Abstract
Obstructive sleep apnoea (OSA) is a common sleep disorder that is associated with significant negative health outcomes including cardiovascular disease, daytime sleepiness, neurocognitive deficits, and increased motor vehicle and workplace accidents. There is wide variation in OSA symptoms and other downstream effects between patients highlighting the need to individualise therapy. Continuous positive airway pressure delivered by a face mask is the gold standard treatment, but adherence to this therapy is poor and improvements in outcomes are often incomplete. A range of alternative treatments are available and may suit different patients. These include behavioural treatments such as weight loss, mandibular advancement using an oral device, sleep posture modification, upper airway surgery, and upper airway muscle stimulation. Towards individualised OSA therapy, novel phenotyping approaches are being developed to identify the specific pathophysiological causes of OSA applying to individual patients. Furthermore, research is underway to help identify patients with OSA at higher risk of daytime sleepiness and adverse cardiovascular and neurocognitive consequences and predict how individuals might respond to treatment. In this article, we review the prevalence, risk factors, and main consequences of OSA; the main treatment modalities available at present; and some new methods for phenotyping patients with OSA that hold promise for a more personalised and effective approach to screening, diagnosis, and treatment.
Epidemiology, Pathogenesis, and Consequences of Obstructive Sleep Apnoea
Prevalence and risk factors for OSA
Obstructive sleep apnoea (OSA) is a common sleep disorder. Early population studies suggested that up to 10% of the middle-aged population1–4 were affected, with as many as 80% of cases undiagnosed.5–7 More recent estimates suggest that there has been a rapid rise in OSA prevalence with more than 20% of middle-aged individuals exhibiting moderate to severe OSA likely driven by increased global obesity as well as more sensitive OSA measurement techniques.8,9 The main features of OSA are snoring and repetitive hypopnoea (reduced airflow) and/or complete collapse (apnoea) of the pharyngeal airway during sleep with associated transient oxygen desaturation (hypoxaemia), carbon dioxide retention (hypercapnia), and repeated arousal from sleep. The major risks factors for OSA are male sex, older age, and obesity.
Pathophysiological phenotypes of OSA
Obstructive sleep apnoea is a heterogeneous disease with variable responses to different treatment modalities, including continuous positive airway pressure (CPAP). The physiological mechanisms contributing to upper airway (UA) instability and increased collapsibility are complex and incompletely understood but appear to be multiple and vary between affected individuals. There are 4 main respiratory phenotypes 10 that are believed to contribute to UA collapse during sleep in OSA. These phenotypes include the following: (1) an anatomically narrower pharyngeal airway, (2) reduced neuromuscular compensation in UA dilator muscles, (3) heightened arousal responses, and (4) an unstable respiratory control system. Therapies targeted at specific OSA phenotypes may enable individualised approaches to optimise treatment success. Several genetic risk markers have also been found to be associated with OSA, and genome-wide association data are now beginning to become available.11,12 In most individuals with OSA, it is likely that multiple traits in combination lead to UA collapse rather than one specific causative factor.
Daytime consequences of OSA
Excessive daytime sleepiness
One of the main clinical symptoms of OSA is excessive daytime sleepiness (EDS). Those with EDS experience an increased propensity or ‘pressure’ to fall asleep, sometimes at inappropriate times (eg, while driving). Excessive daytime sleepiness is associated with negative health outcomes and quality of life. 13 It is also common in other sleep disorders such as circadian rhythm abnormalities and in lifestyle-related chronic sleep restriction.14,15
Current clinical tests to assess daytime sleepiness include the Epworth Sleepiness Scale (ESS) questionnaire, 16 which asks about the person’s propensity to doze (ie, fall asleep) in 8 common situations. Objective measures of sleepiness include the multiple sleep latency test which measures the average time to fall asleep during 4 to 5 daytime nap opportunities and the maintenance of wakefulness test which measures how well subjects can maintain wakefulness when placed in a soporific environment. 17
In OSA, EDS is believed to be the result of sleep fragmentation and/or episodic hypoxia. The relative contribution of these factors to EDS and daytime function in OSA remains uncertain. Excessive daytime sleepiness makes it difficult for many patients with OSA to maintain vigilance and attention, especially in monotonous situations. Although it is clear that patients with OSA as group have EDS, there is growing evidence of substantial inter-individual variation.18–20 Many patients, even among those with severe OSA, report little or no daytime sleepiness, whereas others with milder OSA are dangerously sleepy. Routine clinical polysomnographic measures of OSA severity and sleep quality such as the apnoea/hypopnoea index (AHI), level of nocturnal hypoxaemia, and frequency of sleep microarousals have demonstrated weak and inconsistent relations with measures of self-reported daytime sleepiness (ESS score)21,22 and neurobehavioral function. 23 The Sleep Heart Health Study found that pathological sleepiness, defined as an ESS score >10, affected only 35% of patients with severe OSA (AHI > 30 events/h). 22
Neurocognitive impairment
A recent meta-analysis has concluded, in agreement with previous reports,24,25 that OSA is also associated with deficits in neurocognitive daytime functions including attention, psychomotor function, processing speed, memory, learning, and executive function. 26 These daytime abnormalities are likely driven by disturbance to brain oxygenation during sleep as well as sleep fragmentation through repeated apnoea-induced awakenings24,25 and are associated with abnormalities in brain functional imaging and electrophysiology.27,28
Similar to complaints of EDS, substantial inter-individual variation has also been found for neurocognitive impairment. In the recent Apnea Positive Pressure Long-term Efficacy Study (APPLES), Quan et al 23 reported weak correlations between AHI, oxygen desaturation, and neurocognitive performance in an unadjusted model, and after adjusting for age, sex, and ethnicity, AHI had no significant association with neurocognitive function, whereas oxygen indices remained weakly associated with attention, processing speed, and intelligence. The reason for this heterogeneity of effect of OSA on neurobehavioral function is unclear.
Obstructive sleep apnoea–driven neurocognitive dysfunction has a major impact on driving and workplace safety. Obstructive sleep apnoea is associated with impaired driving performance 29,30 and at least a 2-fold increase in workplace 31 and motor vehicle accident (MVA) risk, 32 with an estimated 1400 road fatalities costing $15.9 billion annually in the United States. 33 Although these aggregate figures are alarming, there is large heterogeneity in driving performance between individual patients, similar to sleepiness and cognitive symptoms, such that the increased MVA risk might apply only to a minority of patients with OSA. 20 At present, assessing the MVA risk of an individual patient with OSA, and their suitability to hold a driver’s license, is challenging. Current clinical metrics of OSA severity and daytime sleepiness are limited in their ability to identify at-risk patients. Novel methods, including quantitative electroencephalography, 34 are being developed that may help clinicians to better identify which patients with OSA are at high risk of alertness failure and driving impairment.
There is also emerging evidence that OSA disturbs balance and gait, potentially increasing the propensity for falls, particularly in older patients.35–37 Falls are a leading cause of injury, disability, and mortality worldwide 38 ; and therefore, OSA therapy may be a potential target to reduce falls risk. Preliminary evidence suggests that OSA therapy may reduce gait instability in OSA. 39 Large prospective studies and randomised trials are needed.
Co-morbid insomnia and OSA
Adding to the challenge of correctly identifying the sleepy patients with OSA who are most in need of treatment is the recent revelation that a significant proportion of patients with OSA have overlapping insomnia. Although, traditionally, OSA is described as a condition that disrupts sleep, leads to EDS, and a tendency for the affected individual to fall asleep rapidly, it is now recognised that insomnia symptoms (difficulty with falling and/or maintaining sleep) often co-occur in OSA. In fact, co-morbid insomnia has been estimated to affect between 39% and 58% of patients with OSA.40,41 In such patients, fatigue may be more prominent than EDS, and adherence to CPAP treatment may be problematic because of a longer than usual sleep onset latency or wakefulness during the night. A combination of cognitive behavioural treatment for insomnia and CPAP therapy may be required in such patients 42,43 to achieve optimal patient outcomes.
Cardiovascular disease and mortality
Hypertension
Associations between OSA and hypertension, cardiovascular disease, and all-cause mortality have been demonstrated in a number of epidemiologic studies.44–49 The prospective, longitudinal Wisconsin Sleep Cohort Study demonstrated a dose-response relationship between baseline OSA severity and development of hypertension within 4 years of follow-up, 50 with 2.9 times higher odds of developing hypertension in those with moderate to severe OSA (ie, AHI ⩾ 15 events/h) compared with those without OSA, after controlling for known confounders. However, an independent association between OSA and hypertension could not be found in 2 other prospective cohort studies,51,52 although differences in the studied populations, in particular, age, sex, and sleep study procedures used, may have contributed to the discrepant results. 53 A high prevalence of OSA (up to 83%) 44 has been reported in patients with resistant hypertension.
Other cardiovascular morbidity and mortality
Experimental studies, including evaluation of biomarkers related to cardiovascular disease risk, have found that OSA is associated with oxidative stress, increased inflammatory mediators 54 (including C-reactive protein, interleukin 6, and tumour necrosis factor α), arterial stiffening, vascular endothelial dysfunction, and elevated catecholamine levels consistent with sympathetic nervous system activation.54–56 Observational studies have also found independent associations between OSA and other cardiovascular disorders including coronary artery disease (CAD), 47 cerebrovascular disease, 57 heart failure, 58 and cardiac arrhythmias, 59 as well as all-cause mortality.48,49,60 A prospective, observational study of the incidence of incident CAD in patients with and without OSA who were free of cardiovascular disease at baseline revealed an increased relative risk of 4.6 (95% confidence interval [CI]: 1.8-11.6) for the development of incident CAD in patients with OSA after 7 years of follow-up, and effective treatment of OSA appeared to reduce the excess risk. 45 A 3-fold increased risk of ischemic stroke was found in men with OSA who were followed up for a median of 8.7 years in the Sleep Heart Health Study, 57 a community-based, prospective cohort study.
Main Treatment Modalities and Their Relative Effectiveness
Continuous positive airway pressure
The main therapy pathways for OSA are summarised in Figure 1. The primary form of therapy for moderate to severe OSA is with CPAP, which has been the predominant form of treatment for OSA since its use was first described by Sullivan et al 61 in 1981. By applying air pressure through a mask secured over the patient’s mouth and/or nose, CPAP creates a pneumatic splint that stabilises the UA and reduces hypoxia and sleep fragmentation associated with repetitive obstructive events. 62 Continuous positive airway pressure therapy has been shown in a number of studies to be effective in improving or preventing many of the health-related consequences of OSA, 63 most notably, daytime sleepiness.
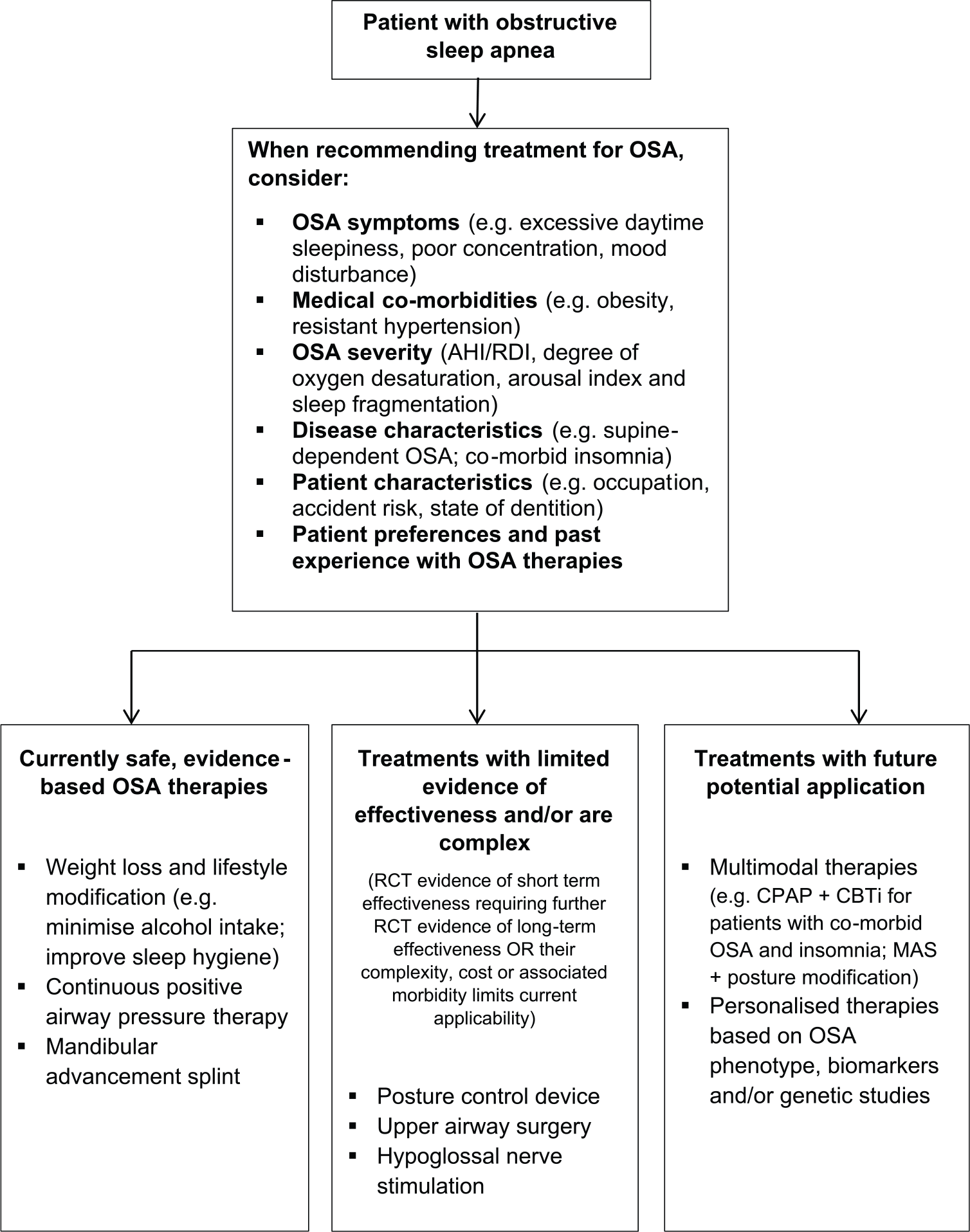
Treatment pathways for obstructive sleep apnoea. AHI indicates apnoea/hypopnoea index; OSA, obstructive sleep apnoea; RDI, respiratory disturbance index.
A meta-analysis of 32 randomised controlled trials (RCTs) on the impact of CPAP therapy on blood pressure which included data from 1948 patients with OSA revealed modest but significant reductions in diurnal systolic blood pressure (SBP) and diastolic blood pressure (DBP) (SBP: 2.58 mm Hg [95% CI: 3.57-1.59], DBP: 2.01 mm Hg [2.84-1.18], respectively). 64 Larger blood pressure decreases have been demonstrated in patients with resistant hypertension, with a meta-analysis of 6 RCTs revealing mean changes in ambulatory SBP of −7.21 (95% CI: −9.04 to −5.38) and DBP of −4.99 (95% CI: −6.01 to −3.96) following CPAP therapy.
Experimental research studies have demonstrated reductions in inflammatory markers related to cardiovascular disease and catecholamine levels following CPAP use in patients with OSA,55,65 as well as reversibility of endothelial dysfunction. 66 Clinical observational studies have reported significant reductions in cardiovascular morbidity and mortality in patients with OSA treated with CPAP. A study of long-term cardiovascular outcomes in men with OSA by Marin et al 47 showed a higher incidence of fatal and nonfatal cardiovascular events in those with severe, untreated OSA compared with OSA patients treated with CPAP, those with untreated mild to moderate OSA, and simple snorers, with similar findings seen in a subsequent study in women. 67 Although these epidemiologic studies have supported a causal association between OSA and cardiovascular disease, RCTs that have evaluated the effect of CPAP in patients with OSA have failed to demonstrate improvements in cardiovascular outcomes following treatment. An earlier RCT that examined the impact of CPAP on the incidence of hypertension and cardiovascular events in 725 consecutive patients showed no significant reduction in these outcomes after a median of 4 years of CPAP therapy. 68 Similarly, an RCT of CPAP treatment of patients with OSA who had recently had a stroke found no reduction in cardiovascular events. 69 Another recent RCT randomised patients with OSA with CAD into auto-CPAP (n = 122) vs no CPAP (n = 122) arms and assessed time to first event of repeat revascularisation, myocardial infarction, stroke, or cardiovascular mortality after a median follow-up of 57 months. 70 There was no difference between the 2 groups at follow-up, although post hoc analyses showed a reduction in CAD in patients who used CPAP for >4 hours per night. The most recent and the largest RCT, the Sleep Apnea Cardiovascular Endpoints (SAVE) Study 71 included 2717 patients with moderate to severe OSA and established cardiovascular disease. Patients were randomised to CPAP plus usual care versus usual care alone. This study also demonstrated no benefit of CPAP on the composite end point of death from cardiovascular causes, myocardial infarction, stroke, and hospitalisation for unstable angina, heart failure, or transient ischemic attack. However, it did show significant improvements in snoring, daytime sleepiness, health-related quality of life, and mood following CPAP treatment. 71 Mean nightly CPAP use in this minimally symptomatic population was low at only 3.3 h/night. Although this was anticipated by the investigators and the study powered accordingly, it is nonetheless possible that low adherence may have contributed to the neutral result. Based on current evidence, CPAP treatment of OSA should be directed at improving patient symptoms and quality of life, rather than with the aim of reducing cardiovascular disease risk.
Partial adherence to CPAP
Despite the ability of CPAP treatment to almost completely abolish UA obstruction, stabilise blood gases, reduce sleep fragmentation, and improve EDS, as many as 29% to 83% of patients fail to adhere to treatment over the long term. 72 A number of side effects limit adherence to CPAP therapy. These include mask problems (skin irritation/allergies, claustrophobia, and mask leak), pressure-related problems (headaches, sinusitis, and rhinitis), and equipment-related issues (noise, smell). 62 In addition, a perceived lack of treatment benefit or dislike of the treatment can lead to poor CPAP adherence. 73 Some of the factors that influence whether or not a patient accepts CPAP treatment over the long term have been evaluated in large clinical trials. These studies have concluded that adherence is related to pre-treatment OSA severity (ie, higher AHI results in better long-term adherence),62,74 mask interface type, and effective humidification (nasal masks and humidification lead to better adherence), 75 whereas CPAP pressure levels do not appear to influence adherence levels.76,77 However, a growing body of evidence has highlighted that in addition to the biophysical factors contributing to CPAP adherence, psychosocial attitudes and patient’s health beliefs may play a more important role in determining adherence.78,79 For example, Sawyer et al 78 reported differences between patients with OSA who adhere to CPAP and those who do not, in perception of risk associated with OSA, symptom recognition, outcome expectations, treatment goals, and perceived treatment facilitators and barriers. Another study has examined predictors of long-term adherence to CPAP therapy in 275 patients with OSA and cardiovascular disease in the SAVE study randomised to the CPAP arm. 80 Continuous positive airway pressure use and side effects at 1 month were the only independent predictors of adherence to CPAP at 12 months. 80 This suggests that early intervention is critical to motivate patients to accept and use CPAP in the longer term.
Effects of CPAP treatment on daytime sleepiness, cognitive function, driving performance, and MVA risk
An important clinical question is how effective is CPAP therapy in improving and/or normalising daytime sleepiness, cognitive function, driving performance, and reducing MVA risk associated with OSA. A number of studies designed to address this question have demonstrated that CPAP treatment is effective in significantly reducing daytime sleepiness, 62 improving driving simulator performance 81,82 and decreasing MVA risk. 83 However, uncertainty remains about whether CPAP improves performance impairments to the level of healthy subjects. Evidence suggest that when compared with controls, only partial improvements in neurocognitive performance, 84 driving simulator performance, 82 and daytime sleepiness85,86 are observed, even when patients are well treated with an average of 6 or more hours of CPAP use per night.85,86 These observations raise the possibility that there may be irreversible central nervous system damage, at least in more vulnerable patients, 20 by the time patients present for treatment, perhaps driven by years of unrecognised repetitive hypoxia and sleep fragmentation.
Telemedicine and CPAP
Recent technological advancements have resulted in rapid growth in the use of smartphones, mobile health applications, and wearable devices worldwide, with increased interest in the development of telemedicine strategies aimed at enhancing education and/or follow-up of patients with OSA, as well as optimising CPAP adherence. Continuous positive airway pressure modems have the capacity to transfer and store information to cloud-based servers, enabling health providers to remotely access an individual’s CPAP data, identify poor CPAP adherence, and intervene when needed. Furthermore, data can be transferred directly to patients via specialised online portals to provide feedback on CPAP use and enhance self-management. Although such technologies have significant potential to improve the care of patients with OSA, monitoring of patient data can be labour-intensive, and only limited evidence currently exists to support their role in patient management. 87 Thus, further research into the effectiveness of telehealth strategies in improving CPAP adherence and other patient outcomes is needed.
Alternative Treatment Options for Obstructive Sleep Apnoea
Although CPAP is the gold standard treatment for OSA, poor overall acceptance and adherence have led to the development of several alternative therapies. These include behavioural modifications (eg, weight loss, avoidance of the supine sleep posture) and treatments such as mandibular advancement splints (MAS) and UA surgery.88,89 Mandibular advancement splints and UA surgery are most frequently prescribed for patients with mild to moderate OSA and those of any OSA severity who are intolerant of CPAP treatment. 90
Mandibular advancement splint
A MAS acts by producing forward protrusion of the mandible which is thought to improve UA patency by increasing the size of the oropharynx and by improving UA muscle tone. It has been shown, particularly in milder OSA, to be effective in reducing AHI, improving sleep, and reducing nocturnal blood pressure.89,91 A number of randomised, controlled studies have demonstrated that oral appliance therapy can produce clinically significant reductions in AHI and improvements in symptoms, including sleepiness and snoring.92–94 Use of a MAS has also been shown to be associated with a significant reduction in mean 24-hour DBP following 4 weeks of treatment compared with a control oral appliance in patients with OSA. 95 A meta-analysis of RCTs of MAS and CPAP reported that both MAS and CPAP were clinically effective in treating OSA in terms of improving ESS scores; however, CPAP therapy produced a greater reduction in AHI than MAS (difference in change −7.48 events/h, 95% CI: −5.77 to −9.19, P < .001). 96 The recently updated 2015 American Academy of Sleep Medicine (AASM) Clinical Practice Guideline for the treatment of OSA and snoring with oral appliance therapy recommends that a MAS, rather than no therapy, should be considered for patients with OSA who are intolerant of CPAP or who prefer alternative therapy; MAS should be made by a qualified dentist using a customised, titratable device; and that follow-up sleep study testing should be conducted to confirm treatment efficacy. 97
UA surgery
Surgical modification of the UA has also been used to treat patients with OSA, including procedures such as uvulopalatopharyngoplasty (UPPP), maxillo-mandibular advancement, laser-assisted uvulopalatoplasty, radiofrequency ablation, and a combination of surgical approaches known as multilevel (or stepwise) surgery. Upper airway surgery is a treatment option sometimes prescribed for OSA; however, the treatment benefit varies widely, and overall, significant benefits have not been consistently demonstrated. 88 A recent systematic review and meta-analysis of the evidence for UA surgery in the treatment of OSA revealed a paucity of high-level, randomised, controlled trial data, with most of the published literature consisting of small case series. 98 In addition, there was a great deal of variability in reported preoperative assessment strategies, surgical techniques, and in postoperative follow-up. Randomised controlled trial evidence of the effectiveness, safety, and cost-effectiveness of UA surgery for OSA is needed. Surgical therapy of OSA is, therefore, generally recommended only for patients who decline or are intolerant of CPAP and in whom oral appliance therapy is ineffective or undesirable. 99
Sleep posture modification
Gravity has major effects on UA tissues such as the tongue and soft palate and on diaphragm position and lung volume. Sleeping in the supine position increases the propensity for UA collapse because of posterior prolapse of the tongue and soft palate; and in the recumbent position, increased abdominal pressure and lower lung volume reduce the stiffness of UA by reducing the downward longitudinal tension exerted by the trachea and other intra-thoracic structures. 100 Sleep posture modification can be an effective form of treatment for patients with positional OSA (ie, OSA which is worse during supine, compared with lateral sleep). A study assessing the effects of the ‘tennis ball technique’ (TBT; ie, wearing a tennis ball strapped to the back during sleep) showed that patients who reported good compliance with TBT had reductions in their supine sleep time and AHI, as well as subjective improvements in sleep quality, daytime alertness, and snoring loudness compared with patients who had stopped using TBT. 101 Another study which used a prospective, randomised, crossover design to compare the effects of positional therapy versus CPAP also showed a reduction in the total amount of supine sleep and in AHI with use of a ball strapped to the back and found improvements in daytime sleepiness (as measured by the ESS), cognitive performance, mood scores, and quality of life measures, which were comparable with those with CPAP treatment. 102 However, long-term compliance with TBT has been shown to be problematic, 103 which may limit its effectiveness as a therapeutic option for OSA. Vibrating, electronic posture control devices worn around the chest or neck are showing promise as an alternative non-CPAP therapy to limit supine sleep in patients with position-dependent OSA, with data from at least 2 studies demonstrating their effectiveness in reducing the percentage of supine sleep, improvements in daytime sleepiness and sleep-related quality of life, and reasonable compliance.104,105
Lifestyle and behavioural modifications
In some patients with OSA, lifestyle and behavioural modifications may be sufficient to effectively control disease and improve symptoms of OSA. Weight loss has been shown to reduce AHI and improve symptoms particularly among patients with mild OSA.106,107 A previous study revealed that a 10% loss of weight was associated with a 26% reduction in AHI. 108 An intensive lifestyle intervention consisting of a very low calorie diet and regular nutritionist visits for 1 year was shown to be effective in achieving significant weight loss and AHI reduction compared with a single counselling session with general dietary and exercise advice as first-line treatment for patients with mild OSA. 107 Although losing weight is challenging for many patients and few achieve sufficient weight loss to significantly improve their OSA and an even smaller number of patients are able to maintain weight loss long term, 90 it is an important treatment goal for most patients with OSA. Weight loss added to CPAP has an incremental beneficial effect on some cardiovascular risk markers. 109 Other behavioural treatments include smoking cessation and reduced alcohol consumption (particularly before bedtime) as these factors have been associated with worsening OSA and OSA-related symptoms.110,111
Lifestyle factors may also contribute to insomnia (difficulty with falling and/or maintaining sleep) which, as outlined above, frequently co-occurs with OSA. Reducing stress and improving sleep hygiene can significantly improve sleep efficiency and increase the success of conventional OSA therapies, although specific behavioural treatments (eg, insomnia-specific cognitive behaviour therapy) may be required for patients with co-occurring chronic insomnia.
Hypoglossal nerve stimulation
Stimulating the genioglossus muscle, the main dilator of the pharynx, via an implanted pacemaker device with electrodes attached to the hypoglossal nerve and triggered to fire on inspiration, has now been shown to be feasible and effective in treating sleep-related UA obstructions in selected patients with OSA 112 and to have sustained long-term beneficial effects. 113 However, the role of this more invasive therapy in the OSA treatment has yet to be established and is likely to appeal to a relatively small proportion of patients who fail or refuse the more conservative approaches outlined above, or those in whom the predominant phenotypic deficit or aetiology is a suboptimal pharyngeal dilator muscle reflex response to airway obstruction (see below).
Overall principals of management
Obstructive sleep apnoea is a common disorder that has all the characteristics of a chronic condition. 105 There are many disease management issues for patients with OSA, including lifestyle factors known to contribute to OSA severity, multiple comorbidities, low CPAP adherence, and socioeconomic problems. 114 Mental health issues such as depression are also associated with sleep apnoea. Poor health literacy is potentially a major barrier to optimal disease control. According to the Adult OSA Task Force of the AASM clinical guidelines in 2009, OSA should be approached as a chronic disease requiring long-term, multidisciplinary management. 115 Integrated care for OSA involves collaboration between health professionals from a variety of disciplines including specialist nurses and primary care physicians. Strategies aimed at promoting patient self-management and improving communication between patients and health care providers, including structured, chronic disease self-management programmes, and use of e-health interventions and related technologies, may be of benefit for patients with OSA. 116
OSA Phenotyping and Personalised Sleep Health
Novel targets for therapy based on pathophysiological phenotype
A major issue in managing patients with OSA is the often substantial inter-individual variability in treatment responses to various modalities, including CPAP. Knowing the contribution of different physiological phenotypes or traits to OSA on an individual level is important as it opens up the potential for targeted and individualised OSA therapies. 117 In some patients, the primary cause of OSA may be an anatomical compromise of the pharyngeal airway; therefore, UA surgery involving UPPP or multilevel surgery may be beneficial as an alternative to CPAP therapy in non-adherent patients. For patients whose predominant deficit is pharyngeal muscle dysfunction, enhancing hypoglossal nerve output, either by drug treatment or by electrical stimulation, could be an option. In other individuals, a low arousal threshold to increasing respiratory drive predisposes them to OSA. When apnoea occurs, the individual must stay asleep long enough for the pharyngeal muscles to activate and restore airway patency thus establishing stable sleep. If arousal occurs too quickly, the individual will cycle between sleeping hypopnoea and waking hyperpnea. 118 Recent evidence suggests that drug therapies may be a good option for patients with OSA in whom low arousal threshold is the primary phenotype for leading to cyclical UA collapse.119–121 The hypnotic, eszopiclone, was shown to raise the arousal threshold to respiratory stimuli in a subset of patients with pre-existing low arousal threshold resulting in an improvement in their OSA, as measured by the AHI. 122
Despite these early promising findings on personalised phenotype-specific approaches to treat OSA, there are still challenges in measuring OSA phenotypes in routine clinical practice. Currently, this requires detailed and specialised physiological investigation 10 which is beyond the reach of most laboratories. 123 However, there has been significant recent progress in this area. Edwards et al 124 reported, for example, that more than 60% of the variance in arousal threshold can be determined using standard polysomnographic data, thereby perhaps establishing which patients might respond to pharmacological manipulation of arousal threshold. Similar approaches are being developed to quantify the contribution of central ventilatory control instability (or ‘loop gain’) to OSA pathogenesis using clinical polysomnography.
Apart from methods to assess the phenotypic traits causing OSA, there have also been recent advances in developing biomarkers to predict treatment responses. A recent example is the prediction of blood pressure response to CPAP treatment. As mentioned above, there is a very high prevalence of OSA (approximately 80%) among patients with resistant hypertension 125 compared with patients with non-resistant hypertension (approximately 40%). 126 Continuous positive airway pressure therapy appears more effective in resistant hypertension and is potentially of much greater benefit in this situation when pharmacologic approaches have been exhausted. However, blood pressure response to CPAP in patients with OSA with resistant hypertension remains highly variable. Being able to distinguish between patients who will respond to CPAP and those who will not, in this setting, would be of considerable clinical value. Sánchez-de-la-Torre et al 127 recently reported that blood pressure responder status to CPAP therapy among patients with OSA with resistant hypertension can be predicted with a high degree of accuracy by measuring the plasma levels of 3 cardio-specific microRNAs.
Information gathered from routine sleep study investigations are also being explored in an attempt to distinguish patients with OSA who are particularly vulnerable to sleepiness and alertness failure.34,128 As we gain new insights and better understanding of the pathophysiology, phenotypic differences and heterogeneity in daytime consequences and treatment response between individual patients with OSA, as well as increasing range of novel therapeutics, the management of obstructive sleep apnoea could soon transition from a general approach to a more precise, individualised approach.
Footnotes
Peer Review:
Six peer reviewers contributed to the peer review report. Reviewers’ reports totalled 439 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AV- initial direction and write up, ongoing editing revisions, DM- overall concept and direction, revisions, CH- write up and editing, CC- Over all direction, write up and editing, revisions.
