Abstract
Co-trimoxazole is an antimicrobial drug gotten from potentiation of sulfamethoxazole with trimethoprim. It is widely used for the treatment of bacterial and protozoan infections in humans. It is also used in veterinary clinics against susceptible microorganisms, but thyroid dysfunction has raised concern especially in dogs. This study aimed to determine the effects of prolonged treatment with co-trimoxazole in euthyroid dogs. Dogs were given co-trimoxazole at 30, 60, and 120 mg/kg body weight at 12-hour intervals for 21 days. Standard procedures were used to assay total T4 and T3, thyrotropin, testosterone, aspartate aminotransferase, alanine aminotransferase, and alkaline phosphatase in serum. The thyroid gland and testes were weighed. In addition, thyroid and liver were examined histologically. Epididymal sperm count was also performed. Co-trimoxazole caused dose-dependent depression of serum thyroxine levels with severe colloid depletion, intrafollicular hemorrhage, hyperplasia, and hypertrophy of the follicular cells. The liver showed vacuolar hepatopathy. Epididymal sperm count was low in the 120 mg/kg–treated group. The study revealed that thyroid hemorrhage and lowered epididymal sperm reserve were new findings in co-trimoxazole toxicity in dogs.
Introduction
For several decades, sulfonamides have been employed in the prevention and treatment of bacterial and protozoan infections in both humans and animals.1,2 Over time, pathogen resistance to sulfonamides has become widespread, and cross-resistance among the different sulfa drugs is considered complete.2,3 Moreover, toxicity/idiosyncrasy associated with sulfonamide treatment is of great concern in medical practice. This led to potentiation of sulfonamides. Co-trimoxazole is a product of potentiation of sulfamethoxazole with trimethoprim. Potentiation not only lowers the quantity of sulfonamides required to inhibit specific susceptible bacteria but also reduces toxicity and microbial resistance. 4 However, a number of reports in dogs have associated clinical signs of hypothyroidism with potentiated sulfonamides.5–7
Prolonged treatment with potentiated sulfonamides has been reported to suppress thyroid function by inhibiting thyroglobulin iodination and coupling of iodotyrosines.8,9 Prolonged administration may be required in certain disease processes such as in staphylococcal pyoderma, in which medication is supposed to be continued for a minimum of 4 to 6 weeks after clinical resolution of pyoderma. 10 In one study, prolonged treatment of dogs with potentiated sulfonamides at doses within the therapeutic dose range (15-30 mg/kg body weight) produced no alterations in serum triiodothyronine (T3) and thyroxine (T4). 11 In another study, trimethoprim alone did not depress thyroid function, suggesting that the sulfonamide component of the combination drug may be the one responsible for lowering the thyroid hormone levels. 6
The antithyroid activity of sulfonamides varies among species. 11 In dogs, potentiated sulfonamide-induced thyroid dysfunction was reported in Labrador and Golden Retrievers,6,7 as well as in mixed breeds. 12 The purpose of the work reported here was to determine the effects of prolonged treatment with co-trimoxazole on the thyroid gland, liver, and epididymal sperm reserve in euthyroid dogs. Assessment of liver function and sperm count may highlight possible direct toxic effects of co-trimoxazole on the liver and gonads, as well as the influence of thyroid dysfunction on hepatic and gonadal functions.
Materials and Methods
Animals
A total of 20 sexually mature male mongrel dogs were used for the study. They were acclimatized in metal cages for 3 weeks, during which period they were vaccinated against rabies and screened for both ectoparasite and endoparasite. They were randomly assigned to 4 groups (A-D) that comprised 5 dogs each. Food and clean drinking water were provided for the dogs at regular intervals. The baseline values for the thyroid hormones (T3 and T4), thyrotropin (TSH), as well as the weight and rectal temperature of the dogs in all the groups were determined before commencement of treatment. Co-trimoxazole was then administered orally to the various groups. Group A was the control and received equal volume of water. Groups B, C, and D received 30, 60, and 120 mg/kg body weight of co-trimoxazole in water, respectively. Treatment was for 21 days at 12-hour intervals.
Ethical approval
The study strictly adhered to the institutional guidelines on research involving animals. Approval was obtained from the Experimental Animal Ethics Committee of the Faculty of Veterinary Medicine, University of Nigeria, Nsukka PG/11/60133.
Sample collection
Blood samples were drawn from the cephalic vein of the dogs in all the groups on days 0, 7, 14, and 21 of treatment into clean glass test tubes and allowed to clot. The samples were centrifuged at 3000 rpm for 10 minutes to obtain clear serum samples which were then decanted into separate sample bottles. The serum samples were split into 2 aliquots for weekly assay of hormones—total T3 and T4, TSH, and testosterone—as well as serum enzymes—aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP). The serum samples were refrigerated until use, and all the analyses were completed within 48 hours of sample collection.
Hormonal assay
Microplate enzyme immunoassay kits (AccuBind; Monobind Inc., Lake Forest, CA, USA) were used for weekly determination of the serum levels of total T3 and T4, TSH, and testosterone for each dog. 13
Serum enzyme assay
Serum AST and ALT levels were determined weekly by colorimetric method using AST and ALT test kits (Química Clínica Aplicada S.A. [QCA], Tarragona, Spain). 14
Serum ALP level was assayed weekly by phenolphthalein monophosphate method, using ALP test kits (QCA). 15
Morphometric studies
The dogs were weighed weekly using a standard weighing balance. The weights were recorded in kilogram. At the end of the 21-day treatment period, 3 dogs in each group were humanely sacrificed by intravenous injection of pentobarbitone solution at the dose of 150 mg/kg. Postmortem examination was performed immediately after euthanasia. Thyroid glands and testes were dissected and weighed. The mean weight of the testes as well as that of the thyroid glands was obtained using a sensitive Mettler weighing balance and recorded in gram. The organosomatic indices of the thyroid and testes were calculated.
Epididymal sperm count
The combined left and right portions of the epididymis (caput, corpus, and cauda) were minced with ophthalmologic scissors and homogenized for 1 minute in 20 mL of physiological saline. The homogenate was filtered using nylon mesh sieve, and 20-µL aliquot of the filtrate was used to charge the improved Neubauer chamber for the appropriate counting of the number of sperm/milliliter of tissue sample.16–18
Histopathotology
Tissue sections were taken from both lobes of the thyroid gland as well as from the liver. The tissues were fixed in 10% neutral buffered formalin for 48 hours, after which they were routinely processed, sectioned with microtome, and stained using hematoxylin-eosin. The tissues were then observed for pathological changes under the light microscope and pictures were taken.
Statistics
Data generated from the study were subjected to 1-way analysis of variance using SPSS 17.0. Variant means were separated using the least significant difference method. Significance was accepted at probability P < .05. The quantitative data were presented as means for each group with standard error.
Results
Effects of co-trimoxazole on the thyroid gland of the dogs
There were no significant variations (P > .05) in the serum T3 levels of all the groups on days 0, 7, and 14, but on day 21, the serum T3 level of group C dogs was significantly (P < .05) lower than that of the group A (Figure 1).

Graph showing serum triiodothyronine T3 (ng/dL) of dogs treated with varied oral doses of co-trimoxazole for 21 days.
Serum thyroxine level in all the treated groups was significantly (P < .05) lower than that of the control from day 7 of the experiment to the end of the experiment (Figure 2).

Graph showing serum thyroxine levels T4 (µg/dL) of dogs treated with varied oral doses of co-trimoxazole for 21 days.
The variations in serum TSH level between the groups were not significant (P > .05) on days 0, 7, and 14, but on day 21, the serum TSH level of the group D dogs was significantly (P < .05) higher than that of the control group (Figure 3). In all the treated groups, however, there was dose-dependent increase in serum TSH level on day 21.
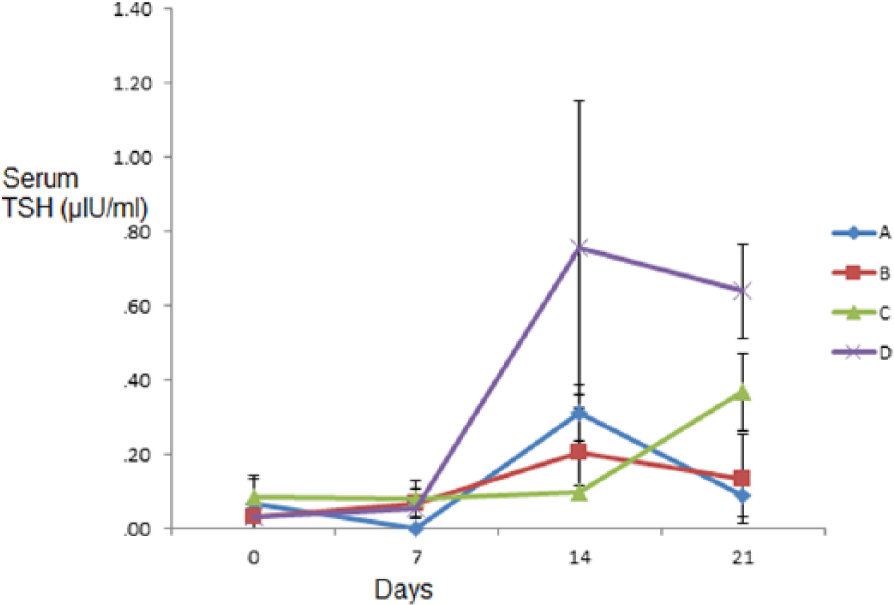
Graph showing serum thyrotropin (TSH) (µIU/mL) of dogs treated with varied oral doses of co-trimoxazole for 21 days.
The relative thyroid weight of all the treated groups were significantly (P < .05) higher than that of the control (Figure 4).

Bar chart showing relative thyroid weight (g) of dogs given varied oral doses of co-trimoxazole for 21 days.
Effects of co-trimoxazole on the serum enzymes of the dogs
Treatment with co-trimoxazole at varied doses as used in this study did not produce any significant variations (P > .05) in the serum AST activity in any of the groups. The mean serum ALT activity was significantly (P < .05) higher in group D when compared with groups A and C on day 14. This higher serum ALT activity in group D was sustained up to day 21, on which it was significantly (P < .05) higher than that of groups A and B (Figure 5).

Graph showing serum alanine aminotransferase (ALT) (IU/L) of dogs treated with varied oral doses of co-trimoxazole for 21 days.
There were no significant variations (P > .05) between the groups in their serum ALP activity on days 0 and 7 of the experiment. On day 14 of treatment with co-trimoxazole, group D had a significantly (P < .05) higher serum ALP activity when compared with all other groups, but on day 21 of the experiment, the serum ALP activity of groups C and D was significantly (P < .05) higher than those of groups A and B (Figure 6).

Graph showing serum alkaline phosphatase (ALP) (IU/L) of dogs treated with varied oral doses of co-trimoxazole for 21 days.
Effect of co-trimoxazole on the testes of the dogs
There were no significant (P > .05) variations in the relative testicular weight of all the groups, although the untreated group had the highest value (Figure 7).

Bar chart showing relative testicular weight (g) of dogs given varied oral doses of co-trimoxazole for 21 days.
There were no significant variations in serum testosterone levels in all the groups on days 0, 7, and 21 of the experiment, but on day 14 of the experiment, the serum testosterone levels of group D dogs were significantly higher (P < .05) than that of the other groups (A, B, and C) (Figure 8).
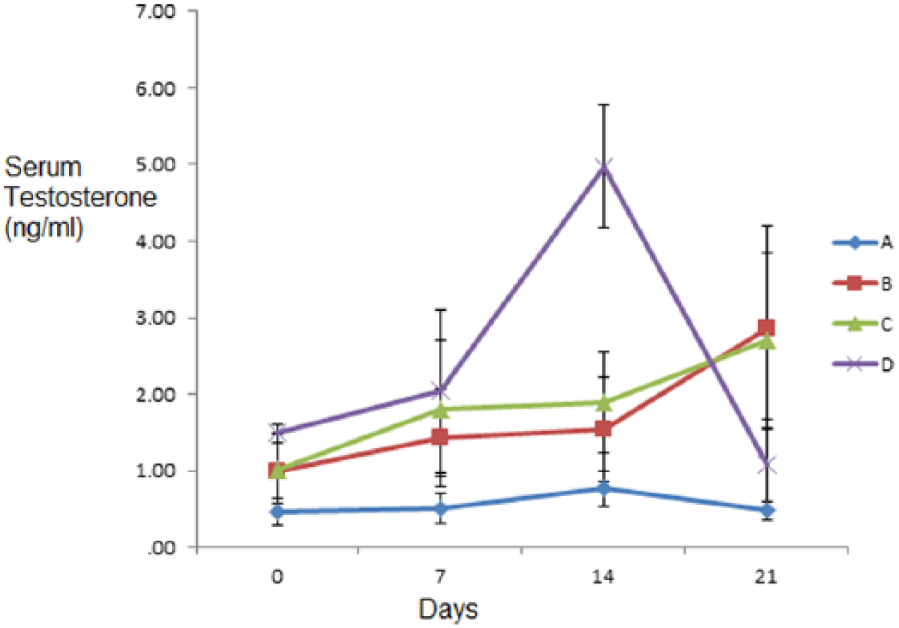
Graph showing serum testosterone (ng/mL) of dogs treated with varied oral doses of co-trimoxazole for 21 days.
The values obtained for the epididymal sperm count were the highest in group B dogs which were significantly (P < .05) higher than the values obtained for all other groups. The epididymal sperm count of the group D was the lowest and significantly (P < .05) lower than that of all other groups (Figure 9).
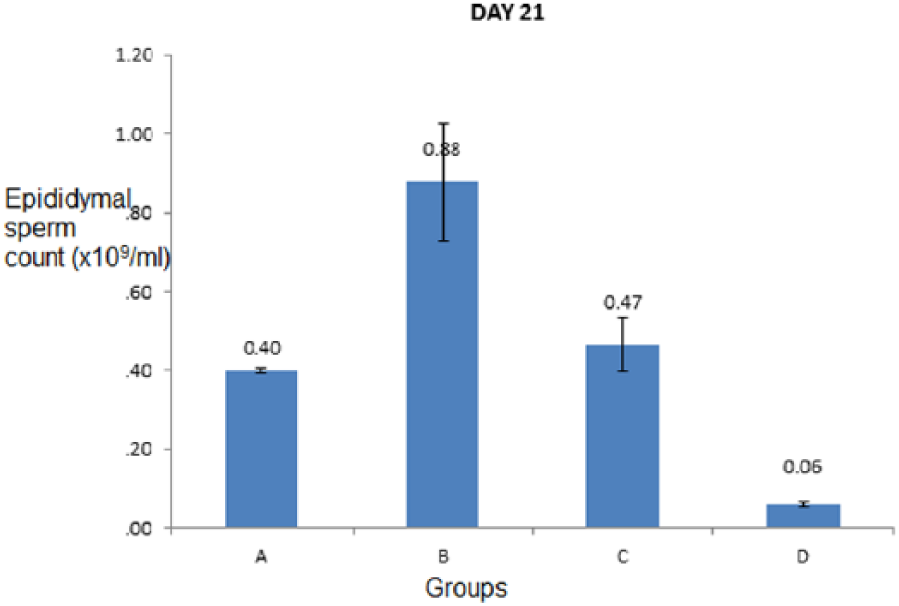
Bar chart showing epididymal sperm count (×109/mL) of dogs treated with varied oral doses of co-trimoxazole for 21 days.
Gross lesions
All the animals in group D had ulcerations of the skin of the underbelly that also affected the scrotal skin (Figure 10), the canthus (Figure 11), and also symmetrical alopecia that affected the caudal half of the body down to the root of the tail (Figure 12), from days 18 to 21. The thyroid glands of group D dogs showed petechial and ecchymotic hemorrhages (Figure 13). Gross lesions in the liver consisted of multifocal, randomly distributed, confluent, 1- to 5-mm diameter areas of pallor (degeneration and necrosis), especially in group D (Figure 14).

Photograph of dog in group D, treated with co-trimoxazole, 120 mg/kg body weight, orally, showing multifocal ulcerative lesions (arrows), affecting also the scrotal skin from day 18 of treatment.
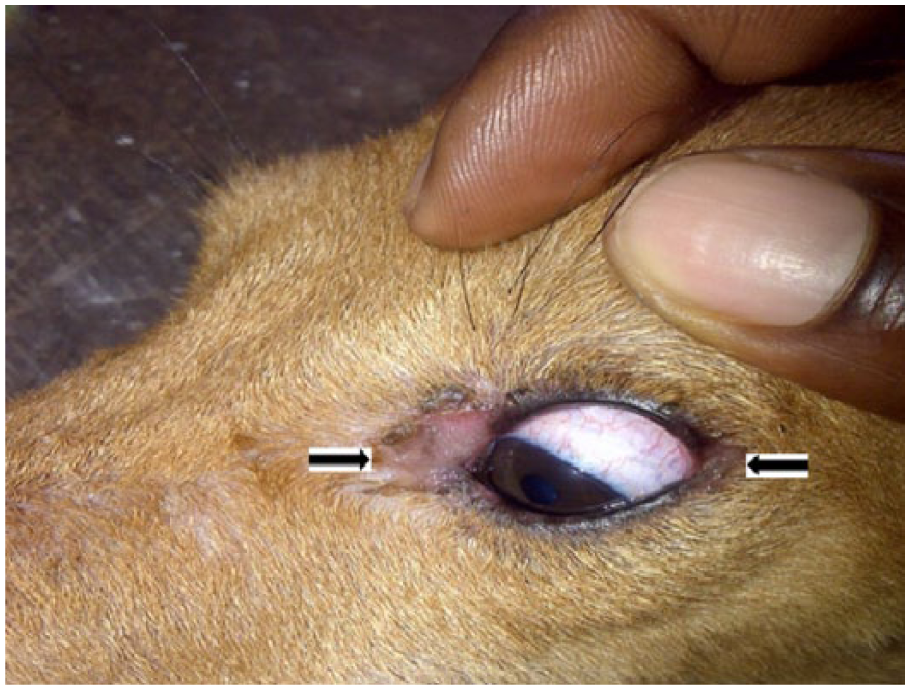
Photograph of dog in group D showing ulcerations that affected both corners of the left eye (arrows).

Picture of dog in group D showing symmetrical alopecia on the caudal half of the body down to the base of the tail (arrows).
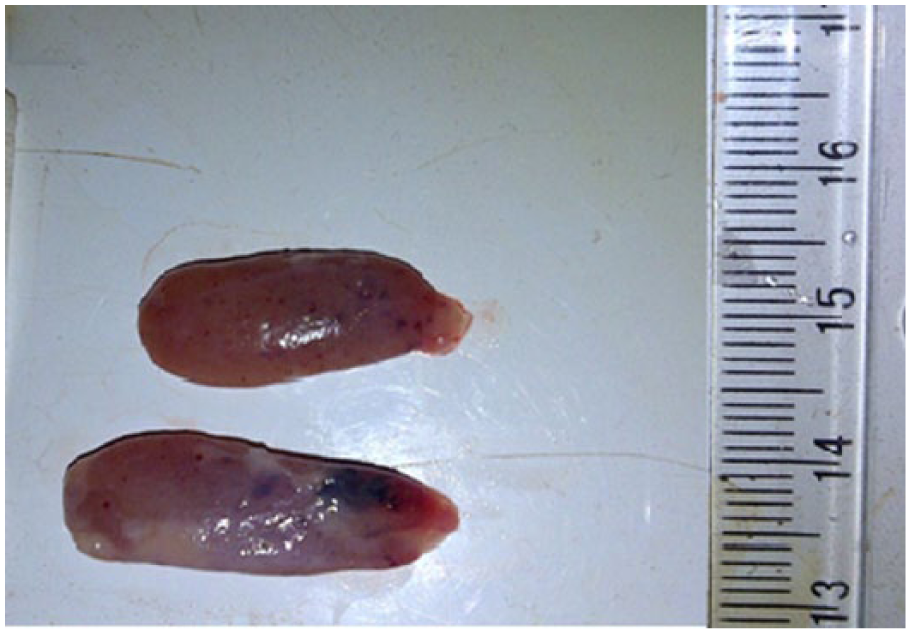
Gross picture of the thyroid gland of one of the dogs treated with co-trimoxazole, 120 mg/kg body weight for 21 days showing severe petechial and ecchymotic hemorrhages in both lobes of the thyroid gland.

Gross picture of the liver (L) of dog treated with 120 mg/kg body weight of co-trimoxazole for 21 days showing multifocal, depressed, and pale areas of hepatocytes necrosis.
Histopathology
Group A showed normal thyroid follicles (Figure 15), but the treated groups showed thyroid follicles that were devoid of colloid in their lumen (Figure 16). In addition, group D showed follicular cell hypertrophy, thickened follicular epithelium, and severe hemorrhage into the follicular lumen (Figure 17).

Normal thyroid follicles (from the untreated group) densely packed with colloid (C) in their lumen. The follicular cells are simple cuboidal in shape (arrows) with large oval-shaped nuclei (hematoxylin-eosin, original magnification ×400).

Thyroid tissue of dogs treated with 30 mg/kg body weight of co-trimoxazole, orally, for 21 days showing thyroid follicles devoid of colloid in their lumen (*) (hematoxylin-eosin, original magnification ×400).
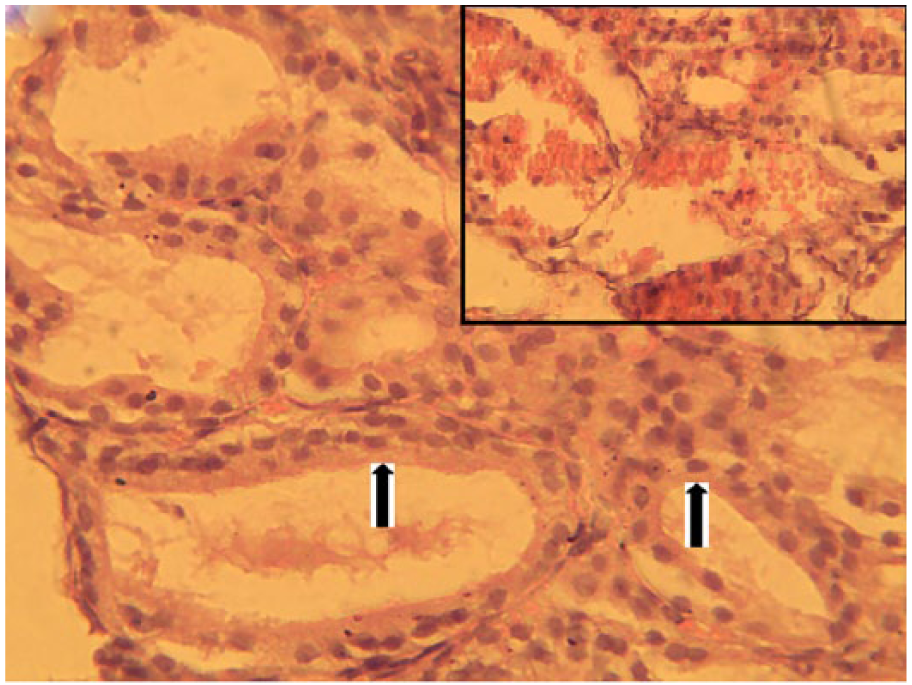
Thyroid tissue of dogs treated with 120 mg/kg body weight of co-trimoxazole showing follicular cells hypertrophy (arrows) and thickening of the follicular epithelium. The follicular lumens are devoid of colloid. Note severe intrafollicular hemorrhage (inset) (hematoxylin-eosin, original magnification ×400).
Lesions in the liver included widespread vacuolar degeneration of hepatocytes, congestion of portal veins (Figure 18), and severe random hepatocellular necrosis and liquefaction with mild to moderate mononuclear cellular infiltration associated with the necrotized hepatocytes (Figure 19).
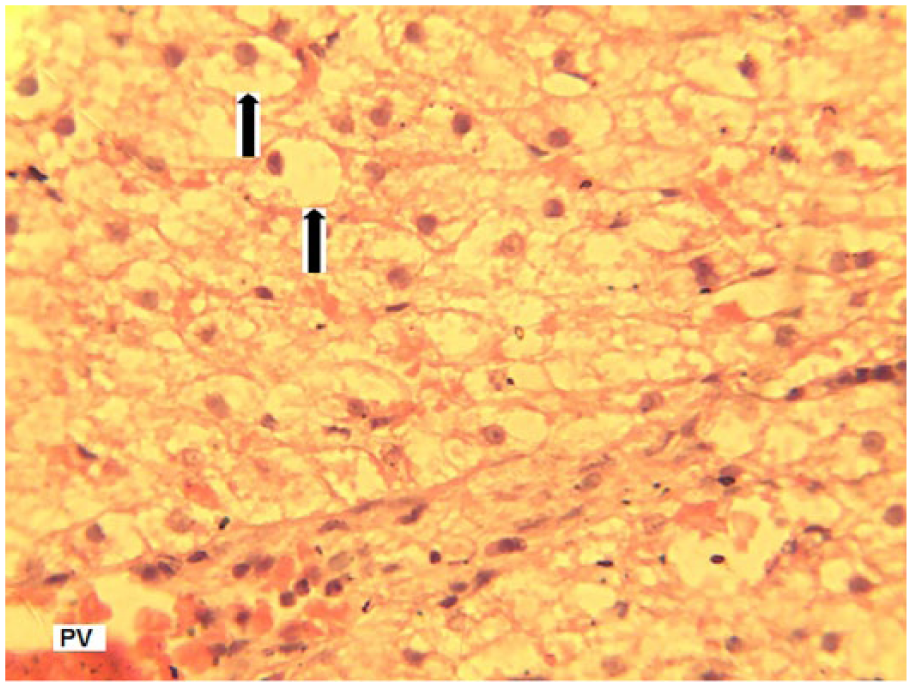
Liver section of dogs treated with co-trimoxazole 60 mg/kg body weight for 21 days showing severe, widespread vacuolar degeneration of hepatocytes (arrows) (hematoxylin-eosin, original magnification ×400).

Liver section of dogs treated with co-trimoxazole 120 mg/kg body weight for 21 days showing severe, random hepatocellular necrosis with liquefaction and infiltration of mononuclear inflammatory cells associated with the necrotized hepatocytes (arrows) (hematoxylin-eosin, original magnification ×100).
Discussion
Co-trimoxazole, as used in this study, caused thyroid dysfunction and led to severe thyroid lesions including hemorrhage. The relative thyroid weights of the treated groups were significantly higher than that of the control group. This may be as a result of thyroid enlargement due to hypertrophy of the follicular cells caused by the sulfonamide treatment. 19 Trimethoprim potentiates the goitrogenic action of sulfonamides. 20
Previous studies have shown that co-trimoxazole decreases serum T4, whereas T3 remains unaffected. 5 This study presented a similar picture except that group C had a significantly lower value for T3 on day 21. Significantly lower T4 values suggested that long-term sulfonamide treatment suppressed thyroid function most likely by impairing thyroglobulin iodination and coupling of iodotyrosines.8,9 Significantly higher values for serum TSH as seen on day 21 in group D further buttressed the existence of a hypothyroid state in the treated groups. Serum TSH increased through a negative feedback mechanism.
Follicular cell hyperplasia and hypertrophy and colloid depletion may be the result of increased and persistent stimulation of the follicles by TSH. The rate of colloid endocytosis by follicular cells exceeds the rate at which they are synthesized. These findings compare with iodine deficiency goiter. 21 Goitrous hypothyroidism was reported with 5 weeks of trimethoprim-sulfamethoxazole therapy in a young dog. 22 Several authors have maintained that sulfonamides are goitrogenous, depressing serum thyroid hormones and elevating serum TSH leading to follicular hypertrophy and hyperplasia.11,21,23,24 The present findings, however, differed from previous studies in the occurrence of severe thyroid hemorrhage seen both grossly and microscopically, especially in group D. The cause of the hemorrhage is not clear. However, depletion of colloid in the follicular lumen and in the cytoplasm of individual follicular cells may have increased diapedesis of red blood cells from the possibly hyperemic interfollicular capillaries into the follicular lumen. Thyroid hyperemia was one of the earliest lesions reportedly caused using sulfonamides. 25
It has been reported that sulfonamide treatment may lead to increased serum values of AST, ALT, and ALP. 26 In this study, AST values did not vary significantly across the groups. Alanine aminotransferase values were significantly higher on days 14 and 21 of treatment in group D alone. However, ALP values were significantly higher on days 14 and 21 in group D and day 21 in group C. It was observed that the elevation of serum ALT and ALP values were dose and time dependent. It is possible that at the doses given to groups C and D, the drug caused necrosis of hepatocytes and release of these serum enzymes into the circulation. Occurrence of liver necrosis in this study is consistent with the findings of other authors.27–29 The present findings suggest that the increased ALT and ALP activities were as a result of drug-induced hepatotoxicity as reported with tetracycline, 30 carprofen, 31 and trimethoprim/sulfonamides. 32 Moreover, focal or diffuse intrahepative cholestasis may have occurred following increase in membrane cholesterol-phospholipid ratio and diminished membrane fluidity which may have affected bile movement out of hepatocytes. 33 The distention of hepatocytes by fatty acids may have further occluded bile canals leading to intrahepatic cholestasis and high ALP activity. Steroid (vacuolar) hepatopathy has been reported in a dog with clinical hypothyroidism associated with co-trimoxazole administration. 12 Furthermore, bile is toxic; irritation caused by its accumulation may have contributed to the high ALT activity and mild inflammatory reaction in the liver.
Group D had significantly higher value for serum testosterone on day 14. However, this was quickly followed by a sharp drop by day 21. Recall that group D also had a significant higher value for serum TSH on day 21. It is possible that the increased need for TSH caused hyperplasia of the anterior pituitary leading to the release of luteinizing hormone (LH) alongside TSH. Luteinizing hormone is required for the production of testosterone by the Leydig cells. 34 Variation in the hypothalamus-pituitary-thyroid axis seems to affect the hypothalamus-pituitary-gonadal axis. 34 Although some authors recorded a decreased value for serum gonadotropins in hypothyroidism and an increased value in thyrotoxicosis, 35 the actual cause and duration of hypothyroidism should be considered as some other authors have linked decreased serum gonadotropins to prolonged hypothyroidism.36,37 However, the cause of hypothyroidism is important because if it is such that does not affect serum T3, then elevation of serum testosterone can be explained. This is because T3 has been reported to increase LH receptors and steroidogenesis of the Leydig cells. 38 Although our study did not assay for serum LH level, elevated testosterone level can be understood from the angle of T3 not being depressed by co-trimoxazole.
The sharp drop in serum testosterone in group D by day 21 may be due to the scrotal lesions presented in Figure 10. Significantly higher values for epididymal sperm count in group B may indicate the dose at which co-trimoxazole has no toxic effects on the gonads. However, group D had the lowest epididymal sperm count despite high testosterone level. This may also be due to a possible elevation of the scrotal temperature associated with the scrotal lesions recorded in group D.
Conclusions
Lesions caused by subacute co-trimoxazole toxicity in dogs used for this experiment were mostly dose and time dependent. However, some adverse reactions were intricately associated with sulfonamide use irrespective of dose. The study reports thyroid hemorrhage and lowered epididymal sperm reserve, potentially as a result of scrotal skin lesions, as new findings in co-trimoxazole toxicity in dogs.
Footnotes
Acknowledgements
The authors would like to thank Mr Obioma Egonu of the Histopathology Laboratory, University of Nigeria, Nsukka, for his technical assistance in tissue processing.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
DCA and VSOS conceived and designed the experiments. DCA and JII analyzed the data and wrote the first draft of the manuscript. DCA, VSOS, and JII contributed to the writing of the manuscript; agree with manuscript results and conclusions; jointly developed the structure and arguments for the paper; made critical revisions and approved final version; and reviewed and approved the final manuscript.
