Abstract
Genotype 3 (GT3) hepatitis C virus (HCV), the second most common HCV genotype worldwide, has emerged as the most difficult-to-cure genotype. Sustained virological response (SVR) rates with new direct-acting antiviral regimens for GT3 patients who are treatment-naïve and noncirrhotic are now similar to those seen in patients with non-GT3 HCV infection. However, GT3 HCV patients who are treatment-experienced or who have cirrhosis, particularly those with decompensated disease, continue to be a more challenging group to treat due to lower SVR rates. Here, we review the current evidence for the treatment of patients with GT3 HCV including current data for patients with GT3 HCV who are HIV coinfected. Future studies in GT3 HCV treatment will need to focus on direct-acting antiviral combinations that improve cure rates and potentially eliminate the need for ribavirin in GT3 patients who have advanced liver disease. The significance of HCV resistance-associated variants in GT3 HCV patients who have failed prior treatment also needs further assessment to help guide re-treatment strategies.
Introduction
A significant genetic diversity exists among the various hepatitis C virus (HCV) strains, resulting in the classification of seven different HCV genotypes. 1 Genotypic differences are now clearly understood to account for differences in treatment outcomes, with genotype 3 (GT3) HCV being identified as the most difficult-to-cure genotype. This is problematic as GT3 HCV is the second most common genotype globally, accounting for 30.1% of all HCV cases or 54.3 million infected people. 2
Significant geographic diversity exists with respect to the frequency of GT3 HCV infection. Three quarters of all GT3 HCV infections occur in South Asia where GT3 HCV is the common genotype. However, GT3 is not uncommon in other regions; a quarter of all HCV infections are due to GT3 in Western Europe and GT3 accounts for 10%–20% of HCV infections in North America.2,3 Further, there is no significant difference in the frequency of GT3 HCV infections when comparing countries of low, middle, or high income. 2
It is further problematic that GT3 HCV is associated with a more accelerated fibrosis progression and a higher risk of hepatocellular carcinoma development than the other HCV genotypes.4,5 These outcomes may be the result of the cytopathic effect of GT3 HCV-induced steatosis.6–8 Steatosis development itself may also explain the lower response rates in patients with GT3 HCV infection. 9
In previous studies, patients with GT3 HCV were grouped together with genotype 2 patients. This made it appear that patients with GT3 were relatively easy to treat when compared to patients with genotype 1 (GT1) HCV infection. However, rapid improvements in our ability to cure patients with GT1 HCV with new direct-acting antiviral (DAA) regimens have served to highlight GT3 as a difficult-to-cure genotype. Further, patients with classic predictors of poor response, including failure of previous treatment and presence of cirrhosis, continue to prove challenging in curing GT3 HCV.
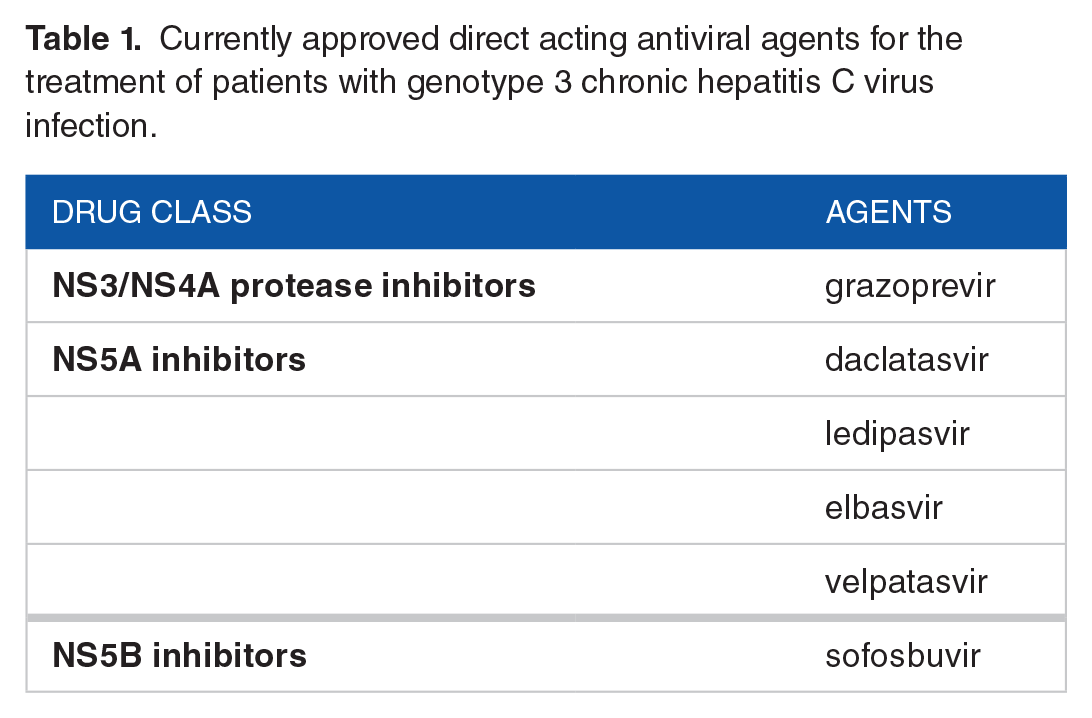
We conducted a search of the available peer-reviewed literature on treatment of GT3 HCV in addition to a search of recent presentations from international congresses published in abstract form (see Table 1 for available DAAs for genotype 3 HCV). Current literature regarding treatment of patients with or without compensated cirrhosis will be reviewed first followed by a discussion of patients with decompensated liver disease and liver transplant recipients. Treatment options in patients coinfected with HIV or who have renal impairment will also be discussed.
Currently approved direct acting antiviral agents for the treatment of patients with genotype 3 chronic hepatitis C virus infection.
DAA therapy in treatment-naïve GT3 HCV patients
The goal in HCV therapy is to achieve a sustained virological response (SVR, SVR24), defined as an HCV RNA level below the threshold of quantification, 24 weeks after the end of treatment. SVR is highly predictive of persistent HCV clearance and is used to define HCV cure. 10 SVR12, an HCV RNA level below the threshold of quantification 12 weeks after the end of treatment, is also highly predictive of HCV cure (SVR12 positive predictive value [PPV] 99.7%). 11 Further, the concordance between SVR12 and SVR24 across genotypes 1–3 is strong, which has resulted in the use of SVR12 as the primary outcome of treatment in recent HCV studies. 12
Pegylated-interferon (PEG) in combination with ribavirin (RBV) and sofosbuvir, an NS5B inhibitor, remains one of the most efficacious regimens for the treatment of GT3 HCV (see Table 2).
Efficacy of direct acting antiviral agents for the treatment of previously untreated patients with HCV genotype 3.
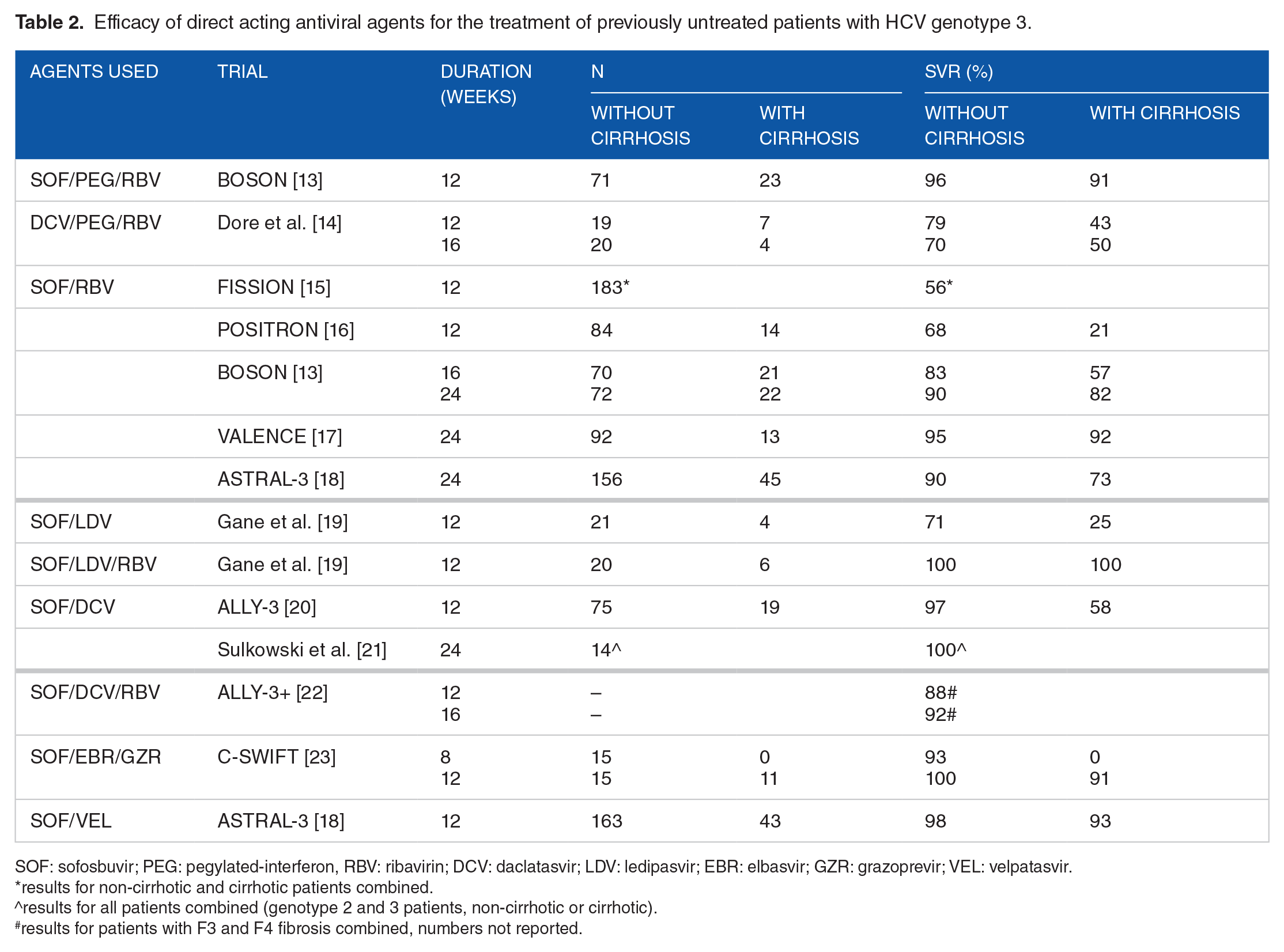
SOF: sofosbuvir; PEG: pegylated-interferon, RBV: ribavirin; DCV: daclatasvir; LDV: ledipasvir; EBR: elbasvir; GZR: grazoprevir; VEL: velpatasvir.
results for non-cirrhotic and cirrhotic patients combined.
results for all patients combined (genotype 2 and 3 patients, non-cirrhotic or cirrhotic).
results for patients with F3 and F4 fibrosis combined, numbers not reported.
This is particularly true for treatment-naïve patients who do not have cirrhosis. In the phase 3 BOSON study, SVR was achieved in 96% (68/71) of treatment-naïve GT3 patients without cirrhosis and 91% (21/23) of patients with cirrhosis after 12 weeks of sofosbuvir, PEG, and RBV. 13 Dore et al 14 assessed the efficacy of 12 and 16 weeks of daclatasvir, an NS5A inhibitor, PEG, and RBV for noncirrhotic treatment-naïve patients with GT3 HCV and reported SVR rates of 79% (15/19) and 70% (14/20), respectively. However, SVR was achieved in only 43% (3/7) and 50% (2/4) of cirrhotic patients who received daclatasvir, PEG, and RBV for 12 and 16 weeks, respectively. 14
Sofosbuvir and RBV without PEG are effective in treatment-naïve patients with GT3 HCV; however, treatment duration and presence of cirrhosis have proven to be critical predictors of cure in treatment-naïve patients. In the FISSION trial, treatment-naïve (both cirrhotic and noncirrhotic) GT3 HCV patients received sofosbuvir and RBV for 12 weeks, with only 55.7% (102/183) of patients achieving SVR. 15 Although an exact breakdown of treatment responses in cirrhotic versus noncirrhotic patients was not provided, noncirrhotic patients were almost three times more likely to achieve SVR than noncirrhotic patients in FISSION. 15 These results were confirmed in the POSITRON trial where SVR was achieved in 68% (57/84) of patients without cirrhosis compared to 21% (3/14) of patients with cirrhosis after 12 weeks of treatment with sofosbuvir and RBV. 16 Sofosbuvir and RBV treatment for 16 weeks in the BOSON trial resulted in SVR of 83% (58/70) and 57% (12/21) in noncirrhotic and cirrhotic patients, respectively. 13
Twenty-four weeks of sofosbuvir and RBV therapy was studied in the BOSON, VALENCE, and ASTRAL-3 trials where SVR rates of 90% (65/72), 95% (87/92), and 90% (141/156), respectively, were obtained in noncirrhotic patients.13,17,18 Corresponding values in each of these trials for patients with cirrhosis were 82% (18/22), 92% (12/13), and 73% (33/45), respectively.13,17,18 Therefore, it is clear that, although highly effective in treatment-naïve patients without cirrhosis, the combination of sofosbuvir and RBV results in a suboptimal treatment response in GT3 HCV patients with cirrhosis. Further, longer treatment duration with sofosbuvir and RBV is required to achieve these results, which increases treatment cost and may carry a higher treatment risk profile.
Sofosbuvir has been further studied in combination with various HCV NS5A inhibitors for the treatment of GT3 HCV. In a phase 2 study by Gane et al, 19 treatment-naïve patients with GT3 HCV received sofosbuvir and ledipasvir with or without RBV for 12 weeks. For patients without cirrhosis, SVR was achieved in 100% (20/20) and 71% (15/21) in the RBV-containing and RBV-free groups, respectively. 19 All six cirrhotic patients who received sofosbuvir/ledipasvir with RBV achieved SVR, whereas only one of four cirrhotic patients in RBV-free group achieved SVR. 19 Sofosbuvir and ledipasvir may, therefore, reduce treatment duration in treatment-naïve GT3 patients but RBV is still required.
Sofosbuvir with daclatasvir appears to be more effective than sofosbuvir with ledipasvir in GT3 HCV patients with cirrhosis. In the ALLY-3 study, treatment-naïve patients received 12 weeks of sofosbuvir and daclatasvir, resulting in an SVR in 97% (73/75) of noncirrhotic patients compared to 58% (11/19) of patients with cirrhosis. 20 Increasing treatment duration of sofosbuvir and daclatasvir to 24 weeks may improve treatment efficacy in GT3 patients; all 14 patients (genotype 2 and 3; with or without cirrhosis) who were treated in one study with sofosbuvir and daclatasvir achieved SVR. 21
The addition of RBV to sofosbuvir and daclatasvir, however, may allow for a shorter treatment duration in patients with advanced fibrosis (Metavir F3) or cirrhosis (Metavir F4). In the ALLY-3+ study, there was no significant difference in SVR between patients with advanced fibrosis or cirrhosis who were treated with 12 (88% SVR) or 16 (92% SVR) weeks of sofosbuvir, daclatasvir, and RBV. 22 SVR was achieved in 92% of all treatment-naïve patients (advanced fibrosis or cirrhosis), whereas 88% of treatment-naïve cirrhotic patients achieved SVR. 22
The combination of grazoprevir, an NS3/4A protease inhibitor, elbasvir, an NS5A inhibitor, and sofosbuvir in treatment-naïve GT3 patients was studied in the C-SWIFT study. A total of 93% (14/15) and 100% (14/14) of treatment-naïve patients without cirrhosis who received grazoprevir, elbasvir, and sofosbuvir for 8 or 12 weeks, respectively, achieved SVR. 23 Cirrhotic treatment-naïve patients were treated for 12 weeks, which resulted in a 91% (10/11) SVR.
The combination of sofosbuvir and velpatasvir, an NS5A inhibitor, was recently shown to be highly efficacious in the treatment of GT3 HCV. In the ASTRAL-3 study, 98% (160/163) of treatment-naïve patients without cirrhosis and 93% (40/43) with compensated cirrhosis achieved SVR after 12 weeks of sofosbuvir and velpatasvir. 18 This RBV- and PEG-free regimen will offer a simple well-tolerated all-oral option for the majority of treatment-naive GT3 patients. The sofosbuvir–velpatasvir combination was further studied in patients with Child-Pugh B cirrhosis in the ASTRAL-4 study as outlined below.
The addition of GS-9857, an NS3/4A protease inhibitor, to sofosbuvir and velpatasvir may allow an even shorter treatment duration in patients with GT3 HCV. In a phase 2 study, 6 weeks of sofosbuvir, velpatasvir, and GS-9857 in 21 treatment-naïve patients with cirrhosis resulted in an SVR rate of 100%. 24 In cirrhotic patients treated for eight weeks with this regimen, the SVR was 94% (17/18). 24
Sofosbuvir in addition to ombitasvir, an NS5A inhibitor, and ritonavir-boosted paritaprevir, an NS3/4A protease inhibitor, may be yet another option for the treatment of GT3 patients. All six noncirrhotic treatment-naïve patients who were treated with this combination for 12 weeks as part of the QUARTZ-II study achieved SVR. 25
The C-CREST study assessed the efficacy of an eight-week combination of grazoprevir, with MK-3682, an NS5B inhibitor, and one of two NS5A inhibitors, either elbasvir or MK-8408, in treatment-naïve noncirrhotic patients with GT3 HCV. The SVR rates in the four treatment arms were: 90% (19/21) with grazoprevir, elbasvir, and MK-3682 (300 mg); 86% (19/22) with grazoprevir, elbasvir, and MK-3682 (450 mg); 95% (20/21) with grazoprevir, MK-8408, and MK-3682 (300 mg); and 91% (20/22) with grazoprevir, MK-8408, and MK-3682 (450 mg). 26 This study shows that a short course of an interferon and RBV-free DAA regimen is possible in noncirrhotic treatment-naïve patients with GT3. The efficacy of this regimen in treatment-naïve patients with cirrhosis needs to be clarified.
SURVEYOR-II, a phase 2 trial, has shown that ABT-493, an NS3/4A protease inhibitor, and ABT-530, an NS5A inhibitor, are also efficacious in the treatment of patients with GT3 HCV. Thirty (27 treatment-naïve) noncirrhotic patients were treated with ABT-493 (300 mg) and ABT-530 (120 mg) for 12 weeks in the part one of SURVEYOR-II. 27 SVR was achieved in 93% of all patients. The treatment duration of ABT-493 and ABT-530 in treatment-naïve noncirrhotic genotype 3 patients was shortened to eight weeks in part two of the SUREYOR-II study. 28 A single patient withdrew consent from the study (overall SVR12 28/29); however, all 28 patients who were included in the modified intention-to-treat analysis achieved SVR with the shortened regimen. 28 All treatment-naïve cirrhotic patients treated for 12 weeks with ABT-493 and ABT-530 with (n = 24) or without (n = 24) RBV achieved an SVR. 29
DAA therapy in treatment-experienced GT3 HCV patients
GT3 patients with prior treatment exposure continue to be a challenging group (see Table 3). In LONESTAR-2, 12 weeks of sofosbuvir, PEG, and RBV resulted in SVR rates of 83% (12/13) for both cirrhotic and noncirrhotic patients. 30 In BOSON, however, the same combination resulted in an SVR of 94% (49/52) in treatment-experienced patients without cirrhosis and 86% (30/35) with cirrhosis. 13 Further, Esteban et al 31 assessed the efficacy of retreating patients who had previously failed sofosbuvir and RBV with 12 weeks of sofosbuvir, PEG, and RBV and showed that SVR could be achieved in 93% (13/14) of noncirrhotic and 88% (7/8) of cirrhotic patients. Interferon-containing regimens, therefore, prove to be highly efficacious even in treatment-experienced GT3 patients who have cirrhosis.
Efficacy of direct acting antiviral agents for the treatment of previously treated patients with HCV genotype 3.
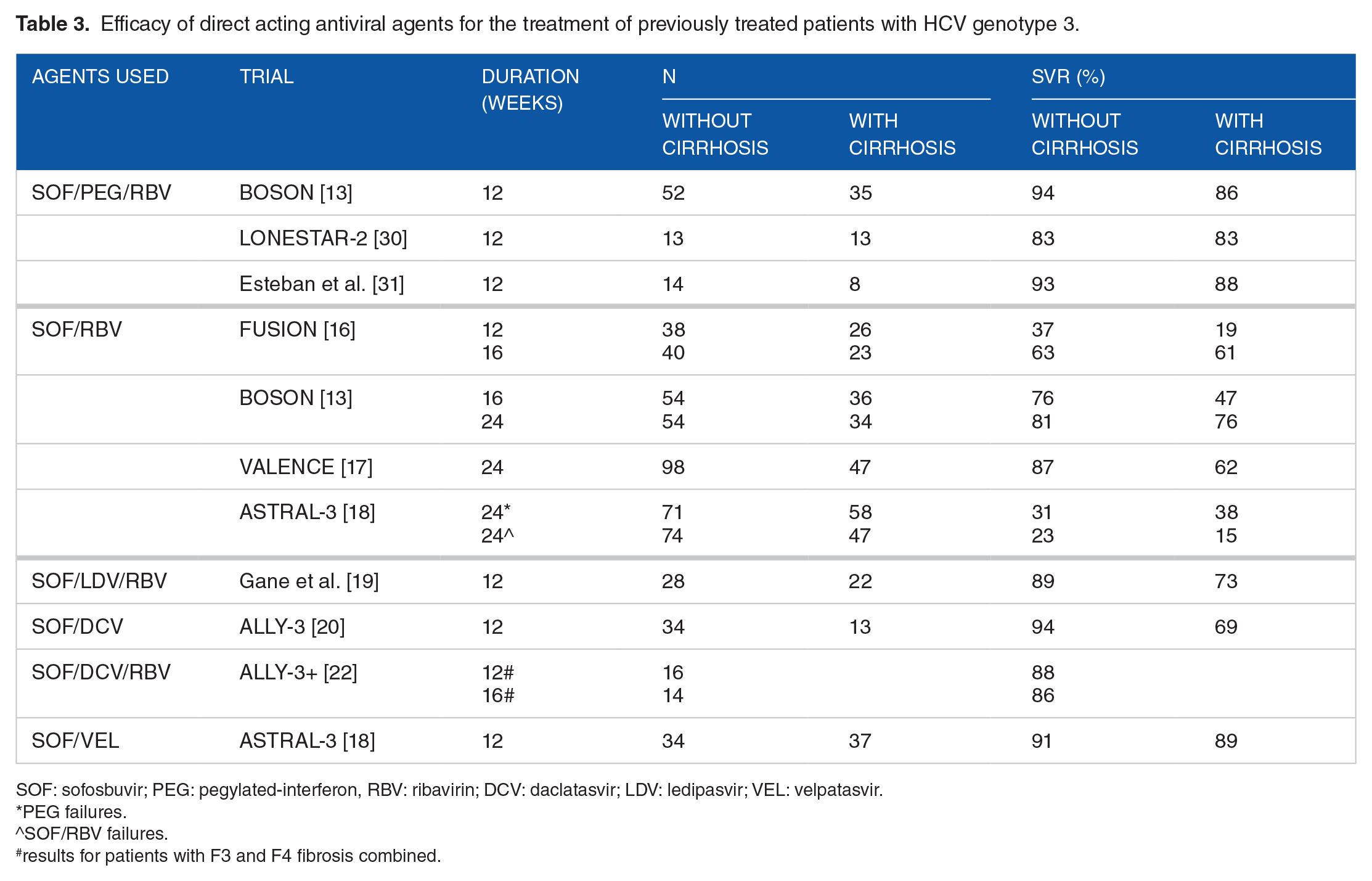
SOF: sofosbuvir; PEG: pegylated-interferon, RBV: ribavirin; DCV: daclatasvir; LDV: ledipasvir; VEL: velpatasvir.
PEG failures.
SOF/RBV failures.
results for patients with F3 and F4 fibrosis combined.
Re-treatment of GT3 HCV patients with sofosbuvir and RBV has been assessed in a number of studies. In FUSION, 12 and 16 weeks of sofosbuvir and RBV resulted in SVR rates of only 36.8% (14/38) and 62.5% (25/40) in noncirrhotic patients and 19.2% (5/26) and 60.9% (14/23) in compensated cirrhotic patients who had previously failed treatment with PEG-based therapy. 16 A longer treatment duration of 24 weeks of sofosbuvir and RBV improved SVR rates in treatment-experienced patients without (86.7%; 85/98) and with compensated cirrhosis (61.7%; 29/47) in the VALENCE study; however, efficacy rates, particularly in patients with cirrhosis, remained poor. 17 The BOSON study showed results similar to FUSION and VALENCE; SVR was achieved in 76% (41/54) and 81% (44/54) of treatment-experienced patients without cirrhosis and 47% (17/36) and 76% (26/34) of treatment-experienced patients with cirrhosis after 16 and 24 weeks of sofosbuvir and RBV. 13
ASTRAL-3 yet again confirmed the suboptimal efficacy of sofosbuvir and RBV in treatment-experienced patients with compensated cirrhosis. In that study, SVR was achieved in 71% (22/31) and 58% (22/38) of GT3 patients without or with cirrhosis who had previously failed an interferon-containing regimen. 18 Re-treatment of patients from the FISSION, POSITRON, and FUSION studies who had previously failed 12 or 16 weeks of sofosbuvir and RBV for a longer duration (24 weeks) resulted in similar SVR rates of 74% (17/23) for those without cirrhosis and 47% (7/15) for those with cirrhosis. 31 Together, these studies support the continued benefit of PEG when added to sofosbuvir and RBV in treatment-experienced cirrhotic patients.
The addition of an NS5A inhibitor to sofosbuvir improves cure rates in treatment-experienced patients with GT3 HCV. Sofosbuvir, ledipasvir, and RBV for 12 weeks resulted in SVR rates of 89% (25/28) in treatment-experienced patients without cirrhosis and 73% (16/22) in those with compensated cirrhosis. 19 In ALLY-3, sofosbuvir with daclatasvir for 12 weeks resulted in SVR rates of 94% (32/34) in treatment-experienced patients without cirrhosis but only 69% (9/13) with cirrhosis. 20 The addition of RBV to sofosbuvir and daclatasvir may help increase response rates in patients with cirrhosis. In ALLY-3+, 88% (14/16) and 86% (12/14) of cirrhotic patients who had previous treatment-experience achieved SVR after 12 and 16 weeks of sofosbuvir, daclatasvir, and RBV, respectively. 22
In ASTRAL-3, sofosbuvir with velpatasvir given for 12 weeks was very effective in treatment-experienced patients resulting in an SVR in 91% (31/34) and 89% (33/37) of patients without and with cirrhosis, respectively. 18 The sofosbuvir and velpatasvir combination will, therefore, offer a 12-week interferon and RBV-free treatment option for treatment-experienced cirrhotic GT3 patients, which has similar efficacy to sofosbuvir, PEG, and RBV.
Patients who did not achieve SVR with 8 or 12 weeks of sofosbuvir/velpatasvir in phase 2 studies were re-treated with 24 weeks of sofosbuvir/velpatasvir plus RBV in the GS-US-342-1553 study. 32 This study included a mix of patients without cirrhosis and those with compensated cirrhosis. Thirteen of 17 (76%) GT3 HCV patients achieved SVR with one treatment failure being due to withdrawal of consent to participate in the study. SVR occurred in 77% of the 13 patients who had a baseline resistance-associated variant (RAV), while all 3 patients without baseline RAVs achieved SVR. 32 These data suggest that a longer treatment duration of sofosbuvir/velpatasvir plus RBV may be an effective treatment strategy for patients who have previously been exposed to the sofosbuvir/velpatasvir combination even in the presence of baseline RAVs.
Even higher cure rates with shorter treatment duration may certainly be possible in this difficult-to-cure population with the addition of the NS3/4A protease inhibitor GS-9857 to sofosbuvir and velpatasvir; all 19 treatment-experienced patients with cirrhosis achieved SVR after being treated with sofosbuvir, velpatasvir, and GS-9857 for only eight weeks. 33 The use of sofosbuvir and velpatasvir with GS-9857 for 12 weeks in 35 treatment-experienced GT3 patients, 48% of whom were cirrhotic, resulted in an SVR of 97%. 34 Seventeen GT3 patients in this study had a previous exposure to at least one DAA class. Baseline RAVs were identified in 60% of the entire cohort (all genotypes); however, the only relapse occurred in a GT3 patient with cirrhosis who had a baseline Y93H RAV. 34
Special populations
Decompensated cirrhosis and post liver transplantation
Sofosbuvir and daclatasvir were studied in a small number GT3 HCV patients with decompensated cirrhosis as part of the ALLY-1 study. Five of six (83%) patients with both compensated and decompensated cirrhosis (Child-Pugh A–C) who were treatment-naïve or treatment-experienced had an SVR after 12 weeks of sofosbuvir and daclatasvir. 35 For patients (treatment-naïve or treatment-experienced) who had undergone liver transplantation, the SVR rate with the same regimen was 91% (10/11). 35
ASTRAL-4 specifically focused on the use of sofosbuvir and velpatasvir in patients with decompensated cirrhosis. For GT3 HCV patients, 12 weeks of sofosbuvir and velpatasvir without or with RBV resulted in SVR rates of 50% (7/14) and 85% (11/13), respectively. 36 A longer treatment duration of 24 weeks of sofosbuvir and velpatasvir without RBV failed to improve SVR (50%; 6/12) indicating the continued need for RBV in GT3 HCV with decompensated cirrhosis patients who are treated with this regimen. 36
GT3 HCV patients with decompensated cirrhosis remain a challenge with suboptimal cure rates when compared to other genotypes. Increasingly, data from heterogeneous real-world cohorts are showing similar efficacy rates in decompensated patients with GT3 HCV as have been described in the setting of clinical trials. There is an ongoing need for larger studies in this difficult-to-cure population to help clarify the best treatment regimen.
HIV coinfected patients
Treatment outcomes for GT3 HCV/HIV coinfected patients are similar to GT3 HCV mono-infected patients. In PHOTON-1, two-thirds of treatment-naïve patients with (4/6) or without (24/36) cirrhosis achieved SVR after 12 weeks of sofosbuvir and RBV. 37 Treatment-experienced patients were treated with sofosbuvir and RBV for 24 weeks, which resulted in SVR rates of 100% (11/11) and 83% (5/6) in noncirrhotic and cirrhotic patients, respectively. 37 In PHOTON-2, treatment-naïve patients were treated for 24 weeks with sofosbuvir and RBV; 49 of 54 (91%) patients without cirrhosis and all 3 treatment-naïve patients with cirrhosis in PHOTON-2 achieved SVR. 38 SVR rates of 92% (24/26) and 78% (18/23) were reported for treatment-experienced coinfected patients without and with cirrhosis, respectively. 38
The use of sofosbuvir and daclatasvir was assessed in a small number of GT3 HCV/HIV coinfected patients in the ALLY-2 study. In noncirrhotic treatment-naïve patients, 8 and 12 weeks of sofosbuvir and daclatasvir resulted in an SVR in 1 of 2 patients and all 6 patients, respectively. 39 All three noncirrhotic treatment-experienced patients achieved SVR after 12 weeks of sofosbuvir and daclatasvir. 39 SVR was achieved in the one treatment-naïve patient and one treatment-experienced patient with compensated cirrhosis who received sofosbuvir and daclatasvir for 8 and 12 weeks, respectively. 39 As seen in other HCV genotypes, these results highlight that GT3 HCV patients coinfected with HIV should now no longer be viewed as a more difficult-to-cure group when compared to mono-infected GT3 HCV patients.
Recent results from ASTRAL-5 study support the use of a 12-week course of sofosbuvir and velpatasvir for the treatment of HCV GT3 patients who are coinfected with HIV. 40 A mix of 12 cirrhotic or noncirrhotic GT3 patients who were treatment-naïve or treatment-experienced were included in ASTRAL-5. SVR was reported as 92% (11/12); the only treatment failure occurred in a patient who withdrew consent from the study. 40
Patients with severe renal impairment
The use of new DAA agents in patients with GT3 HCV and severe renal impairment has not been assessed. Studies to assess the safety and efficacy in this population are greatly needed.
Discussion
The creation of new pan-genotypic DAA therapies has dramatically improved treatment outcomes across all genotypes. However, we are still seeing lower response rates in patients with GT3 HCV, particularly for those who are treatment-experienced or who have cirrhosis. Further, HCV eradication in GT3 patients with decompensated liver disease remains an ongoing challenge with suboptimal rates of cure. Interferon and even RBV-free DAA regimens are proving to be highly efficacious in non-GT3 HCV. However, in GT3 HCV, there is a continued need for RBV in many patient groups, which may prove problematic in patients with advanced liver disease. For example, in the ASTRAL-4 study, the use of RBV clearly increased SVR rates but came with the added cost of higher adverse event rates with 5% of patients receiving 12 weeks of sofosbuvir, velpatasvir, and RBV having to stop treatment compared to only 1% of those receiving 12 weeks of sofosbuvir and velpatasvir. 36
Further, it is important to recognize that sofosbuvir, PEG, and RBV remain the most highly efficacious regimens in treating GT3 HCV, which remains true even in treatment-experienced cirrhotic patients where SVR rates are between 85% and 90%. Thus, we may have identified newer regimens that are highly efficacious and more tolerable in treatment-naïve GT3 patients without cirrhosis but we have not yet completely freed many of our GT3 patients from the use of RBV or even PEG-based therapies.
Our understanding of HCV RAVs and their significance on treatment outcomes continues to grow. Work in GT1 HCV has shown that NS5A RAVs, particularly those at the 93 locus, lead to significantly lower SVR rates. 41 The assessment of RAVs in ASTRAL-3 support lower response rates in GT3 patients who have NS5A RAVs. The presence of any NS5A RAV at baseline in ASTRAL-3 was associated with an SVR rate of 88% compared to 97% in patients who did not have a baseline NS5A RAV. 18 Further, the SVR rate was only 84% in the 25 patients who had the Y93H NS5A RAV at baseline. 18 A recent analysis, however, found that baseline RAVs did not affect the efficacy of a short course of sofosbuvir, velpatasvir, and GS-9857 in a small number of GT3 patients with cirrhosis. 42
It is also important to recognize that NS5A RAVs appear to be more durable than RAVs at other DAA target sites; recent data in GT1 HCV have shown that NS5A RAVs can persist for more than 96 weeks. 43 This will have clinical importance when attempting to re-treat patients with GT3 HCV.
Conclusion
With the introduction of new DAAs, the goals of HCV treatment were to initially increase cure rates. When this proved possible, the next goal was to eliminate interferon-based and even RBV-based therapies altogether. This has become possible for many with GT1 HCV infection, where the next challenge is to reduce treatment duration as low as possible. However, for GT3 HCV, the best regimens continue to involve the use of RBV and, for some patients, even interferon.
Cure rates of 90% in HCV are no longer acceptable; the new goal in HCV therapy is now to cure as many patients as possible with a target no lower than 95%. Unfortunately, this target is still not being met for many patients with GT3 HCV. Lower cure rates in HCV GT3, particularly in patients with cirrhosis or who have previously failed treatment, indicate that our focus on new treatment strategies for HCV is still required. It is hoped that the development of more potent DAAs or the combination of new DAAs with existing DAAs may improve our ability to cure patients with GT3 HCV.
Footnotes
Peer Review:
Three peer reviewers contributed to the peer review report. Reviewers’ reports totaled 287 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Matthew Sadler was funded through Vertex / Canadian Association for the Study of the Liver Clinical Hepatology Fellowship Grant.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kosh Agarwal discloses- Advirosry Committees or Review Panels: Gilead, BMS, Novartis, Janssen, AbbVie; Consulting: MSD, Janssen, Achillion, Intercept; Grant/Research Support: Roche, Gilead, BMS, Arbutus; Speaking and Teaching: Astellas, Gilead, BMS, GSK.
This paper was subject to independent, expert peer review by a minimum of two blind peer reviewers. All editorial decisions were made by the independent academic editor. All authors have provided signed confirmation of their compliance with ethical and legal obligations including (but not limited to) use of any copyrighted material, compliance with ICMJE authorship and competing interests disclosure guidelines and, where applicable, compliance with legal and ethical guidelines on human and animal research participants.
Author Contributions
Conceived and designed the experiments: MS, KA. Analyzed the data: MS, KA. Wrote the first draft of the manuscript: MS. Contributed to the writing of the manuscript: MS, KA. Agree with manuscript results and conclusions: MS, KA. Jointly developed the structure and arguments for the paper: MS, KA. Made critical revisions and approved final version: MS, KA. All authors reviewed and approved of the final manuscript.
Provenance
The authors were invited to submit this paper.
