Abstract
Topiramate (TPM) is effective for multiple seizure types and epilepsy syndromes in children and adults. Topiramate has adverse effects (including cognitive, depression, renal stones), but many of these are low incidence when started at a low dose and slowly titrated to 100 to 200 mg/day. Also, TPM has proven benefit for migraine, obesity, eating disorders, and alcohol use disorders, which can be comorbid in patients with epilepsy and may also be effective in subpopulations within specific psychiatric diagnoses. Recently approved extended-release formulations of TPM (Trokendi and Qudexy in the United States) have reliable data supporting their safety and efficacy for patients with epilepsy. They have potential for more rapid titration within 1 month to 200 mg/day and have better patient retention than TPM immediate-release, but there are no robust double-blind randomized controlled trials comparing the different formulations. We expect the once per day extended-release formulations to improve medication adherence compared with the twice per day formulations. This has significant potential to improve outcomes in epilepsy and the other TPM-responsive disorders.
Keywords
Introduction
Epilepsy is a common neurologic condition, with approximately 1 in 26 people diagnosed in their lifetime.1,2 As pharmacotherapy is the fundamental intervention for seizure control with resultant improvement in quality of life for people with epilepsy (PWE), selecting the appropriate antiepileptic drug (AED) among the 24 currently available AEDs (as of January 2016) is an important but complicated choice. Preferences of the clinician, patient, and payer are the leading factors driving the specific AED choice. These preferences include the main goals of maximizing therapeutic efficacy and minimizing adverse effects while limiting disruption to the patient and his or her caregiver’s lives. Consistent with these goals is the pharmacologic goal to maintain stable, effective, and minimal necessary plasma concentrations to prevent seizures and minimize adverse drug effects.
A major cause for treatment failure is medication intolerance. In total, 10% to 30% of PWE typically do not tolerate a seizure drug due to adverse effects. 3 The options in these cases are to lower the dosage, use another medication, or use a different formulation of the drug. Use of slow-release formulation as opposed to short acting (ie, immediate-release [IR]) can reduce adverse effects while maintaining seizure control. The altered release mechanism in longer acting formulations can result in slower absorption rates and decreased peak concentrations, resulting in fewer adverse effects than those who have peak dose adverse effects. Long-acting formulations (ie, controlled-release [CR], extended-release [ER]) normally decrease dosing frequency, potentially improve medication adherence, improve efficacy associated with trough concentrations, and stabilize blood concentrations. 4
Several formulation principles can be applied to create CR properties for a drug product to promote desired drug release over time. A drug product consists of the active chemical ingredient (the drug) and inert excipients (nondrug ingredients that form the vehicle or formulation matrix). Hydroxypropyl methylcellulose, proteins, and polyvinyl acetate are commonly used excipients in CR formulations to control drug release and dissolution over a long period of time. These excipients are added to fillers, lubricants, disintegrants, and surfactants, which are standard components of IR tablet formulations. Examples of CR formulations include Sinemet, Dexedrine Spansules, Ritalin LA, and Concerta. Other modifications of excipients and delivery vehicles may alter drug release but not necessarily result in a CR product. For example, certain drugs are encapsulated using a hard or soft gelatin. Once the capsule is broken, the drug can be rapidly absorbed. The use of other technologies, such as enteric-coating (eg, with cellulose acetate trimellitate or other substances), can delay drug release with some site and timing specificity (ie, delayed-release [DR]). This may have benefits such as decreasing amount of drug reaching gastric mucosa, promoting capsule breakdown in the small intestine, and decreasing gastric bleeding in the case of enteric-coated aspirin. 5
Therefore, it is important to compare AEDs that have multiple manufacturers or formulations (IR, CR, DR, and ER) as they may have different release properties. For example, to minimize gastrointestinal adverse effects, 1 formulation of valproic acid has an enteric-coating to prevent it from being broken down in the stomach and allows it to be released in the lower gastrointestinal system. This delays peak concentration and decreases adverse effects while extending the duration of action. However, this enteric-coated DR formulation has significantly different Cmax, Tmax, and bioavailability values compared with the ER formulation. 6
In this article, we examine the classic and more recent evidence for topiramate (TPM) in PWE. We place special emphasis on data for the relatively new ER formulations of TPM (Trokendi XR, Supernus Pharmaceuticals, Inc., [Rockville, Maryland] and Qudexy ER, Upsher-Smith Laboratories, Inc. [Maple Grove, MN]) and how it improves adherence. We will also discuss support for TPM-ER’s role in other conditions that often co-occur in PWE, including treatment of migraine, obesity, eating disorders, and alcohol use disorders. Last, we will examine the costs and benefits when using TPM-ER in these multiple conditions. We believe TPM-ER formulations have an important role in epilepsy, and this role may increase in other comorbid conditions after sufficient comparative effectiveness studies and cost-benefit acceptance.
Indications
TPM is an antiepileptic drug marketed for epilepsy as an IR formulation since 1996. This includes monotherapy and adjunctive therapy usage in children 2 years old through adulthood for partial-onset seizures (POS) and generalized tonic-clonic seizures (GTCs). It is also used for other seizure types in Lennox-Gastaut syndrome. 7 Topiramate does not have robust data supporting its use for the treatment of primary myoclonic, absence, or infantile spasm seizures though it may be considered when alternative AEDs are ineffective or not well tolerated. Furthermore, TPM is Food and Drug Administration (FDA) approved for migraines and in combination with phentermine for weight loss.8,9 We will discuss those and select psychiatric usages in the “Indications and Efficacy in Other Conditions” section.
The 2 ER formulations of TPM differ in how they are formulated; the primary goals are to deliver the drug at a predetermined rate slower than that of TPM-IR and to decrease dosing frequency. Trokendi (TKR) was approved by the FDA for epilepsy in August 2013. It is formulated with a combination of IR and 2 types of ER beads using the Microtrol drug delivery system to slow the absorption of TKR more than 20-fold relative to TPM-IR. The Microtrol® technology Supernus Pharmaceuticals, Inc., (Rockville, Maryland) is also used in Carbatrol and Adderall XR (Shire US Inc., Wayne, PA). 10 Qudexy (QXR) was approved by the FDA for epilepsy in March 2014. It is a capsule with coated beads, which results in slow release over a 24-hour period. QXR can be sprinkled onto soft foods.
Pharmacology, Pharmacokinetics, and Dosing
Topiramate works as an anticonvulsant by blocking voltage-dependent sodium channels, augmenting the activity of the neurotransmitter gamma-aminobutyric acid and antagonizing the A-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)/kainite subtypes of glutamate receptors. It is also a weak inhibitor of carbonic anhydrase (CA, types II and IV) isoenzymes, which may modulate pH-dependent activation of voltage- and receptor-gated ion channels.3,10,11 This unique combination of activities may account for its broad-spectrum role against various seizure types.
Topiramate is used in children and adults typically at dosing of 50 to 400 mg/day for epilepsy and for several indications specified above. It is titrated slowly due to increased chance for intolerable adverse effects with rapid titration. For adults, it is often started at 25 to 50 mg/day, titrated by 25 to 50 mg/day every 1 to 2 weeks until the 200 to 400 mg/day range and/or the desired seizure outcome is reached. Twice a day dosing is used for the IR formulation, and once a day dosing is used for the ER formulation. Some drug-sensitive patients, especially those with the potential for pharmacodynamic interactions, may benefit with lower initial dosing, slower titrations, and even lower goal titration range. Children 2 to 9 years old are typically started at 25 mg/day (or 1-3 mg/kg/day), titrated up by 1 to 3 mg/kg/day every 1 to 2 weeks, and maintained at 5 to 9 mg/kg/day divided in twice a day dosing for IR formulation. Monotherapy may have higher targets at 200 to 400 mg/day compared with the weight-based dosing in children. 3 A dose regression analysis examining all available dose-response trials for children and adults found little evidence for major efficacy at dosages greater than 400 mg/day. 12 The recommended adult dosing of both ER formulations is 200 to 400 mg daily for adjunctive therapy; it is suggested as 400 mg once daily for primary GTCs.10,11 There is much anecdotal data in epilepsy practice that patients with less severe epilepsy (ie, low seizure frequency or nondisabling seizures) can have good efficacy with dosages of 100 to 200 mg/day and achieve necessary outcomes. 13 We have seen these dose targets of less than 200 mg/day to be sufficient several times in practice for a variety of nonsevere focal and generalized epilepsies. In a study comparing 50 and 500 mg/day TPM monotherapy, plasma concentrations >9.91 µg/mL were associated with better seizure control than concentrations <9.91 µg/mL. The therapeutic range often includes 2 to 25 µg/mL; concentrations >25 µg/mL result in higher adverse effect rates and minimal additional benefit. 13 Routine measurement of serum drug concentrations is not necessary if patients have good efficacy, no adverse effects, and no interaction concerns.
Patients with renal impairment defined as CrCl <50 mL/min/1.73 m2 should receive half the usual daily dosage. 14 Patients should receive a supplement after hemodialysis. With major hepatic metabolism dysfunction, it is prudent to reduce dosage in noncritical seizure situations and monitor more closely for adverse effects and drug concentrations. 14
There may be an increased risk of hyperammonemia when TPM is used in combination with valproic acid. It is unclear whether these drug interactions occur less frequently with the 2 TPM-ER formulations compared with TPM-IR as no comparative studies have been done, although we expect those interactions to be similar given their mechanism.10,11
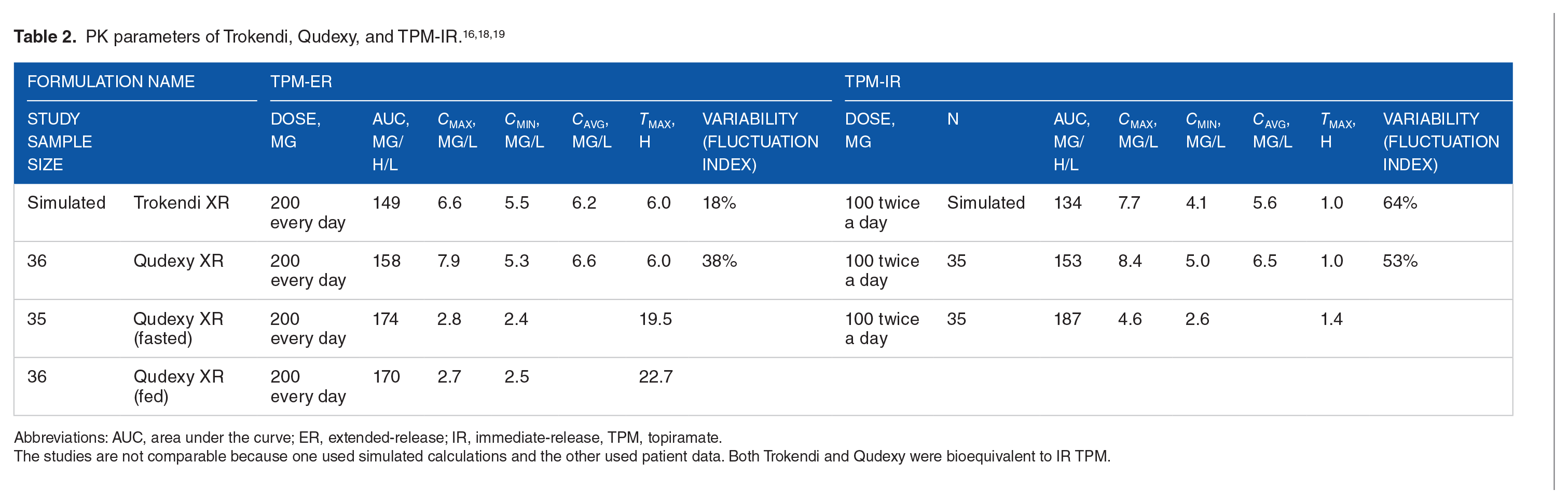
Both TKR and QXR follow linear pharmacokinetics for doses 50 mg and higher. Nonlinear pharmacokinetics were seen at the 25 mg dose due to binding of TPM to CA in red blood cells. 15 High-fat meals can affect Tmax (time to maximum concentration) and Cmax (maximum or peak concentration) but do not appear to change AUC (area under the curve; Table 1).10,11,16 One important aspect to note regarding TKR is that in vitro studies found that alcohol alters the pattern of TPM release from the capsules to potentially lead to higher plasma concentrations early after administration and subtherapeutic concentrations later in the day. 10 Other administration information about QXR and TKR is summarized in Table 1. The QXR capsules can be opened and sprinkled onto soft food with the same bioequivalence as taking an intact capsule, allowing for use in patients with swallowing difficulties. The QXR capsules have also been opened and administered through a feeding tube to minimize the number of daily doses. 17 This sprinkle-use is not supported by the TKR data; however, a few of our patients have opened TKR capsules and mixed them into soft items such as applesauce and yogurt. These few patients have not reported any changes in seizure control. The conversion from TPM-IR to TPM-ER is one to one. 18
Selected features of Qudexy ER and Trokendi XR.
Annual cost is a very crude estimate of price listed by supplier from wholesale acquisition price (which does not include rebates and other incentives that pharmacy receives from supplier before purchasing drug). It was derived from 3 samplings at pharmacies in 2016. Topiramate immediate-release generic cost is approximately $70 to $100 per year.
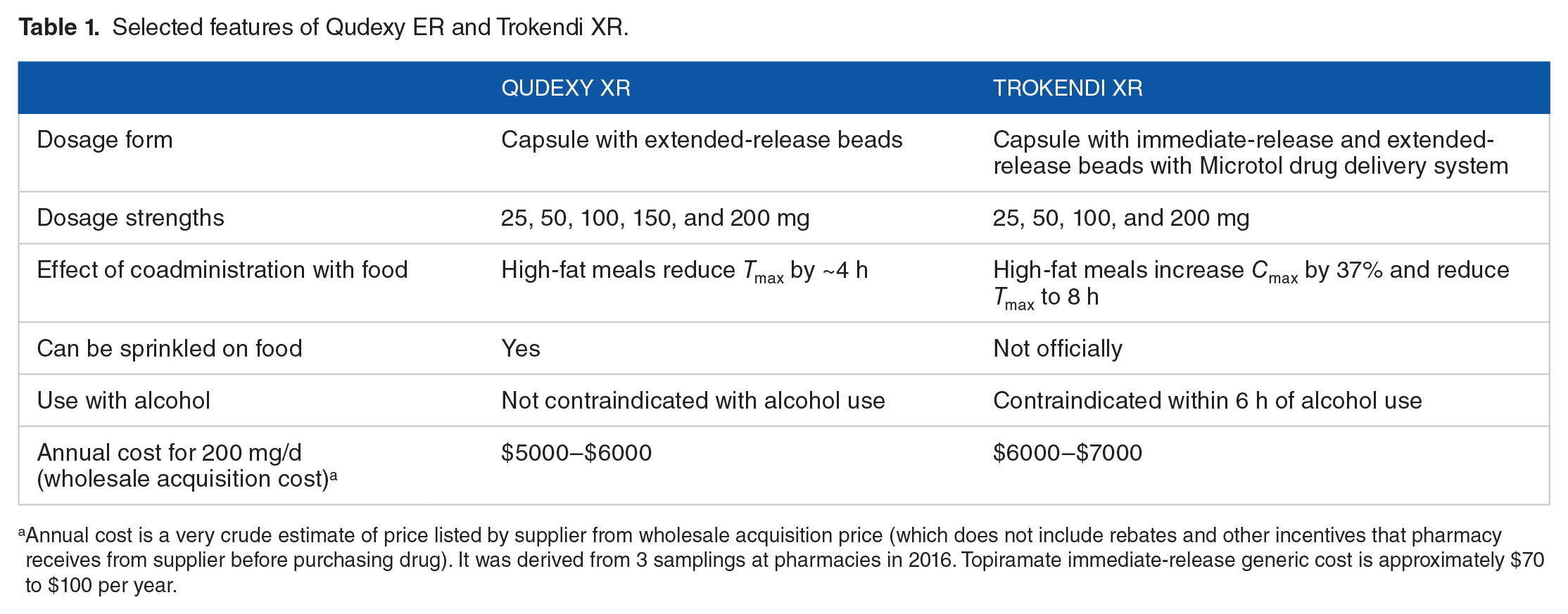
Multiple studies have shown that pharmacokinetics (PK) parameters between TPM-IR vs TKR and TPM-IR vs QXR are similar with equivalent plasma exposure (see Table 2, and Figures 1 and 2). Figures 1 and 2 demonstrate decreased concentration fluctuations and less frequent concentration peaks and troughs over 24 hours when using TKR or QXR compared with TPM-IR 100 mg twice a day. These data came from PWE undergoing formulation change studies.18,19 A randomized, open-label, single-centered, multiple-dose study assessed 38 patients receiving TPM-IR 100 mg administered every 12 hours or QXR 200 mg once daily. Patients were assessed over two 14-day maintenance periods, a crossover period, and 12-day titration period. 18 Although QXR and TPM-IR demonstrated equivalent steady-state, peak and trough exposures, QXR took more time to reach peak concentrations compared with TPM-IR (6 vs 1 h, respectively). QXR also had greater plateau time than TPM-IR (13 vs 4 h, respectively). Furthermore, QXR had a lower peak (Cmax) and higher trough (Cmin) concentration when compared with TPM-IR (5.3 vs 5.0 mg/L; P < .001). There was a 26% decrease in plasma fluctuation index after a single dose of QXR when compared with TPM-IR dosing. Switching between the 2 formulations did not significantly affect TPM concentrations. Therefore, QXR was an equivalent alternative to twice-daily TPM-IR. A simulation study comparing steady-state PK profiles of daily TKR and twice-daily TPM-IR showed that peak-trough fluctuations were higher with once-daily TPM-IR compared with TKR (64% vs 18%, respectively). 19 As seen in Table 2, the Cmax was lower for TKR than for TPM-IR. There are no direct comparisons between the 2 TPM-ER formulations. However, when comparing the simulated PK results of TKR with the actual steady-state concentrations of QXR, TKR has a predicted lower Cmax compared with that of QXR Cmax (6.6 vs 7.9 mg/L, respectively). Lower AUC and average serum concentration are also noted for TKR when indirectly compared with QXR. Both have the same peak dose time of 6 hours.18,19 To what extent these fluctuations of 0.5 to 1.5 mg/L over 24 hours improve or worsen efficacy and adverse effects for large cohorts of PWE is unclear. We consider patients who may most benefit from the conversion of TPM-IR to TKR or QXR to be those (1) having seizures due to medication nonadherence, (2) reporting adverse effects mostly in the 1 to 2 hours after taking TPM, and (3) having seizures correlating with the hours when TPM is at trough concentration.
Abbreviations: AUC, area under the curve; ER, extended-release; IR, immediate-release, TPM, topiramate.
The studies are not comparable because one used simulated calculations and the other used patient data. Both Trokendi and Qudexy were bioequivalent to IR TPM.
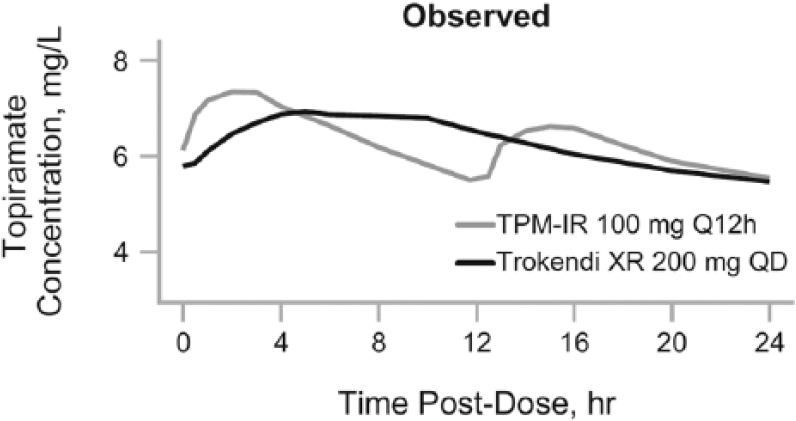
Steady-state TPM concentration vs time. In 49 PWE taking TPM-IR 100 mg twice a day and switched to Trokendi XR 200 mg once daily. IR indicates immediate-release; PWE, people with epilepsy; TPM, topiramate.
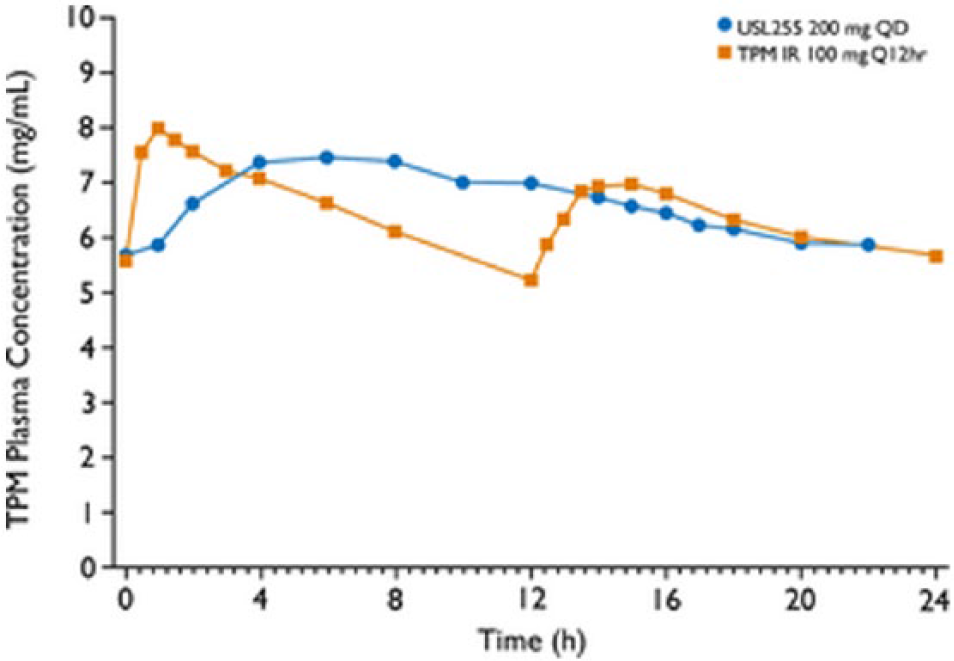
Steady-state TPM concentration vs time. In 36 healthy adults taking TPM-IR 100 mg twice a day and switched to Qudexy ER 200 mg once daily. IR indicates immediate-release; TPM, topiramate.
Hepatic enzyme induction
Topiramate has been shown to be a weak inducer of the hepatic microsomal enzyme system. Hepatic cells treated with high concentrations (>50 µM) of TPM in vitro appeared to induce the microsomal enzyme system. 20 Those in vitro concentrations are many times greater than patients’ therapeutic serum concentrations. Serum concentrations are likely to vary significantly within the population, and the dose beyond which clinically significant hepatic induction happens has not been determined. The induction effect is most likely at doses greater than 400 mg/day. 21 Doses less than 200 mg/day are unlikely to lead to any significant induction. 22 The hepatic microsomal system is involved in several important reactions in the body, including, but not limited to, hormone synthesis, vitamin D metabolism, and cholesterol synthesis. Induction of this system can decrease free concentrations of sex hormones and vitamin D concentrations, increase low-density lipoprotein cholesterol, and cause changes in other markers of cardiovascular risk.23,24
Hepatic induction can also lead to enhanced clearance of several drugs. 23 Closer monitoring of efficacy of drugs induced by high dosages of TPM is a prudent measure. Strong inducers such as phenytoin and carbamazepine have been shown to decrease plasma concentrations of TPM. 25 The clearance of ethinyl estradiol is also increased in the presence of TPM, especially at high doses. As noted in one study evaluating pharmacokinetics of a birth control medication, high-dose TPM is associated with both an increased ethinyl estradiol clearance and an 18% to 30% decreased ethinyl estradiol AUC. 26 Therefore, it would be prudent to use oral contraceptive pills having a higher estrogen content while on TPM 21 ⩾200 mg/day and/or to use a different form of contraception. 21 One should consider decreased efficacy of ethinyl estradiol–containing contraception if menstrual bleeding patterns change.
Conversely, TPM appears to inhibit CYP2C19, an isoenzyme of the CYP450 system. This can manifest as a mild increase in phenytoin concentrations in patients on dual therapy. 27 This usually does not result in clinical toxicity, but it may be judicious to monitor older PWE and patients on a high dose of phenytoin. High dosages of TPM have been reported to increase serum concentrations of lithium and cause toxicity in a few patients through suspected decreased elimination.10,28
Efficacy
Topiramate efficacy for multiple seizure types and epilepsy syndromes has been demonstrated in several high-quality trials since the 1990s. Although concerns for adverse effects may lower its relative ranking, TPM is a first-tier choice for many clinicians. A review of 11 trials indicated that TPM is an effective add-on treatment for drug-resistant partial epilepsy with an estimated response rate (eg, >50% reduction in seizure frequency) of 45% (95% confidence interval [CI], 41.7%-48.2%) compared with 14.6% (95% CI, 11.7%-17.9%) for patients receiving TPM and placebo, respectively. 12 The Standard and New Antiepileptic Drugs (SANAD) study is the largest comparative effectiveness study examining the efficacy and tolerability of TPM-IR compared with several other seizure drugs (carbamazepine, gabapentin, lamotrigine, or oxcarbazepine for POS; valproate or lamotrigine for generalized or unclassifiable epilepsy). Most patients were untreated before entering this unblinded, randomized trial in the United Kingdom. At 1-year follow-up, TPM was equal to or better than the other AEDs in efficacy for the focal epilepsy or generalized/unclassified epilepsy.29,30 At 1-year follow-up, there was a trend without statistical significance of (1) carbamazepine having better efficacy than TPM in the POS arm, 30 and (2) valproate having better efficacy than TPM in the generalized/unclassifiable epilepsy arm. 29 A randomized controlled trial (RCT) by Ramsay comparing phenytoin 300 mg/day with TPM-IR 50 mg twice a day as monotherapy in new-onset adult PWE could not establish that TPM was noninferior to phenytoin in the seizure freedom at 28-day efficacy measure (81% seizure-free for TPM and 90% seizure-free for phenytoin). In addition, phenytoin could not be shown to be superior to TPM (P = .366). One aspect of this study that may have limited TPM was the 100 mg/day maintenance dose. 31
The FDA approval of TKR was based on showing bioequivalence with TPM-IR for AUC, Cmax, and Cmin. A drug is considered bioequivalent as long as the concentrations are within a 90% CI, meaning that concentrations can be 80% to 125% from the compared drug. 32 However, QXR had a phase III, double-blinded, placebo-controlled trial assessing its efficacy compared with placebo as adjunctive treatment for refractory POS in patients taking 1 to 3 concurrent AEDs. 33 This international PREVAIL study included 249 randomized adults (18-75 years old) with relatively severe epilepsy. After an 8-week baseline and 3-week dose titration period, patients received either QXR 200 mg/day or placebo for 11 weeks. For all POS patients, the comparative reductions in POS median weekly frequency for QXR and placebo were 39.5% and 21.6% (P < .001), respectively; similarly, the comparative responder rates for QXR and placebo were 37.9% and 23.2% (P = .013). 33 In the subgroup of patients who had complex partial or secondarily generalized seizures, the QXR formulation was more effective than placebo with a respective 40.6% and 17.7% (P < .001) reduction in seizure frequency. The decreased POS frequency was also maintained in patients receiving 1, 2, and ⩾3 AEDs. Although there were no significant differences in total quality of life scores, there was a significant improvement in seizure worry and nearly twice as many patients receiving QXR had improved Clinician-Reported Global Impression of Change scores. 33
Safety and Tolerability
Like most other AEDs, adverse effects of TPM primarily stem from its direct effects on the central nervous system (CNS). A few adverse effects are related to some unique properties of this drug such as the presence of a sulfa moiety and carbonic anhydrase inhibition (CAI). Idiosyncratic effects are seldom seen with TPM use. No major hematologic or hepatic dysfunction has been reported, though a decline in platelet counts has been reported when TPM is added to valproic acid.34,35 Acute myopia with increased intraocular pressure occurs as a rare complication of TPM use and presents with bilateral eye pain and blurriness. 36 Fortunately, discontinuation of treatment appears to lead to resolution of symptoms. Some of the CNS adverse effects are worse with pharmacodynamic interactions. Some other adverse effects from pharmacokinetic interactions have been mentioned in the Pharmacology, Pharmacokinetics, and Dosing section. There is a paucity of long-term studies specifically looking at the ER preparations of TPM. Nearly all our understanding of the adverse effects stems from studies on TPM-IR. Comments on ER studies are made where applicable.
CNS adverse effects
The most frequently reported adverse effects of TPM are nonspecific symptoms of fatigue, dizziness, somnolence, and psychomotor slowing. 34 Using a much slower titration schedule mirroring current clinical practice has been shown to reduce these adverse effects in comparison with earlier studies, which used a rapid titration and far higher doses. 37 Furthermore, the incidence of adverse effects is lower if concomitant AEDs are tapered as TPM is up-titrated. 38 In the previously cited Ramsay RCT (TPM-IR 100 mg/day vs phenytoin 300 mg/day), 6.8% of patients taking TPM and 13.4% taking phenytoin withdrew from the study in the first 4 weeks due to adverse events. The mean AED blood concentration for TPM was 3.6 µg/mL (SD, 1.34) in the month after initiation. 31 This study supports that a lower maintenance dose can improve tolerability.
In addition to the above reported nonspecific symptoms, studies have also shown a higher incidence of neurocognitive problems with TPM use such as a decline in memory, psychomotor slowing, and language problems. 34 They are often the reason for discontinuation of the drug. When a single 100 mg dose of TPM-IR was administered to 158 healthy volunteers, several aspects of cognition were affected to varying degrees. Moreover, this effect appeared to be independent of age, gender, and level of education. 39 There is some patient specificity to the neurocognitive adverse effects with some patients being more susceptible to this effect. 40 Along the same line of evidence, patients with dominant temporal lobe epilepsy and simple partial seizures appear to be more susceptible to word finding difficulty. 41 A study comparing lamotrigine, gabapentin, and TPM-IR in healthy volunteers showed that there was a significant decline in attention and word fluency in patients on TPM. 42 In PWE previously taking carbamazepine, TPM addition resulted in a worsening of short-term memory and verbal memory.40,43 The risks of cognitive impairment and psychiatric effects are less when the dose is gradually titrated. 44
Very few studies have compared head-to-head different AEDs when used in monotherapy in PWE. One study reported that the use of TPM-IR was associated with a higher incidence of concentration difficulty, language problems, and confusion (11%, 7%, and 6%) at 200 mg/day in comparison with carbamazepine and valproate. 45 However, the same study showed that a lower dose of TPM-IR (100 mg/day) was equally efficacious and better tolerated with comparable adverse effects. A high starting dose and rapid titration to 400 mg/day appear to increase the probability of these adverse effects. 46 The SANAD study showed TPM-IR had more unacceptable adverse events than valproate in the generalized/unclassifiable epilepsy trial arm, 29 and TPM-IR had more unacceptable adverse events than lamotrigine and gabapentin in the POS trial arm.29,30 In those trials, 75 of 226 (33%) patients in the generalized/unclassified arm and 129 of 358 (36%) patients in the POS arm stopped TPM-IR due to unacceptable adverse effects in per-protocol analyses. The mean TPM-IR dose was 150 to 200 mg/day in these subgroups.29,30
Studies exclusively examining the adverse effects of the ER formulation are rare. The pivotal PREVAIL study titrated QXR from 50 to 200 mg/day over 3 weeks using 50-mg weekly increases. Only 10% discontinued due to adverse events in the drug arm vs 3% in the placebo arm; the overall discontinuation rate was 17% vs 9%, respectively, over the 11-week treatment period. 33 These data provide some encouragement that achieving a QXR 200 mg dose is feasible for a large proportion of patients. 33 During the 55-week, open-label extension phase of the PREVAIL study, new-onset neurocognitive adverse effects were reported in 12.9% of the 210 patients treated with QXR, with aphasia accounting for 40.7% of these neurocognitive treatment emergent adverse events (TEAEs). 47 Metabolic acidosis and kidney stones were not reported in patients in this study.33,47,48 In an article that summarized phase I QXR data from 2 studies (25-400 and 600-1400 mg/day), the incidence of nausea in healthy volunteers was 0% in the placebo group; it was 17% (5 of 30) and 12% (5 of 40) for the lower and higher treatment ranges, respectively. Furthermore, there was a dose-dependent incremental reporting of TEAEs (11% for 25 mg, 46% for 400 mg, and 92% in study subjects who received doses of 600-1400 mg). 48 The Cochrane review of TPM-IR RCTs for PWE found 9% (14 of 160) of patients in 3 trials receiving TPM-IR had speech difficulty or language problems. However, these 3 studies were heterogeneous, and dosages were allowed up to 600 mg/day. 12
A manufacturer-funded study using the Controlled Oral Word Association test to assess verbal fluency found statistically significant better performance for those subjects receiving TKR 50 mg daily compared with subjects receiving TPM-IR 50 mg twice a day (P = .0002). This difference favoring TKR over TPM-IR was consistent throughout the 31-day titration to 200 mg/day, but the statistical significance was not maintained at each time point the subjects were tested (P ranged from <.001 to .41) and the trial was not fully blinded. These results come from a meeting poster presentation. 49 Other results for TPM-IR in comparison with QXR come from a meeting poster presentation pooling 3 separate single-center, phase 1, randomized, crossover studies comparing QXR 200 to 400 mg/day with TPM-IR 200 to 400 mg/day in 134 healthy volunteers. These manufacturer-sponsored studies allowed for up-titration periods of 12 to 35 days and maintenance dose observation periods of 7 to 14 days. Adverse effect rates having at least a 5% absolute value difference benefiting QXR compared with TPM-IR in the total treatment period were headache (18.1% vs 24.8%), memory impairment (2.9% vs 9.2%), and anxiety (2.9% vs 8.3%). However, weight loss was more frequently reported in the TPM-IR group than the QXR group (16.5% vs 11.4%). 50 These findings should be examined in larger, prospective, controlled studies appropriately designed for detecting significant differences in adverse events.
Inhibition of carbonic anhydrase
Some of TPM’s adverse effects are explained by its CAI property (CA, types II and IV). Several isoforms of CA are present in the body. This enzyme catalyzes the formation of carbonic acid from carbon dioxide and water and is very important for maintenance of acid-base balance. The clinically relevant effects of this inhibition are paresthesia and an increase in the incidence of renal stones. Paresthesia appears to be more common with monotherapy than when used as an add-on. 34 Renal stones develop in 1.5% of TPM-treated patients, and this is a 2- to 4-fold increase than in the general population. 34 Renal stone formation appears to be more common in men and is more common when other CAIs are combined. A family history of renal stones is also an independent risk factor for formation of renal stones. The CAI in the proximal and distal renal tubules and the resultant renal tubular acidosis are believed to be the causes for the stones. 51 Calcium carbonate and oxalate stones are most commonly reported with TPM use. Despite the above, studies have not shown a clear and consistent relationship between dosage and duration of therapy with TPM and the incidence of renal stones. 51 In general, patients are advised to maintain adequate hydration while on TPM. A low-sodium diet would also likely help by reducing the renal calcium excretion. 52
Topiramate treatment is associated with decreased serum bicarbonate. This is usually a modest effect (average of 4 mEq/L decrease in serum bicarbonate) and for the most part is asymptomatic. In general, this effect is most prominent when the drug is newly initiated and tends to stabilize with continued treatment. One TPM monotherapy treatment trial reported that 15% of patients had serum bicarbonate values <18 mmol/L without attributable adverse effects; this finding was dose-dependent with 9% at TPM 50 mg/day and 22% at 400 mg/day. 53 Patients who have coexisting renal or respiratory dysfunction and who take other medications that affect acid-base regulation should have their serum bicarbonate concentrations monitored while on the TPM. As would be expected, usage of TPM with other CAIs (eg, zonisamide and acetazolamide) can increase the severity of metabolic acidosis. Concurrent use with metformin is contraindicated in patients with metabolic acidosis.
Hypohidrosis has been reported with TPM use, especially in children. 54 Some published cases of hypohidrosis in adults also exist.55-58 Although the exact mechanism of TPM-induced hypohidrosis remains a matter of debate, CAI likely contributes. Hypohidrosis was not reported in the open-label study of the QXR.47,59 However, the study used a low maximal dose (200 mg/day), and this effect may be more prevalent at higher dosages. Formulations of TPM should be considered as a cause in patients with unexplained symptoms of feeling hot or flushed, heat intolerance, decreased sweating, or unexplained persistent fever.54,58
Use in pregnancy
Topiramate is currently a pregnancy class D drug (ie, human and animal data suggest risk). 60 In 2011, the FDA issued a warning regarding the higher incidence of oral clefts in infants who were exposed to the drug in utero. The North American Pregnancy Registry showed a 1.4% prevalence of oral clefts in TPM-exposed infants. 61 In 2014, the registry also reported an increase in the incidence of low birth weight among infants exposed to TPM in utero (17.9%) compared with controls (5.0%). 62 When compared with lamotrigine-exposed babies, TPM-exposed babies had a 221 g decreased birth weight with a 1 cm decreased mean length. 62 This effect appeared to persist despite adjusting for factors such as age, parity, smoking, education, and folic acid intake. The serum concentrations of TPM appear to decline gradually during pregnancy. A study of 12 pregnant women showed that the concentration to dose ratio declined by 30% and 34% during the second and third trimester of pregnancy, respectively. 63 This would necessitate regular monitoring during pregnancy. Potentially, TKR or QXR may minimize peak dose adverse effects and maximize seizure efficacy.
Indications and Efficacy in Other Conditions
Weight loss
Approximately 36% of the US adults are obese. 64 There are no large rigorous epidemiological studies of obesity in epilepsy, but it is assumed to be similar or higher in PWE. A single-center epilepsy study estimated 31% of 554 consecutive adult PWE were obese. 65 Topiramate monotherapy is associated with weight loss, and a TPM combination drug (Qsymia, VIVUS [Campbell, CA]) with the amphetamine-related drug phentermine has an FDA-approved chronic use indication for weight loss in obese patients unable to sustain weight loss despite lifestyle modification.66,67 This phentermine/TPM formulation uses an ER TPM capsule, and the most common maintenance dose is TPM-ER 46 mg/day. In the Qsymia RCT, the mean weight loss was 8% to 10% in the drug group (vs 1% in the placebo). 8 Weight loss on TPM is primarily seen early in treatment, and there is a correlation with higher doses leading to greater weight loss in many patients. 68 Patients experience a decrease in appetite early on in therapy which likely contributes to the weight loss. However, the weight loss appears to continue even after the caloric intake returns to baseline suggesting possible alternate mechanisms behind this effect. 69 In a Veterans Administration (VA) study, women experienced more weight loss than men (43.2% of women vs 29.4% of men). 70 A prospective study looked at weight change in patients with focal epilepsy who were started on TPM-IR (without phentermine). The study showed a mean weight loss of 3 kg at 3 months and 5.9 kg at 1 year. In general, patients who weighed more prior to drug initiation appeared to experience greater weight loss. These patients show a reduction in body fat mass and a favorable metabolic profile such as better glycemic control and total cholesterol levels. 69 The effect appears to continue for about 2 years and at least 1 study showed that the effect tends to stabilize after 2 to 3 years of steady use. 68 In an adjunctive TPM-ER for medically refractory focal epilepsy RCT, subjects taking the TPM-ER lost approximately 2 kg more than placebo at 11-week follow-up (P < .001). 33 There is a paucity of data to derive conclusions about differences in weight loss from TPM-ER vs TPM-IR.
Migraine
Migraine has a prevalence of about 13% of the US general population, and PWE have a 2-fold greater risk of migraine than the general population. 71 They both appear to share a common pathophysiology of neuronal hyperactivity, and they sometimes are linked to each other in time. In fact, the term migralepsy was first used by William Lennox to describe a syndrome where nearly all migraines with aura were followed soon after by a seizure in PWE. 72 One of the main strategies to treat frequent disabling migraines is to use a daily preventive medication to reduce the frequency, duration, and severity of migraine attacks. The preventive medication may also enhance the response of acute treatments and improve a patient’s ability to maintain his or her work and lifestyle during attacks. Topiramate immediate-release was approved for migraine prevention in 2004 and is a well-established first-line preventive medication for migraine. 73 Four class I and 7 class II studies show that maintenance dosages of 50 to 200 mg/day of TPM-IR are effective in migraine prevention. 74 Dosing often starts at 25 mg nightly, and the dose is increased by 25 mg/day/week with a goal of 50 to 200 mg/day based on the response of the patient.
Topiramate extended-release has not been studied formally for migraine prophylaxis. We believe there is opportunity for TPM-ER to improve adherence and quality of life in patients with migraine due to low adherence rates. Oral preventive medications (including TPM-IR) were taken with adherence rates 21% to 80% at 6 months and 35% to 56% at 12-month assessment in a systematic review of 6 observational studies of adults with migraine. 75 Worse outcomes for migraine patients stopping their TPM have been demonstrated. A double-blind RCT examined 514 migraine patients on TPM who were randomized to remain on the drug vs switch to placebo for 6 months. There was 1 additional day of migraine per 4 weeks and 1 additional day of abortive medication usage in the placebo group compared with the TPM group (P < .01 for each measure). 76
Psychiatric disorders
Antiepileptic drugs are commonly prescribed for psychiatric and pain disorders.77,78 The original use of carbamazepine and divalproex sodium for the treatment of bipolar disorders has led to almost all AEDs being studied for multiple psychiatric disorders.77,79,80 However, to date, only 5 AEDs have received either FDA or European Medicine Agency indications for psychiatric disorders: carbamazepine, divalproex sodium, and lamotrigine for the treatment of bipolar disorders; benzodiazepines for the treatment of anxiety disorders, insomnia, and alcohol withdrawal; and pregabalin for the treatment of generalized anxiety disorder. 77 Topiramate is no exception and, though not having received approval for indicated treatment in any psychiatric disorder, TPM has been studied and/or clinically used in the treatment of 32 diagnoses within 13 major Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM-5) categories: affective, psychotic, anxiety, obsessive-compulsive, trauma-related, somatic, impulse control, eating, substance-related and addictive, neurocognitive, neurodevelopmental, paraphilic, and personality disorders (Table 3).77,81 Many of these studies have methodological limitations (eg, case reports; open-label, small sample size; short-duration, unclear randomization/blinding; and nonadjustment for potential confounders including comorbidities and polypharmacy). Appropriately powered and well-controlled studies are required to better determine the use of TPM in psychiatry. Nonetheless, a comprehensive literature review by the authors suggests that TPM may be effective in subpopulations within specific diagnoses.
Clinical psychiatric uses studied for Topiramate

Abbreviations: MDMA, 3,4-Methylenedioxymethamphetamine or ecstasy.
Furthermore, the literature supports the off-label use of TPM in eating disorders,82-88 weight control secondary to psychotropics in different psychiatric diagnoses,89,90 and alcohol use disorders.91-93 As mentioned earlier, TPM-ER combined with phentermine is FDA approved for the treatment of obesity. 66 Obesity, though not a psychiatric disorder, may be a key medical comorbidity in psychiatric patients secondary to adverse psychotropic effects, including second-generation antipsychotics with metabolic syndrome. 94 As such, TPM and TPM-ER may serve in a medical, as opposed to psychiatric, treatment role for psychiatric patients.
Eating disorders. There are controlled experimental data supporting TPM-IR as a therapy in eating disorders. Topiramate immediate-release was effective in reducing binging/purging measures in bulimia nervosa compared with placebo in 2 double-blind RCTs.82,83 Specifically, these combined 10-week trials examined a total of 124 patients (123 women; 1 man; minimum inclusion age 16 years) and showed statistically significant and clinically meaningful improvements in mean binging/purging frequency measures in each study with TPM dosages titrated to 250 mg/day in 1 study and median-daily dose of 100 mg (range: 25-400 mg) in the second study.82,83 There was an approximately 4 kg additional weight loss in the TPM-IR group compared with the placebo. 82 Topiramate is considered a treatment choice after selective serotonin reuptake inhibitors for bulimia nervosa. 84 Similarly, TPM-IR was effective compared with placebo in reducing binge-eating measurements and body mass index for obese patients with binge-eating disorder (BED). One small multicenter double-blind RCT of 73 obese patients (minimum inclusion age 18 years) studying the effect of adjunctive TPM-IR added to cognitive-behavioral therapy (CBT-TPM) compared with placebo added to CBT (CBT-placebo) showed a 5.9 kg additional weight loss in the CBT-TPM treatment group compared with the CBT-placebo treatment group as well as a statistically significant increased remission rate in binge eating. 85 A US-based multicenter RCT study of 407 obese adults with BED showed that TPM-IR (median dose 300 mg/day) significantly reduced weight and several binge-eating measures over 16 weeks compared with placebo. 86 In this large trial, which used a slow TPM titration, the discontinuation rate due to adverse events was 15% in the treatment group vs 8% in the placebo group of which memory difficulty and depression were the most common reasons for discontinuing TPM.
Weight gain associated with psychotropics. A small (n = 43) positive 10-week placebo-controlled double-blind RCT of adjunctive TPM added to olanzapine patients (OLZ-TPM) noted both a statistically significant and clinically meaningful weight loss of 5.6 kg compared with placebo added to OLZ (OLZ-placebo) at endpoint 89 with continued weight loss during an 18-month open-label extension phase with adjunctive TPM. 90
Alcohol use disorders. A small positive 6-week relapse prevention RCT (2 sites, placebo-controlled single-blind) for 52 alcohol detoxified patients found statistically significant improvement in multiple measures comparing TPM (100 mg twice daily) with placebo—greater abstinent and fewer relapsed patients, decreased drinking days, decreased alcohol consumption, greater treatment adherence, and decreased Clinical Institute Withdrawal Assessment for Alcohol Revised scale (CIWA-Ar) withdrawal symptoms. 91 A meta-analysis of 7 placebo-controlled double-blind trials (n = 1125) for alcohol use disorders found a statistically significant increased abstinence and decreased heavy drinking comparing TPM with placebo and trend toward decreased craving with TPM compared with placebo. 92 A 6-month open-label active comparator trial of 182 alcohol-dependent patients found statistically significant decreased composite measures comparing TPM with naltrexone. 93
Prior to using TPM for psychiatric disorders, it is important that the clinician be cognizant of potential negative psychiatric and behavioral adverse effects. In one study of 431 PWE, 23.9% had psychiatric adverse effects, including depression, psychosis, and aggressive behaviors. 95 A recent review of 90 TPM placebo-controlled randomized clinical trials noted the following statistically significant adverse effects: decreased cognition, memory impairment, attention difficulties, and somnolence. 96 Clinicians need to be judicious when prescribing off-label TPM with consideration of slow titration to minimize potential adverse psychiatric effects. 44 Although suicidal behaviors are considered a potential class adverse effect of all AEDs by the FDA, further research has noted contradictory findings and numerous articles have commented on multiple methodological flaws.97-99 For example, a geriatric VA study reported a statistically significant increased risk of suicidal behavioral for patients treated with TPM though confounders included age, specific VA population, gender, and severity/acuteness of psychiatric comorbidities. 100 The most recent large-scale research study which compared the UK Clinical Practice Research Datalink with the Danish National Registries did not find a statistically significant relationship between TPM and suicidal behaviors. 101
The authors are unaware of TPM-ER studies for the treatment of psychiatric disorders; however, its potential off-label use in psychiatry can be extrapolated from TPM studies. The longer duration of activity of ER products would be beneficial in patients with poor compliance or peak dose adverse effects related to TPM-IR. Finally, as medication nonadherence in chronic medical and psychiatric disorders is an important problem affecting therapeutic outcome, dosing strategies (once-daily as opposed to multiple-daily dosing) should be considered to maximize adherence leading to the potential use of TPM-ER rather than TPM-IR.102,103
Factors That Determine Adherence
There are numerous factors that can affect medication adherence. Common ones include adverse effects, lack of efficacy, and frequency of dosing. Medication nonadherence declines as dose frequency increases. 19 A retrospective analysis reported that up to 42% of patients did not take medications for chronic conditions as prescribed. 104 A survey of PWE found 66% missed doses at least once a month, and forgetfulness was the primary reason. 105 A prospective, observational trial of 2031 PWE switching from valproic acid IR to ER resulted in a statistically significant improvement in adherence rates from 40% to 70%. 106 In a pharmacokinetic conversion study of 61 adult patients on TPM, 92% of patients preferred once-daily dosing with TKR and expressed improved treatment adherence.107,108
Tolerability is also a proposed advantage of ER formulations due to lower peak values, reduced peak dose period where adverse events may occur, and fewer fluctuations in drug serum concentrations. A prospective, 3-month, open-label trial comparing carbamazepine IR vs ER in 453 patients found that an adverse event profile score reduced from 37.2 to 31.7 (P < .0001) from an IR to ER switch. 109 Other trials with divalproex, levetiracetam, and oxcarbazepine also found decreases in TEAEs.106,107,110,111
Efficacy is another major factor determining medication adherence to AEDs. A large trial involving 2031 PWE switching from valproate IR to ER had a 19% (P < .001) reduction in number of patients experiencing ⩾1 seizure per month. 106 However, other studies of divalproex demonstrated no change or only slight improvement in efficacy with a given ER formulation rather than IR formulation. Studies of other AEDs such as carbamazepine and lamotrigine IR vs ER formulations have also seen comparable effectiveness in controlling seizure frequency.107,109
Studies of health care insurance claims in PWE have found nonadherence to be associated with significant increases in seizures, ER visits and hospitalizations; higher inpatient costs; and increased mortality. The Research on Antiepileptic Non-adherence and Selected Outcomes in Medicaid (RANSOM) study examining Medicaid claims in adult PWE found nonadherent patients had 3 times higher mortality, 50% higher rate of ER visits, and 86% higher hospitalization rates when controlling for several other factors. 112 Younger age, adverse events, inconvenience, and social stigma are all associated with medication nonadherence. 107
In consideration of AED noncompliance in PWE, a theoretical concern arises that switching from an IR to an ER formulation may result in even lower trough concentrations for patients who continue to miss dosages. There is also a theoretical concern that a missed dose replaced too close to the next scheduled dose that might cause the peak concentration to increase sufficiently to cause toxicity. Multiple computer simulation studies of ER vs IR drug counterparts favor ER formulations because they have a smaller magnitude of concentration fluctuation. A computer simulation study using a population PK model for steady-state concentrations was used to compare strict dosing protocols with those that delayed TKR or TPM-IR doses by 4, 8, 12, 16, and 24 hours. 19 For patients not receiving enzyme-inducing drugs, Cmin was 9% to 31% lower in patients receiving TPM-IR and 6% to 27% lower in patients receiving TKR after the delayed dose. Although the changes were greater in patients receiving drugs that induced TPM metabolism, the overall effects were comparable. There were similar declines in Cmin after omitting the next scheduled dose of TPM-IR (21%) and TKR (27%). If the missed dose was replaced at the same time as the next dose was scheduled (doubling the dose), the TKR Cmax increased by 26% and the TPM-IR Cmax increased by 28%. Therefore, regarding missed doses, there is no concern from these simulations that ER formulations affected patients worse than IR formulations. A missed dose of TKR can be given at the time it is remembered. Patients on enzyme-inducing drugs are more susceptible to larger declines in trough concentrations with missed dosages, and they are also more susceptible to greater increases in peak concentrations with “catch-up, double” dosages. These changes are present with both ER and IR formulations.
As with TPM, pharmacokinetic simulation studies with lamotrigine and valproic acid had similar results. A lamotrigine simulation using data from 44 patients in a previous study found a lower Cmax and higher Cmin with the lamotrigine ER daily formulation compared with the same daily dose of a lamotrigine IR twice-daily formulation. Both formulations had similar changes in Cmax and Cmin after a delayed dose, missed dose, and doubled dose following a missed dose; the greatest changes were in patients receiving enzyme inducers. 113 In fact, concentrations with IR dosing were outside the concentration range of the ER formulation at least 55% of the time. A valproic acid simulation study indicated that a valproic acid ER replacement dose can be taken 12 hours after a missed dose with minimal risk of toxicity and decline in Cmin. 114 Creating guidelines from simulation studies is difficult because some patients may not tolerate changes of 25% from their usual concentrations associated with a missed or replacement dose. A more fine-tuned replacement dose may need to be calculated for the subset of patients who are more sensitive to small changes in concentration, receiving enzyme inducers, or near the next dosing interval.
With the FDA approval of 2 new once-daily formulations of TPM, medication compliance should be improved. There is potential for fewer adverse effects as these formulations have lower maximum concentrations with comparable trough concentrations; however, the magnitude of this benefit, as well as which patients are most likely to benefit, remains unclear and requires further research.
Conclusions
The long-acting forms of TPM, TKR and QXR, are welcome additions to several other FDA-approved ER formulations of AEDs.115,116 These ER formulations include lamotrigine, levetiracetam, oxcarbazepine (Oxtellar XR; Aydan extent), phenytoin, divalproex sodium (Depakote ER), and carbamazepine (Tegretol XR or Carbatrol) for epilepsy. Gabapentin (Gralise, Horizant) has XR versions FDA approved, although these versions are not approved for epilepsy but for postherpetic neuralgia and restless legs syndrome. These options clearly provide more convenience for patients and opportunities for increased compliance. How much these formulations decrease adverse effects compared with the IR formulations is not clear. Large differences in adverse effects seen in large numbers of patients are not expected for TKR and QXR based on the small and limited studies thus far.
Further research is needed using prospective, well-controlled trials examining for improvements in patients converted from TPM-IR to TPM-ER or in patients newly started on TPM-ER. Insurance and other population database cohort or crossover studies examining clinically meaningful outcomes of patients taking TPM-IR vs TPM-ER can provide additional data to test hypotheses and plan for more rigorous and better-controlled clinical trials comparing IR and ER formulations.
We provide the following advice for using TPM-ER rather than TPM-IR in a cost-sensitive health system: Due to the current limited data, significant cost difference, and presumably similar efficacy, TPM-ER should be considered for patients who need once a day dosing to maintain compliance or where peak concentration adverse effects are suspected. As the TPM-ER costs decrease or cost sensitivity decreases, usage of TPM-ER over IR becomes an easier choice. With the current magnitude in cost difference between the generic TPM and TPM-ER formulations, a short trial (eg, 1-6 months) switching from the generic TPM to TPM-ER with a priori determined goals is reasonable. We suggest these goals be significant improvements in compliance, efficacy, or adverse effects such that the patient’s quality of life substantially improves with the formulation switch.
Footnotes
Acknowledgements
We acknowledge Dr. Sunay Shah, PharmD, for his comments and drug cost information.
Peer Review:
Five peer reviewers contributed to the peer review report. Reviewers’ reports totaled 744 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
YW, KS, RM, KRK, and MW wrote the first draft of the manuscript. YW, KS, RM, KRK, MW, and KM contributed to the writing of the manuscript. YW, KS, RM, KRK, MW, and KM agree with manuscript results and conclusions YW, KS, RM, KRK, and MW jointly developed the structure and arguments for the article. YW, KS, RM, KRK, MW, and KM made critical revisions and approved final version. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
As a requirement of publication, author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality, and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.