Abstract
The eradication of tumors remains a critical and challenging issue in medical research. Exosomes (EXOs) derived from various sources have garnered increasing attention in the field of tumor therapy. Among these, cord blood (CB)-derived EXOs may offer unique advantages in combating tumors due to the distinctive properties of CB. This narrative review examines the current application status of CB-derived EXOs in tumor prevention and treatment: in the context of tumor treatment, it focuses on their direct therapeutic application and their use as drug carriers; regarding tumor prevention, it explores their application in cancer vaccines. In addition, it also reviews the specific components of CB from which these EXOs are derived, their particle sizes, characteristic proteins, and the types of tumors to which they have been primarily applied for prevention and treatment. In conclusion, we believe that CB-derived EXOs hold substantial potential for effective tumor prevention and treatment and are likely to emerge as a prominent area of research in the future.
Introduction
Cancer remains a leading cause of death worldwide.1,2 At present, the main therapeutic options for cancer treatment are still surgery, chemotherapy, radiotherapy, immunotherapy, and targeted therapy. 1 However, despite the effectiveness of various treatment strategies, issues such as tumor recurrence, metastasis, and drug resistance remain challenges for effective cancer management. 1
In recent years, exosomes (EXOs) have emerged as a promising focus in tumor therapy research.3,4 Exosomes, the predominant subtype of extracellular vesicles (EVs), are secreted by virtually all cell types.5,6 Extracellular vesicles are membrane-closed vesicle structures, which are found in body fluids such as blood, plasma, and serum 7 and can be divided into 3 main categories according to their size and biogenesis: apoptotic bodies, microvesicles, and EXOs. 8 Exosomes are the smallest EVs due to their diameter of approximately 40 to 160 nm.1,8 Notably, the diameter of EXOs measured by different laboratories has a certain difference, with a minimum diameter of 30 nm and a maximum diameter of 200 nm reported, such as 30 to 150 nm, 9 40 to 150 nm, 10 40 to 200 nm, 11 30 to 200 nm. 12 Exosomes are produced through invagination of the cytoplasmic membrane which results in the formation of multivesicular bodies (MVBs), and inward-budding of endosomes.1,8 Exosomes are released from MVBs which fuse with the cytoplasmic membrane via exocytosis by the vast majority of eukaryotic cells.1,8 Exosomes contain a variety of components, including nucleic acids (DNA, miRNA, mRNA, etc), proteins (receptors, extracellular matrix [ECM] proteins, transcription factors, enzymes, etc), lipids and metabolites, and so on.7,10 Previous studies have shown that EXOs mediate intercellular communication, transfer loaded proteins, lipids and nucleic acids to target cells, and change the biological behavior of recipient cells. 10 Moreover, compared with other nanoparticles, EXOs have lower immunogenicity and higher stability. 13 These characteristics of EXOs make them have the basis of wide application and have high research value. Several clinical trials have explored the possibility of using EXOs for disease diagnosis, treatment, and monitoring, such as their use in tumor therapy and early cancer diagnosis. 4 For example, several studies have been registered in ClinicalTrials.gov for the diagnosis and treatment of pancreatic cancer with EXOs. 14
Exosomes exist in almost all living cells and can be stably present in various body fluids, such as blood including umbilical cord blood (CB), cerebrospinal fluid, bile, saliva, milk, and urine.15,16 As a special human genetic resource, CB contains many heterogeneous mononuclear cells (MNCs) including stem cells, progenitor cells and immune cells, such as hematopoietic stem cells, mesenchymal stem/stromal cells (MSCs), regulatory T cells, monocyte-derived suppressor cells, endothelial progenitor cells, lymphocytes, dendritic cells (DCs). 17 These cells are not unique to CB, but their combination and naive nature give them special powers to perform well in transplantation and regenerative medicine, 17 especially in the treatment of hematological malignancies.17,18
Given these properties, a critical question arises: Do CB-derived EXOs (CB-EXOs) exhibit antitumor potential? Interest in CB-EXOs dates back over 2 decades, with early studies demonstrating that DC-derived CB-EXOs could induce robust immune responses when cocultured with autologous or allogeneic DCs. 19 Since then, CB-EXOs have shown remarkable efficacy across multiple biomedical domains. This narrative review focuses on their applications in tumor prevention and treatment, examining both mechanistic insights and translational progress.
CB-EXOs
It is well known that CB has long been recognized as a rich source of naïve pluripotent stem cells and immature T cells, 20 characterized by low immunogenicity, reduced incidence of immune rejection post-transplantation, and robust regenerative potential.20,21 In addition, since CB is typically considered medical waste, its collection is noninvasive, posing no harm to donors and involving limited ethical concerns.20,21 Correspondingly, CB-EXOs exhibit multiple advantages, including a cell-free morphology, nanoscale size, stability, reduced immunogenicity, enhanced functionality, ease of accessibility, harmlessness to donors, and absence of ethical controversies.20,21 Therefore, compared with stem cells, CB-EXOs are more suitable for clinical applications. 21 Studies have shown that CB-EXOs exhibit therapeutic effects such as enhancing new blood vessel formation wound healing, 22 reversal of liver fibrosis, 23 and repair of lung injury. 24 Regarding the preventive and therapeutic effects of CB-EXOs on tumors, to date we have identified 12 relevant publications. 11 ,25 -35 This narrative review focuses on them. We categorize them into 3 classes: direct antitumor effects, drug carrier functions, and vaccine adjuvant effects. The first 2 pertain to tumor treatment research, and the last one to tumor prevention research. Due to the limited number of studies, the review may not fully capture the complete therapeutic potential or potential limitations of CB-EXOs in cancer prevention and treatment, which is a key limitation of this article.
It is important to note that all CB-EXOs discussed in this study are exclusively derived from CB serum, CB stem cells (eg, CB-MSCs), CB macrophages (M1/M2), or CB-DCs. The CB-EXOs derived from CB cells are all isolated from the culture supernatants of these CB cell populations. Just like EXOs directly from CB serum/plasma, this ensures the therapeutic potential of CB-EXOs is inherently linked to the unique biological properties of CB, such as low immunogenicity and naive cell populations.
Application of CB-EXOs in Tumor Treatment
Direct antitumor effects of CB-EXOs
Exosomes themselves contain a variety of beneficial components, so the direct study of CB-EXOs on the treatment of tumors is undoubtedly the most straightforward idea and the easiest approach to undertake.
Zhu et al 25 investigated the direct effects of CB-EXOs on hepatocellular carcinoma (HCC) cells. The CB-EXOs were derived from CB serum and the diameter of the CB-EXOs was 50 to 100 nm under the transmission electron microscopy (TEM). 25 CD9 and CD63 were detected in EXO lysate, but caspase-3 was not detected. 25 The CCK8 assay results indicated that CB-EXOs could inhibit the viability of liver cancer cells; the results of flow cytometry detection showed that CB-EXOs promoted the apoptosis of liver cancer cells. 25 Although the mechanism of action of CB-EXOs in cancer prevention and treatment remains unclear, 25 the known research indicated that CB plasma was rich in proteins involved in ECM receptor interactions, and these proteins might act on the integrin family through CB-EXOs to participate in cell migration and proliferation, 25 which might be one of the reasons for their inhibition of cancer cell activity and promotion of their apoptosis.
Naeem et al 26 investigated the direct effects of CB-EXOs on CHL-1 melanoma cells and lymphocytes derived from a healthy person. Cord blood–derived EXOs were derived from human unrestricted somatic stem cells (USSCs), which are differentiated from CB-MNCs. 26 Observed under TEM at 250 keV, the direct diameter of CB-EXOs ranges from 30 to 120 nm. 26 Both the CCK8 and Comet assays showed that CB-EXOs reduced the survival activity of CHL-1 melanoma cells, but had no significant effect on healthy cells. 26 Sequencing analysis results showed the presence of let-7 family miRNAs in CBSC-EXOs, and previous studies had shown that let-7 family miRNAs have the ability to simultaneously induce cell death and inhibit cancer progression in several aggressive cancers, including melanoma.26,36 Therefore, Parisa Naeem et al 26 believed that some miRNAs in CB-EXOs exhibit the potential to prevent tumor induction, and these miRNAs may contribute to reducing tumorigenicity and growth of cancer cells.
There was already an evidence that the miR-503 cluster as a whole acted as a tumor suppressor complex in human endometrial cancers. 37 Additional studies showed that miR-503-3p, isolated from EXOs derived from human adipose stem cells, inhibited tumor growth by regulating the proliferation and self-renewal of cancer stem cells. 38 Pan et al 27 investigated the inhibitory effect of CB-EXOs rich in miR-503-3p on endometrial cancer. The CB-EXOs were derived from serum-free supernatants of 2 CB-derived MSCs (CB-MSCs), one of which was transfected with miR-503-3p mimic gene and the other without. 27 These CB-MSCs-derived CB-EXOs (CB-MSC-EXOs) was 30 to 200 nm in diameter under TEM and expressed EXO-specific markers CD63 and CD81. 27 Cell experiments showed that CB-MSC-EXOs were able to inhibit the development of endometrial cancer by inhibiting the mesoderm-specific transcript, 27 and the CB-MSC-EXOs that overexpress miR-503-3p achieved better results. 27 These results were further confirmed by experiments with nude mice. 27
Current research indicates that reactive oxygen species (ROS)-mediated mitochondrial dysfunction represents an effective strategy for inducing pancreatic cancer cell death.39 -44 Studies by Pan et al 45 suggest that the induction of mitochondrial apoptosis may constitute a general mechanism underlying natural killer (NK) cell–mediated killing. Based on these, Yanyun Zheng et al 28 hypothesized that CB NK cell–derived EXOs (CB-NK-EXOs) might suppress pancreatic tumor growth by targeting mitochondrial dysfunction. They employed an in vitro coculture system and an in vivo subcutaneous tumor model to evaluate the antitumor effects of CB-NK-EXOs against pancreatic cancer. 28 Cord blood NK cell–derived EXOs were isolated from the serum-free culture supernatant of CB-NK cells. 28 Transmission electron microscopy revealed that CB-NK-EXOs exhibited an average diameter of 122.7 nm. 28 Western blot analysis confirmed their positive expression of the exosomal markers CD81 and Syntenin, while being negative for Calnexin. 28 In vitro cellular assays demonstrated that CB-NK-EXOs induced mitochondrial apoptosis in PANC-1 cells by altering ROS (P < .0001) and mitochondrial membrane potential levels (P < .0001). 28 The in vivo data from experiments in nude mice further substantiated that, compared with the tumor model group, CB-NK-EXOs treatment significantly reduced tumor volume and weight, while increasing Bax protein expression. 28 These findings indicate that CB-NK-EXOs can enter PANC-1 cells via endocytosis, induce mitochondrial oxidative damage, and inhibit the progression of PANC-1 cells, thereby demonstrating potent antitumor effects against pancreatic cancer. 28
Cord blood–derived EXOs can be obtained through various ways and simple methods, and can inhibit a variety of cancer cells or induce their apoptosis while having no effect on healthy cells, which makes them fully qualified as one of the future options for tumor prevention and treament.
CB-EXOs as carriers
Although the direct application of CB-EXOs can play a role in the treatment of cancer, their effectiveness can be limited by off-target accumulation (eg, in liver/spleen) 46 and inherent heterogeneity. 47 Therefore, engineering CB-EXOs as targeted drug carriers represents a promising strategy to enhance therapeutic efficacy and specificity.
We have already mentioned the effects of EXOs on cancer cells in the previous direct application of EXOs, in which miRNAs may play a key role. For example, let-7 family of miRNAs targets important oncogenes to inhibit tumor growth.26,36 In fact, in addition to inhibiting tumor growth, some other miRNAs promote tumor growth. Blocking the function of these miRNAs can also play a role in cancer suppression. 36 It is known that miRNA-221 is a key noncoding RNA in tumorigenesis and development. 48 In a variety of human malignant tumors, such as colorectal cancer, HCC, prostate cancer and other solid tumors, the expression of miRNA-221 is upregulated, 48 that is, it can promote the occurrence and progression of colorectal cancer. 49 Accordingly, anti-miRNA-221 binds to miRNA-221 through complementary sequences and inhibits the function of miRNA-221 in cancer cells. 29
In addition, Lamp2b is an EXO membrane protein, which is widely used for EXO surface functionalization to achieve targeted delivery. 50 And iRGD peptide can be used as a ligand to bind to tumor cell receptor protein neuropilin-1 (NRP-1) to mediate membrane penetration. 29 Accordingly, Han et al 29 transferred plasmids containing the fusion gene iRGD-Lamp2b into the commercial immortalized human CB-MSCs by lentivirus transfection technology, and then transfected anti-miRNA-221 into the EXOs derived from these CB-MSCs by electroporation to investigate the therapeutic effect of anti-miRNA-221 loaded CB-MSC-EXOs on colorectal cancer. Electron microscope analysis and particle size analysis of EXOs showed that most EXOs were about 100 nm in diameter. 29 The experimental results showed that the EXOs were taken up by tumor cells through the interaction with NRP-1 and inhibited the proliferation and clonal formation of colon cancer cells in vitro. 29 In the tumor mouse model subcutaneously inoculated with HCT116 cells, EXOs loaded with anti-miRNA-221 were obviously enriched at the tumor sites and had good antitumor effects. 29 The molecular mechanism may be that anti-miRNA-221 upregulates the expression level of corresponding tumor suppressor factors. 29
MiR-26a has a bidirectional effect of promoting and suppressing cancer.51,52 Mahati et al 30 investigated its inhibitory effect on liver cancer through the delivery of umbilical CB-EXOs. To better target EXOs, the anti-Glypican 3 (GPC3) and single-stranded variable fragment (scFv) fusion gene was first inserted into immortalized CB-MSCs using lentiviral vectors. 30 GPC3, a membrane-associated glycoprotein, is significantly upregulated in HCC with little or no expression in normal tissues 53 and is considered a potential target for tumor treatment. 30 ScFv maintains antigen-binding activity, low molecular weight, and low immunogenicity and is an excellent drug delivery tool with targeting ability. 30 Exosomes isolated from these CB-MSC culture supernatants carrying anti-GPC3 scFv fusion genes were modified by anti-GPC3 scFv. 30 The Neon electroporation system then introduced miR-26a mimics into these EXOs. 30 Electron microscope and dynamic light scattering (DLS) results showed that the diameter of these drug-carrying EXOs was about 160 nm. 30 The experimental results showed that the anti-GPC3 SCFV-modified EXOs effectively delivered miR-26a to GPC3-positive liver cancer cells, thereby inhibiting cell proliferation and migration by regulating the expression of downstream target genes of miR-26a without appreciable side effects. 30
To evaluate the feasibility of treating ovarian cancer with the powerful chemotherapy drug cisplatin (CIS) in EXOs, Zhang et al 11 developed and compared 2 kinds of EXOs derived from CB-MNC M1 and M2 macrophages, and their differences after loading CIS. CB-MNCs were first obtained from CB by centrifugation, and then CD14+ cells were obtained from them by immunomagnetic bead sorting. 11 Next, CD14+ cells were induced to differentiate into 2 types of macrophages, M1 and M2, using different inducers. 11 Finally, 2 groups of EXOs were isolated from the 2 macrophage culture supernatants, which were named CB-M1-EXOs and CB-M2-EXOs, respectively. 11 Under TEM, both these EXOs were round, with an average diameter of 40 to 200 nm, and both expressed high levels of EXO-related proteins CD63, CD81, and TSG101. 11 However, the average particle sizes of CB-M1-EXOs measured by high-sensitivity flow cytometry (HSFC) and nanoparticle tracking analysis (NTA) were 72.63 ± 23.58 nm and 138.9 ± 49.1 nm, and those of CB-M2-EXOs measured by HSFC and NTA were 70.81 ± 21.24 nm and 144.3 ± 46.9 nm, respectively. 11 Cisplatin was introduced into the 2 kinds of EXOs by ultrasonication method, and the resulting EXOs were named CB-M1-EXO-CIS and CB-M2-EXO-CIS, respectively. 11 It was found that the loading capacities of CB-M1-EXO-CIS (30%) was higher than that of CB-M2-EXO-CIS (28%) by high-performance liquid chromatography analysis. 11 Cell experiments demonstrated that compared with chemotherapy alone, the cytotoxicity of CB-M1-EXO-CIS and CB-M2-EXO-CIS against the CIS-resistant line A2780/DDP cells increased by 3.3 and 1.7 times, respectively. 11 It could be said that CB-M1-EXOs were potential powerful new tools for delivering chemotherapy drugs to treat cancer. 11
Subsequently, Zhang et al 31 further used CB-M1-EXO-CIS to research on the treatment of ovarian cancer and drug resistance. The preparation of CB-M1-EXO-CIS followed the method used in the previous experiment. 11 In this experiment, the diameter of CB-M1-EXOs under TEM is 30 to 100 nm and the surface of CB-M1-EXOs was still rich in EXO-related proteins CD63, CD81, and TSG101. 31 They inoculated mice with A2780/DDP cells subcutaneously to create a mouse model of ovarian cancer to assess the effect of CB-M1-EXO-CIS on tumor growth. 31 The research found that CB-M1-EXO-CIS could target tumor sites of ovarian cancer in vivo, markedly inhibit the growth of subcutaneous transplanted tumors, and prolong the survival time of mice. 31 Compared with ordinary CIS preparations, CB-M1-EXO-CIS significantly enhanced the cytotoxicity of the drug and restored the sensitivity of ovarian cancer cells to CIS. 31 The CB-M1-EXO-CIS might reduce drug resistance by uniquely expressing adhesion proteins CD63, CD81, TSG101, and integrin β1/CD29. 31 Binding of the integrins on CB-M1-EXO-CIS to overexpressed integrin receptors in ovarian cancer cells increased the targeting and binding ability of CB-M1-EXO-CIS. 31
Docetaxel (DTX) is an antitumor drug, which can inhibit microtubule dynamics and destroy cell division, thus effectively inducing apoptosis, and can be applied to the anticancer treatment of different tumors.54,55 As a tumor suppressor gene, miR-125a has been confirmed to be related to tumor metastasis, 56 which can specifically target the HER2/ ErbB2 (epidermal growth factor receptor subfamily) mediated signaling pathway and affect the migration of invasive breast cancer cells. 57 For the convenience of description, we first named the CB-MSC-EXOs untransfected and transfected miR-125a as MSC-EXOs and miR125a-EXOs, respectively. Basak et al 32 encapsulated DTX into MSC-EXOs and miR125a-EXOs by optimized mild sonication-incubation technique to obtain 2 EXOs, MSC-EXO-DTX and miR125a-EXO-DTX, respectively, and subjected the EXOs to mouse triple-negative breast cancer cells (TNBC 4T1) to compare their anticancer effects. The characteristic detection of the CB-MSC-EXOs before the experiment showed that the particle sizes of MSC-EXOs and miR125a-EXOs under the DLS were similar, about 185 nm. 32 However, after loading DTX, the particle sizes become larger, which are 217.60 ± 16.88 nm and 232.50 ± 12.9 nm, respectively. 32 They also have different DTX loading capacities, 9.75 ± 1.84 ng/μg and 8.86 ± 1.97 ng/μg, respectively. 32 In addition, CD63, CD81, and TSG101 were detected in MSC-EXOs lysates, whereas β-actin was almost undetected. 32 In vitro wound healing and transwell invasion experiments confirmed that miR125a-EXO-DTX had the most pronounced anti-metastasis ability compared with free DTX and miR125a-EXOs. 32 In vitro invasion experiments showed that miR125a-EXO-DTX treatment showed a significant decrease in the aggressiveness of TNBC 4T1 accompanied by evident cytoskeletal degradation and nuclear deformation compared with miR125a-EXOs treatment. 32 In addition, the synergistic effect of miR-125a and DTX was confirmed by F-actin and nuclear degradation using confocal and FESEM assay. 32 Therefore, DTX and miR-125a loaded into MSC-EXOs can synergistically play anticancer and antimigration roles. 32
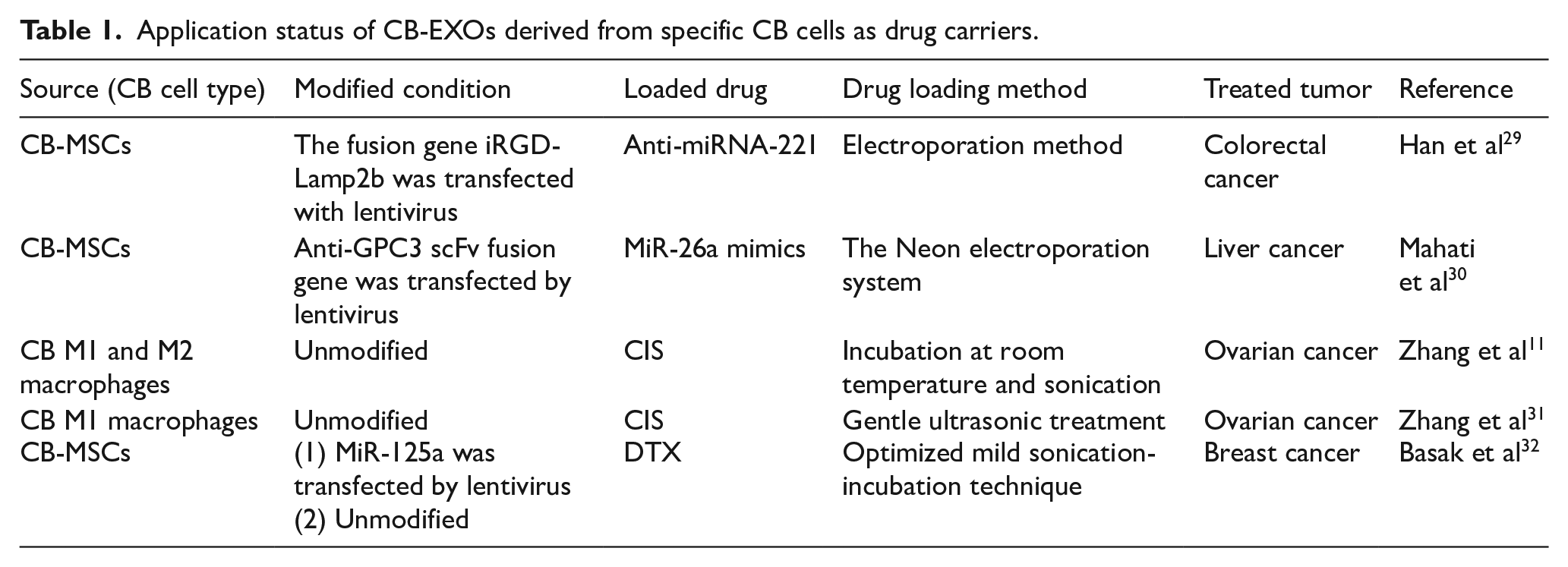
At present, CB-EXOs used as antitumor vectors are all derived from the culture supernatants of CB-MSCs and CB macrophages. Before loading drugs, it is sometimes necessary to modify cells through lentiviruses to transfect certain genes. Loaded drugs are usually introduced directly into the CB-EXOs by physical means such as electroporation and ultrasound (see Table 1). Furthermore, Table 1 demonstrates the diversity of both therapeutic agents loaded into CB-EXOs and the tumor types targeted. Despite the limited number of current studies, these findings suggest a broad potential for CB-EXOs in cancer therapy.
Application status of CB-EXOs derived from specific CB cells as drug carriers.
Application of CB-EXOs as Vaccines in Tumor Prevention
Cord blood–derived EXOs not only demonstrate unique potential in the treatment of some cancers, but can also be developed into vaccines to prevent tumorigenesis.
As early as 2006, Chinese scientists, Du et al 33 at Zhengzhou University, have started the research of EXO vaccines. 33 they used recombinant human stem cell factor (rhSCF), recombinant human granulocyte–macrophage colony-stimulating factor (rhGM-CSF) and recombinant human interleukin-4 (rhIL-4) to induce CB-MNCs to become DCs. Human gastric adenocarcinoma cell BGC823, human esophageal cancer cell Eca109, human chronic myeloid leukemia cell line K562, human promyeloid leukemia cell line HL60 (both K562 and HL60 are leukemia cell lines) were cultured with supernatant to prepare soluble tumor antigen and stimulate DCs. 33 Exosomes were obtained by centrifuging DC culture supernatant. 33 Under 40 000× TEM, the EXOs were round particles with a diameter of 80 to 100 nm and a complete membrane structure. 33 Their surface markers included MHC-II, CD40, CD80, CD86, and CD54. 33 The analysis of mixed lymphocyte reaction and cytotoxic T lymphocyte (CTL) killing activity showed that the EXOs secreted by CB DCs loaded with BGC823, Eca109, K562, and HL60 membrane antigens could effectively stimulate T-cell proliferation and enhance CTL killing activity. 33
Several years later, Guan et al 34 from YING DU’s team at Zhengzhou University still used rhSCF, rhGM-CSF, and rhIL-4 to induce CB-MNCs to become DCs. After that total RNAs and lysates of BGC823 as tumor-associated antigens were cocultured with these DCs. 34 Then, DCs with high expression of MHC-I, MHC-II, CD34, CD40, CD80, CD86, CD11c, and CD54 proteins were obtained, and CB-DC-EXOs are isolated by centrifugation in their culture supernatant. 34 Under TEM, these CB-DC-EXOs were 40 to 90 nm in size and expressed DC surface markers MHC-II, CD40, CD80, CD86, and CD54. 34 Cell and mouse experiments have shown that these CB-DC-EXOs can stimulate T-cell proliferation, produce effective cytotoxicity, and induce more effective antitumor immunity in vivo. 34
Than et al 35 specifically conducted anticancer studies on CB DCs and their EXOs stored at low temperature. They obtained CB DCs by inducing differentiation of CB-MNCs by GM-CSF and IL-4 in vitro, and then obtained CB-DC-EXOs from these CB DCs after stimulating them with lung cancer cell A549 lysate. 35 Observed under TEM, the particle sizes of these CB-DC-EXOs were about 60 to 100 nm (judged by the figure in the paper). 35 They had high expression of CD9 and CD63 and low expression of CD86. 35 The results of cell experiments showed that CB DCs stimulated by the lysate of lung cancer cell A549 and their CB-DC-EXOs could induce the proliferation of allogeneic T cells, and could also make allogeneic peripheral blood MNCs have stronger cytotoxicity to A549 lung cancer cells, demonstrating its anticancer efficacy. 35
As small noncellular vesicles, CB-DC-EXOs can effectively present antigens to T cells and are more stable and easier to preserve for a long time than cell-based vaccines, even from readily available cryopreserved CB-MNCs.33 -35 These advantages make them promising to be a more effective DC-based vaccine. Used for inducing antitumor immunity.33 -35
Discussion
Summative analysis of the application of CB-EXOs in tumor prevention and treatment
So far, CB-EXOs used for tumor prevention and treatment mainly come from CB stem cells (especially CB-MSCs),26,27,29,30,32 CB immune cells (CB macrophages,11,31 CB DCs,33-35 and CB-NK cells), and CB serum, 25 among which, CB-MSCs are relatively most used. Existing studies had shown that EXOs derived from different CB components or the same source but different preparation may have different particle size and specific proteins, and even different detection and application scenarios can show differences in particle size (Table 2). The variations in particle size and marker proteins among CB-EXOs primarily stem from inherent differences in CB sources (including CB physiological/pathological status and biogenesis pathways of EXOs) and their compositional heterogeneity, as well as methodological discrepancies in isolation and detection techniques. 58 The inherent biological variability among individual CB units naturally confers heterogeneity and functional diversity to the EXOs derived from them. This fundamental characteristic results in poor reproducibility when attempting to produce EXOs with identical profiles, potentially leading to significant variations in clinical efficacy. However, with advancements and standardization in EXO isolation and characterization technologies—including the application of precision sorting techniques 59 and engineering modifications 60 —it should be possible to substantially reduce their heterogeneity, thereby enabling better clinical applications.
Characteristics of different CB-EXOs in the prevention and treatment of various tumors.

< 1 > The particle size of drug-loaded EXOs. < 2 > The particle size detected by DLS.
At the same time, it can also be seen that CB-EXOs have a wide range of applications in the prevention and treatment of tumor types, which indicates that CB-EXOs may be one of the main forces in the future tumor prevention and treatment. See Table 2 for details. Particle sizes of non-drug-loaded EXOs were measured by TEM unless otherwise specified. It should be particularly noted that in order for CB-EXOs to function better, especially when they are used as targeted carriers, their modification is indispensable. Some modification examples can be referred to in Table 1.
The composition characteristics of CB-EXOs
Studies have found that the composition of CB-EXOs was different from that of EXOs from adult peripheral blood, especially in protein and miRNAs, which might be an important reason why CB-EXOs can better prevent and treat tumors. The analysis of exosomal miRNAs in CB plasma and adult peripheral blood plasma by small RNA sequencing showed that 65 miRNAs were differentially expressed (46 were upregulated). Some of these miRNAa target genes were enriched in cancer-related signaling pathways.61,62 For example, miR-122-5p had a cancer-suppressing effect,63 -65 and its content in EXOs of CB plasma was 4 times that of adult peripheral blood plasma. 61 Peñas-Martínez et al 62 found that 131 proteins were differentially expressed in EXOs from CB plasma compared with EXOs from adult plasma, of which 83 proteins were overexpressed. Of the 83 highly expressed proteins, α2-macroglobulin and clathrin, and α2-macroglobulin (A2MG) could inhibit tumors,66,67 and clathrin also had an auxiliary effect on tumor suppression in some cases.68,69 Of course, the principle of tumor prevention and treatment by CB-EXOs is very complex, and the molecular mechanisms involved in different tumors, EXOs, and treatment strategies may also be different, which needs to be gradually understood in future in-depth studies.
Analysis of the advantages and disadvantages of CB-EXOs
Cord blood–derived EXOs inherit the characteristics and low immunogenicity of primitive cells in CB. Compared with cell therapies that carry risks of differentiating into undesired cell/tissue types and tumorigenicity, 70 CB-EXOs are noncellular, incapable of differentiation, and have not been observed to induce tumors, 71 rendering them safer. In addition, they possess intrinsic homing capabilities and can be engineered into targeted therapeutics or drug carriers, enabling precise and efficient treatment. Moreover, they hold potential not only for tumor treatment but also for cancer prevention. Furthermore, they raise minimal ethical concerns and can be stored physically, facilitating convenient application.
Although CB-EXOs are predominantly isolated using repeated centrifugation, specific protocols vary significantly across research groups. Combined with source variability and other factors, this results in substantial heterogeneity among CB-EXOs. To date, no unified standards for their preparation, characterization, and quality control have been established, which compromises the consistency and predictability of their efficacy in tumor therapy and prevention.
In addition, for clinical translation, CB-EXOs must be producible at scale and maintain stable performance under accessible and appropriate storage conditions for extended periods. However, robust evidence supporting their stability and scalable manufacturing under these conditions is currently limited.
Conclusion
In summary, CB-EXOs demonstrate substantial dual potential: for tumor treatment, through direct cytotoxic effects and engineered drug delivery systems; for tumor prevention, via vaccine-induced antitumor immunity. Their unique composition, including tumor-related miRNAs and proteins, enhances specificity in both therapeutic and preventive applications. However, the heterogeneity and variability of EXOs have limited their research and application. Future research must address these issues to advance CB-EXOs into clinical trials.
Overcoming current challenges represents an imperative yet promising path toward harnessing the full clinical potential of CB-EXOs in both cancer prevention and therapy. Continued exploration of their molecular mechanisms and large-scale production methods will be critical to unlocking their full therapeutic potential and enabling clinical translation.
Footnotes
Acknowledgements
The authors would like to acknowledge Professors An Hong and Xiaojia Chen of Jinan University, and Xuehua Lin from The Affiliated Panyu Central Hospital of Guangzhou Medical University, for their help during the revision stage of this article.
Authors’ Note
None of the authors is a member of the journal’s Editorial Board.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Chen Huang made a substantial contribution to the concept or design of the work; Yongsheng Li made a substantial contribution to the concept or design of the work, and drafted the article or revised it critically for important intellectual content; Baozhong Zhang made a substantial contribution to the interpretation of data; Yukuan Tang made a substantial contribution to the analysis of data; Yuyang Huang made a substantial contribution to the acquisition of data; Wei Wei approved the version to be published.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Medical Science and Technology Foundation of Guangdong Province (A2024625), Guangzhou Municipal Science and Technology Program key Projects (202103000002), Panyu Science and Technology Planning Project (2023-Z04-019), and Guangzhou Health science and technology project (no. 20241A010112).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
