Abstract
Clevidipine is a third-generation dihydropyridine calcium channel blocker approved in 2008 by the Food and Drug Administration for parenteral therapy of arterial hypertension. The high degree of lipophilicity of clevidipine provides a rapid onset of action and ease of titration to reach the desired clinical effect. Since its introduction into clinical practice, clevidipine has been shown to be safe and effective for perioperative use in patients undergoing procedures in which rapid and effective blood pressure control is essential. In 2011, clevidipine received institutional approval for intraoperative use during neurosurgical procedures and for postoperative blood pressure control in the Post-Anesthesia Care Unit and Surgical Intensive Care Unit during the first 24 hours after surgery. This review describes the efficacy and safety of intravenous clevidipine in intraoperative blood pressure management during neurosurgical procedures in 11 patients over a period of 3 months. The evaluation revealed that lower rates of clevidipine infusion than previously documented in the literature achieved blood pressure control within the desired limits during surgery while avoiding dose-related adverse reactions.
Background
Numerous pharmacologic agents are available for treating intraoperative hypertension including direct vasodilators (nitroglycerin and sodium nitroprusside), β-adrenergic blockers (esmolol), α/β-adrenergic blockers (labetalol), calcium channel blockers (nicardipine), and peripheral µ postsynaptic receptor antagonist plus central serotonin 5-hydroxytryptamine receptor agonist (urapidil). Although these drugs have been effective for the treatment of intraoperative hypertension, there are reported unfavorable features associated with each agent. 1 Nitroglycerin, a commonly used vasodilator, acts predominantly on capacitance vessels and therefore may reduce the preload and cardiac output, with undesirable results in patients with already compromised renal and cerebral perfusion. 1 Tachyphylaxis may develop with prolonged administration of nitroglycerin. β-Adrenergic blockers (ie, esmolol, labetalol) are useful in mitigating perioperative hypertension; however, their use is associated with a reduction in heart rate (HR) which can be deleterious in patients with preexisting bradycardia or heart block. 1 Sodium nitroprusside may induce cyanide toxicity, particularly when high doses are used for prolonged time intervals.
Dihydropyridine calcium channel blockers are promising agents for optimal intraoperative blood pressure (BP) control. Nicardipine is commonly used intraoperatively to control BP. Depending on the mode of administration (continuous intravenous infusion versus bolus dosing), its use may result in infusion of relatively large fluid volume. 1 Nicardipine is also more selective to coronary versus systemic vasculature. 2 Due to its short onset of action of 10 minutes, intravenous infusion of nicardipine can be easily titrated to maintain BP within desirable ranges.2,3 In neurosurgery, Powers et al 4 showed that nicardipine has the advantage of lowering mean arterial pressure by >15% with no significant decreases in cerebral blood flow. The authors concluded that cerebral blood flow autoregulation is preserved with intravenous nicardipine therapy. Furthermore, intra-arterial nicardipine improves cerebral blood flow as measured by computed tomography perfusion in patients with post-subarachnoid hemorrhage vasospasm. 5
Clevidipine is a third-generation dihydropyridine calcium channel blocker approved in 2008 by the Food and Drug Administration as a parenteral agent for the treatment of arterial hypertension. Clevidipine is a highly lipophilic drug with a quick onset of action of 2 to 4 minutes, allowing for rapid titration to reach the desired clinical effect. Clevidipine’s fast metabolism by plasma and tissue esterases provides a short duration of action, even in patients with reduced hepatic and renal function. Clevidipine may be continued for up to 24 hours into the postoperative period if needed, with transition to oral medications once tolerated.6,7
Due to the lipid emulsion formulation of clevidipine, there are inherent risks of hypertriglyceridemia, pancreatitis, and contamination. 1 Based on lipid load restrictions, no more than 1000 mL or an average of 21 mg/h of clevidipine infusion is recommended per 24-hour period. The maximum administration rate should not exceed 32 mg/h. Finally, its use may be limited by increased costs over the other antihypertensive agents used perioperatively. Of note, effective BP control with clevidipine versus other antihypertensive medications including sodium nitroprusside, nitroglycerin, and nicardipine was associated with a 7% reduction to the costs of hospitalization in a group of surgical patients. 8
Since its introduction into clinical practice, clevidipine has been shown to be safe and effective for perioperative use in patients undergoing procedures in which rapid and effective BP control is essential.9–13 Studies have shown that target BP values may be rapidly achieved with intraoperative infusion of clevidipine; this is of special importance in specific areas of surgery such as cardiac and neurological surgery and in cases at high risk for the development of hypertension-related complications.14,15
In 2011, clevidipine was approved by the Pharmacy and Therapeutics Executive Committee at The Ohio State University Wexner Medical Center (OSUWMC) for intraoperative use during neurosurgical procedures and in the Post-Anesthesia Care Unit (PACU) and Surgical Intensive Care Unit (SICU) for postoperative BP control during the first 24 hours after surgery. The purpose of this study was to describe the efficacy and safety of intravenous clevidipine in intraoperative BP management during neurosurgical procedures.
Materials and Methods
A single-center, retrospective, observational study was performed to describe the safety and efficacy of intraoperative clevidipine use during neurosurgical procedures. The study was approved by The Ohio State University Institutional Review Board. All the patients who received clevidipine at the OSUWMC between August 30, 2011 and November 30, 2011 were evaluated. To evaluate the safety and efficacy of intravenous clevidipine administered during neurosurgery, data related to perioperative BP control in patients treated with this medication were collected.
The following information was collected: demographics (age, sex, and weight), type of procedure, start time of procedure, and BP measurements (preinduction BP, goal systolic BP [SBP], intraoperative BP at 30-minute increments). Blood pressure goals were defined as documented by the neuro-anesthesiologist in the patient’s electronic medical record. Medication data related to BP control, including antihypertensives given prior to and during clevidipine infusion, details related to the clevidipine infusion (time of initiation, rates and timing of titration of infusion, time and reason for any discontinuation of clevidipine), medications administered if clevidipine was discontinued for hypotension, and concurrent infusions for sedation (propofol or dexmedetomidine) were collected. In addition, the location and time of patient transfer to either the PACU or SICU were recorded.
Patients were instructed to take their usual antihypertensive medications with a sip of water on the morning of surgery. Based on the values obtained in the preoperative holding area, BP was treated following a standard protocol. In patients without a diagnosis of chronic hypertension, clevidipine was administered as first-line treatment to achieve the desired reduction in SBP. In patients with a diagnosis of chronic hypertension, a dose of intravenous labetalol (if HR > 70 bpm) or hydralazine (if HR < 70 bpm) was administered. If BP remained greater than the patient’s goal SBP, a clevidipine infusion was initiated. Clevidipine was initiated at a starting rate of 1 to 2 mg/h and was titrated to achieve the desired BP goal. Clevidipine was titrated by doubling the dose at 90-second intervals initially. Once the desired BP goal was reached, any subsequent necessary upward titration was performed by increasing the rate by less than double every 5 to 10 minutes.
Statistical Analysis
Analysis was conducted using SAS 9.4. A mixed-effects regression model was used to determine changes in rate, SBP, and diastolic BP for the 11 patients over the 240-minute time period captured in 30-minute increments. The mixed-effects regression model was used to accurately account for the repeated measure nature of the data.
Results
A total of 11 patients (8 men and 3 women), with a median age of 54.5 (interquartile range [IQR]: 38-92) years were included for evaluation. The median weight was 87.5 (IQR: 52-123) kg. Four patients (45.5%) had a past medical history of chronic hypertension managed prior to admission with a β-blocker (3 patients), angiotensin-converting enzyme inhibitor (3 patients), and/or a thiazide diuretic (3 patients). None of the patients who received clevidipine had any documented contraindications to the drug, including allergies to soybeans, egg products, or defective lipid metabolisms such as pathologic hyperlipidemia, lipoid nephrosis, and acute pancreatitis.
In all, 6 patients (54.5%) underwent placement of a deep brain stimulator, 3 (27.2%) underwent a neurosurgical excision of mass, and 2 (18.1%) underwent an aneurysmal clipping. Of the 6 patients undergoing deep brain stimulator, 4 patients had a diagnosis of Parkinson disease, whereas 1 patient received the therapy for tremor related to traumatic brain injury and the other had idiopathic essential tremor. All patients achieved goal BP with the first recorded intraoperative BP measurement at 30 minutes (Figure 1).
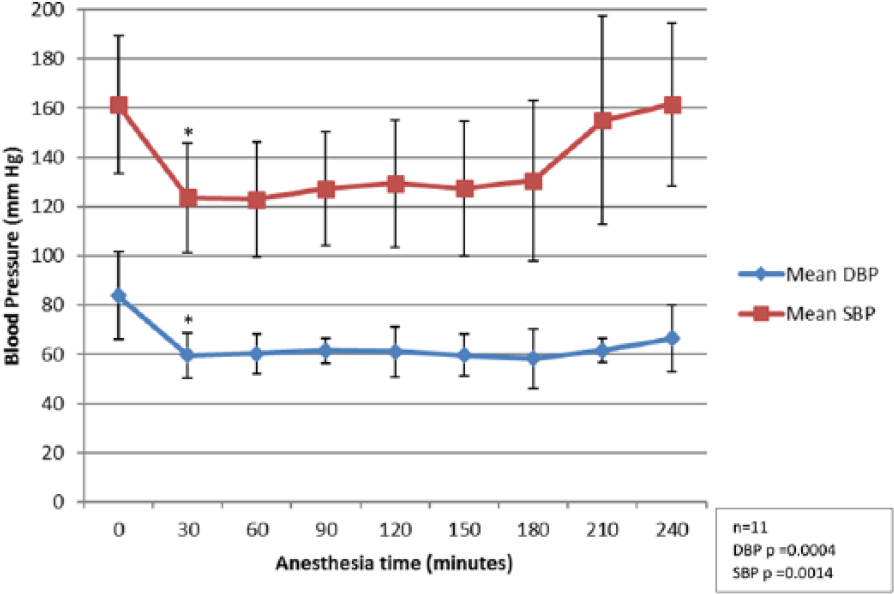
Intraoperative blood pressure values during clevidipine infusion. DBP indicates diastolic blood pressure; SBP, systolic blood pressure.
The mean clevidipine infusion rate with standard deviation associated with effective control of BP at 30 minutes was 6.06 ± 5.03 mg/h. In 4 patients, clevidipine was discontinued after approximately 2 hours from the initiation of infusion due to hypotension (n = 3) and blood loss (n = 1). No further intervention was required for any of the 3 hypotensive events. Out of the 11 patients, 1 received a concomitant infusion of nitroglycerin, and 1 received intravenous labetalol. The highest rate of infusion among the 11 patients was 20 mg/h (Figure 2). Clevidipine infusion was discontinued at the end of surgery for all patients.
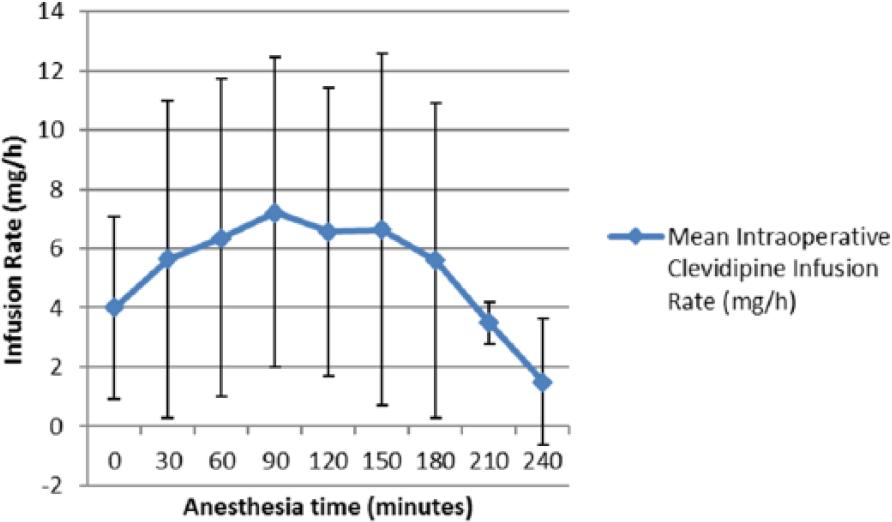
Mean intraoperative clevidipine infusion rates (mg/h).
Concurrent sedation included the following: propofol infusion (n = 1), dexmedetomidine infusion (n = 2), both propofol and dexmedetomidine (n = 4), and no continuous infusion sedation (n = 1).
Postoperatively, 6 patients were transferred to PACU, 2 patients were transferred to SICU, and 2 patients’ medical records did not provide any transfer information.
The mixed-effects regression model showed that there were statistically significant differences in SBP (P = .0014) and diastolic BP (P = .0004) (Figure 1) and rate (P = .0387) (Figure 2) over the given time period. Figure 1 shows a statistically significant decrease in SBP and diastolic BP from baseline when compared with time points after 30 minutes.
Discussion
Previous literature demonstrates that up to 25% of patients with chronic hypertension undergoing surgery will experience elevated perioperative BP.14,16 Intraoperative arterial hypertension is associated with the risk of serious complications, similar to those experienced by patients diagnosed with chronic hypertension. Controlling BP during surgery is an essential requirement for anesthesia management. 1
In this study, all patients achieved goal BP within 30 minutes (Figure 1). The infusion rate required for BP control with 30 minutes was 6.06 ± 5.03 mg/h, which is lower than the rates reported in previous publications. 13 Although clevidipine was discontinued in 4 patients due to hypotension or blood loss, no further intervention was required for any of the 3 hypotensive events. The maximum permitted infusion rate of 32 mg/h was not exceeded in any of the 11 cases (Figure 2). No patient required clevidipine infusion postoperatively.
The safety and efficacy of intraoperative clevidipine have been evaluated in different trials, with Evaluation of Clevidipine in the Perioperative Treatment of Hypertension Assessing Safety Events (ECLIPSE) trial being the first large-scale study that compared the effectiveness of clevidipine with other antihypertensive drugs. 10 The study design included 3 parallel multicenter open-label trials randomizing cardiac surgery adult patients in a 1:1 fashion based on an antihypertensive agent received (clevidipine:nitroglycerine/sodium nitroprusside/nicardipine).1,10 The data suggested that clevidipine was effective at maintaining SBP within 75 to 145 mm Hg preoperative and 65 to 135 mm Hg intraoperatively.1,10 In addition, in a multicenter, double-blind, placebo-controlled study, Efficacy Study of Clevidipine Assessing its Preoperative Antihypertensive Effect in Cardiac Surgery 1 (ESCAPE-1) (n = 105), patients received 0.5 mg/mL clevidipine or placebo of 20% lipid emulsion for a minimum of 30 minutes preoperatively. 12 The trial concluded that the clevidipine group experienced a significantly lower treatment failure rate, achieving the targeted BP in a median time of 6 minutes. 12
Although the maximum recommended infusion rate for clevidipine is 32 mg/h, most studies have reported success with lower doses. The maximum allowed rate of clevidipine infusion in ESCAPE-1 study was 8 µg/kg/min (for example, for a 75 kg patient equivalent to 36 mg/h). However, 60% of the patients achieved goal BP ranges at rates lower than 3.2 µg/kg/min (equivalent to 14.4 mg/h in a 75 kg patient). The ECLIPSE trial reported a median infusion rate of 3.1 µg/kg/min (equivalent to 14 mg/h in a 75 kg patient). Although our study was performed on a smaller scale (n = 11), we were able to demonstrate lower infusion rates to reach target intraoperative BP values, which further supports the use of clevidipine at lower doses in the neurosurgical population. Of note, 7 of the 11 patients evaluated did not have a diagnosis of chronic hypertension, and those with chronic hypertension were instructed to take their home antihypertensive agent(s) the day of surgery.
Tobias and Hoernschemeyer 6 reached their target intraoperative mean arterial pressure (50-65 mm Hg) within 5 to 10 minutes in a group of adolescents using clevidipine infusion rate of 0.5 to 1 µg/kg/min, titrated by 0.5 to 1 mg/kg/min increments every 2 to 3 minutes. None of the patients experienced excessive hypotensive events that required intervention, and BP measurements returned to baseline within 5 to 10 minutes of stopping clevidipine infusion. 6 Despite differences in age and chronic hypertension between our population and this study’s population, we were able to achieve target BP goal with a similar infusion rate.
Our study has several limitations worth noting. Based on the small patient population size and retrospective study design, the causal relationship between dosing strategy and safety or efficacy may not be established. This study was limited to the perioperative management of hypertension in neurosurgical patients; therefore, utility of this dosing strategy in other populations may not apply.
Conclusions and Recommendations
Based on our experience with the intraoperative use of clevidipine for neurosurgical procedures at OSUWMC and published scientific evidence, lower doses of clevidipine infusion should be considered for the perioperative BP control in the neurosurgical setting. Lower doses may achieve BP control within the desired limits during surgery while avoiding dose-related adverse reactions.
Footnotes
Acknowledgements
The authors acknowledge Muhammad Shabsigh, MD, visiting scholar at The Ohio State University Wexner Medical Center for his editorial support.
Peer Review:
Eight peer reviewers contributed to the peer review report. Reviewers’ reports totaled 951 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
