Abstract
Background:
Vietnam is an endemic region for nasopharyngeal cancer (NPC), with approximately 70% of cases diagnosed at stage III-IVa and 90% presenting with undifferentiated histology closely related to Epstein-Barr virus (EBV) infection. However, no studies have identified biomarkers predictive of recurrence and metastasis. This study aimed to determine the cut-off concentrations of cell-free (cf) EBV DNA for predicting recurrence and metastasis in NPC.
Methods:
A longitudinal descriptive study was conducted on 58 patients with stage III-IVa undifferentiated NPC between August 2021 and August 2024. We analysed the predictive value of cf EBV DNA concentrations at pre-treatment (preEBV) and post-treatment (postEBV, 6 months postEBV [EBV6], 12 months postEBV [EBV12]) in relation to disease progression and survival outcomes.
Results:
Of the 58 patients, 23 experienced recurrence and/or metastasis. The recurrence prediction cut-off concentration was 3980.0 copies/mL for preEBV (area under the curve [AUC] = 0.705, 95% confidence interval [CI]: 0.499-0.911, Se = 80.0%, Sp = 64.6%, odds ratio [OR] = 7.294, 95% CI: 1.389-38.307, accuracy [ACC] = 67.2%); 12.5 copies/mL for EBV6 (AUC = 0.783, 95% CI: 0.600-0.966, Se = 77.8%, Sp = 75.0%, OR = 10.5, 95% CI: 1.839-54.242, ACC = 75.5%); and 5.5 copies/mL for EBV12 (AUC = 0.766, 95% CI: 0.577-0.954, Se = 83.3%, Sp = 78.0%, OR = 17.778, 95% CI: 1.835-172.219, ACC = 78.7%). The metastasis prediction cut-off concentration for EBV12 was 42.5 copies/mL (AUC = 0.748, 95% CI: 0.502-0.994, Se = 66.7%, Sp = 80.5%, OR = 8.250, 95% CI: 1.278-52.254, ACC = 78.7%). Cut-off concentrations of preEBV, EBV6, and EBV12 were independent predictors of survival outcomes (except for EBV6). All results were statistically significant (P < .05).
Conclusions:
In advanced-stage NPC-undifferentiated subtype, preEBV, EBV6, and EBV12 serve as predictive biomarkers for recurrence and metastasis. Among these, EBV12 demonstrated the highest predictive value.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is among the most common malignant tumours of the head and neck, originating from the nasopharyngeal epithelia, and is particularly prevalent in Southeast Asia.1-3 According to the International Agency for Research on Cancer, approximately 120 434 new cases of NPC were reported worldwide in 2022, with Vietnam accounting for approximately 3.1%.4,5 Advances in radio-chemotherapy and imaging have substantially improved treatment outcomes and control rates in early-stage disease (stages I and II).6,7 However, recurrence and metastasis remain common in advanced stages (III and IV), accounting for 30% to 40% treatment failures and cancer-related deaths.3,8 In Vietnam, most patients with NPC were diagnosed at advanced stages.9,10 Metastasis is frequent owing to the network of lymphatic capillaries in the head and neck region. 11 Recurrence and metastasis of advanced-stage undifferentiated NPC mainly occur within the first 2 years after chemotherapy and radiotherapy. 5
Metastasis and recurrence refer to the occurrence of local or regional relapse or newly detected lesions, confirmed either by nasopharyngeal biopsy or 2 imaging diagnostics, following a complete response to initial treatment and occurring >3 months after treatment completion. 8 In patients with recurrence/metastasis of NPC, the prognosis is very poor, and treatment options are limited. 12 Recurrence and metastasis represent complex and challenging clinical scenarios, posing substantial hurdles in post-treatment surveillance, diagnosis, and choice of secondary treatment.12,13 Early diagnosis of recurrence/metastasis is essential to improve prognosis and treatment effectiveness.7,14 Management of these cases should be conducted at specialised oncology centres with expertise in multidisciplinary care.8,12 Treatment options, including surgery, re-irradiation, re-chemotherapy, and immunotherapy, can improve overall survival (OS) outcomes.12,13 Therefore, identifying predictive markers for recurrence/metastasis would considerably improve outcomes and prognosis, particularly for patients with advanced-stage disease.3,11,15,16
Undifferentiated carcinoma of the nasopharynx, or non-keratinising squamous cell carcinoma, accounts for approximately 95% of NPC cases and is strongly associated with Epstein-Barr virus (EBV) infection.1,8,15,17 The concentration of plasma cell-free (cf) EBV DNA correlates with tumour burden and is considered a highly sensitive and specific tumour marker for monitoring patients with NPC after radiotherapy. 15 ,17-19 Studies have reported that in the advanced stages, cf EBV DNA concentration has a significant prognostic impact on patients with NPC, especially after treatment. 11 Most reports have proposed significant cut-off values for cf EBV DNA, with pre-treatment concentrations commonly reported at 1500 copies/mL or 4000 copies/mL and post-treatment levels at 0 copy/mL.11,15 However, other post-treatment time points have not been systematically evaluated. In Vietnam, previous studies only described the pre-treatment concentrations of cf EBV DNA and their role in NPC pathogenesis, without examining their association with treatment response or recurrence/metastasis.20,21 In addition, studies assessing EBV DNA levels during chemoradiotherapy and follow-up – which may offer more accurate prognostication of local recurrence and/or metastasis (lr&/m) in undifferentiated subtypes of NPC – remain limited. This study aimed to evaluate the association between cf EBV DNA concentrations at 4 time points during chemoradiotherapy and follow-up in patients with advanced-stage NPC, and their relationship with recurrence, metastasis, and other clinical outcomes.
Materials and Methods
Study design
The study was designed as a longitudinal, descriptive investigation. Based on existing evidence, the incidence of lr&/m in patients with advanced-stage NPC (III, IVa) was estimated at 30% to 45%.15,22,23 To calculate the required sample size, we applied the standard formula for estimating a population proportion:
Patients
This study included 58 patients with advanced-stage NPC (stage III or IVa) treated at the Vietnam National Cancer Hospital, Tan Trieu branch, between August 2021 and August 2024. This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (see Supplementary File for the completed STROBE checklist). Inclusion criteria were as follows: (1) histologically confirmed World Health Organization 4th edition non-keratinising squamous cell carcinoma, undifferentiated subtype 1 ; (2) clinical stage III or IVa based on the 8th edition International Cancer Control/American Joint Committee on Cancer (AJCC) staging system1,24; (3) treatment with CCRT, with or without induction chemotherapy (IC); (4) complete response after radiotherapy, as assessed the Response Evaluation Criteria in Solid Tumours (RECIST), version 1.1 (see Supplementary file), and no recurrence/metastasis within 3 months after postradiotherapy 25 ; (5) availability of EBV DNA concentration measurements at 4 distinct time points: pre-treatment (preEBV), postradiotherapy 1 month (postEBV, corresponding to treatment response evaluation), and at 6 months (EBV6) and 12 months (EBV12) after treatment response evaluation, or until recurrence and/or metastasis was diagnosed; (6) adequate clinical information documented in the medical records. Exclusion criteria: (1) No follow-up records.
The outcomes of this study included patient demographics and clinical characteristics, specifically, age, sex, smoking status, disease stage, treatment method, disease-free survival (DFS), OS, recurrence, and metastasis. Staging was based on the 2018 AJCC classification. 24 Disease-free survival was calculated from the date of treatment commencement to when recurrence or distant failure was first recorded, or, in cases of local and/or regional persistent disease, to the first follow-up visit. Overall survival was measured from the date of treatment initiation to the last follow-up or death from any cause. Both DFS and OS were calculated using the method of Kaplan-Meier. 26
Treatment
The treatment plan followed the guidelines of the National Comprehensive Cancer Network (NCCN) guidelines, Version 3, 2021. 27 All patients underwent CCRT, with or without IC. Radiotherapy was delivered in daily fractions of 1.80 to 2.14 Gy, 5 days/week, to a total dose of 59.4 to 72 Gy to the primary tumour, 54 to 65 Gy to the involved areas of the neck, and 50 Gy to uninvolved areas. Induction chemotherapy consisted of cisplatin or cisplatin with docetaxel, 5-FU, administered every 3 weeks for 2 or 3 cycles. Concurrent chemoradiotherapy involved either cisplatin or carboplatin, administered triweekly (on days 1, 22, and 43) or weekly until the end of radiotherapy. Treatment response was evaluated 1 month following radiotherapy, based on the RECIST, version 1.1. 25
Quantification of plasma EBV DNA
Plasma EBV DNA was measured using real-time quantitative polymerase chain reaction (PCR), following the previous published protocol. 20 Briefly, primers targeting the BamHI-W region were designed and validated using Oligo Primer Analysis Software (Molecular Biology Insights), in which the forward and reverse primer sequences were 5′-CCAGACGAGTCCGTAGAAGG-3′ and 5′-AGCCTAATCCCACCCAGACT-3′, respectively. No cross-reactivity with other viruses or genomic DNA was detected, as confirmed using the BLAST sequence. Plasma EBV DNA was analysed using a real-time PCR system (Rotor-Gene Q), and concentrations were expressed as EBV genome copies per mL of plasma. Plasma cf EBV DNA concentrations were recorded at 4 time points: baseline (preEBV), 1 month post-CCRT (postEBV), 6 month post-CCRT (EBV6), and 12 month post-CCRT (EBV12). All blood tests were performed at the Department of Genomics and Cytogenetics, Institute of Biomedicine and Pharmacy, Vietnam Military Medical University, Hanoi, Vietnam. Cut-off concentrations for each time point were determined using receiver operating characteristic (ROC) curve analyses. The thresholds used to distinguish between low and high EBV DNA were preEBV (3980 copies/mL), EBV6 (12.5 copies/mL), and EBV12 (5.5 and 42.5 copies/mL).
Follow-up and evaluation of recurrence/metastasis
The primary endpoints of this study were the rate of metastasis or recurrence, along with the cf EBV DNA concentrations before and after treatment. All patients underwent routine follow-up after therapy, including clinical assessments and cf EBV DNA testing every 6 months for up to 12 months post-treatment. Follow-up evaluations included physical examination of the head and neck, nasopharyngeal endoscopy, chest radiography, abdominal ultrasound, EBV DNA serological testing, magnetic resonance imaging (MRI) of the nasopharynx and neck, and whole-body positron emission tomography/computed tomography (PET/CT). Additional evaluations, such as MRI, nasopharyngeal/lymph node biopsy, lymph node needle aspiration, and PET/CT, were performed when recurrence or metastasis could not be diagnosed using previous methods. Recurrence and metastasis were confirmed by fine-needle aspiration and pathological examination of biopsy specimens, or diagnosed based on at least 2 imaging diagnoses. 27
Ethical statement
All participants provided written informed consent and agreed to participate in the study. The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of the 103 Military Hospital, Vietnam Military Medical University (Approval No. 1811/CNChT-HĐĐĐ, dated July 23, 2021).
Statistical analysis
Statistical analyses were performed using IBM SPSS version 22.0 (IBM; Armonk, New York). All statistical tests were 2-sided, and a P value of <.05 was considered statistically significant. Receiver operating characteristic curve analysis and the Youden index were used to calculate the optimal cutoff value of cf EBV DNA concentrations at each time point to predict recurrence/metastasis, selecting thresholds that offered the best balance between sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), accuracy, likelihood ratio (LLR), and odds ratio (OR). Survival rates were calculated using the Kaplan-Meier method. The log-rank test (in univariate analysis) and Cox regression model (in multivariate analysis) were used to examine prognostic factors affecting DFS and OS, with results reported at a 95% confidence interval.
Results
The characteristics of the patients with NPC are summarised in Table 1. The study included 38 male and 20 female patients, with no statistically significant difference in outcomes between sexes (P > .05). Among the participants, 38 patients (65.5%) had stage III disease and 20 (34.5%) had stage IVa disease (34.5%), with T4 and N2 being the most common. The mean gross tumour volume (GTV) was 17.444 cm3, and the highest percentage of patients had tumours <20 cm3. In total, 96.6% of patients underwent IC, among whom 67.3% were treated with Gem + Cis, and 22.4% with 5FU/docetaxel/cisplatin. Concurrent chemoradiotherapy included 70.7% of patients treated with intensity-modulated radiotherapy (IMRT), and 96.6% received Cis. The follow-up time was 18 to 26 months. Disease-free survival was >12 months in 77.5% of cases. During the follow-up, 23 patients (39.7%) experienced disease progression, including 10 cases of recurrence (17.2%), 15 cases of metastasis (25.9%), and 2 cases of both (3.4%). The mean pre-treatment cf EBV DNA concentration was 50 847.71 copies/mL (range: 0-1 813 187.00). The concentration at postEBV was 304.59 copies/mL (range: 0-4656.00). At EBV6 and EBV12, the mean concentrations were 634.81 copies/mL range (0-4656.00) and 250.29 copies/mL range (0-3467.00), respectively (Table 1).
Patient characteristics.

Abbreviations: CF, docetaxel/cisplatin; Cis, cisplatin; DFS, disease-free survival; GTV, gross tumour volume; IMRT, intensity-modulated radiation therapy; TCF, 5FU/docetaxel/cisplatin; VMRT, volume of rotating intensity modulated; 3D, conformal radiotherapy.
Male.
Female.
p (1,2) value between male and female.
Using ROC analyses and the Youden index, we determined the optimal cf EBV DNA cutoff concentrations for predicting recurrence at different time points (Figure 1): 3980 copies/mL at baseline (preEBV; Figure 1A), 271.5 copies/mL at postEBV (Figure 1C), 12.5 copies/mL at EBV6 (Figure 1B), and 5.5 copies/mL at EBV12 (Figure 1D). The predictive performances of preEBV, EBV6, and EBV12 were acceptable, 0.7 < area under the curve (AUC) < 0.8, P < .05 (Figure 1). The sensitivity and specificity at pre-treatment were 80.0% and 64.6% (Figure 1A), respectively (P < .05), and those at EBV6 and EBV12 were 77.8% and 75.0% (Figure 1B) and 83.3% and 78.0% (Figure 1D), respectively (P < .05). However, the postEBV concentration showed no predictive value (AUC < 0.6) and was not statistically significant (P > .05), with sensitivity and specificity of 30.0% and 87.5%, respectively (Figure 1C).

ROC analyses of cf EBV DNA concentrations for detecting local recurrence at different time points. (A) ROC analysis of preEBV concentration for detecting local recurrence. (B) ROC analysis of EBV6 concentration for detecting local recurrence. (C) ROC analysis of postEBV concentration for detecting local recurrence. (D) ROC analysis of EBV12 concentration for detecting local recurrence.
The cutoff values of cf EBV DNA concentration were used to analyse recurrence in patients. Plasma cf EBV DNA concentrations exceeding the thresholds for preEBV, EBV6, and EBV12 were significantly associated with recurrence, occurring in 80.0%, 77.8%, and 83.3% of the patients with recurrence, respectively. These associations were statistically significant (P < .05, Fisher exact test), with diagnostic accuracies of 67.2%, 75.5%, and 78.7%, respectively. All cutoff concentrations corresponded to a quite low risk of having recurrence, 2 < LLR(+) ⩽ 5, and a low risk of non-recurrence, 0.5 > LLR(−) ⩾ 0.2, except for postEBV, 1 > LLR(−) ⩾ 0.5. Patients with pre-treatment cf EBV DNA concentration ⩾ cut-off had a 7.294-fold higher risk of relapse, and those exceeding the EBV6 and EBV12 thresholds had 10.5-fold and 17.778-fold higher risks, respectively (Table 2).
Pre-treatment and post-treatment cf EBV DNA concentrations in patients with locoregional recurrence of NPC.

Abbreviations: ACC, accuracy; LLR, likelihood ratio; PPV, positive predictive value; NPV, negative predictive value; OR, odds ratio; Se, sensitivity; Sp, specificity.
Using ROC analyses and the Youden index calculate the cutoff concentration of cf EBV DNA at other time points for predicting metastasis (Figure 2), we determined the concentrations for pre-treatment and post-treatment cf EBV DNA as 21 860.5 copies/mL (Figure 2A) and 271.5 copies/mL (Figure 2C), and for EBV6 and EBV12 as 1.5 copies/mL (Figure 2B) and 42.5 copies/mL (Figure 2D), respectively. The cutoff values for EBV6 and EBV12 concentrations were quite acceptable (0.6 < AUC < 0.7) (Figure 2B and D). The corresponding sensitivity and specificity were 70.0% and 69.8% for EBV6 (Figure 2B), and 66.7% and 80.5% for EBV12 (Figure 2D); however, the difference was not statistically significant (P > .05).

ROC analysis of cf EBV DNA concentrations for detecting metastasis at different time points. (A) ROC analysis of preEBV concentration for detecting metastasis. (B) ROC analysis of EBV6 concentration for detecting metastasis. (C) ROC analysis of postEBV concentration for detecting metastasis. (D) ROC analysis of EBV12 concentration for detecting metastasis.
Using previously determined cutoff values for cf EBV DNA concentration, we analysed their predictive value for metastasis across different patient subgroups. cf EBV DNA concentrations exceeding the EBV6 and EBV12 thresholds significantly predicted metastasis, occurring in 70.0% and 66.7% of patients with metastasis, respectively (P < .05). The diagnostic accuracies for EBV6 and EBV12 were 69.8% and 78.7%, respectively. The cutoff concentrations of EBV6 and EBV12 corresponded to a quite low risk of metastasis, 2 < LLR(+) ⩽ 5. In contrast, preEBV and postEBV showed lower predictive utility, 1 < LLR(+) ⩽ 2, suggesting weaker diagnostic performance. The cutoff concentrations of EBV6 and EBV12 were associated with a very low risk of not having metastasis, 0.5 > LLR(−) ⩾ 0.2, in contrast with preEBV and postEBV, 1 > LLR(−) ⩾ 0.5. Patients with EBV6 and EBV12 concentrations ⩾ cutoff had a 5.385-fold and 8.250-fold higher risk of metastasis, respectively (Table 3).
Pre-treatment and post-treatment cf EBV DNA concentrations in patients with metastatic NPC.
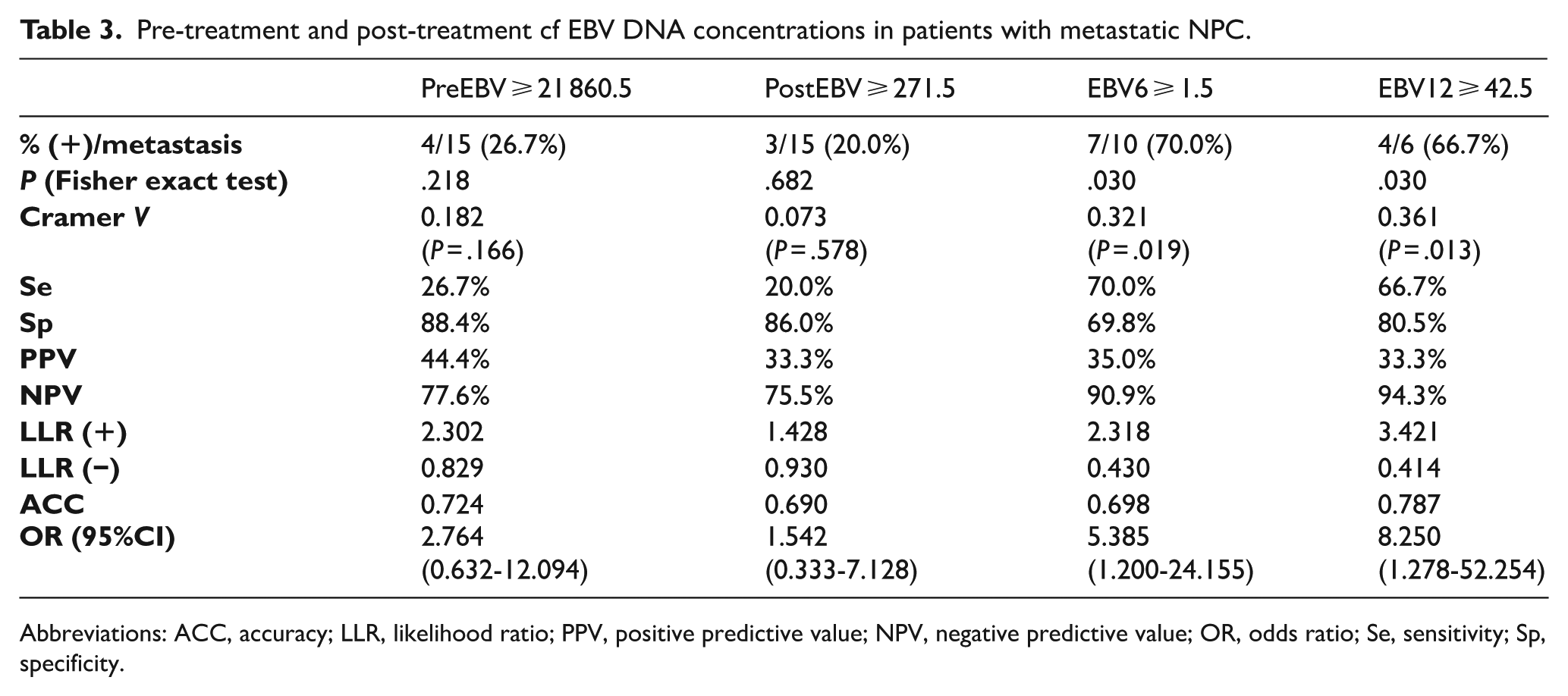
Abbreviations: ACC, accuracy; LLR, likelihood ratio; PPV, positive predictive value; NPV, negative predictive value; OR, odds ratio; Se, sensitivity; Sp, specificity.
The analysis evaluated the correlations between age, disease stage, GTV, and cf EBV DNA concentration with survival outcomes, specifically DFS and OS. Multivariate analysis identified the concentration patterns of preEBV, EBV6, and EBV12 levels as independent predictors of DFS (P < .05), with EBV12 at a cutoff of 5.5 copies/mL emerging as the strongest independent predictor of DFS (Table 4).
Univariate and multivariate analyses of clinical characteristics for disease-free survival (DFS) in the overall cohort.
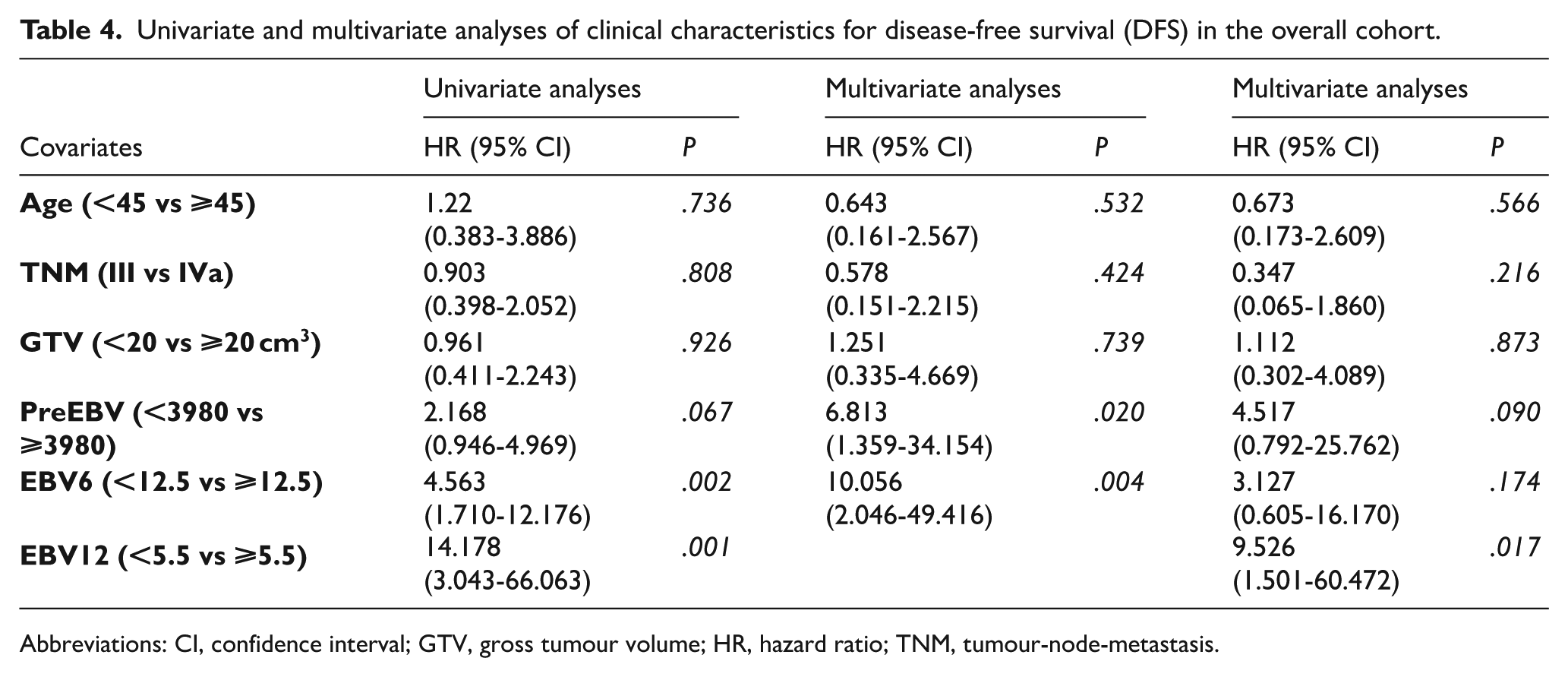
Abbreviations: CI, confidence interval; GTV, gross tumour volume; HR, hazard ratio; TNM, tumour-node-metastasis.
In addition, multivariate analysis identified cf EBV DNA concentrations at preEBV and EBV12 as independent predictors of OS (P < .05), with EBV12 at a cutoff of 5.5 copies/mL emerging as the strongest independent predictor of OS (Table 5).
Univariate and multivariate analyses of clinical characteristics for overall survival (OS) in the overall cohort.

Abbreviations: CI, confidence interval; GTV, gross tumour volume; HR, hazard ratio; TNM, tumour-node-metastasis.
We analysed the univariate association between cf EBV DNA levels at preEBV, EBV6, and EBV12 and survival outcomes in the validation set, using previously determined cut-off values (Figure 3): 3980 copies/mL at preEBV (Figure 3A and D), 12.5 copies/mL at EBV6 (Figure 3B and E), and 5.5 copy/mL at EBV12 (Figure 3C and F). Statistically significant differences in both DFS and OS were observed between the high and low cf EBV DNA groups at each time point (P < .05).

Effect of cf EBV DNA concentrations at different treatment time points on disease-free survival (DFS) and overall survival (OS). (A) Kaplan-Meier curve for DFS based on preEBV. (B) Kaplan-Meier curve for DFS based on EBV6. (C) Kaplan-Meier curve for DFS based on EBV12. (D) Kaplan-Meier curve for OS based on preEBV. (E) Kaplan-Meier curve for OS based on EBV6. (F) Kaplan-Meier curve for OS based on EBV12.
Discussion
This study aimed to evaluate the prognostic value of plasma cf EBV DNA concentrations in predicting recurrence and/or metastasis. We designed a longitudinal, descriptive investigation study using a comprehensive and independent sampling approach without interfering with the treatment processes of the patients. The primary variables included recurrence/metastasis status following confirmed complete treatment response and the predictive value of cf EBV DNA concentrations. In Vietnam, and specifically at the Vietnam National Cancer Hospital – Tan Trieu campus, NPC treatment currently utilises techniques such as volumetric-modulated arc therapy, IMRT, and 3-dimensional conformal radiotherapy. Chemotherapy regimens are implemented based on the NCCN guidelines but are influenced by factors such as drug procurement through bidding processes, regional variations, physician preferences, and patient conditions. Therefore, this study did not evaluate the efficacy of any specific treatment regimen. Instead, we focused on treatment adherence and the conditions under which patients were confirmed to have achieved a complete response, based on receipt of the full prescribed dose. Globally, studies on the role of plasma cf EBV DNA in NPC have assessed its significance independently of treatment modalities, emphasising compliance with radiation dosage and chemotherapy schedules.8,11,15,19,22 In addition, studies employing other approaches have identified prognostic factors for disease progression and post-treatment status;3,16,28 however, cf EBV DNA has been recognised as a superior marker and remains the most extensively studied.11,29,30 The ultimate goal of this study was to establish a clinically applicable threshold for plasma cf EBV DNA concentrations to optimise post-treatment monitoring in conjunction with existing diagnostic tools.
The cf EBV DNA concentrations correlate with tumour burden and are used in the follow-up of NPC.29,31 The association between pre-treatment cf EBV DNA concentrations and survival has been evaluated in several studies.15,32 This study evaluated the best prognostic value for recurrence and metastasis of plasma cf EBV DNA concentration at different time points before and after treatment. Cutoff values at time points: preEBV, postEBV, EBV6, and EBV12 were analysed for positive rate, sensitivity, specificity, NPV, accuracy, LLR, and OR.
The cutoff value of preEBV in predicting recurrence was 3980 copies/mL. This concentration was observed in 80.0% of patients who experienced recurrence and was statistically significant. The high sensitivity (80.0%) and relatively low specificity (64.6%) indicate a risk of false positives, indicating that some patients with no risk of relapse still had high levels before treatment. Therefore, preEBV level has limited potential for predicting relapse and should be combined with factors such as disease stage, age, and sex. On the other hand, an NPV of 93.9% indicated that most patients with EBV levels below the threshold did not experience relapse, supporting the utility of this threshold in excluding relapse. With an accuracy of 67.2%, the risk of relapse in patients with levels above the threshold was low but significant, requiring close monitoring. The risk of relapse was 7.294 times higher in this group. These findings confirm that pre-treatment EBV level is a good prognostic factor for relapse. In previous studies, most of the cutoff values of pre-treatment cf EBV DNA ranged from 1500 copies/mL11,30,33 to 4000 copies/mL.34-36 Our analysed identified threshold is consistent with those reports, particularly in its predictive significance for survival variables (DFS and OS). 35 However, this study only focused on patients with advanced-stage disease (III-IVa based on the AJCC 8th edition classification), which may increase the reliability of the identified cutoff values in predicting recurrence for this subgroup. We also specifically analysed the significance of the cutoff value in diagnosing recurrence, rather than limiting the analysis to survival prediction models. The cutoff postEBV concentration was not statistically significant in predicting metastasis (AUC = 0.497, P = .972 > .05). However, this value was only observed in 20.0% of patients with metastasis after radiotherapy. In the multivariate analysis of factors predicting DFS and OS, we analysed the cutoff concentration for preEBV, EBV6, and EBV12 alongside clinical variables, including age, disease stage, and GTV, demonstrating that preEBV, EBV6, and EBV12 were significant predictors.
The cutoff values of EBV DNA concentration after radiation therapy 1 month (postEBV) in this study were not statistically significant in predicting recurrence and metastasis. This may be because when evaluating the treatment response after 1 month of chemotherapy and radiotherapy, EBV DNA levels mostly decreased to 0 copies/mL, reducing the ability to distinguish between risk groups. Recurrence factors may be related to changes in EBV DNA concentrations at later time points. In most previous studies, the postEBV cutoff value was also 0 copy/mL. 37 Some studies found that the prognostic value of postEBV concentrations was higher than that of pre- and post-treatment concentrations. These authors suggested that re-detectable EBV after radiotherapy may indicate persistent disease, highlighting the need to consider testing for post-treatment EBV concentrations in patients. 36 In this study, we excluded patients who did not achieve a complete response after radiotherapy. All included patients were assessed as having a complete response in both the tumour and regional lymph nodes according to RECIST 1.1 criteria, with no evidence of new tumours or metastases for at least 3 months after completing chemotherapy and radiotherapy. Thus, we excluded patients with residual tumours, disease progression, or no response to treatment after chemotherapy and radiotherapy.
EBV6 and EBV12 concentrations demonstrated good predictive value for recurrence but are not statistically significant for predicting metastasis. The thresholds for predicting recurrence were 12.5 and 5.5 copies/mL, respectively (P < .05), whereas the thresholds for predicting metastasis were 1.5 and 42.5 copies/mL (P > .05). Among these, the EBV6 threshold was met in 77.8% of patients with recurrence, with a sensitivity of 77.8% and specificity of 75.0%. A balance between sensitivity and specificity allowed for a reasonably accurate distinction between patients with and without recurrence. The NPV of 94.3% shows that this test is very effective in excluding recurrence, as most patients below the threshold are not at risk of recurrence. An accuracy of 75.5% indicates that while the possibility of recurrence in the above-threshold group is low, close monitoring remains necessary. Notably, the risk of recurrence was 10.5 times higher in the above-threshold group than in the low-threshold group. Therefore, this is an important factor in predicting the prognosis of patients 6 months after treatment.
EBV12 showed the highest predictive value for recurrence, with a strong AUC, and low risk of recurrence among patients below the threshold. The sensitivity was 83.3% and specificity was 78.0%, indicating that the high sensitivity ensures the detection of most recurrences, whereas the specificity helps to minimise false positives. The NPV was 97.0%, ensuring that most patients below the threshold would not experience recurrence. The accuracy was 78.7%, making this test comparable to the 6-month marker; patients above the threshold should be closely monitored. The risk of recurrence was 17.778 times higher in the above-threshold group; the increased risk of recurrence was obvious, confirming that EBV12 was a strong prognostic indicator. EBV6, at a threshold of 1.5 copies/mL, had a sensitivity of 70.0% and specificity of 69.8%. EBV12, at a threshold of 42.5 copies/mL, achieved a sensitivity of 66.7% and specificity of 80.5%. These results suggest that EBV6 and EBV12 can be used as tools to identify the risk of metastasis, particularly when the values exceed the threshold. Patients with EBV6 levels above the threshold had a 5.385-fold higher risk of metastasis, whereas those with EBV12 levels above 42.5 copies/mL had an 8.250-fold increased risk, indicating that EBV6 and EBV12 are more effective in predicting metastasis than preEBV and postEBV. In particular, their defined thresholds can help identify patients at high risk of metastasis. The marked difference in risk between groups above and below the threshold highlights their potential value in prognostic evaluation and metastatic management.
However, owing to the limited sample size and follow-up duration, larger studies are required to validate the prognostic significance for metastasis. Previous studies have assessed EBV DNA concentrations at a few post-treatment time points but did not establish their prognostic value for recurrence/metastasis.10,38,39 In a study by He et al, 19 a 3-month cf EBV DNA concentration cutoff was 211 copies/mL was associated with poor survival time with OS showing hazard ratio (HR) = 4.71 (95% CI: 2.519-10.27, P < .001) and DFS showing HR = 2.71 (95% CI: 1.085-6.766, P < .001). Other authors have also recommended the use of cf EBV DNA concentrations to assess treatment response and survival outcomes.19,23,38,40 Regarding the predictive value of cf EBV DNA concentration at 6 and 12 months postEBV, the OR were 5.385 (95% CI: 1.20-24.166) and 8.250 (95% CI: 1.278-52.254), respectively. The sensitivities were 70.7% and 66.7%, with NPVs of 90.9% and 94.3%, P < .05. In multivariate analysis, EBV12 remained an independent prognostic factor for OS and DFS (P < .05). Consistent with these findings, studies by Li et al 15 and Liu et al 41 demonstrated that dynamic changes in EBV DNA concentration at different times before and after radiation serve as independent prognostic indicators in patients with NPC (P < .001).
In clinical practice, detecting recurrence early after treatment is essential.13,15,38 Previous studies have confirmed that plasma EBV DNA is a highly sensitive and noninvasive biomarker for predicting recurrence and metastasis. Based on our findings, we strongly recommend long-term follow-up and the development of a more extensive database that includes pre-treatment and multiple post-treatment time points for cf EBV DNA measurement. We examined cf EBV DNA concentration and evaluated the rate of disease progression, metastasis, recurrence, and other survival times. Patients with cf EBV DNA concentration above the defined cutoffs at 6 and 12 months postEBV should undergo closer surveillance. Those with higher plasma cf EBV DNA levels at ⩾2 post-treatment time points should be given more attention, including advanced imaging and/or other physical examinations to exclude recurrence or metastasis.
Briefly, when comparing the recurrence/metastasis prediction values at the 4 time points, preEBV showed predictive significance; however, its accuracy was relatively low, possibly due to confounding factors such as disease stage and tumour size. PostEBV had no predictive value, probably because most patients had very low cf EBV DNA levels after treatment. EBV6 demonstrated high predictive value, with a clear distinction between the recurrence/metastasis and non-recurrence/metastasis groups. EBV12 also retained significant prognostic value, particularly highlighting the importance of long-term monitoring. A preEBV threshold of 3980 copies/mL may aid in risk stratification and inform the development of a more intensive follow-up strategy for high-risk patients. The EBV6 and EBV12 thresholds – 12.5 and EBV12 5.5 copies/mL, respectively – should be regularly assessed through plasma cf EBV DNA quantification at their respective time points and integrated into the follow-up protocols for patients with NPC.
This study had some limitations. First, the sample size (58 cases) was adequate to assess cf EBV DNA levels across multiple time points; however, it was insufficient to explore more complex dynamic patterns. Second, the long-term follow-up strategy proposed could not be fully validated. Therefore, prospective, multicentre, randomised clinical trials with larger sample sizes and extended follow-up durations are needed to validate these findings.
Conclusions
This study established cutoff values for plasma cf EBV DNA concentrations in predicting recurrence and metastasis among patients with advanced-stage undifferentiated NPC in Vietnam. We found that the cut-off concentrations of preEBV, EBV6, and EBV12 demonstrated statistically significant predictive value for both recurrence and metastasis. These thresholds were also independent predictors of DFS and OS, except for EBV6 in relation to OS. Notably, EBV6 and EBV12 demonstrated better predictive value for recurrence, metastasis, and survival outcomes than preEBV did. Therefore, assessing plasma cf EBV DNA concentrations every 6 months during the first 2 years after CCRT should be encouraged in routine clinical practice to enhance follow-up outcomes.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251375893 – Supplemental material for Predictive Value of Plasma Epstein-Barr Virus DNA Concentration for Recurrence and Metastasis in Advanced-Stage Nasopharyngeal Carcinoma: A Longitudinal, Descriptive Investigation
Supplemental material, sj-docx-1-onc-10.1177_11795549251375893 for Predictive Value of Plasma Epstein-Barr Virus DNA Concentration for Recurrence and Metastasis in Advanced-Stage Nasopharyngeal Carcinoma: A Longitudinal, Descriptive Investigation by Huong Do Lan, Thuan Nghiem Duc, Ba Nguyen Van, Nam Thanh Quan, Dang Nguyen Van, Tuan Dinh Le, Son Tien Nguyen and Binh Van Nguyen in Clinical Medicine Insights: Oncology
Supplemental Material
sj-docx-2-onc-10.1177_11795549251375893 – Supplemental material for Predictive Value of Plasma Epstein-Barr Virus DNA Concentration for Recurrence and Metastasis in Advanced-Stage Nasopharyngeal Carcinoma: A Longitudinal, Descriptive Investigation
Supplemental material, sj-docx-2-onc-10.1177_11795549251375893 for Predictive Value of Plasma Epstein-Barr Virus DNA Concentration for Recurrence and Metastasis in Advanced-Stage Nasopharyngeal Carcinoma: A Longitudinal, Descriptive Investigation by Huong Do Lan, Thuan Nghiem Duc, Ba Nguyen Van, Nam Thanh Quan, Dang Nguyen Van, Tuan Dinh Le, Son Tien Nguyen and Binh Van Nguyen in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
We thank all the patients who participated in this study. We are grateful to all the medical staff in the Department of Head and Neck Radiation Oncology, Vietnam National Cancer Hospital, Hanoi, Vietnam, and the Institute of Biomedicine & Pharmacy, Vietnam Military Medical University, Hanoi, Vietnam, for their help in performing the present study. We would like to thank Editage (![]() ) for English language editing.
) for English language editing.
Ethical Considerations
This study was conducted in accordance with the ethical standards of the Military Hospital 103, Vietnam Medical Military University Committee, and the 1964 Helsinki Declaration, as revised in 2013, and its later amendments or comparable ethical standards. Ethical approval for this study was obtained from the Military hospital 103 Ethics Committee, under the approval number 1811/CNChT-HĐĐĐ date 23 July 2021. Informed consent was obtained from all the participants.
Consent to Participate
Informed consent was obtained from all the participants.
Consent for Publication
Written informed consent for publication of the clinical details and images was obtained from the patient.
Author Contributions
Conceptualisation: Huong Do Lan, Nam Thanh Quan, Thuan Nghiem Duc, Ba Nguyen Van, and Dang Nguyen Van; Data curation: Huong Do Lan, Nam Thanh Quan, Thuan Nghiem Duc, and Ba Nguyen Van; Formal analysis: Huong Do Lan, Nam Thanh Quan, Thuan Nghiem Duc, Ba Nguyen Van, and Tuan Dinh Le; Funding acquisition: Nam Thanh Quan, Thuan Nghiem Duc, and Binh Van Nguyen; Investigation: Huong Do Lan, Thuan Nghiem Duc, Ba Nguyen Van, Dang Nguyen Van, Nam Thanh Quan, and Tuan Dinh Le; Methodology: Huong Do Lan, Nam Thanh Quan, Thuan Nghiem Duc, and Ba Nguyen Van; Project administration: Nam Thanh Quan, Thuan Nghiem Duc, Son Tien Nguyen, and Ba Nguyen Van; Resources, Huong Do Lan; Software: Huong Do Lan, Nam Thanh Quan, Thuan Nghiem Duc, Ba Nguyen Van, and Dang Nguyen Van; Supervision: Thuan Nghiem Duc, Ba Nguyen Van, Dang Nguyen Van, Nam Thanh Quan, and Tuan Dinh Le; Validation: Huong Do Lan, Nam Thanh Quan, Thuan Nghiem Duc, Ba Nguyen Van, and Tuan Dinh Le; Visualisation: Huong Do Lan, Nam Thanh Quan, Thuan Nghiem Duc, and Ba Nguyen Van; Writing – review and editing: Huong Do Lan, Nam Thanh Quan, Thuan Nghiem Duc, Binh Van Nguyen, and Tuan Dinh Le; All the authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All patients were informed about the study. The patients and their representatives signed a consent form to participate in this study. Patients agreed to allow the research team to use their information, images, and test results for research purposes and publication.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
