Abstract
Background:
Benzodiazepine (Bz) exposure has been identified as a risk factor of community-acquired pneumonia (CAP) in some observational studies, but this remains controversial. This study was designed to quantify the risk of CAP associated with treatment with Bz.
Methods:
All individuals ⩾14 years of age registered in any of 3 primary health care providers in our area between January 2011 and May 2013 were included in the study. This resulted in a population of 51 912 individuals who contributed to a total of 1 496 680 person-months of observation. Previously anonymized data for each participant were obtained from their personal health records and the official prescription database. The primary outcome measures were the incidence of CAP during the study period and the relative risk (RR) that could be attributed to Bz exposure.
Results:
A total of 696 CAP cases were diagnosed. Incidence density was 12.4 cases per 1000 person-years in individuals exposed to Bz and 4.51 cases per 1000 person-years in those who were not. Benzodiazepine exposure increased the risk of CAP in the whole population (RR: 2.76, 95% confidence interval: 2.35-3.25) and in all the evaluated subgroups. Stratified analysis showed an interaction only with age (RR: 2.99 in patients under 65 years and 1.78 in those aged 65 or older). Benzodiazepine exposure was associated with an excess 0.79 cases of CAP per 100 person-years.
Conclusions:
Benzodiazepine exposure increases the risk of CAP. Given the clinical relevance of CAP, prescribers should be aware of this potentially preventable risk and consider it while newly prescribing Bz.
Community-acquired pneumonia (CAP) is a leading cause of morbi-mortality and health care–related expense worldwide. 1 Among multiple risk factors for CAP identified in epidemiologic studies2,3 stand certain drug exposures. The magnitude of the risk of individual agents, however, remains controversial. A recently published review, for example, concluded that an association between benzodiazepine (Bz) exposure and adverse respiratory outcomes may be reasonably supported, 4 but the results from individual studies are conflicting. Benzodiazepine exposure has been found to be protective from,5,6unrelated to,7-11 or even a risk factor of12-14 CAP. The main features and results of these studies are summarized in Table 1.
Description of the main features of studies evaluating the association between Bz exposure and pneumonia.
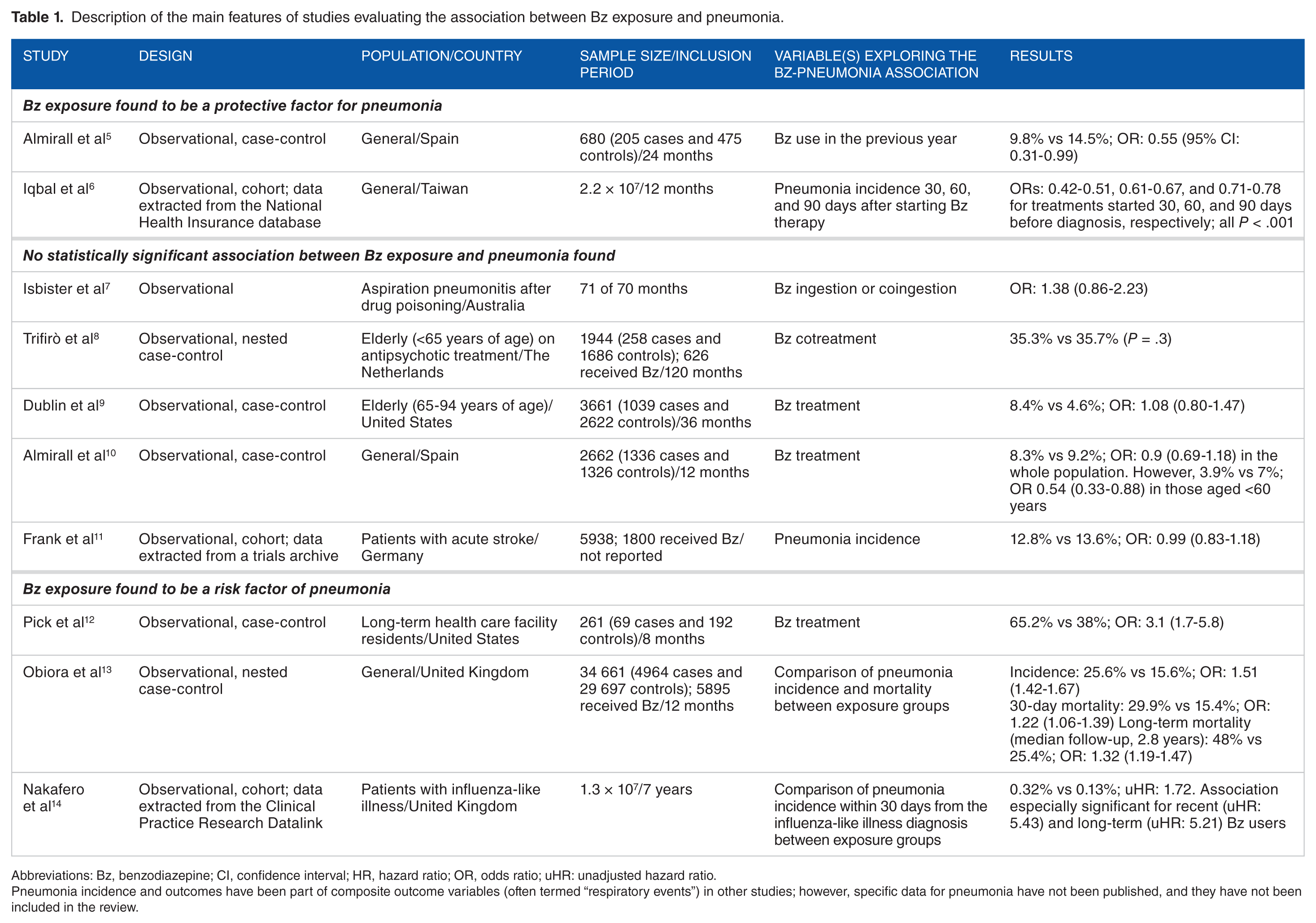
Abbreviations: Bz, benzodiazepine; CI, confidence interval; HR, hazard ratio; OR, odds ratio; uHR: unadjusted hazard ratio.
Pneumonia incidence and outcomes have been part of composite outcome variables (often termed “respiratory events”) in other studies; however, specific data for pneumonia have not been published, and they have not been included in the review.
Benzodiazepines are widely prescribed 15 modulators of the GABAA receptor that have anxiolytic, hypnotic, muscle relaxant, and antiepileptic properties. Benzodiazepine may induce muscle relaxation in the upper respiratory tract and depression of the swallowing reflex, which lead to pharyngeal dysfunction and increase the risk of aspiration into the airway. 16 Moreover, the presence of GABAA receptors on immune cells has immunomodulatory effects (most notably the inhibition of production of inflammatory cytokines) that have been well documented in preclinical models, but not in subsequent clinical studies.17-19
Because exposure to Bz is increasing, its potential association with a serious condition such as CAP becomes a matter of significant public health concern. We designed an observational study in which, in accordance with the results of previous studies by our group, we hypothesized that Bz exposure would be protective from CAP. This study was designed to test this hypothesis in a large nonselected population under usual clinical care.
Methods
A protocol for a prospective population-based cohort study with retrospective data collection from electronic databases was designed and approved by the Clinical Research Ethics Committee of the Hospital de Mataró.
The study population was composed of 51 912 inhabitants from the catchment area aged 14 years or over who were registered at any of the primary care facilities of the Consorci Sanitari del Maresme (CSdM) between January 1, 2011 and May 31, 2013. The CSdM is the public health care provider in the central area of the Maresme County (Barcelona, Spain). About 99% of the area’s population is registered at their corresponding primary health care facility.
Besides Bz exposure and CAP, study variables included gender, age, previous chronic obstructive pulmonary disease (COPD) or asthma, and antipneumococcal vaccination. These variables were chosen among the risk factors of CAP reported in the medical literature20-22 because we considered they were the ones most likely to be available for most participants in the electronic databases from which the information was to be retrieved.
Demographic and clinical data were collected from public electronic health records. Information on Bz prescription (including the date it had occurred) was collected from the pharmacy administrative database. Both sources were cross-linked using a “personal identification code” from the Catalan Health Service.
Interest drugs included those with ATC codes N05BAxx (diazepam [xx = 01], potassium clorazepate [05], lorazepam [06], bromazepam [08], clobazam [09], ketazolam [10], alprazolam [12], and pinazepam [14]), N05CDxx (flurazepam [01], triazolam [05], lormetazepam [06], midazolam [08], brotizolam [09], quazepam [10], and lorazepam [11]), N05CF01 (zopiclone), N05CF02 (zolpidem), and N05CM02 (clomethiazole).
A participant was considered to be “exposed” if he or she had a prescription for any of the interest drugs issued before the date of diagnosis of a CAP, and no mention that the drug had been discontinued before that same date was available in the pharmacy database. Conversely, participants were considered to be “nonexposed” either if no Bz had been prescribed within the study period or (if such a prescription existed) it had been issued after the diagnosis of CAP or the drug had been discontinued before the diagnosis of CAP.
A CAP was considered to have occurred if a general practitioner (GP) had entered this diagnosis in the electronic health record of any participant. The way in which the diagnoses had been made (eg, based on clinical suspicion only or with ancillary testing, such as radiographic/microbiological confirmation) and their accuracy were not assessed. Similarly, the previous diagnosis of COPD or asthma and the vaccination status were obtained from the GPs’ notes in the patients’ electronic records, and no specific validation of the retrieved information was performed.
Participants were followed up to the date in which the first episode of CAP was diagnosed, to the date they moved out of the catchment area or died, or to the last day of the study period (May 31, 2013).
The risk of developing a CAP was measured through its incidence density (ID; count of new cases per 100 person-years). The association between Bz exposure and CAP was explored through the relative risk (RR; risk ratio between the exposed and the nonexposed cohort). The impact of Bz exposure on CAP burden was estimated through the attributable risk (AR; difference in ID between the exposed and the nonexposed cohort) and the etiologic fraction (EF; AR/ID in the exposed group). An RR was considered statistically significant if its 95% confidence interval (CI) excluded unity. To assess confounding and interaction, all these analyses were performed not only for the overall population, but they are also stratified by age, gender, COPD/asthma diagnosis, and pneumococcal vaccination status. When comparing RR between groups, a difference was considered statistically significant if their 95% CI did not overlap. A statistically significant difference between the RR of the strata of a specific study group was interpreted as evidence for an interaction between this specific feature and the risk of CAP conferred by Bz exposure.
Data handling and analysis were performed using the PASW Statistics for Windows, version 18.0 (SPSS Inc., Chicago, IL, USA) software package.
Given the observational nature of the study, its retrospective design, and the exclusive use of anonymized data from electronic databases, the Clinical Research Ethics Committee considered that specific informed consent was not necessary.
Results
Data from 25 204 men and 26 708 women (who contributed to a total of 727 122 and 769 558 person-months of observation, respectively) were obtained. Their median age was 44 years (minimum 14, maximum 107).
During the study period, 696 cases of CAP were diagnosed; 210 of them (30.2%) had been exposed to Bz. The ID in the nonexposed cohort was 0.45 cases (95% CI: 0.41-0.49) per 100 person-years, whereas that in the exposed cohort was 1.24 cases (1.08-1.41) per 100 person-years. Thus, exposure to Bz resulted in an excess of 0.79 cases of CAP per 100 person-years. Based on these data, the RR was 2.76 (2.35-3.25) and the EF was 63.8%. The effect of Bz exposure on CAP incidence for the overall study population and stratified by age, gender, previous COPD/asthma, and pneumococcal vaccination status is presented in Table 2. This effect was significant in all evaluated strata. There was no interaction between Bz exposure and either gender or a previous diagnosis of COPD or asthma.
Incidence densities for community-acquired pneumonia (according to benzodiazepine exposure status) and risk assessment of the exposure in the whole study population and in epidemiologically relevant strata.
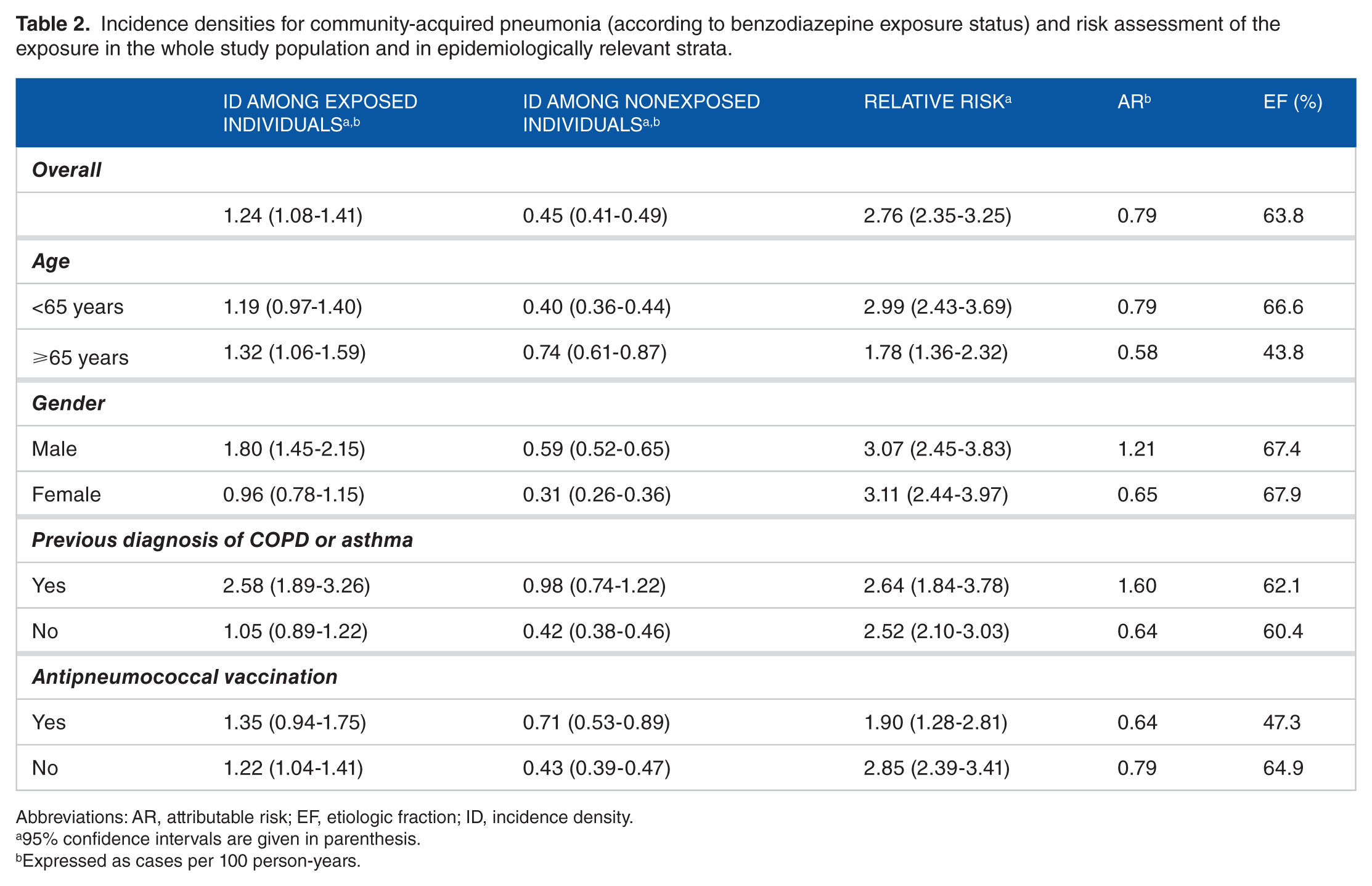
Abbreviations: AR, attributable risk; EF, etiologic fraction; ID, incidence density.
95% confidence intervals are given in parenthesis.
Expressed as cases per 100 person-years.
In total, 287 (41.2%) cases required hospital admission; 8 of them were admitted twice and 1 was admitted 3 times.
Discussion
In contrast to our hypothesis, this study shows that exposure to Bz significantly increases the risk of CAP. Based on its overall AR and the 95% CI of ID in exposed and nonexposed individuals, between 99 and 169 (ie, between 47.1% and 80.5%) CAP cases might have been prevented among Bz-exposed individuals, if exposure to these drugs could have been completely avoided.
The main strengths of this study are its prospective design and the fact that nearly the whole population ⩾14 years of age residing in the catchment area was included and followed for the whole study period. Moreover, data were collected directly from electronic records in which information is entered by trained professionals. Thus, the risk of selection and recall biases seems extremely low.
However, the fact that prescription was used as a surrogate of exposure is a significant limitation of our study, especially because the accuracy of prescription databases regarding Bz treatments may be suboptimal. 23 Moreover, the medical literature suggests that a record of a CAP diagnosis in a patient’s electronic health record is relatively reliable if a hospital admission was required, 24 but the accuracy in the outpatient setting has not been assessed. Thus, the quality of the analyzed data may not be optimal in all cases (eg, the incidence of CAP may have been overestimated by accepting the attending physician’s diagnosis even if a clinical suspicion had not been radiologically confirmed). However, we consider that given the fact that the potential for misclassification in either Bz exposure or CAP diagnosis is likely nondifferential (ie, there is no reason to suppose that it should occur at different frequencies in the exposed vs nonexposed or in the CAP cases vs noncases groups), these acknowledged limitations could dilute the magnitude of the studied association, but not bias it.
On top of this, we limited the number of study variables, so aspects that could be clinically important (such as specific Bz agent or a potential interaction of drug exposure with dysphagia) could not be explored and residual confounding cannot be excluded.
Our present results conflict with those of our previous studies.5,10 A possible explanation to this divergence is their different design. Our previous studies were case-control studies in which both exposures and outcomes were carefully verified (we used personal questionnaires to assess exposures and required x-ray confirmation of the CAP) for each participant; thus, they relied on a limited sample size, but the quality of the data was high. This study included a large population (in fact, the only exclusion criterion was age <14 years), but the resulting large number of study participants precluded the possibility to verify the accurateness (ie, the quality) of the data. It must be stated, however, that this study has a fairly large sample size and a very low attrition rate, is prospective, and its results are consistent through various strata and biologically plausible; for all these reasons, its results reinforce the hypothesis that Bz exposure is a risk factor of CAP.
Notwithstanding, evidence in favor or against the existence of an association between Bz exposure and CAP is still far from conclusive, and further studies to explore it are needed. In our opinion, the most appropriate design would be a sufficiently powered cohort study in which the accuracy of exposures and outcomes (eg, through personal drug questionnaires and chest x-rays) was assured. Such a study could provide valuable information not only on the actual existence of this association but also on other important unresolved issues, such as whether this would be a class- or an agent-specific effect, whether there is a dose-response relationship, and whether Bz exposure interacts with other risk factors for CAP.
The increased risk of CAP in individuals exposed to Bz, coupled with the previously reported association between Bz exposure and a higher mortality from CAP, should raise concern about this potentially preventable risk factor among prescribers. Consequently, unless the disclosed association can be convincingly refuted by future research, this risk should be considered when assessing the risk-benefit ratio of starting a treatment with Bz. 25
Footnotes
Peer review:
Five peer reviewers contributed to the peer review report. Reviewers’ reports totaled 705 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
JA and M S-P designed the study, AL collected the data, M S-P and EP analyzed and interpretaed the data, DS wrote the first draft, all authors critically reviewed the article and approved its final version.
