tolerability profile in terms of less agitation and less likelihood of causing akathisia. 16 In addition, higher affinity for 5-HT2A receptors compared with aripiprazole might suggest a lower rate of akathisia with brexpiprazole 17 (see Table 1). Furthermore, brexpiprazole has greater affinities for 5-HT1A, α1B, and α2C receptors compared with aripiprazole, 13 making brexpiprazole more similar to other second-generation antipsychotics than aripiprazole: this might have clinical implications in terms of improved efficacy, although this remains to be established. 19 The main structural differences between brexpiprazole and aripiprazole are shown in Figure 1.
Binding affinities of brexpiprazole and aripiprazole.
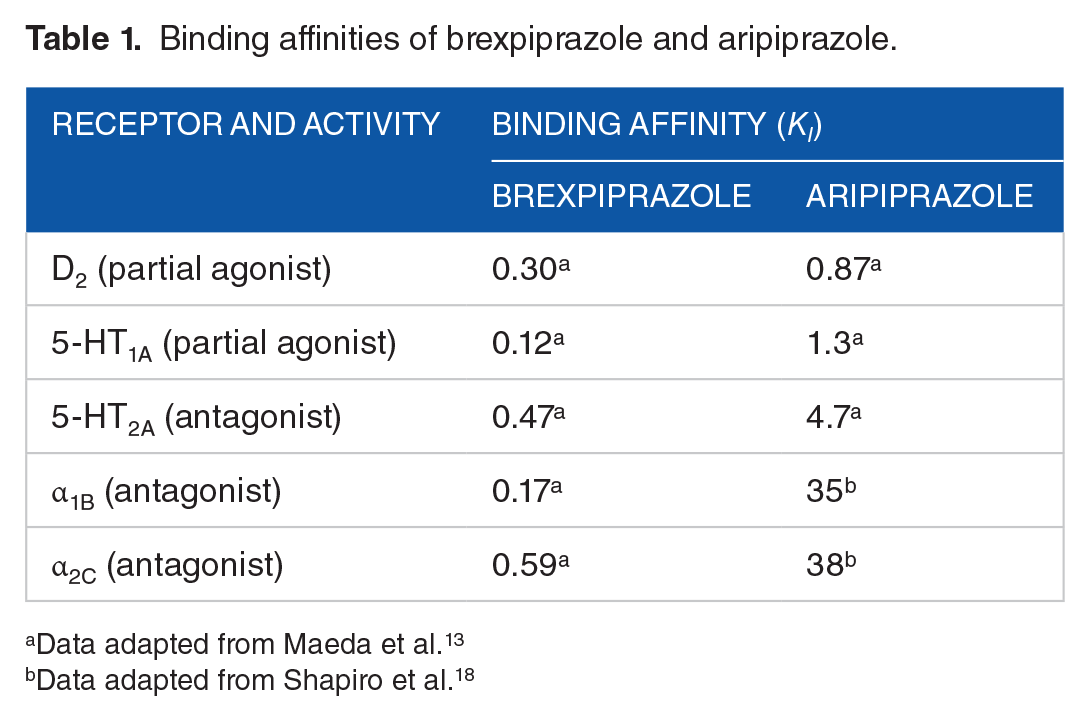
Data adapted from Maeda et al. 13
Data adapted from Shapiro et al. 18
Structural formulae of aripiprazole and brexpiprazole.
Pharmacokinetics
Following a single oral dose administration, the peak plasma concentration of brexpiprazole occurs within 4 hours, with 95% absolute bioavailability and steady-state concentrations occurring within 10 to 12 days of once-daily oral dosing. 20 Brexpiprazole exposure increases in a dose-dependent manner and is not significantly affected by concomitant intake of a standard high-fat meal; therefore, brexpiprazole can be taken with or without food. 20 Brexpiprazole has high protein binding (>99%) that is not affected by hepatic or renal impairment or by the administration of warfarin, diazepam, or digitoxin. 20
The metabolism of brexpiprazole is mediated by the hepatic cytochromes CYP3A4 and CYP2D6. 20 The major metabolite, DM-3411, represents 23% to 48% of brexpiprazole exposure in plasma at steady state. However, DM-3411 has no role in the therapeutic effects of brexpiprazole. 20
After a single oral dose of radiolabeled brexpiprazole, around 25% of the dose was recovered in urine and 46% was recovered in feces. 20 Less than 1% and approximately 14% of unchanged brexpiprazole was recovered in the urine and feces, respectively. The terminal elimination half-lives of oral brexpiprazole and DM-3411, following multiple once-daily doses, were 91 and 86 hours, respectively. 20
Compared with CYP2D6 normal extensive metabolizers and patients with normal hepatic and renal function, brexpiprazole concentration is higher in CYP2D6 poor metabolizers and in patients with moderate to severe hepatic impairment (Child-Pugh score ⩾7) or moderate to end-stage renal impairment (creatinine clearance <60 mL/min). 20 Concomitant use of brexpiprazole with a strong inhibitor of (1) CYP3A4 (eg, clarithromycin), (2) CYP2D6 (eg, fluoxetine), or (3) the combination of a strong/moderate CYP3A4 inhibitor with a strong/moderate CYP2D6 inhibitor increases the concentration of brexpiprazole. Coadministration of brexpiprazole with a strong CYP3A4 inducer (eg, rifampicin) may result in decreased brexpiprazole concentration. 20
Therapeutic Efficacy of Adjunctive Brexpiprazole in MDD
The approval of adjunctive oral brexpiprazole in patients with MDD is based largely on 2 pivotal phase 3, randomized, double-blind trials, conducted between 2011 and 2013, known as “PYXIS” and “POLARIS”21,22 (Figure 2).
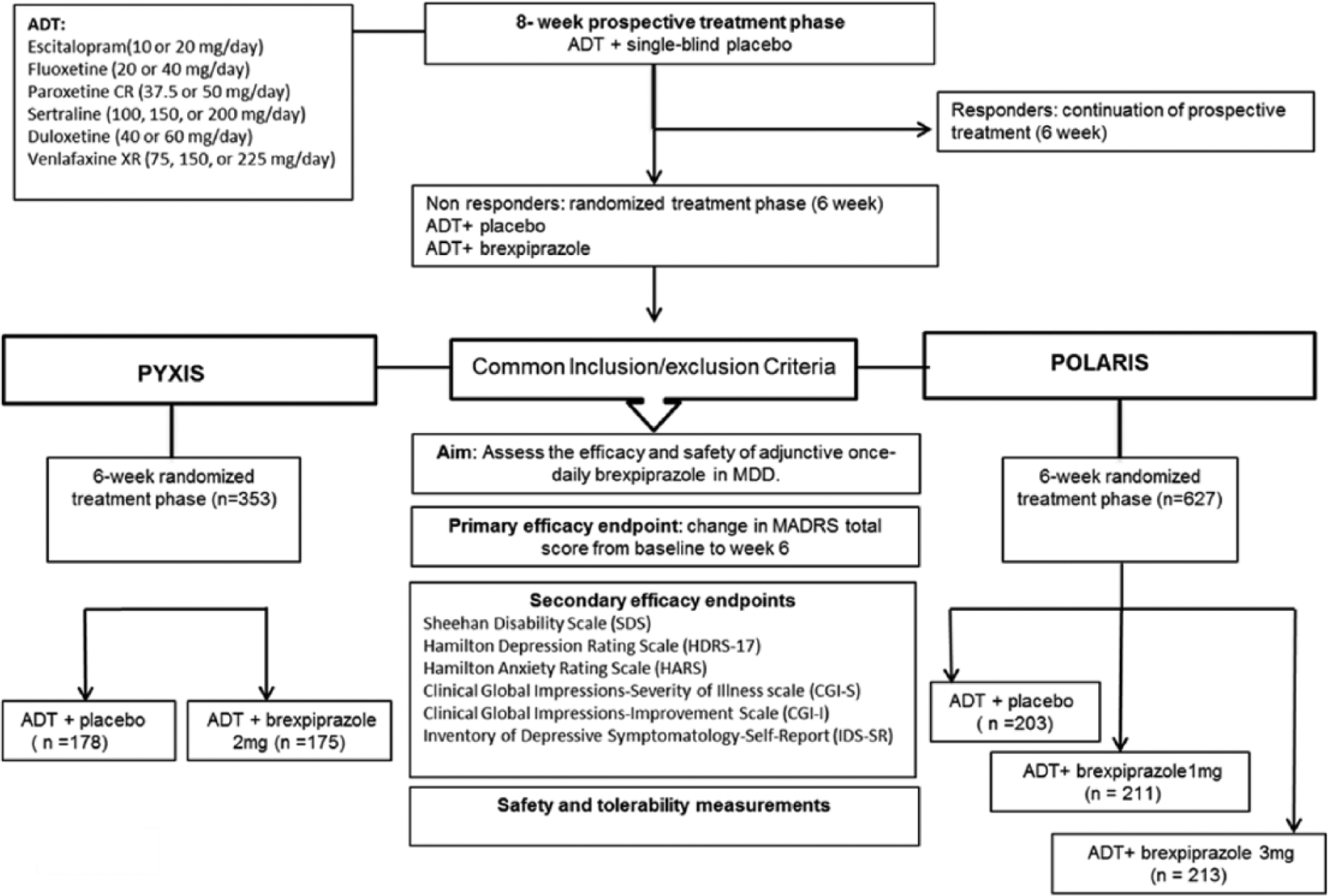
Brexpiprazole phase 3 pivotal trials: study design.
Both trials evaluated the short-term efficacy and tolerability of adjunctive brexpiprazole in adult patients with MDD. Eligible participants demonstrated inadequate responses to adequate doses of up to 3 historically reported ADTs. They also failed to respond to a single-blind, 8-week, open-label prospective trial of one of several antidepressants, before being eligible to participate in the 6-week double-blind randomized treatment phase (see the works by Thase et al21,22 for further details of inclusion and exclusion criteria). During the randomized treatment phase, participants received either an adjunctive fixed dose of brexpiprazole or placebo: the “PYXIS” study evaluated adjunctive brexpiprazole 2 mg 21 once daily, whereas the “POLARIS” study assessed the efficacy of adjunctive once-daily brexpiprazole 1 and 3 mg. 22 A protocol amendment was made during the enrollment phase for both trials to provide more rigorous criteria for inadequate response during the 8 weeks of prospective treatment, to exclude subjects who were not consistently nonresponders to ADT (see Table 2 for eligibility criteria).
Eligibility criteria for the double-blind randomized treatment phase in the “PYXIS” and “POLARIS” trials.

Abbreviations: CGI-I, Clinical Global Impressions-Improvement Scale; HDRS-17, 17-item Hamilton Depression Rating Scale; MADRS, Montgomery-Åsberg Depression Rating Scale.
Initial eligibility criteria for the randomized phase.
Additional criteria after the revision of the protocol.
In total, the study population consisted of 353 patients in the “PYXIS” trial and 627 patients in “POLARIS,” based on the final protocol.19,20 Participants in both trials had similar baseline demographic and clinical characteristics; the mean age of these patients was 45 years, and mean MADRS total score at baseline was 26; reflecting patients with MDD with moderate severity.
Primary efficacy
After 6 weeks of adjunctive brexpiprazole, 2 and 3 mg doses (but not brexpiprazole 1 mg), significantly separated from placebo, based on MADRS total score21,22 (see Table 3).
Mean change from baseline to week 6 in the Montgomery-Åsberg Depression Rating Scale total score.

Abbreviations: ADT, antidepressant treatment; CI, confidence interval; LS, least squares.
P < .01; **P < .001 vs ADT + placebo.
Secondary efficacy end points
All dosages of adjunctive brexpiprazole (1, 2, and 3 mg) demonstrated significant separation from placebo on Sheehan Disability Scale (SDS) total score, which was a key secondary efficacy variable.21,22 Scores from baseline to week 6 on social life and family life subscales significantly favored brexpiprazole treatment groups over placebo groups.21,22 However, none of the brexpiprazole groups had significant impact on work and school functioning21,22 (see Figures 3 and 4).
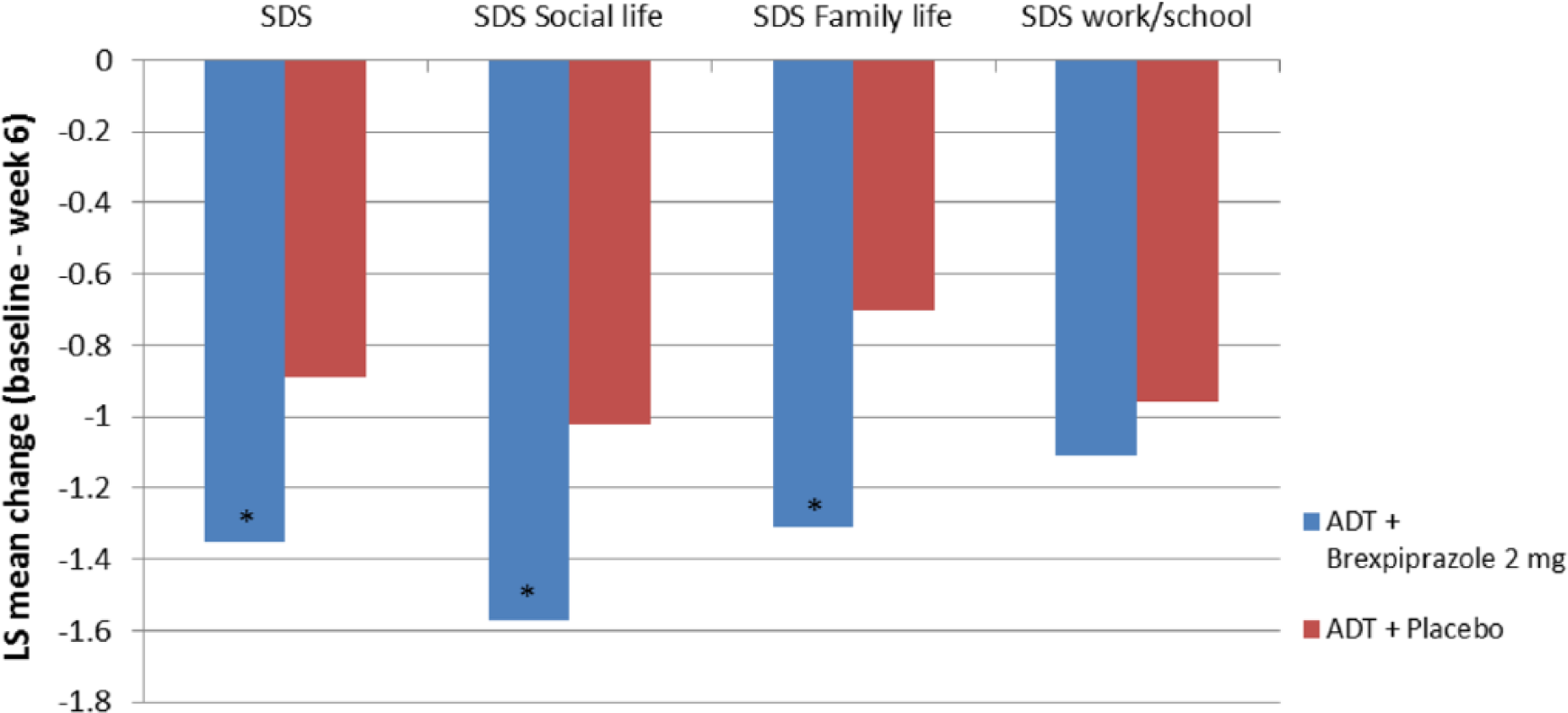
Mean change from baseline to week 6 in the SDS total and subscale scores in the “PYXIS” study. ADT indicates antidepressant treatment; LS, least squares; SDS, Sheehan Disability Scale.
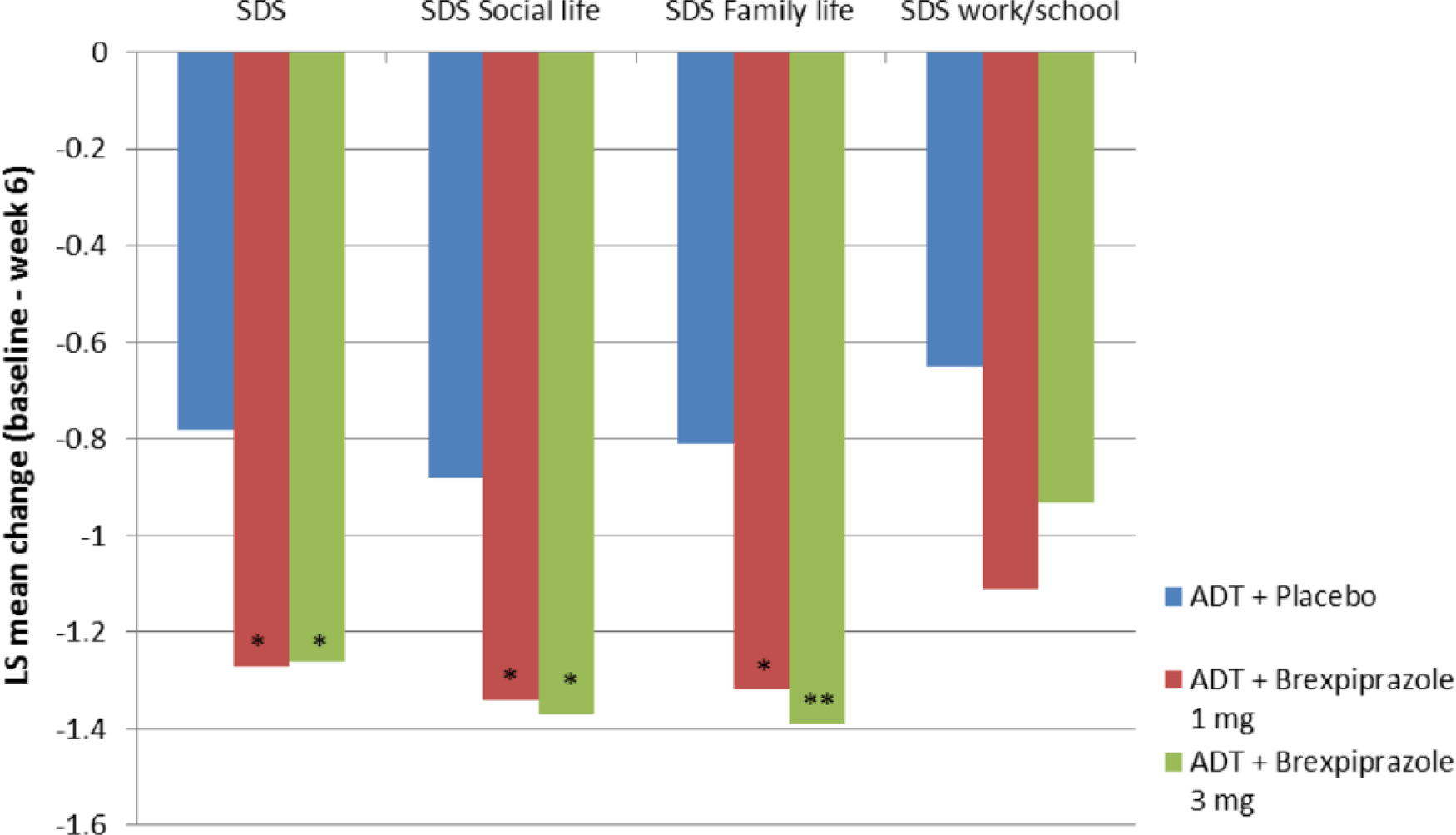
Mean change from baseline to week 6 in the SDS and subscales in the “POLARIS” study. ADT indicates antidepressant treatment; LS, least squares; SDS, Sheehan Disability Scale.
Adjunctive brexpiprazole 2 and 3 mg manifested significant reduction in mean scores from baseline compared with placebo on the following secondary outcome measures: 17-item Hamilton Depression Rating Scale (HDRS-17), Hamilton Anxiety Rating Scale (HARS), Clinical Global Impressions-Improvement (CGI-I) and CGI-Severity (CGI-S) at week 6.21,22 In contrast, adjunctive brexpiprazole 1 mg separated from placebo only on CGI-I. 22 Patients who received adjunctive brexpiprazole 1 or 3 mg showed significant improvement in the Inventory of Depressive Symptomatology-Self Report (IDS-SR) scale scores compared with placebo 22 but not among patients in the “PYXIS” study 21 (see Table 4).
Mean change from baseline to week 6 in other secondary efficacy end points.
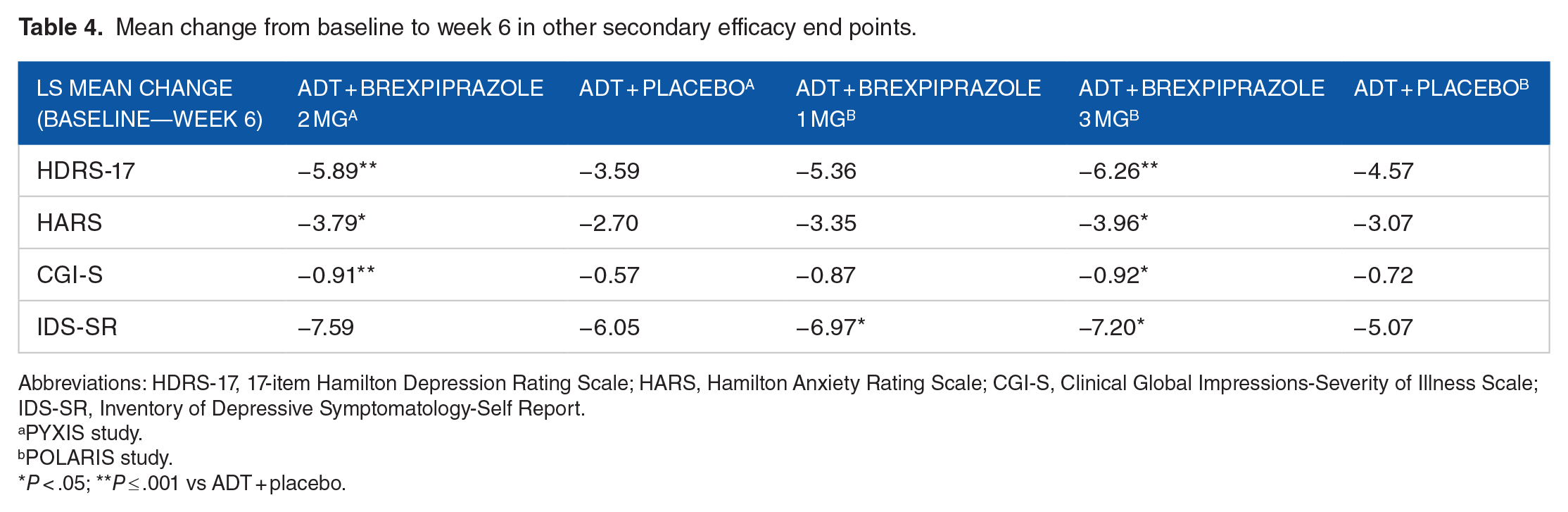
Abbreviations: HDRS-17, 17-item Hamilton Depression Rating Scale; HARS, Hamilton Anxiety Rating Scale; CGI-S, Clinical Global Impressions-Severity of Illness Scale; IDS-SR, Inventory of Depressive Symptomatology-Self Report.
PYXIS study.
POLARIS study.
P < .05; **P ⩽ .001 vs ADT + placebo.
Regarding responder rates, there was a higher percentage of responders at end point (as defined by ⩾50% reduction in MADRS score) in the adjunctive brexpiprazole treatment groups at all doses compared with ADT + placebo groups, and there was a higher proportion of responders (as defined by as core of ⩾3 on CGI-I) in the brexpiprazole 2 and 3 mg treatment groups but not in the brexpiprazole 1 mg or placebo groups.21,22 However, none of the brexpiprazole treatment groups separated from placebo on remission (defined as patients with MADRS total score ⩽10 and ⩾50% reduction in MADRS from baseline)21,22 (see Figure 5A and B). Pooling the data from both studies, the difference between brexpiprazole and placebo responders was 23.2% vs 14.5%, resulting in a number needed to treat of 12 (95% confidence interval [CI]: 8-26). 23
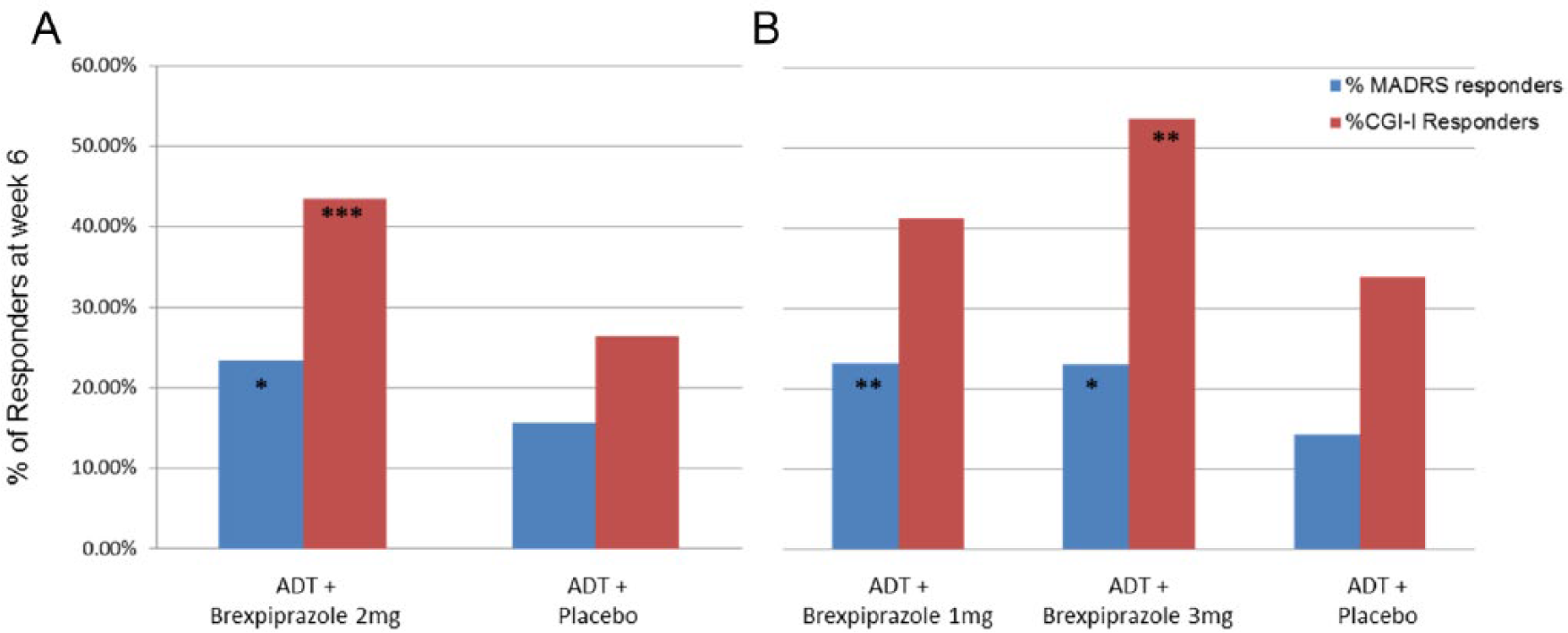
Percentage of responders at week 6 (efficacy population per final protocol): (A) PIXYS study and (B) POLARIS study. ADT indicates antidepressant treatment. CGI-I indicates Clinical Global Impressions-Improvement Scale; MADRS, Montgomery-Åsberg Depression Rating Scale.
Safety and Tolerability of Adjunctive Brexpiprazole in MDD
In this section, we highlight the findings among all patients who received at least one dose of brexpiprazole during the randomized treatment phase (safety population). All doses of adjunctive brexpiprazole were generally well tolerated, and most treatment-emergent adverse events (TEAEs) were mild to moderate in severity21,22 (see Table 5).
Most common treatment-emergent adverse events associated with adjunctive brexpiprazole 1, 2, and 3 mg.

Abbreviations: ADT, antidepressant treatment; TEAEs, treatment-emergent adverse events.
Safety population (all patients receiving at least one dose of brexpiprazole during randomized treatment phase).
POLARIS study.
PIXYS study.
Pooled data adapted from Nelson et al. 23
Moderate weight gain was noted as one of the most common TEAEs across both trials, and mean increases in baseline bodyweight at week 6 were significantly higher among adjunctive brexpiprazole groups compared with placebo groups (see Figure 6). Bodyweight gain of ⩾7% was seen in 4.9%, 4.8%, and 1.8%, respectively, in groups treated with adjunctive brexpiprazole 1, 2, and 3 mg, compared with 2.6% among placebo group in the “PYXIS” and 0.9% among placebo group in the “POLARIS.”21,22
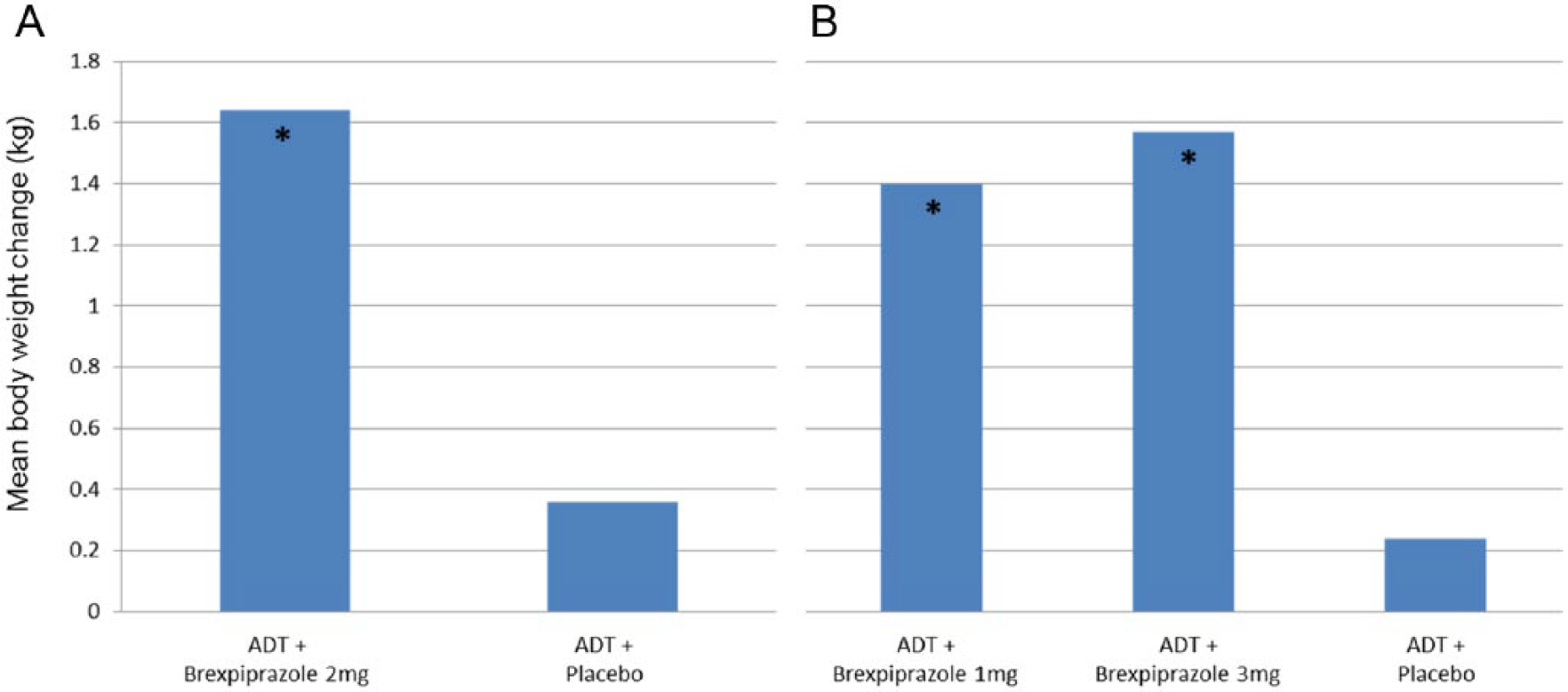
Mean bodyweight change at week 6: (A) PIXYS study and (B) POLARIS study. ADT indicates antidepressant treatment.
Regarding EPS, akathisia was most frequently reported across both trials, with a higher incidence among brexpiprazole recipients than in placebo groups (4.4%, 7.4%, and 13.5% for brexpiprazole 1, 2, and 3 mg, compared with 1% and 2.3% for placebo groups in the PYXIS and POLARIS, respectively). Adjunctive brexpiprazole 2 or 3 mg was associated with small but statistically significant changes in 2 of the 3 rating scales used to evaluate EPS21,22 (see Figure 7). Analysis of pooled data from these 2 trials concluded that akathisia incidence was dose related and usually occurred early in treatment (within 2-4 weeks). 24
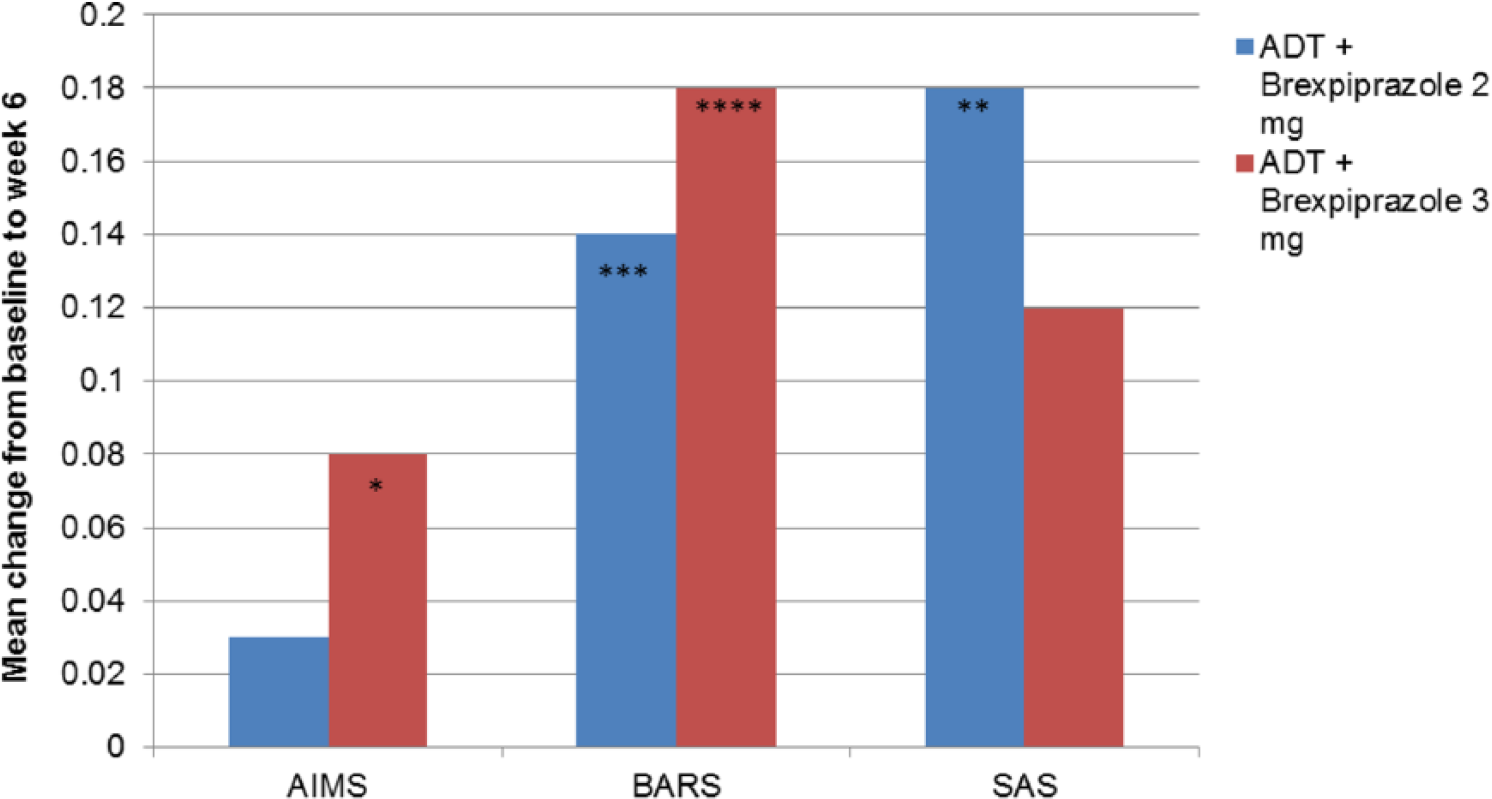
Mean change in extrapyramidal symptom rating scales at week 6. ADT indicates antidepressant treatment; AIMS, Abnormal Involuntary Movement Scale; BARS, Barnes Akathisia Rating Scale; SAS, Simpson-Angus Scale.
Overall, adjunctive brexpiprazole resulted in low levels of sedating or activating side effects.21,22 Also of note, none of the 3 doses of adjunctive brexpiprazole was associated with clinically relevant adverse effects on prolactin, metabolic, or liver parameters. Nor were there differences between brexpiprazole and placebo groups in electrocardiogram and vital signs.21,22
There were no reports of emergent sexual dysfunction in either trial, and there was no incidence of emergent suicidal ideation in either brexpiprazole or placebo groups over the 6-week period.21,22 In a pooled analysis of safety data from both studies, a total of 3% in the brexpiprazole groups and 1% in the placebo groups discontinued due to adverse reactions, 20 equating to a number needed to harm (NNH) of 53 (95% CI: 30-235). 23
Preliminary data on long-term safety and tolerability of adjunctive brexpiprazole in MDD were pooled from 2 open-label, flexible-dose 52-week trials 24 (data cutoff date of May 15, 2015 as one study was still ongoing). The 2 most frequently reported TEAEs were weight gain (25.5%) and akathisia (10%). Weight increase of ⩾7% was reported in 30.3%, suggesting that adjunctive brexpiprazole is associated with a moderate risk of weight gain during continuation and maintenance treatment. Throughout these 2 long-term studies, a total of 14% patients discontinued brexpiprazole treatment due to TEAEs, 3.6% of these patients withdrew because of weight gain, and 0.9% due to akathisia. 24
Switching to Adjunctive Brexpiprazole
An open-label trial explored the effect of adjunctive brexpiprazole in patients with MDD, who failed to respond to at least one ADT monotherapy and had an inadequate response to ⩾1 adjunctive (augmentation with aripiprazole, quetiapine, or stimulants) or combination therapy. 25 Patients discontinued their adjunctive or combination medication at least 5 days before the start of a 6-week treatment trial with brexpiprazole as an adjunct to their current ADT. 25 In total, 51 subjects (aged 18-65 years), who completed the study, showed improvement in depressive symptoms, as evidenced by significant reduction on MADRS total score from baseline, irrespective of prior adjunctive or combination therapy (all P’s < .0001). In addition, overall improvement in disability and cognitive and physical functioning with brexpiprazole based on significant reduction in all SDS domains (P < .0001) and Massachusetts General Hospital-Cognitive and Physical Functioning Questionnaire (P < .0001) were reported. 25 Fatigue was reported in 14.8% and akathisia in 8.2% of patients. None of the patients had ⩾7% increase in bodyweight. 25
Adjunctive Brexpiprazole in MDD-Related Comorbidities
Effectiveness, safety, and tolerability of adjunctive brexpiprazole in patients with MDD experiencing common MDD-related comorbidities were evaluated in combined subanalyses of “PYXIS,” “POLARIS,” and several open-label trials.
MDD with anxious distress
In post hoc analyses of the “PYXIS” and “POLARIS” trials, 550 (55.6%) subjects were classified according to criteria for MDD with anxious distress at baseline. At week 6, patients with MDD with anxious distress who received adjunctive brexpiprazole 2 or 3 mg (but not brexpiprazole 1 mg) significantly separated from placebo groups based on MADRS total score (P < .01). 26 However, in patients without anxious distress, adjunctive brexpiprazole 1 and 3 mg were associated with significant reduction in MADRS total score compared with placebo (P < .01 and .05, respectively), but there was no significant difference for adjunctive brexpiprazole 2 mg (P = .1101). 26 The incidence of akathisia and other activating side effects such as restlessness and agitation was similar in patients with and without anxious distress. 26
Anxiolytic effects of adjunctive brexpiprazole were explored in a small open-label, flexible-dose study involving patients with MDD, aged 18 to 65 years, who had inadequate response to ADT monotherapy and concurrent anxiety (a total score of ⩾20 on HARS at baseline). 27 After 6 weeks of therapy with adjunctive brexpiprazole 1 to 3 mg and target dose 2 mg daily (mean prescribed dose 2.1 mg daily), depressive and anxiety symptoms improved markedly as demonstrated by significant reduction on MADRS and HARS total scores (P < .0001). Patients also showed improvement on SDS and subscales (social life, family life, and work/school life). 27
MDD with irritability
The effect of brexpiprazole on comorbid irritability was evaluated in a post hoc analysis of 811 (82%) patients in the “PYXIS” and “POLARIS” trials. 28 Irritability (score of ⩾1 on the Irritable Mood item 6 of the IDS-SR) was associated with greater illness severity as evidenced by higher MADRS total score, compared with the nonirritable subgroup. 28 In patients with irritability, all dosages of adjunctive brexpiprazole (1, 2, and 3 mg) demonstrated higher efficacy than adjunctive placebo, as shown by significant reduction in MADRS total score from baseline to week 6 (P < .01). 28 Although adjunctive brexpiprazole 3 mg (but not brexpiprazole 1 or 2 mg) significantly separated from placebo on MADRS total score in patients with higher level of irritability (IDS-SR item 6 score ⩾2), these authors caution against the significance of these results due to uneven number of patients with and without irritability. 28
In a separate exploratory open-label trial, 54 adult patients with MDD who had an inadequate response to ADT and met criteria for irritability, defined by a score of ⩾2 on item 6 of a 30-item IDS clinician version (IDS-C30), received adjunctive brexpiprazole (target dose was 3 mg, and mean prescribed dose was 2.4 mg daily) for 6 weeks. 29 Irritability significantly improved as measured on self-reported scales and clinician-rated IDS-C30 (P < .0001), as well as depressive symptoms demonstrated by significant reduction on MADRS total score at week 6. In addition, patients showed improvement in impulsivity, anger, cognition, and physical functioning based on other patient-rated scales (P < .0001). Following discontinuation of adjunctive brexpiprazole, patients were followed up for 4 weeks during which they continued their ADT. During this period, patients showed relapse of their depressive and irritability symptoms but were not to same severity level reported at baseline. 29
MDD with sleep disturbances
Effect of adjunctive treatment with brexpiprazole on sleep disturbances in patients with MDD and inadequate response to ADT has also been evaluated in 44 patients who received open-label treatment for 8 weeks with brexpiprazole (target dose 3 mg daily) + ADT. 29 Eligible subjects scored ⩾8 on the Insomnia Severity Index and had <85% sleep efficiency during 2 consecutive nights of polysomnography (PSG) monitoring. 30 After 8 weeks of adjunctive treatment, patients reported improvement in sleep, daytime alertness, cognition, and physical functioning as supported by PSG and several measurements. Both MADRS total score and CGI-S score decreased significantly at week 8 reflecting improvement in depressive symptoms (P < .05). 30
Adjunctive Brexpiprazole in Special Populations
Young adults with MDD who are at work/school
A 12-week open-label trial enrolled 47 young adults (aged 18-35 years), with MDD who had an inadequate response to ADT, and were actively engaged in a school/work setting. 31 Adjunctive brexpiprazole 1 to 3 mg (target dose 2 mg) produced significant improvement in depressive symptoms, manifested by significant reduction on MADRS total score (P < .0001), and supported by favorable secondary outcomes on measures such as CGI-S, HDRS-17, and Kellner Symptom Questionnaire. In addition, concurrent improvements on work productivity, social, and occupational functioning were reported, as evidenced by significant reduction from baseline on SDS (absolute score and all subscale scores) (P < .0001), and other patient-rated instruments such as Work Limitations Questionnaire and Social Adaptation Self-Evaluation Scale (SASS). 31 Weight gain ⩾7% from baseline occurred in 10.5% of patients, akathisia was reported in 6.4%, and changes from baseline to week 12 for Simpson-Angus Scale (SAS), Abnormal Involuntary Movement Scale (AIMS), and Barnes Akathisia Rating Scale (BARS) were small and considered not to be clinically relevant. 31
Geriatric and pediatric populations
The authors of a report on safety, tolerability, and pharmacokinetics of adjunctive brexpiprazole in the treatment of elderly patients (aged 70-85 years) diagnosed with MDD concluded that pharmacokinetic properties of a single oral dose of brexpiprazole (maximum daily dose was 3 mg for 14 days) were generally similar to those in adult populations. 20 An open-label, flexible-dose trial evaluated the therapeutic effectiveness and long-term safety and tolerability of adjunctive brexpiprazole (1-3 mg/d) in 132 elderly patients (⩾65 years) diagnosed with MDD who had inadequate response to ⩾1 ADT. After 26 weeks of treatment with adjunctive brexpiprazole 1-3 mg (mean prescribed dose 1.8 mg/d), participants showed clinically meaningful improvement in their depressive symptoms and social function, as assessed by MADRS, CGI-S, and SASS. The most common TEAEs were as follows: fatigue (15.2%), restlessness (12.9%), bodyweight gain of ⩾7% (12.3%), and increased appetite (9.8%). The authors concluded that adjunctive brexpiprazole was generally well tolerated, and TEAEs were consistent with those reported in the “PYXIS” and “POLARIS” trials.
Safety and effectiveness of adjunctive brexpiprazole in pediatric patients have not been established, although in the study of brexpiprazole in young adults at work or school (aged 18-35 years), there were no initial or emergent suicidal thoughts or behaviour. 31 Nevertheless, the brexpiprazole prescribing information includes a generic warning about the risk of increased suicidal thoughts and behaviors among antidepressant-treated patients aged 24 years or younger. 20
Comparison Between Brexpiprazole and Aripiprazole in MDD Trials
To our knowledge, no head-to-head trials comparing the efficacy and safety of brexpiprazole versus aripiprazole have been reported in patients with MDD. Therefore, in this section, we can only compare pivotal clinical trials for each drug versus placebo, and this comparison is limited by methodological differences (brexpiprazole trials assessed efficacy and safety of fixed doses, whereas aripiprazole trails were dose flexible). In terms of efficacy, after 6 weeks of adjunctive therapy, both agents significantly separated from placebo, based on MADRS total score (see Table 6).
Mean change from baseline to week 6 in the Montgomery-Åsberg Depression Rating Scale total score (findings from different trials).

Abbreviation: ADT, antidepressant treatment.
P < .01; **P ⩽ .001 vs ADT + placebo.
Although akathisia was the most common TEAE reported with both agents, the incidence was substantially lower among patients receiving adjunctive brexpiprazole21,22 compared with those treated with adjunctive aripiprazole.32,33 Adjunctive treatment with either of these agents was associated with greater weight gain than placebo21,22,32,33 (see Table 7). Neither brexpiprazole nor aripiprazole displayed clinically relevant adverse effects on sexual function, prolactin, or metabolic parameters.21,22,32,33
Incidence of akathisia and % of subjects with ⩾7% bodyweight gain among patients treated with adjunctive brexpiprazole or aripiprazole (findings from different trials).
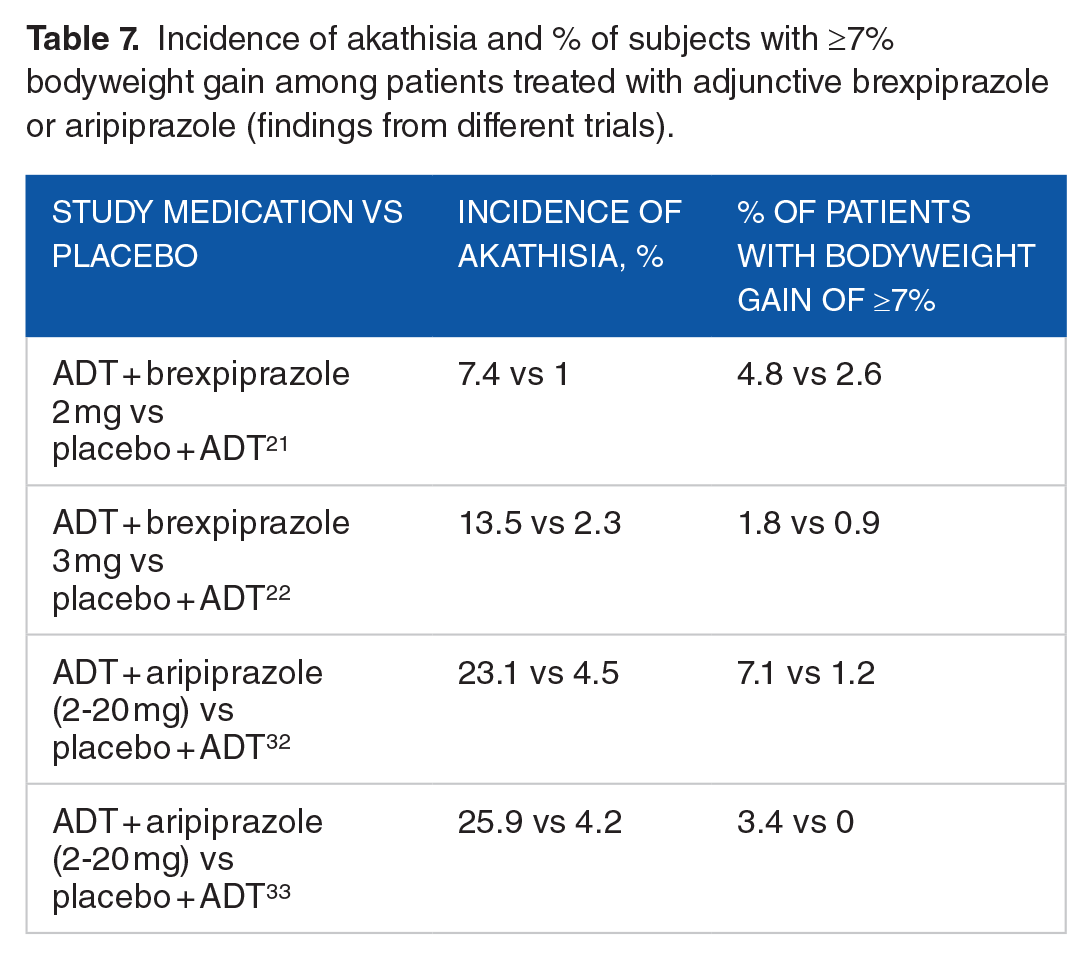
Lower incidence of akathisia with brexpiprazole compared with aripiprazole in MDD was noted by Citrome, 34 yielding a NNH of 5 for aripiprazole vs 15 for brexpiprazole. This is further supported by findings from open-label, long-term safety and tolerability trials. Akathisia occurred in 10% of patients treated with adjunctive brexpiprazole (mean dose: 1.6 mg/d) over 52 weeks 24 (section “Safety and tolerability of Adjunctive Brexpiprazole in MDD”). This compares with 26.2% of patients treated with adjunctive aripiprazole (mean dose: 10.1 mg/d) for up to 52 weeks. 35 In addition, mean increase in bodyweight from baseline to week 52 was slightly lower with adjunctive brexpiprazole compared with adjunctive aripiprazole (3.2 vs 4.4 kg).24,35 At 52 weeks, the percentage of patients with ⩾7% weight gain was 30.3% with adjunctive brexpiprazole 24 (section “Safety and tolerability of Adjunctive Brexpiprazole in MDD”), which is similar to that previously reported for adjunctive aripiprazole (36.6%). 35
Limitations
This review is limited by the relatively small number of adequately powered clinical trials and the absence of trials including an active reference compound. The relatively recent availability of brexpiprazole in only a few countries (United States, Canada, Australia, and Japan) as of August 2017 limits evidence from “real-world” clinical experience. There are also inherent complications in interpreting the efficacy and tolerability of any pharmaceutical agent in combination with other drugs (in this case, antidepressants) with multiple pathways of drug metabolism regulated by distinct genetic polymorphisms.
Conclusions
Atypical antipsychotics should be considered as a heterogeneous group of agents, each with a unique pharmacodynamic and pharmacokinetic profile, which is reflected in disparate levels of effectiveness and tolerability. There is a persistent, unmet need for effective and well-tolerated augmentation strategies for patients with MDD who experience an inadequate response to standard ADTs. Brexpiprazole offers a novel option for such patients, with approved efficacy and safety based on current available research data. However, head-to-head randomized trials involve brexpiprazole, and other agents indicated for adjunctive treatment of MDD would provide valuable comparative data to guide physicians in clinical settings.
Footnotes
Acknowledgements
The authors acknowledge continuing research support from the Ontario Brain Institute to the Canadian Biomarker Integration Network in Depression. The opinions, results and conclusions are those of the authors and no endorsement by the Ontario Brain Institute is intended or should be inferred. They also thank Ms Kishwar Ali for her creation of figures and tables throughout the manuscript.
Peer review:
Three peer reviewers contributed to the peer review report. Reviewers’ reports totaled 623 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Al Shirawi receives fellowship funding from the Ministry of Health, Sultanate of Oman. Dr Kennedy has received research funding or honoraria from the following sources: Allergan, AstraZeneca, BMS, BrainCells Inc., Brain Canada, Clera, CIHR, Eli Lilly, Janssen, Lundbeck, Lundbeck Institute, OMHF, Ontario Brain Institute, Otsuka, Pfizer, Servier, St. Jude Medical, Sunovion, and Xian Janssen.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MAS and SHK conceived the concepts, conducted the review and jointly developed the structure of the paper. MAS wrote the first draft of the manuscript. NEE and MAS independently carried out literature reviews. All authors contributed to the subsequent revisions of the manuscript and approved the final version.
