Abstract
Background and aim:
Pediatric patients undergoing cardiac surgery prior to 1992 in Denmark were at risk of hepatitis C virus (HCV) infection through donor blood used in extracorporeal circulation. HCV screening became possible in donors in 1991, eliminating the risk of iatrogenic infections. No formalized screening has been conducted for patients receiving non-screened blood, potentially leaving some with undetected HCV infection.
Objectives:
This study aimed to determine the prevalence of chronic HCV infection in this group of patients and offer treatment to those affected.
Design:
Nationwide cross-sectional study.
Methods:
Between 2020 and 2023, 1645 individuals who underwent pediatric heart surgery before 1992 in Denmark were identified. Participants were invited for HCV screening using anti-HCV-antibody and HCV-RNA tests. Patients testing positive for HCV were referred to direct-acting antiviral (DAA) treatment.
Results:
Of 1645 patients identified, 571 consented to participate, and 246 completed HCV screening. Two individuals tested positive for chronic HCV infection, resulting in a prevalence of 0.8%. Both patients were asymptomatic for many years before treatment and successfully cleared the virus after DAA treatment.
Conclusions:
The 0.8% prevalence of HCV in this cohort is higher than in the general Danish population although lower than in similar studies from the U.S. and Germany. This may reflect Denmark’s practice of unpaid blood donation, reducing infection risks. Targeted screening for at-risk cohorts exposed to transfusions before 1992 could aid in HCV detection and treatment, potentially preventing long-term liver complications.
Registration:
The study was approved by the Capital Region Ethics Board in Denmark (j.nr. H-18062088).
Keywords
Introduction
In Denmark, approximately 3000 open heart surgeries are performed annually, with around 200 of these procedures involving pediatric patients.1,2 During open heart surgery, the use of extracorporeal circulation (ECC) for cardiopulmonary bypass is essential to ensure optimal surgical conditions. For pediatric patients, priming the extracorporeal circuit with a liquid solution typically requires donor blood. This necessity has historically exposed children to the risk of iatrogenic contamination with various bloodborne viruses, such as human immunodeficiency virus (HIV) or hepatitis C virus (HCV).
Blood donation was made possible in Denmark in 1932 3 and has been common practice long before screening of virus infections became possible, leaving a significant period during which the risk of viral transmission was present. In 1991, it became possible to screen donor blood for HCV, effectively eliminating the risk for iatrogenic infection. However, children undergoing open heart surgery before 1992 remain at risk of being infected with HCV, as no formalized screening program has been conducted for these patients in Denmark until now. 4 This concern is underscored by several studies demonstrating an increased risk of HCV infection among pediatric patients undergoing open heart surgery compared to the general population.5 -8
HCV is a bloodborne virus that primarily affects the liver. Often, the infection is asymptomatic or presents with mild symptoms. 9 However, approximately 65% of those infected will develop a chronic HCV infection, with a significant risk of increased liver-related morbidity and mortality due to liver cirrhosis and eventually hepatocellular carcinoma (HCC),10 -12 especially among individuals with concurrent risk behaviors, such as alcohol or intravenous drug abuse.13,14 The annual risk of developing HCC ranges from 2% to 7%15,16 with a median survival time of 8.4 months. 17 Cirrhosis caused by HCV infection acquired during childhood is thought to have slower progression compared to HCV infections acquired in adulthood. 18 However, a UK cohort found that cirrhosis developed in 32% a median of 33 years after infection. 19 Consequently, proactive detection and treatment are crucial to prevent significant liver damage.
In 2014, a new treatment era of chronic HCV was initiated. 20 Direct-Acting Antivirals (DAAs) work by inhibiting HCV specific proteins that are vital for the replication of the virus. 21 The combination of DAAs is either dependent of the HCV genotype or pan-genotypic, and the treatment can effectively eradicate the virus in 95% to 100% of the patients. 22 This treatment prevents the inflammatory response and likely halts the progression to cirrhosis and potentially liver cancer, except in case of cirrhosis development before DAA treatment, in which cases risk of HCC persists. Since November 2018, all people living with HCV infection have been eligible for curative therapy with DAAs in Denmark, regardless of disease progression. 9 Additionally, curative DAA therapy is now available in granule formulations, enabling treatment of children as young as 3 years old. 23
The prevalence of HCV in the Danish population is very low, and has recently been estimated to approximately 0.21% in a registry study. 24 Additionally, a Danish study conducted in 1990 found that 0.3% of blood donors were HCV antibody positive in 2 random samples from 1982 and 1989, respectively. 25 The low prevalence is likely due to free access to health care in Denmark, the absence of financial incentives for donating blood, and the generally high standard of living across the entire population. Therefore, the prevalence of HCV infection among Danish patients who underwent open heart surgery in childhood before 1992 is expected to be very low. Nonetheless, it is crucial to consider that people living with HCV may be severely ill without suspicion of HCV infection as the underlying cause and could potentially serve as a source of contamination for others. Thus, understanding the prevalence and implications of HCV infection in this patient population is of significant importance.
The objective of this study was to define the prevalence of HCV infection in patients who underwent open heart surgery in childhood prior to 1992. We aimed to identify patients infected with HCV with the intention to offer them curative therapy.
Patients and Methods
This cross-sectional study was conducted from 2020 to 2023. The study focused on individuals who underwent congenital heart surgery during childhood, at any timepoint before 1992, at 1 of the 2 tertiary centers, located in either Aarhus or Copenhagen, where pediatric heart surgery historically has been offered in Denmark. 26
Eligible patients were identified in 2020 through the Danish National Patient Registry (NPR), a comprehensive national database established in 1977. The Danish NPR receives data on all patients who have had contact with any public hospital in Denmark. From 1977 to 1994, the registry used ICD-8 diagnosis codes to classify patient information. In 1994 the system was updated to use ICD-10 codes. 27 It is possible to find surgeries performed before 1977 in the Danish NPR. These were likely registered when it became possible, either because the patient underwent subsequent surgery or had continuous follow-ups related to their earlier procedures. Eligibility criteria for inclusion were patients who were registered in the NPR with 1 or more ICD-8 procedure codes ranging from opr30000 to opr32990, prior to 1992, and before reaching the age of 18 years. Furthermore, eligible patients had to have been on ECC during surgery. If medical record material detailing the procedure was unobtainable, the patient was asked to confirm a sternotomy scar. Deceased or internationally relocated patients were excluded.
In Denmark healthcare is cost free for the individual since the hospitals are funded by Danish taxpayers. Every individual residing in Denmark is assigned a 10-digit civil personal registration number (CPR-number). The CPR registry contains information about address and marital status, which enables researchers to contact patients via mail. Recently, communication with citizens has transitioned to the e-Boks system, an encrypted, digitally based mailbox accessible to all Danish residents. 28
In this study, patients were contacted via their e-Boks in November 2020 and invited to participate in the project. Those who consented, granted permission for access to their digital or paper medical records to establish eligibility, including previous blood tests and information on any prior HCV treatment. Patients who did not respond received an automatic reminder via e-Boks 14 days later.
The eligible patients, who had not undergone previous HCV screening, was invited to undergo testing for HCV with a venous blood sample between 2021 and 2023. Blood samples were collected either during routine follow-ups at their local cardiology outpatient clinic, by local laboratories, or by their general practitioner. Samples were promptly analyzed, and the result was registered in the patient’s digital medical record. The blood sample was analyzed for anti-HCV antibodies with enzyme-linked immunosorbent assay (ELISA) with reflex-testing of HCV-RNA and -genotyping through polymerase chain reaction (PCR) in case of a positive anti-HCV antibody result. Patients with confirmed HCV infection, defined as positive HCV-RNA, were referred to their local department of infectious diseases with the intention of starting DAA treatment. In addition, diagnostics and monitoring programs outlined in the Danish National Guidelines for the Treatment of Hepatitis C were followed, 9 including an ultrasound-based transient liver-elastography used to measure liver stiffness and assess fibrosis. 29 All test, examinations and treatments were free for the individual patient and covered by the Danish healthcare system. The outcome of treatment was documented in the patient’s digital medical journal and the patient was considered cured, in case of a sustained virologic response, defined as HCV-RNA equal to 0, 12 weeks after end of treatment. 9
This study was conducted in accordance with the principles outlined in the Declaration of Helsinki and was approved by the Capital Region Ethics Board in Denmark (j.nr. H-18062088).
Statistical analysis
A descriptive statistical analysis was conducted to summarize the demographics of the population using RStudio by R Core Team (2024), version 2024.04.2+764 (2024.04.2+764; R Core team, 2024). 30 As this study is descriptive with the intention of summarizing and describe a phenomenon, the use of P-values and significance testing were not applicable.
Results
A total of 1645 patients, who underwent pediatric cardiac procedures before 1992 in Denmark, were identified. Of these, 571 (34%) consented to participate, 5 (<1%) requested more information, 39 (2%) declined, and 1030 (63%) patients never replied (Figure 1). Among the consenting participants, 407 (71%) met the eligibility criteria and were included for HCV screening. Of these, 90 (22%) had prior HCV antibody tests done, and the remaining 317 (78%) were offered HCV screening, with 1 year to complete the blood test. Despite this, only 156 patients completed the HCV screening, leaving 161 patients untested. In total, 246 (60%) of the eligible consenting participants completed HCV screening and hence, had an available HCV antibody test result.

Overview of screening and inclusion process. It shows how many patients were identified and asked for consent, how many said yes to participate and of these how many were eligible.
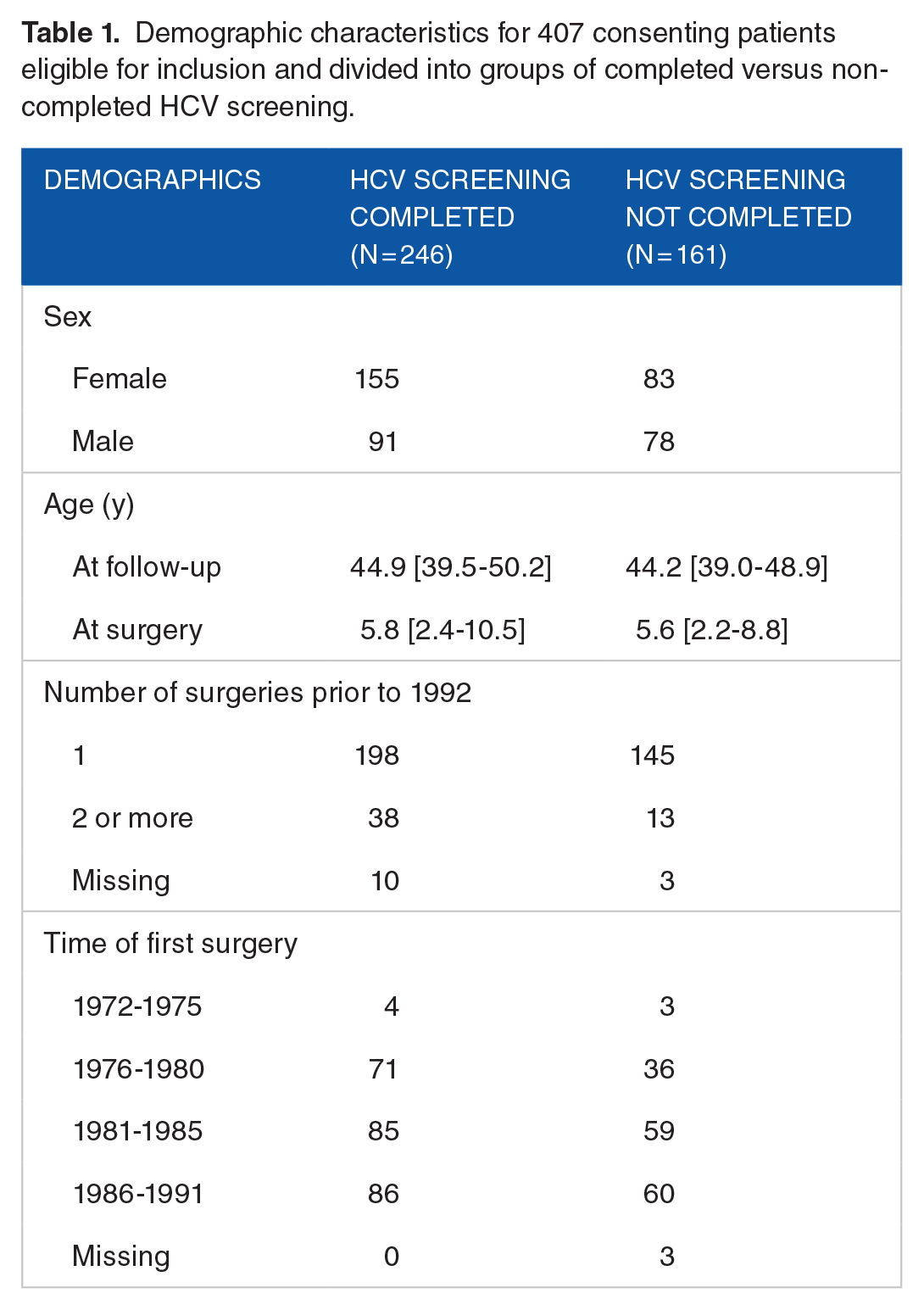
For the group that completed screening, the median age at inclusion was 44.9 years [IQR = 39.5-50.2], and the median age at time of surgery was 5.8 years [IQR = 2.4-10.5], 155 (63%) were female, and 38 (15%) had undergone 2 or more cardiac surgeries prior to 1992 (Table 1). The most frequent procedures performed between 1972 and 1992 were septal defect repairs (56%) followed by valve surgery (20%), including pulmonal stenosis (10%) and aortic stenosis (5%; Table 2).
Demographic characteristics for 407 consenting patients eligible for inclusion and divided into groups of completed versus non-completed HCV screening.
Types and number of cardiac surgical procedures performed in the included cohort. Some patients underwent multiple procedures, resulting in a total number of procedures greater than the number of patients (407).

Among the 246 patients who completed HCV screening, 244 tested negative while 2 patients had tested positive for HCV-antibodies and HCV-RNA, indicating chronic HCV infection. Both of these individuals were among the 90 participants who had undergone HCV antibody testing prior to this study and hence, had already been tested positive prior to inclusion. This represents a prevalence of HCV infection following cardiac surgery in childhood of 0.8%.
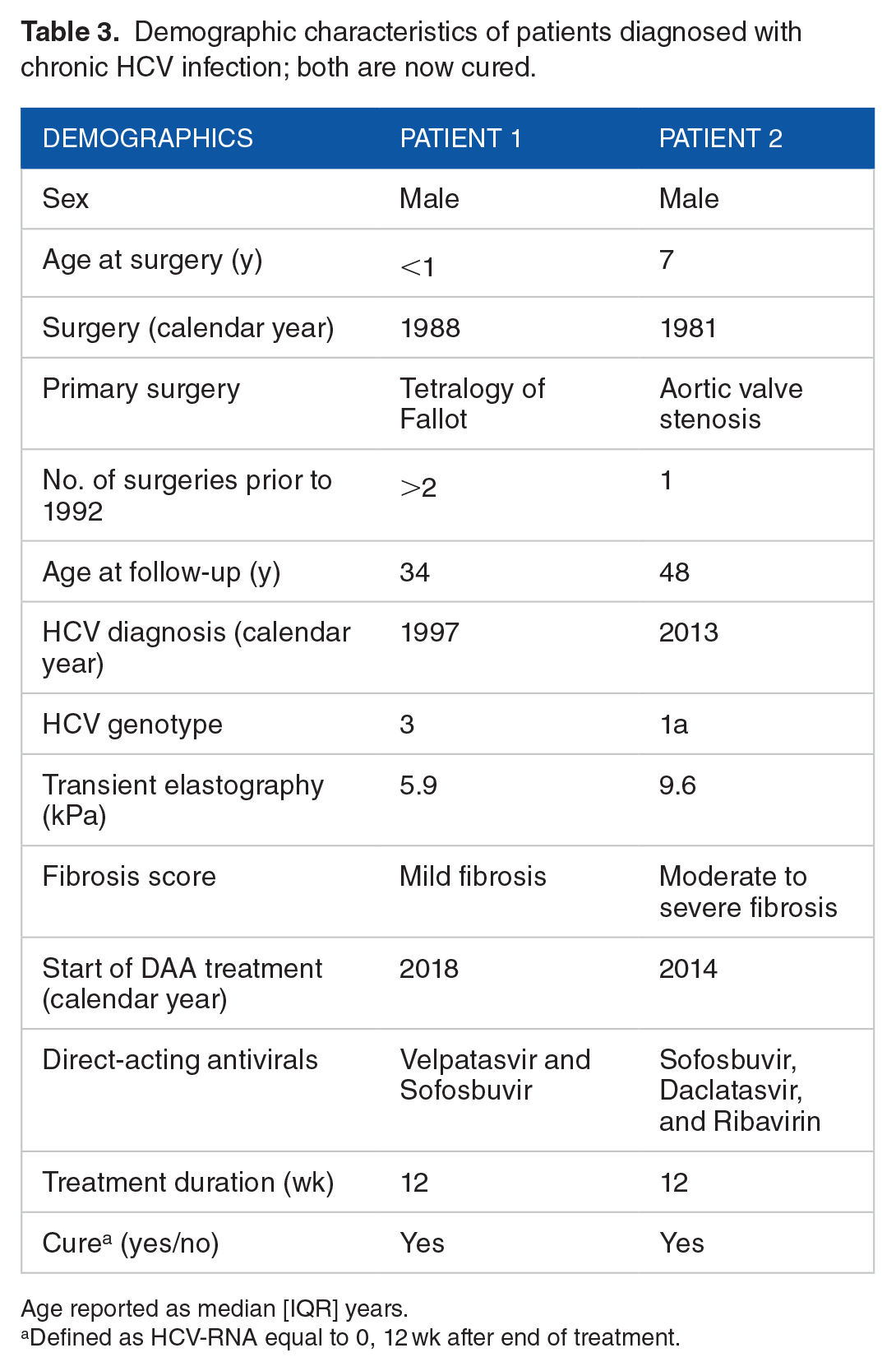
The 2 patients diagnosed with chronic HCV infection were both male, neither had displayed any risk behaviors, nor had a family history of HCV ruling out perinatal transmission. Based on their clinical history, infectious disease specialist concluded that they were likely to have contracted HCV during their cardiac surgery in childhood. Patient 1 underwent surgery for tetraology of fallot before the age of 1 year in 1988. The exact age at surgery is unknown as the original surgical records could not be retrieved, but other medical records confirm the procedure took place in 1988, the year of his birth. He was diagnosed with chronic HCV, genotype 3, in 1997. He was predominantly asymptomatic until 2018 when he developed pruritus due to elevated bilirubin levels. Treatment with the combination of velpatasvir and sofosbuvir was initiated in 2018. The patient achieved viral clearance and was declared cured in 2019 (Table 3). Patient 2 underwent surgery for aortic valve stenosis at the age of 7 years in 1981. He was diagnosed with chronic HCV, genotype 1a, in 2013. This patient was also predominantly symptom free, except for excessive tiredness and elevated alanine transaminase (ALT) levels. Combination treatment with sofosbuvir, daclatasvir, and ribavirin was initiated in 2014 and viral clearance was achieved later same year (Table 3).
Demographic characteristics of patients diagnosed with chronic HCV infection; both are now cured.
Age reported as median [IQR] years.
Defined as HCV-RNA equal to 0, 12 wk after end of treatment.
Prior to DAA treatment, both patients had a transient liver-elastography done to assess the level of liver fibrosis and potentially cirrhosis. 29 Neither had developed cirrhosis, however, patient 2 had a fibroscan that revealed a stiffness of 9.6 kPa signifying moderate to severe liver fibrosis.29,31
Discussion
In this Danish nationwide cross-sectional study, we detected a prevalence of chronic HCV infection of 0.8% among individuals who underwent cardiac surgery during childhood before 1992. This prevalence is slightly higher than the 0.3% prevalence reported among the Danish blood donors around the same time 25 and the current estimated prevalence of approximately 0.21%. 24 However, the observed prevalence in this study is noticeably lower than prevalences reported in similar studies from The U.S. and Germany, where HCV infection ranged from 4.8% to 9.5%5,6,8 and 14.6%, 7 respectively.
The notable difference in HCV prevalence between Denmark and these countries cannot be explained by the prevalence of HCV in the general population at the time, which was approximately 1.8% in The U.S. 32 and 0.4% in Germany. 33 A likely contributing factor is the historical practice of paid blood donations in the U.S. and Germany, which may have incentivized more vulnerable and higher-risk populations to donate, leading to greater transmission of bloodborne infections.34 -36 In contrast, Denmark’s system of voluntary, unpaid blood donation likely helped maintain a lower risk of infections.
The 2 individuals identified in this study who developed chronic HCV infection both remained largely asymptomatic until adulthood. This observation aligns with the existing literature suggesting that HCV infection acquired in childhood may follow a less aggressive course, with lower rate of chronic infection (<50%), milder symptoms, and a delayed onset of liver-related complications, compared to infections acquired later in life.37,38 In contrast over 65% of adults infected with HCV will advance to chronic hepatitis,11,12,37 with more than 20% developing severe complications such as decompensated cirrhosis or HCC within 20 years of initial infection. 39 Interestingly, the patients in this study had active infection for more than 30 years before they started treatment, reinforcing the potential for delayed disease progression in childhood-acquired cases.
It is also important to consider that up to approximately 35% of individuals exposed to HCV spontaneously clear the infection.10,40 This is thought to be more common in younger age 40 and when vertically infected.18,41 Some individuals will even experience seroreversion, becoming anti-HCV negative with time,38,42 which could potentially explain the relatively low prevalence observed in this study. The combination of spontaneous clearance and the lower virulence of childhood infections may have contributed to the overall findings.
Strengths and limitations
A key strength of this study is its nationwide scope, made possible by Denmark’s highly digitalized society. The Danish NPR enables access to comprehensive data material, while the CPR registry ensures that all individuals, including those who have passed or moved abroad, are accounted for, which provides accurate and efficient tracking of the study population. Denmark’s healthcare system, which is free of charge for the individual, guarantees that every child in need of heart surgery received it, without financial limitations. Additionally, the ability to offer free HCV blood testing further strengthens the study, as no participant was excluded due to financial reasons.
Some limitations must also be considered. Nearly two-thirds of the identified patients did not respond to the invitation to participate, and ultimately only 246 completed HCV screening. This leaves a noteworthy number of untested individuals, which introduces the potential for survival bias. Due to strict data protection regulations in Denmark, it was not possible to gather any information on these individuals without explicit consent. This made it impossible to know whether the non-responding group was particularly fragile or exhibited different risk profiles and health outcomes. Consequently, the findings may not fully represent the entire cohort. In light of the notably higher HCV prevalence reported in similar cohorts from the U.S. and Germany, our study may have missed additional HCV-positive patients if the untested group carried the same risk. However, it seems unlikely that survival bias alone could account for such discrepancy. A relevant aspect is that blood donors in Denmark are not financially compensated, reducing the risk of donation-related biases in infections rates, supporting the reliability of the findings.
Relevance and implications of the findings
The findings of this study provide valuable insights into the long-term risk of HCV infection among individuals who underwent pediatric heart surgery before 1992. The slightly higher prevalence of HCV compared to the general population suggests that this group is at a somewhat elevated risk, making it worth considering targeted screening for similar cohorts. Our study raises awareness of a potential risk that healthcare providers should keep in mind when treating patients who were exposed to blood transfusions prior to 1992. Given the availability of a highly effective treatment with DAAs, early identification and treatment of chronic HCV infection in such cohorts could prevent progression to serious liver disease. While this study only identified 2 cases of chronic HCV infection, it emphasizes the importance of awareness in at-risk groups and underscores the need for further research to explore risks and outcomes in similar cohorts.
Conclusion
In this nationwide Danish study, we identified a chronic HCV prevalence of 0.8% among individuals who underwent pediatric heart surgery prior to 1992. While this prevalence is slightly higher than in the general population, it is notably lower than reported in similar studies from the U.S. and Germany. The difference is likely attributable to historical blood donation practices, with paid donations in those countries contributing to higher transmission rates. Despite the low prevalence, the potential for long-term HCV complications highlights the importance of considering targeted screening for at-risk cohorts exposed to transfusions before 1992.
