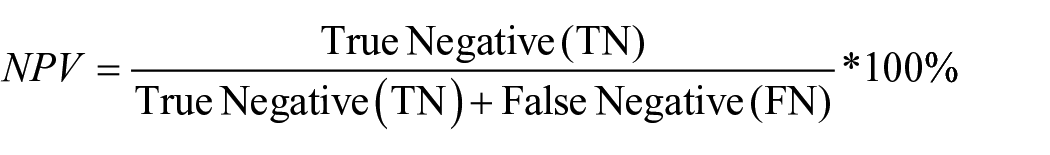Abstract
Background:
Accurate intraoperative navigation remains challenging in gastrointestinal tumor surgery. This study aims to explore the potential of optical coherence tomography (OCT) and endoscopic ultrasonography (EUS) in enhancing the accuracy of intraoperative navigation.
Methods:
This single-center diagnostic study was conducted at Northern Jiangsu People’s Hospital from August to December 2024. This study is part of a prospective study, and more patients will be recruited in the future. The normal tissues, tumor tissues, vascular tissues, adipose tissues and lymph nodes of 12 patients with gastrointestinal tumors were collected for OCT and EUS scanning, and the image differences were compared. In addition, 43 lymph node specimens from 18 randomly selected patients were scanned by OCT and EUS, and pathological reports were used as the baseline. The results of OCT and EUS were compared retrospectively.
Results:
There are notable differences between OCT and EUS imaging. EUS can capture images of the entire tissue layer, making it more effective than OCT in assessing tumor invasion depth. Conversely, OCT serves as an “optical biopsy,” providing higher precision in the early stages of lesions. Our further research indicates that the combined application of these modalities holds significant potential. In a double-blind evaluation of 43 lymph nodes from 18 randomly selected patients, OCT demonstrated a sensitivity of 81.25%, specificity of 88.89%, and a positive predictive value (PPV) of 81.25%. In comparison, EUS showed a sensitivity of 87.50%, specificity of 85.19%, and a PPV of 77.78%. The combined use of OCT and EUS achieved a sensitivity of 93.75%, specificity of 92.59%, and a PPV of 88.24%.
Conclusions:
The combined use of OCT and EUS can enhance sensitivity and specificity, thereby improving the accuracy of intraoperative navigation.
Trial registration:
Chinese Clinical Trial Registry (No. ChiCTR2400088875); Registered 28 August 2024, first patient enrolled 20 September 2024.
Keywords
Introduction
Gastric cancer (GC) and colorectal cancer (CRC) are among the most prevalent malignancies affecting the digestive tract. According to a 2022 report from the International Agency for Research on Cancer, a branch of the World Health Organization, CRC accounts for 9.3% of global cancer-related deaths, ranking second, while GC accounts for 6.8%, placing fifth. 1 This trend is particularly pronounced in China, where, in 2022, their mortality rates ranked fourth and third. 2 The elevated incidence and mortality of gastrointestinal cancers in China significantly hinder the nation’s healthcare progress and social development. Thus, it is essential to not only implement early screening measures to reduce the incidence of gastrointestinal tumors but also to adopt systematic, comprehensive, and evidence-based treatment approaches to lower cancer mortality rates. As research into gastrointestinal tumors progresses, treatments such as traditional Chinese medicine, neoadjuvant therapy, surgery, postoperative chemotherapy, immunotherapy, genetic testing, and molecular targeted therapy have unquestionably become pivotal elements in the comprehensive management of these cancers.3-5 However, surgical intervention remains the primary curative approach for gastrointestinal tumors. On one hand, preserving gastrointestinal function in cancer patients, particularly in low rectal cancer surgeries, requires minimizing tissue resection to facilitate anal preservation. On the other hand, precise resection of the tumor must be achieved. Furthermore, during the dissection of surrounding lymph nodes, it is essential to remove as many positive lymph nodes as possible while preserving functionally intact and noninvaded lymph nodes, thereby providing a foundation for subsequent immunotherapy.
To achieve thorough clearance of the tumor and the positive lymphatic tissue surrounding it, the fluorescence properties of indocyanine green (ICG) under near-infrared (NIR) excitation have been identified and gradually applied in lymph node imaging. 6 This method enhances the regional lymph node clearance rate; however, ICG is insufficiently comprehensive to provide complete lymph node visualization. Optical coherence tomography (OCT) is an emerging biomedical optical imaging technology that enables high-resolution tomographic imaging of fine structures within biological systems. Optical coherence tomography can achieve image resolutions between 1 and 15 μm, 1 to 2 orders of magnitude finer than standard ultrasound (US) imaging. The penetration depth of OCT images is governed by optical scattering and can reach up to 5 mm in tissue. It functions as an “optical biopsy,” offering cross-sectional images of tissue structures at the micron scale. 7 In recent years, OCT has demonstrated its utility in detecting various malignancies, particularly gastrointestinal diseases. 8 When employing OCT for the detection of colonic dysplasia, the sensitivity reaches 92%, with a specificity of 84%. These results underscore the substantial potential of OCT in the early detection of precancerous lesions and the initial stages of CRC. 9 Optical coherence tomography also holds significant potential for real-time intraoperative assessment of lymph node status, potentially reducing unnecessary lymph node excisions. Preliminary studies have shown that OCT can distinguish between metastatic and nonmetastatic lymph nodes during surgery, with a sensitivity of 64.8% and a specificity of 73.3%. 10 When combined with artificial neural networks for real-time detection of lymph node metastasis, sensitivity increases to 98.9%, specificity to 98.8%, and the overall diagnostic accuracy reaches 90.1%. 11 Endoscopic ultrasonography (EUS) is considered the most accurate imaging modality for the local staging of gastrointestinal tumors, particularly in esophageal cancer, GC, and pancreatic cancer. Endoscopic ultrasonography is extensively used in the evaluation of submucosal tumors, including gastrointestinal stromal tumors (GIST), by accurately identifying the layer of origin and echogenic characteristics, thus aiding in the assessment of malignant potential. Studies have shown that EUS achieves a sensitivity of up to 95% and a specificity of 72% in the diagnosis of GISTs. 12 Endoscopic ultrasonography is frequently combined with fine-needle aspiration (FNA) for the detection of malignant lymph node metastasis. The EUS-FNA has demonstrated high accuracy, particularly in lung cancer and gastrointestinal tumors. In lymph node diagnostics, EUS-FNA achieves a sensitivity of 98.3% and a specificity of 100%. 13 Endoscopic ultrasonography serves as a “guiding light” for FNA, further suggesting its potential to become a “guiding light” for OCT in similar applications. Clinical studies have demonstrated the feasibility of combining OCT with US for applications in complex areas such as biliary tumors. This combined approach can be used to detect early malignant changes, such as dysplasia and neoplasm formation, as well as to assist in tumor staging. 14 This article examines the feasibility of using OCT and EUS imaging technologies to enhance lymph node dissection rates during surgery.
Materials and Methods
This diagnostic accuracy study adheres to the CONSORT-2010-Checklist (Supplementary File S4).
Patients
The patients who fulfilled the following inclusion criteria were recruited for the study: Patients must have a confirmed preoperative diagnosis of malignancy, with no history of preoperative radiotherapy or chemotherapy that could impact outcomes. Both male and female patients aged 18 to 75 years are eligible. An expected survival period of at least 3 months is required, along with at least 1 measurable tumor lesion. Patients must not have major organ dysfunction or significant coagulation disorders and must be able to tolerate surgery. The exclusion criteria were as follows: Patients with intestinal obstruction, extensive intra-abdominal adhesions, or significant intra-abdominal inflammation are excluded, as well as those with functional impairments in major organs, such as the heart, kidneys, liver, or brain. Exclusion also applies to patients with cachexia, severe coagulation disorders, uncontrolled or severe cardiovascular and pulmonary diseases, or those with unstable vital signs due to any cause. Vulnerable populations—including individuals with mental illness, cognitive impairment, critical illness, minors, and pregnant women—are ineligible for participation. The complete process is illustrated in Figure 1.

Flow chart of the experiment. Sorting out gastrointestinal malignancies. Tissue was scanned using OCT and EUS. The results were compared with pathological sections. The feasibility and accuracy of OCT and EUS will be judged for lymph node dissection and incisal margin determination. OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.
This prospective study was conducted between 28 August 2024 and 20 December 2024 at Northern Jiangsu People’s Hospital Affiliated to Yangzhou University (a tertiary teaching hospital in Jiangsu Province, China). The study protocol was approved by the hospital’s Medical Ethics Committee (Approval No.2024ky183). The project has been registered in the Chinese Clinical Trial Registry (ChiCTR2400088875). This study is part of a prospective study, and more patients will be recruited in the future. Intraoperative OCT and EUS scans will be performed, and the prognosis of patients will be followed up. It was conducted in accordance with the Declaration of Helsinki and the Code of Practice for the Quality Management of Pharmaceutical Clinical Trials. All patients provided written informed consent before enrollment.
OCT technique
Model and Specifications: OCT (ILUMIEN OPTIS); Guidewire (Dragonfly OPTIS C408645). We connected the guidewire to the OCT and scanned the specimen. The video was exported to a DVD and the images were captured using a computer.
Tumor tissue, normal tissue, lymphoid tissue, adipose tissue, and lymph nodes: The specimens fixed with formalin are scanned using OCT probes. The images are exported in the form of DVD videos. To avoid influencing the patient’s postoperative pathological staging, the lymph nodes are returned to the pathology department after scanning. The remaining specimens are immediately fixed and preserved with formalin after scanning.
EUS technique
Model and Specifications: Olympus Endoscopic Ultrasonography (EUS) Image Processing Device (EU-ME2 PREMIER PLUS); Endoscopic Ultrasound (EUS) Probe (Olympus UM-3R 20MHZ).
Tumor tissue, normal tissue, lymphoid tissue, adipose tissue, and lymph nodes: The specimens fixed with formalin are immersed in water, scanned using an EUS probe, and images are collected using the associated equipment. The images are exported via mobile devices. Likewise, to prevent any impact on the patient’s postoperative pathological staging, the lymph nodes are returned to the pathology department after scanning. The remaining specimens are promptly fixed and preserved with formalin following the scanning.
The hematoxylin and eosin staining process
Tissue sections are first deparaffinized and hydrated through a series of xylene and alcohol washes. The sections are then stained with hematoxylin, which binds to nucleic acids and provides a blue-purple color to cell nuclei. Following a rinse, the sections are treated with eosin, which stains cytoplasmic and extracellular components pink. Finally, the sections are dehydrated through graded alcohols, cleared with xylene, and coverslipped for microscopic examination.
Immunohistochemistry procedure
First, tissue sections are deparaffinized and rehydrated through xylene and graded alcohols. Antigen retrieval is then performed by heating in a citrate buffer solution to expose CK19 epitopes. After cooling, sections are treated with a blocking agent to prevent nonspecific binding. The CK19-specific primary antibody is applied, followed by incubation with a secondary antibody conjugated to horseradish peroxidase. The DAB chromogen is then added, producing a brown precipitate in areas expressing CK19. Finally, sections are counterstained, dehydrated, cleared, and coverslipped for microscopic evaluation.
Image evaluation and processing
Both OCT and EUS images were jointly captured by 2 experienced endoscopists, while pathological tissue sections were scanned and recorded using NDP.view2.
Following image acquisition, 2 experienced pathologists compared and evaluated the imaging correlations, determining the histological characteristics of various tissues. Subsequently, the team discussed and collaboratively established diagnostic and differential criteria by comparing, observing, and summarizing the characteristics of OCT and EUS images.
Subsequently, We used a double-blind method to randomly collect 43 lymph nodes from 18 patients. The team members would assess the nature of the lymph nodes in accordance with the characteristics and differences of OCT and US imaging, and ultimately calculate the sensitivity and specificity based on the final pathological outcomes.
Statistical analysis
The sample size calculation procedure is shown in Supplementary Figure 3 in the Supplementary material. The statistical analysis in this study primarily evaluated the diagnostic performance of 3 methods: OCT, EUS, and the combined OCT-EUS approach. Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated to compare the differences in diagnostic accuracy among these methods.
Sensitivity is defined as the proportion of correctly identified positive cases, calculated using the formula:
Specificity is defined as the proportion of correctly identified negative cases, calculated using the formula:
PPV represents the probability that a positive test result is truly positive:
NPV represents the probability that a negative test result is truly negative:
Results
Baseline characteristics
All patients in this study underwent 3 types of imaging evaluations: OCT, EUS, and the combined OCT-EUS. Baseline characteristics were consistent across all 3 evaluation groups, as they involved the same patient cohort. Therefore, these baseline characteristics were collectively reported without performing statistical comparisons between the groups.
To investigate the potential of OCT and EUS in defining surgical margins, 12 patients were enrolled in this study (Details are provided in Supplementary Figure 1 in the Supplementary material), including 7 men (58.3%) and 5 women (41.7%). The mean age was 70.0 ± 6.3 (range: 60-78) years. The mean height was 164.2 ± 7.4 cm, and the mean weight was 58.6 ± 10.1 kg. In terms of hematological analysis, the mean red blood cell count was 3.92 ± 0.66 × 1012/L, and the mean hemoglobin level was 116.5 ± 21.8 g/L. The mean white blood cell count was 6.09 ± 1.39 × 109/L, and the mean albumin level was 37.5 ± 5.9 g/L. For tumor markers, the mean levels of carbohydrate antigen 19-9 (CA19-9), alpha-fetoprotein (AFP), and carcinoembryonic antigen (CEA) were 8.1 ± 6.8 U/mL, 4.06 ± 2.25 ng/mL, and 3.93 ± 2.14 ng/mL, respectively. Missing values for tumor markers were excluded from the analysis . Regarding tumor staging, based on the cTNM classification, the patients were categorized into stages T2-T3, N0-N1, and Mx. Similarly, according to the pTNM classification, the patients were distributed across stages T2-T3, N0-N1, and M0-Mx (Table 1).
Baseline characteristic of 12 patients.
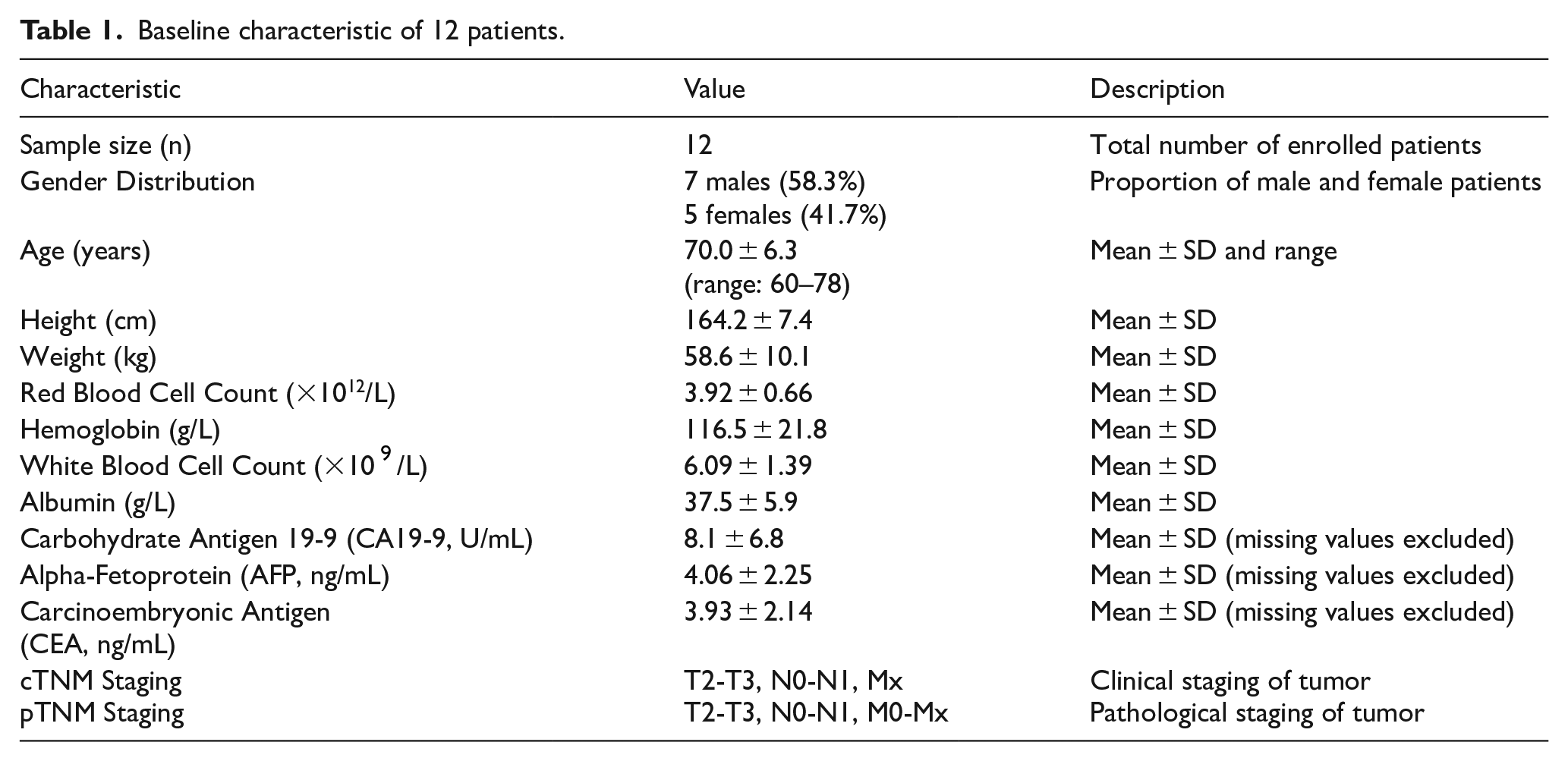
ICG has difficulty in providing a comprehensive visualization of lymph nodes
As shown in Figure 2, we observed during the surgery that some lymph nodes did not fluoresce after ICG injection. To precisely define the surgical resection margins, thoroughly clear the regional lymph nodes, and differentiate tissue types, we tested ex vivo samples, including tumor tissue, normal tissue, lymphatic tissue, vascular tissue, and adipose tissue.
Intraoperative visualization of lymph nodes (A) and non visualized lymph nodes (B).
We collected 12 sets of sample tissues and categorized them by TNM staging, including 5 groups for GC and 7 groups for colon cancer. Optical coherence tomography and EUS were used to scan the mucosal and serosal layers of the tissue samples, which were subsequently processed into wax blocks for pathological sectioning. Clear and representative images were selected for observation, comparison, and analysis, with the results summarized as follows:
OCT can scan fat and blood vessels more clearly compared with EUS
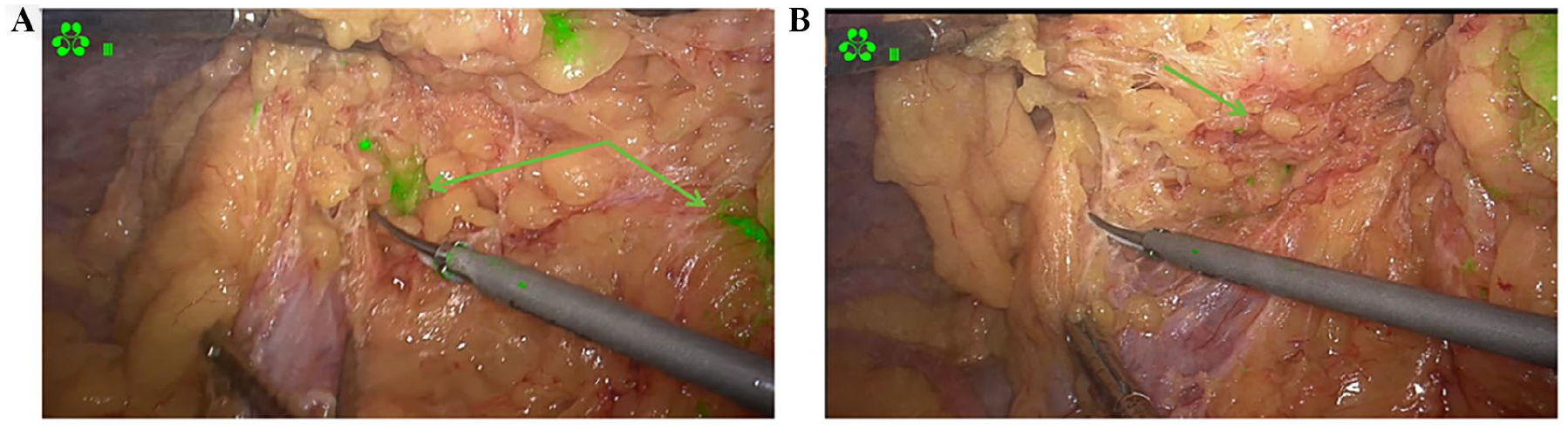
To differentiate from OCT and EUS images of tumor tissue, we also scanned adipose and vascular tissues (as shown in Figure 3). Pathological sections confirmed the nature of the tissues as adipose and vascular (Figure 3E). Optical coherence tomography images of adipose tissue (Figure 3A) revealed shadows, uniform coloration, and granular structures. The EUS images of adipose tissue displayed a uniform, consistent high-echo signal (Figure 3B). Vascular tissue appeared as a black shaded area (Figure 3C, green arrow), which was not clearly visible on the EUS images (Figure 3D).

Images of adipose tissue and vascular tissue. (A and B) OCT, EUS and pathology images of adipose tissue. (C and D) OCT, EUS and pathology images of vascular tissue. (E) Pathological sections of fat and blood vessels (HE staining). OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.
OCT and EUS have respective advantages in scanning normal gastric tissue
Before analyzing the tumor tissue images, we first scanned normal tissue using OCT and EUS (Figures 4 and 7). To further distinguish tumor tissues, we separately scanned the mucosal layer (Figure 4A and B) and serosal layer (Figure 4C and D) of normal gastric tissue. Both the superficial and deep layers of normal gastric mucosa are visible on OCT (Figure 4A) and EUS (Figure 4B) images, appearing as uniform, consistently dense shadows. The serosa, connective tissue, and intrinsic muscle layers are present (Figure 4C and D), with OCT images showing greater clarity compared with EUS.

Images of normal gastric tissue. (A and B) OCT and EUS images of gastric mucosal layer. (C and D) OCT and EUS images of gastric serosal layer. (E) Whole layer pathological section of stomach. OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.
OCT and EUS each have their own merits in scanning gastric tumor tissue
We scanned tissues with postoperative pathological diagnoses of T2 (Figure 5C, D, G and H) and T3 (Figure 6C, D, G and H). Figure 5 shows T2 stage images. Compared with Figure 4A and C, OCT images reveal uneven low-density shadows beneath the mucosal layer (Figure 5A, green arrows) and uniform density shadows in the serosal layer (Figure 5E), indicating tumor presence under the mucosa but no invasion of the serosal layer (Figure 5C and D). The EUS images display uneven high-density shadows in the mucosal, submucosal, and intrinsic muscle layers (Figure 5B and F, green arrows). Figure 6 shows the T3 stage images, with uneven low-density shadows visible in the submucosal layer on the OCT image (Figure 6A, green arrow), and uneven density shadows present in the serosal layer (Figure 6E, green arrow), indicating tumor invasion of the subserosal tissue, which is confirmed by pathological sections (Figure 6G and H). Uneven high-density shadows are visible in the mucosal, submucosal, intrinsic muscle, and serosal layers on the US images (Figure 6B and F). We observed that compared with EUS, OCT provides clearer delineation of tissue layers. The CK19 immunohistochemical staining of 2 cases of gastric malignancies in T2 and T3 stages exhibited uniformly intense brown-yellow staining, consistent with the hematoxylin and eosin (H&E) staining results. This further corroborates the potential of OCT and EUS. However, OCT is slightly limited in terms of scanning depth.

Images of gastric cancer tissue (TNM:T2). (A-D) OCT, EUS, and pathology images of gastric mucosal layer. (E-H) OCT, EUS, and pathology images of gastric serosal layer. OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.

Images of gastric cancer tissue (TNM:T3). (A-D) OCT, EUS, and pathology images of gastric mucosal layer. (E-H) OCT, EUS, and pathology images of gastric serosal layer. OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.
Images of normal colorectal tissue
In addition to gastric tissue, we also examined intestinal tissue. The following images display the mucosal layer (Figure 7A and B) and serosal layer (Figure 7C and D) of normal intestinal tissue. The OCT image of normal intestinal tissue reveals uniform and consistent density shadows, with the vascular lumen visible in the serosal layer scan (Figure 7C, green arrow). The lack of clear layering in EUS images may be attributed to poor tissue sample fixation and challenges in achieving accurate endoscopic US scans. This suggests that OCT may offer greater convenience compared with EUS scanning.

Images of normal colorectal tissue. (A-B) OCT and EUS images of colorectal mucosal layer. (C-D) OCT and EUS images of colorectal serosal layer. (E) Pathological sections of normal colorectal tissue (HE staining). OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.
Analysis of CRC tissue images
Subsequently, we scanned tissues confirmed by postoperative pathology as T1 (Figure 8C, D, G, and H) and T3 (Figure 9C, D, G, and H). Compared with the normal image in Figure 7, Figure 8 shows T3 stage images, with OCT revealing irregular low-density shadows on the mucosal surface and regular density shadows on the serosal surface (Figure 8A and E, green arrows). Endoscopic ultrasonography indicated tumor invasion in the submucosal layer, with no invasion observed in the lamina propria or serosal layers, which is consistent with pathological sections. Figure 9 shows T3 stage images, with OCT revealing low-density shadows in the mucosal and serosal layers (Figure 9A and E, green arrows), and EUS revealing high-density shadows (Figure 9B and F, green arrows), all consistent with pathological results and this was further validated through immunohistochemical analysis.
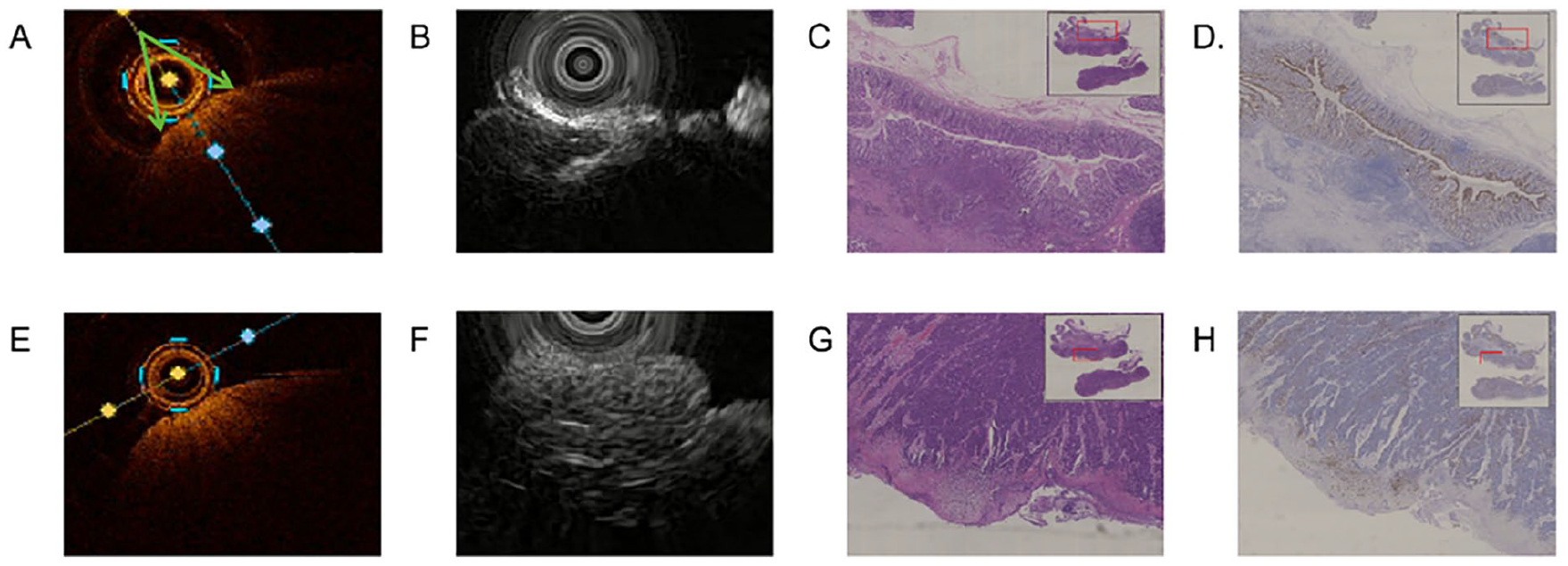
Images of colorectal cancer tissue (TNM:T1). (A-D) OCT, EUS, and pathology images of colorectal mucosal layer. (E-H) OCT, EUS, and pathology images of colorectal serosal layer. OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.
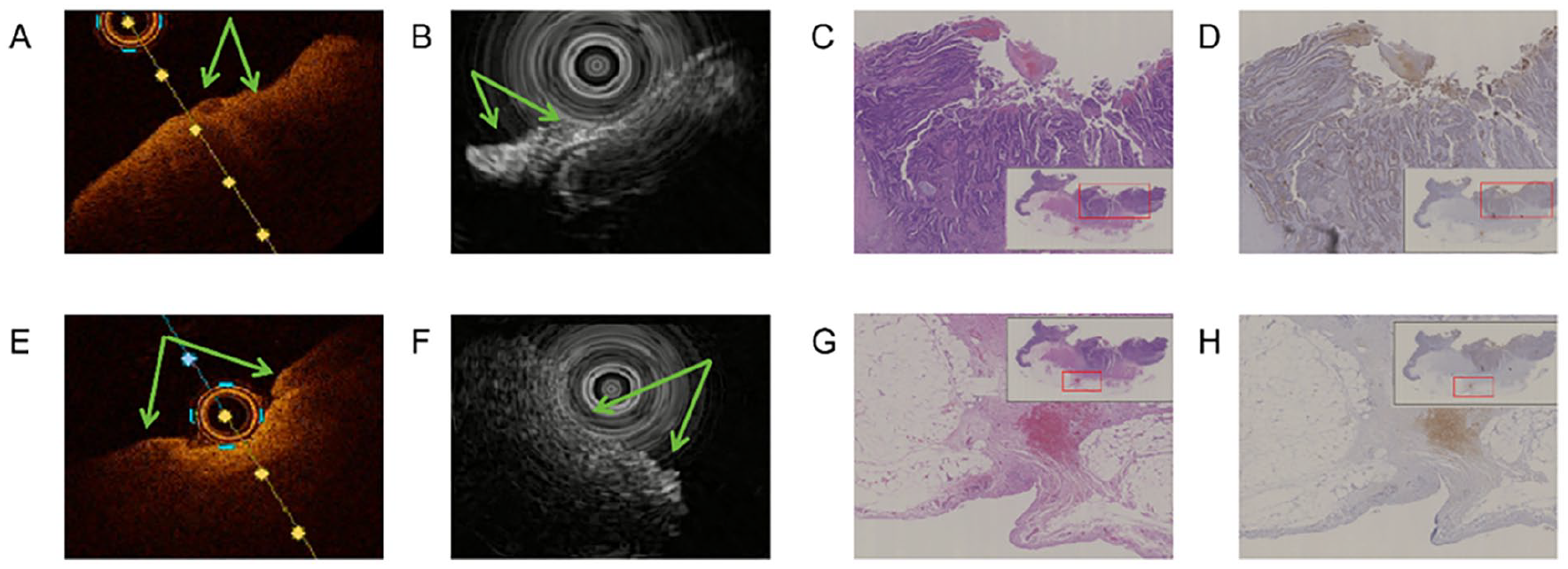
Images of colorectal cancer tissue (TNM:T3). (A-D) OCT, EUS, and pathology images of colorectal mucosal layer. (E-H) OCT, EUS, and pathology images of colorectal serosal layer. OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.
The scanning of lymph nodes to assess the feasibility of OCT and EUS-assisted lymph node dissection
To evaluate the feasibility of OCT and EUS in assisting lymph node dissection, we conducted further scans on lymph nodes (Figure 10). Immunohistochemical analysis of normal lymph nodes showed no CK19(+) expression (Figure 10D), and H&E staining revealed no significant abnormalities (Figure 10C). Optical coherence tomography imaging displayed uniform and consistent density shadows without abnormal low-density regions (Figure 10A), and EUS showed no abnormal discontinuous high-density shadows (Figure 10B). In positive lymph nodes, normal lymph node structures were disrupted, and structural loss was observed (Figure 10G). Immunohistochemical staining revealed CK19(+) expression (Figure 10H). Optical coherence tomography imaging showed irregular low-density shadows (Figure 10E, green arrow), while EUS demonstrated discontinuous abnormal high-density shadows (Figure 10F).

Images of negative and positive lymph nodes. (A-D) OCT, EUS, and pathology images of negative lymph nodes. (E-H) OCT, EUS, and pathology images of positive lymph nodes. OCT indicates optical coherence tomography. EUS indicates endoscopic ultrasound.
Finally, we summarized the imaging characteristics of OCT and EUS into a table (Table 2).
Comparison of imaging characteristics between OCT and EUS.

EUS, endoscopic ultrasound; OCT, optical coherence tomography.
To further investigate the ability of OCT and EUS to detect lymph nodes, we used a double-blind method to randomly enroll 18 patients, collecting 43 lymph nodes (Details are provided in Supplementary Figure 2 in the Supplementary material), including 10 men (55.6%) and 8 women (44.4%). The mean age of the cohort was 66.9 ± 11.3 years, ranging from 40 to 81 years. The mean height was 161.9 ± 8.4 cm, and the mean weight was 59.2 ± 9.2 kg. In terms of hematological parameters, the mean red blood cell count was 3.95 ± 0.65 × 1012/L, the mean hemoglobin level was 117.3 ± 26.4 g/L, the mean white blood cell count was 5.98 ± 1.55 × 109/L, and the mean albumin level was 40.8 ± 5.0 g/L. Regarding tumor markers, the mean level of carbohydrate antigen 19-9 (CA19-9) was 12.31 ± 12.91 U/mL, alpha-fetoprotein (AFP) was 2.27 ± 1.47 ng/mL, and carcinoembryonic antigen (CEA) was 3.29 ± 2.46 ng/mL. Missing values for tumor markers were excluded from the analysis. Regarding tumor staging, based on the cTNM classification, the patients were distributed across stages T1-T4, N0-N1, and Mx. Similarly, according to the pTNM classification, the patients were distributed across stages T1-T4, N0-N1, and M0-Mx. These baseline characteristics were consistent across the 3 imaging evaluation groups (OCT, EUS, and OCT-EUS), as they involved the same patient cohort, and no statistical comparison was performed between the groups. The final results are summarized in Table 3.
Comparison of lymph node detection rates between OCT and EUS.

EUS, endoscopic ultrasound; FN, False
Discussion
Optical coherence tomography can provide cross-sectional imaging with micrometer resolution, but its tissue penetration depth is limited. This technology is mainly used as the gold standard for the diagnosis of retinal diseases and to evaluate the characteristics of coronary plaques and stent adherence. Endoscopic ultrasonography combined with endoscopy and high-frequency US has the ability to penetrate deep tissue and is mainly used to evaluate the pancreatic biliary system. By judging the depth of tumor invasion (T stage) and lymph node metastasis (N stage); To achieve accurate sampling in EUS-FNA (fine-needle aspiration).
To investigate the feasibility of using OCT and EUS to accurately define surgical margins and assist in lymph node dissection, we scanned various tissues at different stages. Our findings indicate that both OCT and EUS are beneficial for surgical treatment. Compared with OCT, EUS can quickly scan the entire layer of tumor tissue; however, it requires fixed samples during the scanning process and is susceptible to interference. Moreover, the success of EUS procedures is highly dependent on the operator’s skill and experience. The accuracy of EUS largely hinges on the technical proficiency of the operator, as well as their ability to expertly interpret US images. 15 Extensive hands-on practice is required to achieve a high level of accuracy, with studies indicating that an operator must perform 225 to 250 EUS procedures to become proficient. This undoubtedly raises the level of difficulty and sets higher expectations for our surgeons. 16 This limitation may pose challenges when scanning tissues intraoperatively. We also compared OCT images at different stages, aiming to use OCT to determine tumor staging, assess patient prognosis, and further define the extent of surgical resection. However, our research revealed that OCT cannot reliably determine tumor staging, especially in samples from the T3 and T4 stages (Figures 5A, 6A, 8A, and 9A). Previous studies have shown that during surgeries for breast cancer and brain cancer, OCT can provide real-time imaging of tissue architecture, guiding resection margins and ensuring surgical precision. Our research further validates the potential application of OCT in gastrointestinal tumors. 17 We found that compared with EUS, OCT can accurately identify gastrointestinal tumor tissues regardless of the tumor stage. This capability is instrumental in accurately determining whether the surgical margin is positive or negative, potentially solving the issue of “how much to cut and how much to preserve” during surgery. Thus addressing the limitations of the ICG tracer to enable precise intraoperative navigation.
In our exploration of OCT’s role in assisting lymph node dissection, we discovered that OCT can more clearly differentiate lymph node structures and distinguish lymph nodes from adipose tissue with greater clarity (Figures 3A, 10A and E). This advantage may address the issue of incomplete lymph node dissection during surgery. Due to its ability to provide “optical pathological examination,” OCT holds significant promise in this context.
We further confirmed the advantages of combining multiple imaging techniques, providing robust statistical support for the clinical efficacy of OCT combined with EUS. This combination not only excels in detecting positive cases but also significantly reduces false-positive results, thereby enhancing the overall diagnostic accuracy. In terms of diagnostic indices, OCT combined with EUS demonstrated superior sensitivity (93.75%) and NPV (96.15%), which are critical in minimizing missed diagnoses and enhancing diagnostic reliability. These findings suggest that the combination of OCT and EUS leverages the strengths of both methods, potentially integrating complementary diagnostic features to achieve higher accuracy. Clinically, the high sensitivity and NPV of the combined method may reduce the risk of undertreatment and improve patient outcomes by ensuring a more reliable identification of lymph node pathology. Therefore, despite the lack of statistical significance in certain tests, the overall diagnostic performance of OCT combined with EUS highlights its potential as a preferred diagnostic strategy in clinical settings. While promising for real-time navigation, the therapeutic impact of OCT-EUS integration requires further prospective survival analyses.
In the future it is possible to integrate electronic gastroscopy (EGS), EUS, and OCT in our approach. This technique combines the visual capabilities of EGS with the “sharp eyes” of EUS and OCT to complement and enhance EGS performance. The value of OCT has been validated in other tumors,17-19 and we predict that this method could improve the detection rate of early gastrointestinal tumors. After pathological analysis confirms the nature of the lesion, for patients who meet the criteria for radical tumor resection (RTR), we may use OCT and EUS to improve the R0 resection rate.20,21 During the anastomosis process, we will use ICG and NIR to predict the likelihood of anastomotic leakage, allowing for the implementation of preventive measures in advance. 22 After completing nutritional support and other treatments, the specific chemotherapy or radiotherapy plan will be selected according to the patient’s actual condition. Conventional chemoradiotherapy effectively controls tumor lesions and significantly improves patient survival rates. However, the cytotoxic effects of chemoradiotherapy inevitably cause damage to normal tissues, leading to a range of side effects. 23 To address this, multiple technologies are possible to reduce the damage of chemotherapeutic agents to normal tissues. Chemotherapy-loaded nanoparticles combined with ICG offer not only excellent photodynamic and anticancer effects but also reduce cytotoxicity to normal tissues through NIR-based therapy.24,25 We aim to combine OCT with nanoparticle drug delivery to monitor tumor blood perfusion and structural changes in the tissue.
However, there are still limitations to this experiment, as pathology, as the “gold standard” for clinical diagnosis, cannot be completely replaced by OCT. In this study, OCT equipment was originally used for vascular imaging, and the resolution of scanning gastrointestinal tumor tissue was relatively average. There are studies indicating the possibility of “artifacts” in OCT imaging. 4 Therefore, OCT can be used to determine the extent of resection during surgery, but rapid frozen pathology remains the “gold standard.” In addition, the sample size in this study is relatively small, necessitating further validation through large-scale studies and investigations across various tumor types. Future research may explore the use of novel imaging tracers or the integration of advanced imaging technologies to further enhance intraoperative accuracy.
Conclusion
In conclusion, this study demonstrates that the combined application of OCT and EUS significantly enhances diagnostic sensitivity, specificity, PPV, and NPV. These findings provide clinical evidence supporting the integration of dual-modal imaging with laparoscopy for achieving precise intraoperative navigation.
Supplemental Material
sj-doc-1-onc-10.1177_11795549251363829 – Supplemental material for A Partially Randomized Diagnostic Trial of OCT-EUS Integration: Enhancing Surgical Margin Delineation and Lymph Node Dissection in Oncological Resection
Supplemental material, sj-doc-1-onc-10.1177_11795549251363829 for A Partially Randomized Diagnostic Trial of OCT-EUS Integration: Enhancing Surgical Margin Delineation and Lymph Node Dissection in Oncological Resection by Yutao Wang, Lu Zhao, Jiahao Zhao, Xuanyu Wei, Daorong Wang, Jun Liu, Linjie Hu, Bing Xu, Jun Ji, Jiayi Zhang, Chaowu Chen and Dong Tang in Clinical Medicine Insights: Oncology
Supplemental Material
sj-tif-2-onc-10.1177_11795549251363829 – Supplemental material for A Partially Randomized Diagnostic Trial of OCT-EUS Integration: Enhancing Surgical Margin Delineation and Lymph Node Dissection in Oncological Resection
Supplemental material, sj-tif-2-onc-10.1177_11795549251363829 for A Partially Randomized Diagnostic Trial of OCT-EUS Integration: Enhancing Surgical Margin Delineation and Lymph Node Dissection in Oncological Resection by Yutao Wang, Lu Zhao, Jiahao Zhao, Xuanyu Wei, Daorong Wang, Jun Liu, Linjie Hu, Bing Xu, Jun Ji, Jiayi Zhang, Chaowu Chen and Dong Tang in Clinical Medicine Insights: Oncology
Supplemental Material
sj-tif-3-onc-10.1177_11795549251363829 – Supplemental material for A Partially Randomized Diagnostic Trial of OCT-EUS Integration: Enhancing Surgical Margin Delineation and Lymph Node Dissection in Oncological Resection
Supplemental material, sj-tif-3-onc-10.1177_11795549251363829 for A Partially Randomized Diagnostic Trial of OCT-EUS Integration: Enhancing Surgical Margin Delineation and Lymph Node Dissection in Oncological Resection by Yutao Wang, Lu Zhao, Jiahao Zhao, Xuanyu Wei, Daorong Wang, Jun Liu, Linjie Hu, Bing Xu, Jun Ji, Jiayi Zhang, Chaowu Chen and Dong Tang in Clinical Medicine Insights: Oncology
Supplemental Material
sj-tif-4-onc-10.1177_11795549251363829 – Supplemental material for A Partially Randomized Diagnostic Trial of OCT-EUS Integration: Enhancing Surgical Margin Delineation and Lymph Node Dissection in Oncological Resection
Supplemental material, sj-tif-4-onc-10.1177_11795549251363829 for A Partially Randomized Diagnostic Trial of OCT-EUS Integration: Enhancing Surgical Margin Delineation and Lymph Node Dissection in Oncological Resection by Yutao Wang, Lu Zhao, Jiahao Zhao, Xuanyu Wei, Daorong Wang, Jun Liu, Linjie Hu, Bing Xu, Jun Ji, Jiayi Zhang, Chaowu Chen and Dong Tang in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
None.
Ethics approval and consent to participate
This study was approved by the Medical Ethics Committee of Northern Jiangsu People’s Hospital (Approval No. 2024ky183) in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants for both the procedures and use of their anonymized data for research purposes.
Consent for publication
All participants and patients provided written consent for publication of de-identified clinical data. Original consent forms are archived at the institution.
Author contributions
All authors met the following 4 ICMJE criteria for authorship:
1. Study conception & design: Daorong Wang, Dong Tang contributed to the research protocol development and methodology selection.
2. Data acquisition/analysis: Yutao Wang, Jiayi Zhang, Jiahao Zhao, Xuanyu Wei, Lu Zhao performed specimen collection, imaging procedures, and pathological correlation assessments.
3. Manuscript drafting/revision: Yutao Wang wrote the original draft, with critical intellectual revisions by Chaowu Chen, Bing Xu, Jun Ji, Jun Liu, Linjie Hu on diagnostic accuracy interpretation.
4. Final approval: All authors reviewed the manuscript and approved the submitted version, agreeing to be accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Social development project of the key R & D plan of Jiangsu Provincial Department of science and technology [No. BE2022773], the Graduate Research- Innovation Project in Jiangsu province [No.SJCX21_1644], the Academic Science and Technology Innovation Fund for College Students [No. 202011117056Y], the Social Development-Health Care Project of Yangzhou, Jiangsu Province [No. YZ2021075], and High-level talent “six one projects” top talent scientific research project of Jiangsu Province [No. LGY2019034], the Graduate Research-Innovation Project in Jiangsu province [No. SJCX22_1816]. The funding bodies had no role in the design of the study; in the collection, analysis, and interpretation of the data; and in the writing of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
Restrictions apply to the availability of these data due to patient privacy protections.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.