Abstract
Chronic hepatitis C is a common cause of liver-related morbidity and mortality. Ledipasvir/sofosbuvir is a combination of 2 direct-acting antiviral agents that has been approved for use in patients with genotype 1, 4, 5, or 6. This approval is based on multiple phase 3 studies in which the rate of sustained virologic response exceeded 90% for 12 or 24 weeks of treatment, depending on the patient population. For some patients, the addition of ribavirin is required. Ledipasvir/sofosbuvir is well tolerated, with the most commonly reported adverse events being fatigue and headaches.
Keywords
Introduction
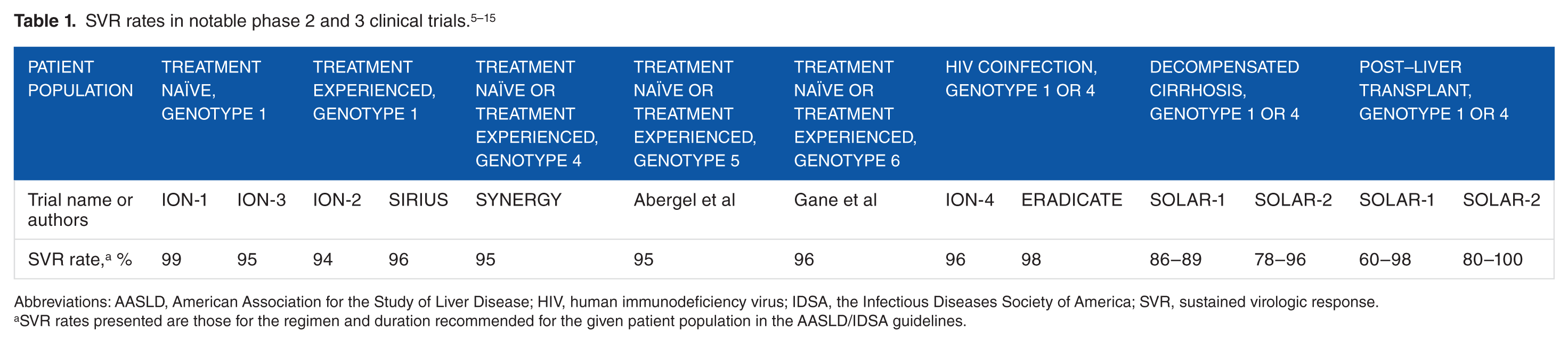
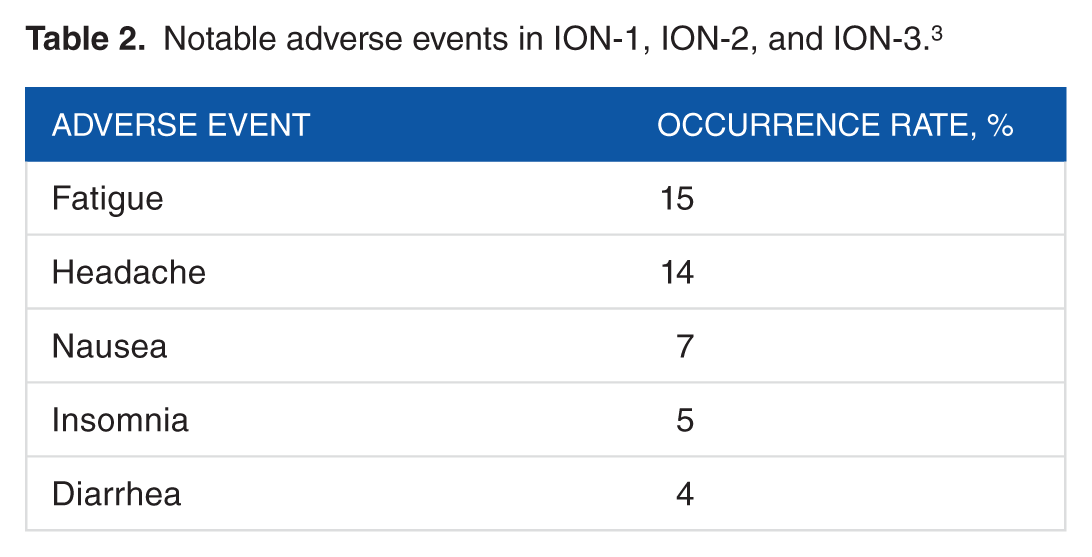
Chronic hepatitis C virus (HCV) infection affects about 170 million people globally and can lead to significant morbidity and mortality, including cirrhosis and its complications (variceal hemorrhage, ascites, and hepatic encephalopathy), as well as hepatocellular carcinoma. 1 Previous HCV treatment regimens involved multiple injections of interferon in combination with multiple ribavirin pills per day; treatment durations of up to 48 weeks; significant adverse events, including but not limited to flu-like symptoms, psychiatric symptoms, and hematologic abnormalities; and less-than-optimal success rates. 2 In the past several years, direct-acting antivirals (DAAs), which target specific structural and functional viral proteins, have significantly increased the chances of treatment success. 2 One such DAA coformulated combination is ledipasvir/sofosbuvir. Ledipasvir inhibits the formation of NS5A replication complex, and sofosbuvir inhibits the NS5B polymerase enzyme. 3 The dual mechanism of action serves to effectively terminate viral replication. The combination tablet is indicated to be taken once daily between 12 and 24 weeks of treatment, with or without ribavirin. 3 The goal of chronic HCV treatment is sustained virologic response (SVR), defined as an undetectable HCV viral load at least 12 weeks after the completion of treatment. 4 The American Association for the Study of Liver Disease (AASLD) and the Infectious Diseases Society of America (IDSA) released guidelines that place ledipasvir/sofosbuvir as a preferred treatment option for multiple patient populations with chronic hepatitis C. 4 The objective of this article is to review the efficacy and safety of ledipasvir/sofosbuvir for chronic HCV infection treatment in 6 phase 2 and 4 phase 3 trials. The efficacy rates are presented in Table 1, whereas the most common adverse events are presented in Table 2.
Abbreviations: AASLD, American Association for the Study of Liver Disease; HIV, human immunodeficiency virus; IDSA, the Infectious Diseases Society of America; SVR, sustained virologic response.
SVR rates presented are those for the regimen and duration recommended for the given patient population in the AASLD/IDSA guidelines.
Notable adverse events in ION-1, ION-2, and ION-3. 3
Clinical Studies
Studies in treatment-naïve genotype 1 patients
Ledipasvir/sofosbuvir was studied in 2 phase 3 randomized trials for treatment-naïve genotype 1 patients: ION-1 and ION-3. ION-1 examined 4 treatment arms in 865 patients: ledipasvir/sofosbuvir for 12 weeks, ledipasvir/sofosbuvir plus ribavirin for 12 weeks, ledipasvir/sofosbuvir for 24 weeks, and ledipasvir/sofosbuvir plus ribavirin for 24 weeks. 5 In total, 67% of the enrolled patients had genotype 1a and 16% had cirrhosis. 5 The SVR rates of the 4 arms were 99%, 97%, 98%, and 99%, respectively. 5 Only 3 patients experienced treatment failure: 1 relapse in the 12-week ledipasvir/sofosbuvir arm, 1 relapse in the 24-week ledipasvir/sofosbuvir arm, and 1 virologic breakthrough in the 24-week ledipasvir/sofosbuvir arm. 5 The remaining patients not counted as achieving SVR were either lost to follow-up (10 patients across all arms) or withdrew consent (3 patients across all arms). 5 In all, 2 of the 3 relapses had viral NS5A resistance-associated variants (RAVs), whereas 1 patient with virologic relapse and 1 with virologic breakthrough did not. 5
ION-3 randomized 647 treatment-naïve, genotype 1a and 1b, noncirrhotic patients to 1 of the 3 treatment arms: ledipasvir/sofosbuvir for 8 weeks, ledipasvir/sofosbuvir plus ribavirin for 8 weeks, and ledipasvir/sofosbuvir for 12 weeks. 6 The SVR rates for the treatment arms were 94%, 93%, and 95%, respectively, with respective relapse rates of 5%, 4%, and 1%, indicating noninferiority of 8 weeks of treatment. 6 In all, 9 of the 15 patients with relapse had NS5A RAVs at baseline. 6 Currently, the AASLD/IDSA guidelines recommend ledipasvir/sofosbuvir for 12 weeks in treatment-naïve genotype 1 patients with either no cirrhosis or compensated cirrhosis. 4
Based on the results of ION-3, some health systems, such as the Department of Veterans Affairs, have explored use of 8 weeks of ledipasvir/sofosbuvir in treatment-naïve, noncirrhotic, HCV monoinfected patients with a baseline viral load of less than 6 million IU/mL. 16 More recent analyses have shown that patients who are African American, obese, and/or viremic at week 4 of therapy may be less likely to achieve SVR with 8 weeks of treatment.17–20
Studies in genotype 1 patients previously treated with interferon and ribavirin, with or without boceprevir or telaprevir
Ledipasvir/sofosbuvir was studied in ION-2, which enrolled 440 genotype 1a and 1b patients previously treated with peginterferon and ribavirin with or without a protease inhibitor, such as boceprevir or telaprevir. 7 Twenty percent of enrolled patients had compensated cirrhosis, and more than half of all patients had previously been treated with a regimen that included a protease inhibitor. 7 Patients were randomized to receive ledipasvir/sofosbuvir for 12 weeks, ledipasvir/sofosbuvir plus ribavirin for 12 weeks, ledipasvir/sofosbuvir for 24 weeks, or ledipasvir/sofosbuvir plus ribavirin for 24 weeks. 7 The corresponding SVR rates were 94%, 96%, 99%, and 99%, respectively. 7
The SIRIUS study enrolled 155 genotype 1a and 1b patients with compensated cirrhosis who had unsuccessful prior treatment with a protease inhibitor–based regimen. 8 The patients received either ledipasvir/sofosbuvir plus ribavirin for 12 weeks or ledipasvir/sofosbuvir alone for 24 weeks. 8 The SVR rates were 96% and 97%, respectively. 8 Based on these study results, ledipasvir/sofosbuvir for 12 weeks is a preferred regimen for noncirrhotic patients who failed treatment with peginterferon and ribavirin, with or without boceprevir or telaprevir. 4 The AASLD/IDSA guidelines recommend the addition of ribavirin for patients with compensated cirrhosis. 4
Studies in patients previously treated with other DAAs
To date, there have been no large, phase 3, randomized controlled trials investigating treatment with ledipasvir/sofosbuvir in patients who have failed HCV regimens with DAAs besides boceprevir and telaprevir. However, small studies and post hoc analyses including this patient population have been presented and published. In one study, 14 noncirrhotic genotype 1 patients who failed the (no longer preferred) regimen of sofosbuvir plus ribavirin for 24 weeks received treatment with ledipasvir/sofosbuvir for 12 weeks with all patients achieving SVR. 21 Another study investigated ledipasvir/sofosbuvir plus ribavirin for 12 weeks in 51 genotype 1 patients who had failure with sofosbuvir plus ribavirin with or without peginterferon. 22 Twenty-seven percent of patients had cirrhosis, and all but 1 patient achieved SVR. 22 The only patient who did not achieve SVR was incorrectly reported to have genotype 1 HCV infection but actually had genotype 3 infection. 22 A small study investigated retreatment in patients who failed a prior course of ledipasvir/sofosbuvir. 23 The 41 patients evaluated were treated with 24 weeks of ledipasvir/sofosbuvir, and SVR rates corresponded to whether the patient had an NS5A RAV at baseline: 100% SVR for patients without RAVs and 60% for patients with RAVs. 23 Currently, the AASLD/IDSA guidelines recommend deferring retreatment in DAA-experienced patients unless treatment is necessarily urgent to wait for additional data to guide retreatment recommendations. 4 For those patients in whom treatment is urgent, the guidelines recommend testing for baseline NS3/4A and NS5A RAVs. Ledipasvir/sofosbuvir plus ribavirin for 24 weeks is recommended for patients without NS5A RAVs, but adding another DAA to ledipasvir/sofosbuvir as salvage therapy or using alternative DAA regimens may be an option. 4
Studies in patients with other HCV genotypes
Ledipasvir/sofosbuvir was studied in SYNERGY, a phase 2a trial of HCV genotype 4 monoinfected patients who were treatment naïve or treatment experienced with interferon/ribavirin. 9 In total, 21 patients were studied, 7 with cirrhosis, and after 12 weeks of treatment, the SVR rate was 95%, with the only nonsuccessful patient withdrawing from the study. 9 Similarly, a phase 2 trial investigated ledipasvir/sofosbuvir efficacy in HCV genotype 5 monoinfected patients who were treatment naïve or treatment experienced with an interferon-containing regimen. 10 In total, 41 patients were enrolled, 9 of whom had cirrhosis, and after 12 weeks of treatment, the SVR rate was 95% with 2 patients who relapsed. 10 Efficacy in genotype 3 and 6 patients was established in an open-label study with 12 weeks of treatment. 11 Genotype 3 treatment-naïve patients as well as all genotype 6 patients received ledipasvir/sofosbuvir alone, whereas genotype 3 treatment-experienced patients received ledipasvir/sofosbuvir plus ribavirin. 11 The SVR rates for genotype 3 patients were 64% for those not receiving ribavirin and 100% for those receiving ribavirin; the SVR rate for genotype 6 patients was 96%. 11 These data support the AASLD/IDSA guideline recommendation of ledipasvir/sofosbuvir for 12 weeks in patients with genotype 4, 5, or 6 infection; currently, ledipasvir/sofosbuvir is not recommended in genotype 3 infection.
Studies in HIV/HCV coinfected patients
ION-4 enrolled 335 human immunodeficiency virus (HIV)/HCV coinfected patients with HCV genotype 1a, 1b, or 4 and whose HIV medication regimens included tenofovir; emtricitabine; and efavirenz, rilpivirine, or raltegravir. 12 All patients received 12 weeks of ledipasvir/sofosbuvir. 12 In total, 55% of patients were treatment experienced, and 20% had cirrhosis. 12 The SVR rate was 96% and was similar regardless of any baseline characteristics. 12 Similarly, in the ERADICATE study, HIV/HCV coinfected patients with HCV genotype 1 received ledipasvir/sofosbuvir for 12 weeks. 13 All 50 enrolled patients were treatment naïve and noncirrhotic. 13 The SVR rate was 98%, with 1 patient experiencing relapse. 13 The AASLD/IDSA guidelines recommend ledipasvir/sofosbuvir in HIV/HCV coinfected patients in the same way as a patient with only HCV would be managed, but special consideration for drug-drug interactions between HIV and HCV drugs should be paid. 4
Studies in patients with decompensated cirrhosis and/or liver transplant recipients
The SOLAR-1 trial studied HCV genotype 1 or 4 patients with decompensated cirrhosis or those who had received a liver transplant. 14 Patients received ledipasvir/sofosbuvir plus ribavirin for either 12 or 24 weeks. The SVR rate for pretransplant patients was 86% to 89%, the SVR rate for posttransplant patients without cirrhosis or with compensated cirrhosis was 96% to 98%, the SVR rate for posttransplant patients with Child-Pugh B cirrhosis was 86% to 88%, and the SVR rate for posttransplant patients with Child-Pugh C cirrhosis was 60% to 75%. 14 The SOLAR-2 trial followed a similar protocol, enrolling 333 genotype 1 or 4 patients with decompensated cirrhosis and/or those who had received a liver transplant and treating them with ledipasvir/sofosbuvir plus ribavirin for 12 or 24 weeks. 15 The SVR rates for patients with decompensated cirrhosis were 78% to 96%, with the lowest SVR rates in patients with Child-Turcotte-Pugh (CTP) score C. 15 The SVR rates for genotype 1 posttransplant patients exceeded 90%, with the exception of patients with CTP C (50%-80%). Genotype 4 patients had slightly lower SVR rates of 78% to 94%. 15 Therefore, the AASLD/IDSA guidelines recommend ledipasvir/sofosbuvir with low starting doses of ribavirin for 12 weeks in patients with genotype 1 or 4 infection and decompensated cirrhosis. 4 For those patients who cannot be given ribavirin, the AASLD/IDSA guidelines recommend ledipasvir/sofosbuvir for 24 weeks. 4
Safety
Adverse effects
Ledipasvir/sofosbuvir is tolerated well by most patients with few notable adverse effects. Some of the most common adverse effects observed in clinical trials are similar to those experienced by patients infected with chronic HCV due to the disease itself. A pooled safety analysis from 3 clinical trials conducted in genotype 1 HCV with compensated liver disease both with and without cirrhosis showed that 0%, less than 1%, and 1% of patients who received 8, 12, and 24 weeks of therapy, respectively, permanently discontinued ledipasvir/sofosbuvir due to adverse events.5-7 The most common adverse effects observed in at least 5% of patients included fatigue, headache, nausea, diarrhea, and insomnia. Some of these events occurred with increasing incidence with longer durations of treatment. Fatigue occurred in 16%, 13%, and 18% of patients who received ledipasvir/sofosbuvir for 8, 12, and 24 weeks, respectively. For the same treatment durations, headache occurred in 11%, 14%, and 17% of patients, respectively; nausea occurred in 6%, 7%, and 9% of patients, respectively; diarrhea occurred in 4%, 3%, and 7% of patients, respectively; and insomnia occurred in 3%, 5%, and 6% of patients, respectively. Patients with chronic HCV genotype 4, 5, or 6 had similar rates of observed adverse events based on pooled data from 3 open-label trials.9-11 Adverse events that occurred in at least 10% of these non–genotype 1 patients included asthenia (18%), headache (14%), and fatigue (10%). 3
Adverse events were observed in other select populations studied in clinical trials. Patients who were treatment experienced with compensated cirrhosis most commonly experienced asthenia, headache, and fatigue. 3 Patients with decompensated cirrhosis and/or were liver transplant recipients notably experienced decreases in hemoglobin to less than 10 g/dL likely due to receipt of ribavirin in addition to ledipasvir/sofosbuvir as part of the HCV treatment regimen. 3 Lactic acidosis has been observed in patients with decompensated cirrhosis who received other sofosbuvir-based regimens in combination with ribavirin. Risk factors for the development of lactic acidosis included impaired renal function and more advanced liver disease. 24 Another case report described the development of liver toxicity, hyperbilirubinemia, and jaundice in a patient with decompensated cirrhosis and HIV coinfection who received ledipasvir/sofosbuvir and ribavirin combination therapy. 25 In patients with decompensated liver disease, it is a challenge to determine adverse events attributable to drug effect versus other variables. 3 These patients are at increased risk of additional progression to advanced liver disease and death. 3 Finally, in patients coinfected with HIV-1, the most common adverse effects observed in at least 10% of patients included headache and fatigue. 3
Laboratory abnormalities that were observed in clinical trials for ledipasvir/sofosbuvir include elevations of bilirubin and lipase. Increases in bilirubin to greater than 1.5 times the upper limit of normal were observed in 3%, less than 1%, and 2% of patients treated with ledipasvir/sofosbuvir for 8, 12, and 24 weeks, respectively. In patients treated with ledipasvir/sofosbuvir plus ribavirin for 12 weeks, bilirubin elevations of greater than 1.5 times the upper limit of normal were observed in 11% of patients. 8 Creatine kinase was assessed in the ION-4 trial, and it was found that asymptomatic elevations of at least 10 times the upper limit of normal were observed in 1% of patients treated with ledipasvir/sofosbuvir for 12 weeks. 12 Decreases in hemoglobin to less than 10 and 8.5 g/dL during ledipasvir/sofosbuvir plus ribavirin treatment for 12 weeks were observed in 38% and 13% of patients, respectively. Ribavirin was discontinued in 11% of these patients. 3 Because the onset of ribavirin-associated anemia is within the first 2 to 4 weeks of treatment, it is not surprising that these rates of anemia are similar to those seen in patients treated with pegylated interferon and ribavirin. 26
Cases of symptomatic bradycardia, some requiring pacemaker intervention, in addition to fatal cardiac arrest have been reported in patients receiving amiodarone coadministered with ledipasvir/sofosbuvir during the postdrug approval period. 27 Postmarketing reports submitted by the manufacturer Gilead Sciences and from the FDA Adverse Event Reporting System (FAERS) database suggest a causal association. 28 Patients taking β-blockers or with underlying cardiac abnormalities and/or advanced liver disease may be at an increased risk of bradycardia with coadministration of amiodarone. 28 Bradycardia has usually occurred within hours to days of ledipasvir/sofosbuvir initiation; however, this adverse effect has been observed up to 2 weeks following initiation of therapy. 28 If a patient must receive amiodarone coadministered with ledipasvir/sofosbuvir, patients must be counseled about the serious risk of symptomatic bradycardia.3,28 In these cases, it is also recommended that cardiac monitoring be performed in the inpatient setting for the first 48 hours of coadministration followed by 2 weeks of outpatient cardiac monitoring.3,28
Drug interactions
Ledipasvir and sofosbuvir are substrates of P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP). 3 P-glycoprotein inducers, such as rifampin and St. John’s Wort, may decrease ledipasvir and sofosbuvir plasma concentrations, leading to decreased efficacy; thus, coadministration with P-gp inducers is not recommended. 3 Other agents that may decrease ledipasvir and sofosbuvir concentrations leading to decreased therapeutic effect include anticonvulsants, such as carbamazepine, phenytoin, phenobarbital, and oxcarbazepine, and other antimycobacterials, such as rifabutin and rifapentine. 3 Coadministration of ledipasvir/sofosbuvir with these agents is not recommended.
The concentration of ledipasvir is decreased by acid suppressants due to decreased solubility of ledipasvir at higher pH concentrations. 3 Accordingly, antacids, such as aluminum and magnesium hydroxide, should be separated from ledipasvir/sofosbuvir administration by 4 hours. 3 H2 receptor antagonists such as famotidine may be administered simultaneously with ledipasvir/sofosbuvir or 12 hours apart at a dose not exceeding the equivalent of famotidine 40 mg twice daily. 3 Proton pump inhibitors at doses comparable with omeprazole 20 mg or lower may be administered simultaneously with ledipasvir/sofosbuvir under fasting conditions. 3
Other agents whose concentrations are affected by ledipasvir/sofosbuvir include tenofovir, digoxin, and rosuvastatin. Tenofovir is a substrate of P-gp and BCRP, whereas ledipasvir is an inhibitor of both these drug transporters. Patients receiving the antiretroviral tenofovir without an HIV protease inhibitor boosted by ritonavir or cobicistat should be monitored for tenofovir-associated adverse reactions due to increased concentrations of tenofovir. 3 Increased tenofovir concentrations may still be observed when the following antiretroviral regimens, including an HIV protease inhibitor with ritonavir or cobicistat, are coadministered with ledipasvir/sofosbuvir: atazanavir plus either ritonavir or cobicistat, darunavir plus either ritonavir or cobicistat, or lopinavir/ritonavir. 3 If coadministration of these agents is required, monitoring for tenofovir-associated adverse events, most notably renal toxicity, should occur. 3 Nephrotoxicity has been associated with concomitant use of ledipasvir/sofosbuvir and tenofovir in a patient who was coinfected with HCV and HIV. 29 Acute tubular necrosis and acute interstitial nephritis were revealed by renal biopsy with an increase in serum creatinine concentration from a baseline of 1.0 to 10 mg/dL 8 weeks after initiating ledipasvir/sofosbuvir. The authors suggested that this adverse reaction occurred due to increased tenofovir exposure resulting from a drug interaction with ledipasvir. They recommended close monitoring of markers of tubular dysfunction, including urinary phosphorous excretion and renal injury at baseline and week 4 of therapy in coinfected patients treated with this combination of antiretroviral and DAA therapy. 29
The concentration of digoxin may be increased if coadministered with ledipasvir/sofosbuvir due to ledipasvir’s inhibition of P-gp. Therapeutic drug monitoring of digoxin is recommended. 3 In addition, ledipasvir inhibits BCRP and may significantly increase the concentration of rosuvastatin, a BCRP substrate, leading to myopathy and rhabdomyolysis. Coadministration is not recommended. 3
Some medications without a known drug interaction with ledipasvir/sofosbuvir include methadone and antiretrovirals, including abacavir, darunavir/ritonavir, dolutegravir, and raltegravir. 3 Although there is not an anticipated significant drug-drug interaction with the immunosuppressants cyclosporine and tacrolimus, therapeutic drug monitoring of the immunosuppressants is prudent.3,4
Use in renal impairment
Ledipasvir/sofosbuvir does not require a dosage adjustment when used in patients with mild-to-moderate renal impairment; however, its safety has not been established in severe renal impairment (estimated glomerular filtration less than 30 mL/min/1.73 m2) or end-stage renal disease requiring hemodialysis. A dosage adjustment is not available for these cases, and the fixed-dose combination should be avoided. 3 A case of biopsy-proven acute interstitial nephritis associated with the use of ledipasvir/sofosbuvir was recently reported. The patient presented in this report also had diabetic nephropathy; however, the clinical presentation, pathology findings, and time course of renal impairment were consistent with ledipasvir/sofosbuvir use. 30 Furthermore, renal clearance is the main route of elimination for the predominant sofosbuvir metabolite GS-331007. 3 Although caution has not been recommended, there have been reports documenting the risk of lactic acidosis and hepatic decompensation during sofosbuvir treatment in patients with chronic kidney disease. 24
Conclusions
In summary, ledipasvir/sofosbuvir is a safe and effective treatment option for patients with genotype 1, 4, 5, or 6 chronic HCV, including those with HIV/HCV coinfection, those with decompensated cirrhosis, and those who have received a liver transplant. Ledipasvir/sofosbuvir has been recommended as a preferred treatment regimen for each of these patient groups in the AASLD/IDSA guidelines. Further studies to determine effective treatment options for patients who have failed treatment with ledipasvir/sofosbuvir are warranted.
Footnotes
Peer review:
Six peer reviewers contributed to the peer review report. Reviewers’ reports totaled 1153 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
LMCK and JJ conceived and designed the manuscript, analyzed the data, wrote the first draft of the manuscript, contributed to the writing of the manuscript, agree with manuscript results and conclusions, jointly developed the structure and arguments for the paper, and made critical revisions and approved the final version. All authors reviewed and approved the final manuscript.