Abstract
Fertility preservation is an increasingly important discipline. It requires close
coordination between reproductive medicine specialists, reproductive biologists,
and oncologists in various disciplines. In addition, it represents a particular
health policy challenge, since fertility-protection measures are to be
understood as a treatment for side effects of gonadotoxic treatments and would
therefore normally have to be reimbursed by health insurance companies.
Therefore, it is inevitable that fertility-preservation activities should
organise themselves into a network structure both as a medical-logistic network
and as a professional medical society. The necessary network structures can
differ significantly at regional, national, and international level, as the size
of the regions to be integrated and the local cultural and geographical
conditions, as well as the political conditions are very different. To address
these issues, the current review aims to point out the basic importance and the
chances but also the difficulties of fertility-protection networks and give
practical guidance for the development of such network structures. We will not
only discuss network structures theoretically but also present them based on
three established, different sized networks, such as the Danish Network
(www.rigshospitalet.dk), representing a centralised network in a
small country; the German-Austrian-Swiss network
Introduction
Since the birth of the first child after ovarian tissue transplantation, fertility protection in women and men has developed into a new and clinically relevant independent field. 1 This field has a number of special features.
On one hand, the discipline is interdisciplinary, since fertility protection requires close coordination between reproductive medicine specialists, reproductive biologists, and oncologists in various disciplines.
On the other hand, some fertility-preserving measures such as transplantation of ovarian tissue, in vitro maturation of oocytes, maturation of oocytes from ovarian tissue, and the cryopreservation of testicular tissue from prepubertal boys are still in clinical or even scientific development and therefore require a high degree of specialisation on the part of the centres involved.2-5
In addition, this new specialist field represents a particular health policy challenge, since fertility-protection measures are to be understood as a treatment for side effects of gonadotoxic treatments and would therefore normally have to be reimbursed by health insurance companies, which is not yet the case in many countries.
Due to these special features of this field, it is inevitable that all the scientific, clinical, and, if necessary, even health care policy areas involved organise themselves into a network structure both as a medical-logistic network and as a professional medical society.
The necessary network structures can differ significantly at regional, national, and international level, as the size of the regions to be integrated and the local cultural and geographical conditions, as well as the political conditions are very different.
Therefore, in this article, we would like to point the basic importance and the chances, but also the difficulties, of fertility-protection networks and give practical guidance for the development of such network structures. We will not only discuss network structures theoretically but also present them based on three established different sized networks. These three networks have different goals and different logistic structures and thus cover the possible range of possible network structures.
The selected networks are as follows:
The Danish Network (www.rigshospitalet.dk). This network is a centralised network for the practical implementation of specific fertility-preserving techniques such as the cryopreservation and transplantation of ovarian tissue in a small country.
The German-Austrian-Swiss network
The Oncofertility Consortium (www.oncofertility.northwestern.edu). This is a decentralised, internationally oriented network that primarily serves the transfer of knowledge among its members.
Network Structure
The structure of a network depends on the following mostly given and therefore unchangeable conditions and the self-imposed goals:
Given conditions for the establishment of a network
Size of the region to be networked.
Transport-logistical development of the region.
Density and area coverage of reproductive medicine centres.
Willingness of the centres and doctors to cooperate
Political and financial support.
Health care policy conditions.
These conditions are largely unchangeable. Accordingly, the desired network structures must take these conditions into account and integrate them into the network concepts.
Goals for the establishment of a network
A nationwide supply with specialised centres should be established.
Individual, especially not yet fully established or experimental reproductive techniques such as cryopreservation and transplantation of ovarian tissue and cryopreservation of testicular tissue from prepubertal boys should be centralised.
Regular information events should be carried out by the participating centres and associated disciplines.
A data register is to be established.
The establishment of a good regional or national network should try to achieve all these goals. Only partial implementation is possible in some regions and countries.
Structural composition of networks
Networks are often modular; the number of modules depends on their size (Figure 1).

Networks are frequently set up as multimodular structures. Small modules (blue) such as infertility centres or hospitals integrate gynaecologists and oncologists etc. (green). Several of these small modules (blue) are organised as a medium-sized network (orange) such as regional or small national networks. Several of these medium-sized modules (orange) are organised as a large network with a centralised body (red) which organises registries, scientific activities, conferences, and so on.
The conditions and intentions of these modules are different (Table 1). The smallest modular unit is usually a reproductive medicine centre or a clinic that networks regionally or within the clinic with oncologists. Patients are referred to the reproductive medicine centres directly by the oncologists. The therapy decision is often based on direct bilateral communication. The reproductive medicine centre documents the treatments so that the data can later be passed on to a registry.
Characteristics of the different sized networks.
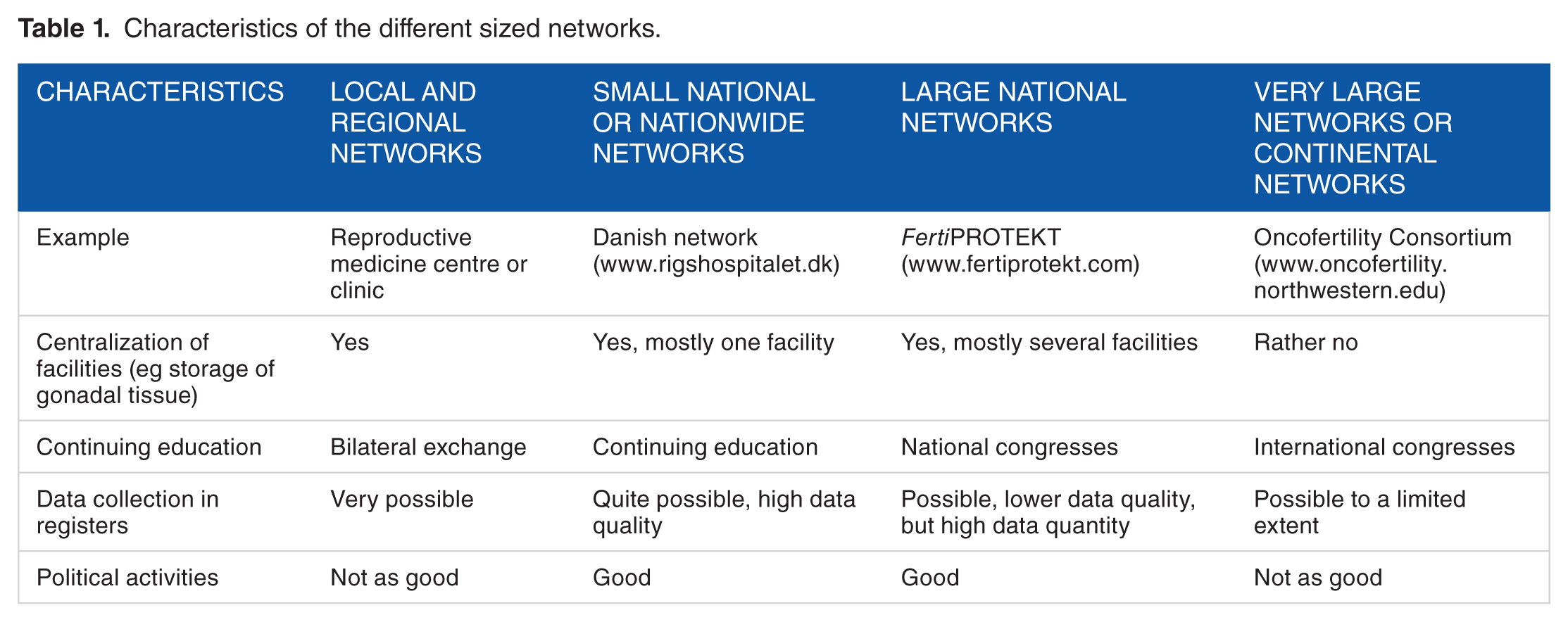
The next medium-sized modular stage is a union of local units into a small national or large regional network. An example of such a network is Denmark (www.rigshospitalet.dk). The centres know each other, and personal communication is possible. Data from the local units are merged into a register, which is relatively easy to create due to its limited size. It is easily possible to establish centralised, highly specialised facilities, for example, cryopreservation of gonadal tissue. The establishment of such centralised facilities allows high-quality fertility-protective techniques, scientific evaluation, good transparency of activities, and thus also health care policy initiatives. Due to short travel distances, shorter training courses can be organised with the help of oncologists. The strengths of these medium-sized networks lie in the possibility of being able to collect high-quality data, as detailed data documentation is usually possible.
In very large regions or larger countries, several network structures are
combined into one large network. One such example is the
In addition, it is possible to combine several of these networks for data collection and professional exchange. Examples of such international networks are the ‘Oncofertility® Consortium’ (www.oncofertility.northwestern.edu), the Special Interest Group ‘Fertility Preservation’ of the European Society of Human Reproduction and Embryology (ESHRE) (www.eshre.eu/Specialty-groups/Special-Interest-Groups/Fertility-Preservation.aspx), and the ‘International Society for Fertility Preservation’ (ISFP) (www.isfp-fertility.org).
Obstacles to Creating Networks
The explanations have shown that networks are clinically, scientifically, and politically of great importance. In practice, however, they are often difficult to implement.
What are the reasons for this?
In most cases, networks can only be implemented in regions and countries that have good medical, technical, and infrastructure care. Thus, the topic of fertility protection can only be of importance if sufficient oncological care is guaranteed. If these requirements are not met, network-based care with fertility-protective measures is hardly possible.
In industrialised, well-developed countries, there are no infrastructural obstacles to the establishment of networks. Nevertheless, it is often difficult to build networks. The main obstacle is the ‘human factor’ (Table 2).
The human factor as the main obstacle in the establishment of networks.

When setting up a network, priority should be given to examining what can motivate active participation. University centres are often more interested in scientific activities and cooperation, private centres more in economic advantages, and the use of networks as an advertising platform. Possible motivations for active participation must be identified and integrated into the network programmes. The willingness to participate in the network can often be increased by a democratic voice. If there is a danger that the right to have a say in the network will hamper its development, a democratically elected network board can also make sense to promote the development and expansion in a targeted manner with a small group of board members.
Financing of Networks
The financing of networks differs regionally and nationally. The start-up financing for setting up a network may differ.
Only a few thousand Euros were initially available from a pharmaceutical company for
the
In contrast, the United States initially made 22 million dollars available for the founding of the Oncofertility Consortium.
Undoubtedly, generous start-up financing is advantageous for the establishment of a network. Far more decisive is not the amount of funding but the initiative and willingness of a few people.
The following are necessary to start a network:
An initiation meeting with as many reproductive medicine centres as possible.
The development of a network name and logo.
A website that can be created largely free of charge by network members with IT experience.
An online documentation tool which can be created largely free of charge by network members with IT experience.
Regular (eg annual) continuing education events.
Introduction of the Danish Network – Centralised Network for Smaller Countries/Large Cities
History
Inspired by research efforts three decades ago from a large number of people including Roger Gosden, Outi Hovatta, Kutluk Oktay, and David Baird, it became clear that it would be potentially clinically feasible to freeze human ovarian tissue with the intention of replacing it later to restore ovarian function. At the end of the last century, encouraging results emerged from primate studies and from transplantation of human ovarian tissue.6,7 This sparked the first clinical initiatives of freezing ovarian tissue in the United Kingdom and Belgium. In Denmark, our laboratory conducted a number of mouse studies, in which different cryoprotectants and freezing protocols were evaluated. 8 However, Danish legislation clearly stated that it was illegal to transplant ovarian tissue to a woman. In 1998, we had a direct correspondence with Danish Minister of Health and he concluded that there were no restrictions on freezing ovarian tissue from a woman as long as only autologous transplantation was considered. There was no time limit on the storage period and normal medical rules applied for use of this technique including replacement of frozen thawed tissue. In addition, the Minister of Health informed that similar rules applied for testicular tissue. This was basically a very liberal rule that placed this new technique in the context of normal medical practice and the concept of ‘do no harm’.
At the end of the last century, our own laboratory had a more than 40-year-old tradition for studying the physiology of and working with human ovaries as the only laboratory in Denmark focussing on female reproduction. We started out with a clinical service freezing ovarian tissue at our local hospital in 1999, and soon thereafter, other parts of Denmark also wanted to start. We shortly realised that it would be more effective if we could centralise this service, which at that time was only performed occasionally. Instead of doing few cases per centre, we reasoned that it would provide a better service if the activity was centralised to the laboratory in which the knowledge and expertise of dealing the human ovaries was already available. On this background, we developed what is now known as the Danish concept for freezing ovarian tissue ‘the woman stays the tissue moves’ (Figures 2 and 3).9-12

Number of cryopreservations of ovarian tissue per year in the collaborating centres covering all of Denmark and the very southern part of Sweden.

Yearly number of cryopreservations of ovarian tissue (grey bar) and number of transplantations of frozen/thawed ovarian tissue (black bar).
Structure
Cancer treatment in Denmark is centralised mainly to three university hospitals located in different parts of Denmark. Each of these hospitals also has a fertility clinic which focus on in vitro fertilisation (IVF) treatment and other assisted reproductive technology (ART) procedures. In addition, our network now also includes Skåne Region of Sweden, in which the University Hospital of Lund and Malmø are members. These two regions have entered a formal agreement, allowing patients from one country to be treated in other country in the area of reproductive medicine. The Swedish side also has a fertility clinic that is coordinating the local counselling. The Skåne Region is now connected to Copenhagen via a bridge, and the transport time from the local hospital to the central laboratory is maximally 1 to 2 hours by car and lower from other parts of Denmark. Importantly, all these clinics are public-funded hospitals, and patients receive treatment for free paid for by the taxes, including extraction of tissue, freezing, storage, and transplantation. Furthermore, if ART is needed after transplantation, this will also be covered by the public health care system. Basically, any Danish woman or women in the Skåne Region of Sweden, who potentially may benefit from these procedures and who qualify for these treatments (ie age, diagnosis, and clinical evaluation) should have it offered and then it is her decision to accept or decline the offer.
In each of these fertility clinics, one or two consultants have specialised in fertility preservation. Patients potentially requiring fertility preservation identified in the oncological, haematological, or other departments in which patients are exposed to potential gonadotoxic treatment are referred to the consultants of the fertility clinic where they immediately get a consultation. Here, the different options are discussed with the patient including ovarian stimulation with cryopreservation of mature oocytes, excision, and cryopreservation of ovarian tissue or doing nothing. Depending on the clinical evaluation and the patient’s wishes, a plan for fertility preservation is agreed upon. In case freezing of ovarian tissue is planned, a date will be agreed upon with the central laboratory performing the cryopreservation and the surgeons excising the ovarian tissue at the local hospital.
The surgical intervention to extract the tissue is normally the first operation on that day, and the tissue will be able to reach the central laboratory in maximally 4 to 5 hours. After excision of the ovarian tissue, the surgeon will bring it to the local laboratory, where the tissue is placed in a 50-ml tube with basal medium that goes into a flamingo-box filled with crushed ice to maintain temperatures around 0°C. The box is transported to the central laboratory that checks for the presence of ice and processes the tissue immediately after arrival.
After cryopreservation, the tissue will be stored at the central laboratory and kept in liquid nitrogen until potential use or until the patient decides otherwise for her tissue. The central laboratory is accredited by the Danish authorities to conduct this treatment including a licence according to the European Union (EU) tissue directive. We have collaborated with the competent Danish authorities to formulate guidelines for new clinics and networks starting out to cryopreserved ovarian tissue. 13 If the patient request transplantation, the tissue will be transported in liquid nitrogen to the local hospital, where the surgical procedure of replacing the tissue will take place.
The clinical follow-up of transplanted patients is performed by the local hospital including monitoring of whether the patient becomes pregnant, experience relapse and so on.
Representatives from the participating clinics in the network will meet to discuss the service and results to align policies and various other matters when needed. We are setting up a framework for a database containing all information on patients who have ovarian tissue cryopreserved, which will provide valuable information looking forward. The competent Danish authorities secure implementation of the EU tissue directive and also function as advisers to the political system. We have discussions with authorities and have direct political contacts where we try to modify the Danish regulations to allow storage of tissue for social indications and for postponing menopause at the cost of the patient herself.
Financial support
Initially, the project was financed from external funding by the Danish Cancer Society, but around the last 10 years, the treatment has been recognised as an established treatment that the public health care system covers. As the technique is almost exclusively performed in the public system of hospitals that accounts for more than 99% of activity in the medical field anyway, the patient categories who have this technique offered for free have been limited to patients with a risk of iatrogenic-induced follicle loss and infertility plus patients with a genetic condition that may render them infertile prematurely.
Scientific focus
The scientific focus in Denmark has during recent years been on the surplus medulla tissue that contains growing follicles that do not sustain freezing (Table 3). Normally this tissue is discharged, but we have ethical permission to ask women for donation of this surplus tissue for research purposes. Furthermore, we can ask women who have cortical stored and who do not wish to continue storage for permission to use the tissue for research purposes. This has resulted in an unprecedented access to normal ovarian tissue both fresh and frozen from women at various ages and has allowed us to study human folliculogenesis.
Scientific focus and examples of corresponding publications with the participation of the Danish network.

Lately, we are focussing on establishing an optimised platform for human in vitro maturation (IVM), since the surplus medulla tissue contains immature oocytes which may become a surplus fertility-preservation option in case metaphase II (MII) oocytes can be generated in sufficient numbers with sufficient quality. We are in the situation that IVM is not considered a standard procedure in Denmark, and we are legally unable to use the MII oocytes generated for clinical purposes. 47
Key points for success of the network
The most important task of the network has been to establish a patient friendly and patient-oriented treatment offer that provided stable, reliable, and well-documented results in terms of follicle survival in connection with the freezing procedure and renewed and improved ovarian function after transplantation. Our service has provided these results to a large extent:
Following transplantation frozen/thawed ovarian tissue more than a total of 130 times; the tissue has consistently provided renewed and improved ovarian function for variable time periods mainly depending on the initial follicular density, patient age, and amount of tissue transplanted. Except for one case, where a woman in her mid-thirties had only three pieces of cortical tissue frozen, which did not result in ovarian function after transplantation.
It has been a focus area to employ a freezing technique with as good a follicular survival rate as possible. We have recently published a new quantitative method to evaluate follicular survival following a period of freezing and found a survival rate of on average 84%, with a 91% rate of healthy follicles in unfrozen control samples. 70 Furthermore, survival rates were constant over a period of freezing lasting 17 years and similar irrespective of whether the tissue was transported or not.
During several different interviews, patients have expressed great satisfaction with the treatment offer and the ability to stay in the local environment at difficult times having just faced a cancer diagnosis. The psychological impact of having tissue stored has a massive positive impact on many patients.
What can be further improved and further challenges
We have now recently documented that the quantitative survival of follicles during the freezing process is very high showing that only a minor part of follicles is lost during the freezing process. 70 In contrast, the follicle loss during transplantation due to poor vascularization, ischemia, and reduced oxygen tension accounts for the clear majority of follicle demise. We are therefore now trying to improve follicle survival during the initial stages of revascularization by interfering with the processes that lead to follicle atresia.
Furthermore, one aspect that can be optimised and which is of obvious importance for the patients is the speed at which fertility preservation is executed. They may suffer a potential deadly disease and require in many instances gonadotoxic treatment as fast as possible. We are now trying to monitor the speed at which the procedure is executed and whether there may be cases where we can reduce the time needed to perform the fertility-preservation procedure.
Introduction of Ferti PROTEKT Network – a Partly Decentralised
Network for Large Countries
History
At the start of this millennium, various scientific endeavours in the field of
fertility protection already existed in Germany,71,72 but there was still no
coordinated counselling and care of patients. Therefore, at the initiative and
invitation of Prof M. von Wolff (then Department of Gynaecological Endocrinology
and Fertility Disorders, Heidelberg) and Prof M. Montag (then Department of
Gynaecological Endocrinology and Reproductive Medicine, Bonn), 30 university
reproductive medical centres met in Heidelberg in May 2006 and founded the
However, the increasingly well-known national and international network was
initially not a founded scientific society and was in danger of, for example,
being insufficiently recognised and acknowledged in professional policy
discussions or in the development of guidelines by other (including
interdisciplinary) scientific societies. Therefore, in 2015, the decision was
made to create a scientific society. The
Centres that would like to become members of the
Structure
The


Number of documented counsellings and treatments performed by the large
German-Austrian-Swiss network
The structural characteristics shown in Table 1 are represented as follows in
the network
Centralisation of facilities
This applies to the centralization of cryobanks for ovarian tissue in the
The cryobanks have established a transport logistics system which plans that the peripheral member centre providing advice and the indication for cryopreservation of ovarian tissue orders a special transport container from the cryobank and has it transported to the relevant centre. The latter sends the tissue immediately postoperatively in this container in a nutrient medium on ice (ie under defined conditions) to the cryobank overnight, where it is prepared and cryopreserved.
Continuing education
From 2006 to 2018, 2-day working meetings were held annually at different
locations, organised logistically and in terms of content by the board of
the network and a representative of the member centre of each venue. In
addition to individual lectures, the programme included workshops on various
fertility-protection methods. From 2018, these meetings will only be held
every 2 years because
Data collection in registers
Data are currently entered in a self-created register, which is accessible to
the member centres via the intranet of the
Political activities
Financial support
When the
Since the founding of the
Scientific focus
The scientific strengths lie in register analyses, studies with tissue stored in centralised cryobanks, smaller multicentre studies, and the compilation of practically oriented recommendations (Table 4).
Scientific focus and examples of corresponding publications with the
participation of the

See also keyword «Fertiprotekt» in PubMed (www.ncbi.nlm.nih.gov).
Key points for success of the network
Establishment of numerous and qualified counselling and therapy centres
The greatest merit of the network is the consolidation and coordination of many advisory member centres, which helps patients and oncologists, to find a contact person for advice on fertility-protection measures even at short notice, who can also implement them promptly if they so wish.
Coordination of content of counselling and therapies
Through studies, publications of recommendations and joint exchange among member centres and other specialist areas, the network has created a basis for the content-related consultation and implementation of fertility-protection therapies. The annual 2-day national member meetings, in which approximately 200 persons participate, are an essential element for the coordination of content.
Documentation of consultations and therapies in a register
A register for fertility-protective consultations and therapies has existed since 2007. Due to the high quantity of data and the further improvement in quality in the future (through data entry via the national IVF register), there have been and will be opportunities in the future to clarify scientific questions. In addition, the amount of data is an argumentation aid, for example, in the enforcement of cost absorption by health insurance funds.
Initiation, implementation, and support of studies
The close cooperation and common interests in the network enable a timely agreement on the initiation of studies and their implementation. Scientific questions from member centres can be dealt with conceptually within a short period of time and can be investigated by joint prospective or retrospective studies on larger amounts of data.
Definition of standards and publication of recommendations
Joint working meetings in combination with our own study data and international
experience form the basis for the constantly developing standards discussed in
the network, as well as published recommendations for counselling and therapy in
the context of fertility protection. The first recommendations were published in
2011 with ‘online access’ to make them available to as many readers as possible
free of charge.
90
The update was carried out in 2018 with a paper on
indications based on selected diseases and another on fertility-protective
techniques.91,92 The association also financed the online access for these
two publications. A German Austrian and Swiss guideline on fertility
preservation was prepared and published
93
by the AWMF with the
contribution of
What can be further improved and further challenges
At a strategy meeting in April 2018, the executive board of the
Introduction of the Oncofertility Consortium – A Decentralised Global Network
History
Oncofertility is a discipline that merges oncology with fertility and has moved rapidly from the purview of individual champions to an integrated field that has become standard of care in many institutions. 94 Oncofertility as a field has developed in parallel to the many life-preserving advances in oncologic care, including earlier diagnostics and the emergence of targeted cancer therapies, methods to reduce radiation dose and field, and localised surgical procedures. Addressing the complex treatment plans, general health, and quality of life issues that concern young cancer patients whose fertility may be threatened by their disease or its treatment is a priority, and the Oncofertility Consortium has led efforts in this area for almost 15 years.94-104 Northwestern University was first funded as a Specialised Cooperative Centre Programme in Reproductive Research (SCCPIR) in 2003, and the Centre for Reproductive Research focused on understanding structure-function relationships in reproductive science. This centre provided a mechanism to bring new perspectives to reproductive science from ancillary disciplines (eg bioengineering and structure biology). In 2007, we transitioned this fundamental science to a National Institutes of Health (NIH) Roadmap Interdisciplinary Research Consortium – the Oncofertility Consortium – to specifically address the intractable problem of fertility-preservation options for young female cancer patients. 94 In 2012, we broadened the scope of the Oncofertility Consortium to include the Centre for Reproductive Health After Disease, whose mission is to protect and preserve the reproductive health – including fertility, endocrine health, sexuality, or the ability to carry an offspring to term – of women at reproductive risk after disease or treatment of disease.
Structure
The multidisciplinary, international approach to the Oncofertility Consortium consists of research sites Oncofertility Professional Engagement Network (OPEN) Members, and key members of the multidisciplinary oncofertility team. The Oncofertility Consortium includes stakeholders across the width and depth of the academy, across departments and institutions, and includes expert core facilities and sophisticated human specimen collection and use and disseminates our work to patients, families, and the public. These are meaningful interactions that are fostered by the presence of a strong organisation like the Oncofertility Consortium and through leadership at the academic and staff level.
Centralisation of facilities
The Oncofertility Consortium Network spans six continents, including 40 countries around the globe and 97 sites in the United States (Figure 6). The OPEN works to engage researchers and clinicians both domestically and aboard, and these communities converge at the Annual Oncofertility Consortium Conference held in Chicago, Illinois, each year.
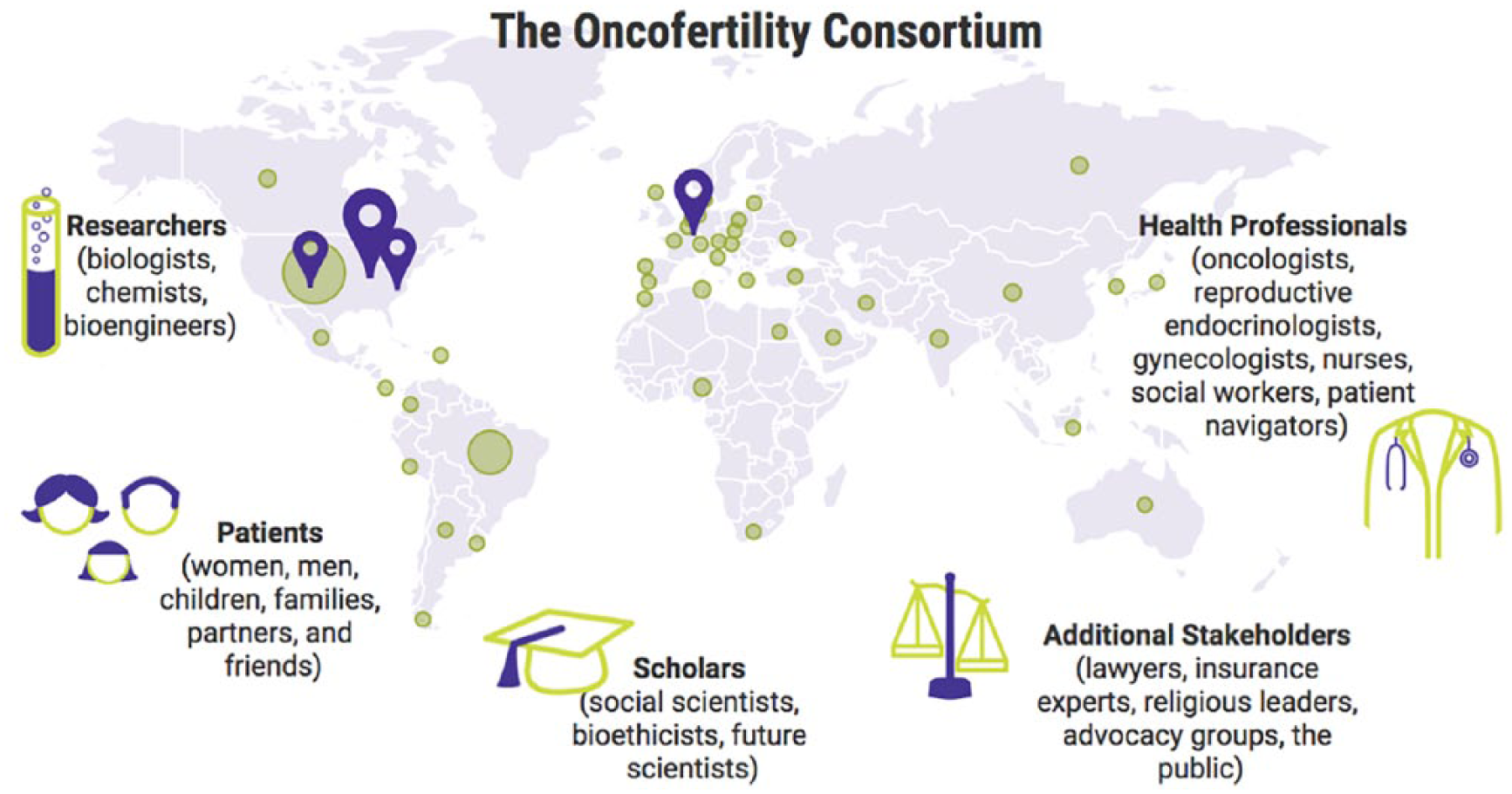
Decentralised structure of the Oncofertility Consortium.
Continuing education
Education is a hallmark of the Oncofertility Consortium’s programming. We have created numerous field forming and changing tools that have catalysed the growth of oncofertility. These items include the first comprehensive oncofertility textbook, training videos, and educational materials that are enduring but also updatable. These products are listed below in more detail. In addition to these, we also house institutional review board (IRB) documents that can be used as templates for those wanting to start an oncofertility programme, a Follicle Culture Handbook for basic scientists, and many other materials that enable faster adoption of best practices by members of the broader field. Through the use of Facebook, Twitter, and other social media modalities, the Oncofertility Consortium facilitates the ability of our projects to be communicated in a way that enables the public to see how the work is progressing in a lay-friendly manner.
Annual Oncofertility Consortium Conference
The Oncofertility Consortium hosts an annual conference to convene the field and set priorities for the upcoming year. Oncofertility Consortium focuses on providing attendees at the annual Oncofertility Conference the ability to connect with colleagues from around the world to share research and clinical case studies to facilitate a rapid pace of growth within the field and expand resources to non-malignant conditions. The Oncofertility Conference is a place where the field-wide advances are shared through traditional lectures, hands-on training, and small group sessions. The variety of education settings address the wide range of education levels and backgrounds represented at the conference.
Fellow Education Day Symposium
At the annual Oncofertility Conference, we host an annual Fellow Education Day Symposium. The purpose of the course is to educate fellows on fertility-preservation options and survivorship care for cancer patients across the reproductive life cycle. The course will also model a team approach to fertility-preservation care. The course is comprised of didactic lectures given by leaders in the field interspersed with complex clinical cases that will be reviewed in interdisciplinary teams. Participants are supplementary materials for review before course attendance to facilitate an interactive ‘flipped classroom’ approach to team-based learning. This course uses e-learning modules that were created with the American Society for Reproductive Medicine (ASRM). 105
Oncofertility textbooks
Together with other colleagues in the field, we have published seven books to encompass the areas of basic science, ethics religion and the law, medical practice, communication strategies, paediatric and disorders of sexual development populations, and non-oncologic and other non-malignant fertility threatening conditions, as well as the first of its kind Oncofertility Textbook, which includes didactics. The hope is these books, which aggregate everything we know in the field, serve as a starting point for material that will become integrated into the major oncology, internal medicine, and reproductive texts of our professions.
Oncofertility Saturday Academy
The Oncofertility Science Academy was created in 2007 as a way to introduce underserved high school girls from the Chicagoland area to science and medicine by engaging them in hands-on lab and clinical activities on the Northwestern University medical campus. Northwestern’s OSA programme impacted more than 275 students with 5 students securing oncofertility research internships or employment within the Woodruff Lab; a true example of training the next generation of future clinicians and scientists. The Oncofertility Saturday Academy (OSA) model addresses the gap in reproductive science education at the high school level and provides an adaptable education model that can be implemented across multiple institutions. Currently, OSA curricula are available at Northwestern University; University of Pennsylvania; University of California, San Diego; and Oregon National Primate Research Centre and has impacted 545 high school girls nationally. Learning goals of OSA include providing students with hands-on laboratory and clinical activities, incorporating art modules for learning scientific and medical information in a new format and developing relationships with scientists, doctors, and other professionals. 106
Data collection in registers
The Oncofertility Consortium does not have any formal registries or patient data collection.
Political activities
As the Oncofertility Consortium is housed within Northwestern University, the university policy limits lobbing activities and the Consortium’s ability to directly contact politicians and participate in many political activities. However, the Oncofertility Consortium works with other politically motivated groups, like the Alliance for Fertility Preservation, to push forward legislative activities in any capacity within its scope. The Oncofertility Consortium is the great convener in the United States and helps to make critical connections among members of the oncofertility community, like lawyers, patients, and politicians, to ensure that fertility-preservation coverage is attainable in each state. Currently, there are five states in the United States that require insurance companies to cover oncofertility and fertility-preservation services for cancer patients.
Financial support
Most of the funding for the Oncofertility Consortium’s activities comes from the NIH and the Eunice Kennedy Shriver Institute for Child Health and Human Development (NIH/NICHD). Its efforts are currently supported by the Centre for Reproductive Health After Disease (P50HD076188) from the NIH National Centre for Translational Research in Reproduction and Infertility (NCTRI). The annual Oncofertility Consortium Conference has been funded for years by an NIH grant (5R13HD063248), as well as institutional funds from the Robert H. Lurie Comprehensive Cancer Centre at Northwestern University and the Office of the President. In addition to these funds, we have secured other funding from industry partners including EMD Serono, Merck, Ferring, Walgreens, and Reprotech Ltd. We have targeted a variety of sources to enable our success and made innovation and invention in communication methods a main mission of our programme.
Scientific focus
Mechanisms underlying the fertility threat of life-preserving cancer drugs (Figure 7, Table 5).
Methods for cryopreservation (freezing), storing, and growing ovarian and gonadal tissue.
In vitro follicle grown and oocyte maturation using a three-dimensional environment.
Communication barriers between cancer patients and health care providers.
Ethical and legal concerns regarding the use of fertility-preservation technologies in cancer patients.

10 years of transforming traditional bench science in the Oncofertility Consortium network.
Scientific focus and examples of corresponding publications with the participation of the Oncofertility Consortium network.

Key points for success of the network
Because of the intrinsic value in creating diverse networks and collaborations, the Oncofertility Consortium continues its efforts to connect local centres of excellence and create a strong global network of diverse collaborators, many of whom may not have worked together otherwise. The Oncofertility Consortium supports interaction between global and local partners to create momentum for clinical activities (shared protocols and patient case studies, inclusion of allied health professionals), research (sharing results, both failures and successes, in ways that hasten work), and meeting patient needs (educational websites, patient decision tools, and patient navigator). By facilitating these interactions, the Oncofertility Consortium ensures the coordinated effort of the global oncofertility community in conducting cutting-edge research that can continue to be rapidly translated to the clinic and establish an evidence-based standard of care.
What can be further improved and further challenges
There are a number of common barriers that are commonly identified by OPEN members and the Oncofertility Consortium. These barriers include lack of insurance coverage and high out-of-pocket costs for patients, lack of awareness among providers and patients, cultural and religious constraints, and lack of funding to help to support oncofertility programmes. Despite these barriers, many opportunities exist to grow the field of oncofertility. Continuing to engage stakeholders around the globe and expand the efforts of the Oncofertility Consortium will aid in the acceptance of oncofertility on a global level thus accelerating the pace of research from bench to bedside to babies.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MVW designed the manuscript. All authors prepared the manuscript and revised the final version.
