Abstract
Background:
This study aimed to determine the prognostic effect of the serum vitamin B12 level/CRP ratio (BCR) on patients diagnosed with solid cancer.
Methods:
Patients
Results:
The median age of the 344 patients included in the study was found to be 60.9 (range: 27-88) years. Around 56.6% (n = 174) of the patients had nondistant metastatic group and 49.4% (n = 170) had metastatic group. The median follow-up period of the patients was found to be 46.8 (range: 4-63) months. In the metastatic group, mortality was statistically significantly 2.1 times higher in those with a BCR cut-off value <3.5 (95% confidence interval [CI]; 1.4-3.2, P < .001). In the nonmetastatic group, mortality was statistically significantly twice higher in those with a BCR cut-off value <3.8 (95% CI; 1.4-3.3, P < .001). In addition, mortality was statistically significantly higher in high-risk patients in the nonmetastatic group (63.8% (n = 188) vs 45.5% (n = 156) (P < .001). The mOS period for patients in the high-risk group was 42.8 ± 1.4 (95% CI: 40.1-45.5) months, the mOS period for patients in the low-risk group was 51.5 ± 1.3 (95% CI: 49.0-54.0) months, and the mOS period for the entire group was 46.8 ± 1.0 (95% CI: 44.8-48.7) months (P < .001).
Conclusıons:
In conclusion, our study has shown that BCR, which has not been reported in the literature to date, is one of the cheapest and easily accessible inflammation markers that can determine prognosis in cancer patients.
Introduction
As survival times in cancer increase, the search for prognostic and predictive markers continues to increase day by day. Recently, as the characteristics and microenvironment of cancer cells have begun to be further elucidated, various studies have shown that nutrition (albumin), immune status (lymphocyte), inflammation markers (neutrophil, platelet, C-reactive protein (CRP)), serum vitamin levels, combined formulations of these values include immunonutritional indices (neutrophil to lymphocyte ratio (NLR), platelet to lymphocyte ratio (PLR), Prognostic Nutrition Index (PNI), CONUT etc.) have prognostic and predictive value in cancer, in addition to traditional prognostic factors such as histological subtype, disease stage, Functional scales (Karnofsky, Eastern Cooperative Oncology Group (ECOG)).1,2
Also, recent studies have indicated that vitamin B12 and CRP may also be prognostic and predictive markers.
As it is known, cancer-related inflammation negatively affects overall survival (OS). C-reactive protein, an inflammatory marker, is synthesized by hepatocytes in response to proinflammatory cytokines, such as interleukin 6, interleukin 1, and tumor necrosis factor-α, and in the literature, CRP is one of several inflammatory biomarkers associated with OS in patients with cancer. Studies have shown that high CRP levels are associated with early death after diagnosis. 3
Vitamin B12 (cyanocobalamin) that is another possible marker, a structurally complex, nonpolymeric biomolecule that plays an important role in a variety of biological processes, including the synthesis and methylation of deoxyribonucleic acid (DNA), and is a cofactor for 2 key enzymes in the conversion of homocysteine to methionine. 4 Although high B12 levels are seen in benign cases of liver diseases, long-term renal failure, autoimmune or inflammatory diseases, and Gaucher disease, studies have shown that B12 levels are also increased in myeloproliferative diseases, myeloid malignancies and hematological malignancies such as lymphoma, some solid cancers such as stomach, liver, pancreas, lung cancer, and liver metastatic malignancies.4 -8 The mechanisms that may link these conditions to elevated serum vitamin B12 levels are not fully understood, but it is thought that they may be related to release from damaged cells into the circulation (liver disease), decreased clearance (renal failure, liver disease), increased serum levels of tasacobalamin II or other transcobalamins, or increased haptocorrin. 9 The association between high levels of circulating transcobalamins and neoplastic or inflammatory diseases suggests that these proteins may act as an “acute phase reactant.” It has also been associated with high B12 levels in some cancers, tumor mass, or granulocytic immune response.1,8 Based on this information, studies on the prognostic value of B12 levels in cancer patients have reported an association between high serum vitamin B12 levels or hypercobalaminemia (HCbl) HCbl and poor prognosis in patients with solid tumors and hematological malignancies.1,10
Although both CRP and B12 have been shown to have prognostic value in studies, serum vitamin B12/CRP Index (BCI) (B12 [pmol/L] × CRP [mg/L]) was associated with a worse prognosis in the original study by de Geissbühler et al 1 and in the validation study by Kelly et al. 11
Based on this information, we aimed to reveal the prognostic value of the B12/CRP ratio (BCR) at the time of diagnosis in patients with solid cancer, assuming that the BCR, which has not been investigated in the literature to our knowledge, could be a prognostic biomarker.
Materials and Methods
Study design and population
In this retrospective study, patients diagnosed with solid cancer between 2016 and 2019 at the Dokuz Eylül University Faculty of Medicine, Department of Internal Medicine, Medical Oncology Department and who had serum CRP and vitamin B12 values at the time of diagnosis were evaluated. The inclusion criteria of the study were determined as follows: (1) being diagnosed with solid cancer, (2) receiving treatment for at least 3 months, (3) Having CRP and B12 values at the time of diagnosis, (4) Having complete data, (5) Being male and female aged 18 and over, (6) Being an outpatient. Among the solid cancer types, patients diagnosed with lung cancer, breast cancer, gastrointestinal system (GIS) cancers, hepatobiliary cancers, gynecological cancers, and urinary system cancers were included in the study. The exclusion criteria of the study were determined as follows: (1) Receiving vitamin B12 replacement in the last 6 months, (2) Being diagnosed with stomach cancer, (3) Being diagnosed with autoimmune and autoinflammatory diseases, (4) Having protein-losing enteropathy, (5) Having myeloproliferative or lymphoproliferative diseases, (6) Having glomerular filtration rate (GFR) <45 mL/min/1.73 m2, (7) Having acute or long-term liver disease (AST and ALT > 3 times), and (8) having diabetes mellitus diagnosis using metformin. All patients meeting the study criteria were consecutively included in the study. Therefore, there was no patient selection or bias since all patients who met the study inclusion criteria were included in the study. Demographic and clinicopathological characteristics of the patients at the time of diagnosis, complete blood count, biochemical laboratory values, vitamin B12 and CRP values, and patient death information were recorded retrospectively from the hospital database.
Ethical approval
This study was designed retrospectively in accordance with the principles of the Declaration of Helsinki, and the use of participant data was permitted without obtaining informed consent with the permission of the hospital administration. The requirement for written consent was waived by the Institutional Review Board/Ethics Committee because permission was obtained from the hospital administration to use patient data. Based on this, the study was approved by the Dokuz Eylül University Faculty of Medicine Non-Interventional Research Ethics Committee. (Date: March 1, 2021 /No: 2021/07-31). The reporting of this study conforms to the STROBE statement.
B12/CRP ratio
B12/CRP ratio was calculated by dividing the serum vitamin B12 (pmol/L) level by the CRP (mg/L) value at the time of diagnosis (BCR: value of vitamin B12 (pmol/L) / value of CRP (mg/L)). In our laboratory, the vitamin B12 level is in pg/mL and is converted to pmol/L in the index using the conversion factor 0.738. The unit of the CRP level is mg/dL in our laboratory and is converted to mg/L and used in the BCR calculation method. For nondistant metastatic group BCR cut off value was 3.8 while for metastatic group BCR cut off value was 3.5. In both the group nondistant metastasic and the metastatic group, those above these cut-off values were defined as low risk, and those below were defined as high risk.
Response and toxicity assessment
Tumor staging was performed according to “Eighth Edition of American Joint Committee on Cancer (AJCC) and the Union for International Cancer Control (UICC) TNM stage classification”. 12 Response assessments. It was made according to the “Response Evaluation Criteria in Solid Tumors (RECIST) v1.1 guidelines”. 13 Toxicity assessments were made according to the National Cancer Institute Common Toxicity Criteria (NCI-CTC). 14
Statistical analysis
Demographic characteristics, clinicopathological features, and blood sample results were collected from the hospital database. The Shapiro-Wilk Francia test was used to evaluate the conformity of the data to normal distribution, and the Levene test was used to evaluate the homogeneity of variance. In comparing 2 independent groups according to quantitative data, the Independent-Samples T-test was used with Bootstrap results, while the Mann-Whitney U-test was used with the Monte Carlo simulation technique. In comparing categorical variables with each other, Pearson chi-square, linear-by-linear association and Fisher-Freeman-Holton tests were tested using the Monte Carlo Simulation technique and Fisher Exact test Exact results, and the column ratios were compared with each other and expressed according to the Benjamini-Hochberg corrected P value results. Receiver operating characteristic (ROC) curve analysis was performed to determine the cut-off value of BCR in the entire population, in the nondistant metastatic and metastatic groups. Progression-free survival (PFS) was defined as the time from the date of starting of systemic treatment until progression, relapse, death, or last follow-up, which ever came first. Overall survival was defined as the time from the date of diagnosis to death or last follow-up. The median follow-up time in the study was calculated using the reverse Kaplan-Meier. Kaplan-Meier (product limit method)—Log Rank (Mantel-Cox) analysis was used to examine the effects of variables on survival times according to the determined cut-off value. For the nondistant metastatic and metastatic groups, gender, BCR, lactate dehydrogenase (LDH)/albumin, mean platelet volume (MPV), NLR, and PLR risk factors were evaluated with univariate and multivariate regression analyses. Quantitative variables are expressed in the tables as mean ± SS (standard deviation) and median (Minimum/Maximum or IQR25/75), while categorical variables are shown as n (%).
All statistical analyses were performed with the SPSS Statistics 24.0 for iOS software program (SPSS, Inc., Chicago, IL, USA), and variables were evaulated at a 95% confidence interval and statistical significance was determined as P < .05.
Results
Patients and clinicopathological characteristics
The median age of the 344 patients included in the study was 60.9 (range: 27-88) years, and although the distribution of male and female genders in the population was almost close to each other, the number of male patients was slightly higher at 52.3% (n = 180). Most of the patients (86.6%, n = 298) had ECOG PS 0, and the most common cancers in the study population were lung cancer, pancreatic cancer, breast cancer, colon cancer, rectal cancer, and prostate cancer, respectively. The nonmetastasis group and the metastatic group in the study were distributed evenly (50.6% (n = 174), 49.4% (n = 170), respectively). Clinicopathological features of the patients are shown in Table 1.
Clinicopathological features of the patients.

Survival analysis
The mean follow-up period in the study population was 46.8 (range: 4-63) months, and 55.5% (n = 191) of all patients died during the follow-up period. Median OS in the entire group was 46.8 ± 1.0 (95% CI: 44.8-48.7) months. The mortality rate in metastatic group patients was statistically significantly higher than in nonmetastatic patients (77.6% vs. 33.9%, respectively) (P < .001).
Effect of BCR on survival
The median serum vitamin B12 level of all patients included in the study was 292.5 (75-2000) pg/ml, and the median CRP value was 9.1 (0.5-412) mg/dL. The median vitamin B12 and CRP values of the deceased patients were statistically higher than those who survived, and a statistically significant relationship was found between vitamin B12 and CRP levels and mortality (B12: 335 pg/mL (97-1500 pg/mL) vs 262 pg/mL (75-2000 pg/mL), P = .002; CRP: 12.6 mg/dL (0.5-412) vs 5.6 mg/dL (0.6-283), P < .001).
The BCR cut-off value was determined as 3.8 according to ROC analysis in the nonmetastatic group (Figure 1). When the relationship between BCR value and mortality in this group was evaluated, mortality was twice as high in patients with BCR value below 3.8, which was statistically significant (95% CI; 1.4-3.3 P < .001). Around 63.8% of high-risk (n = 188, BCR < 3.8) patients and 45.5% of low-risk (n = 156) patients died, and this difference was statistically significant (P < .001). The median OS of patients in the low-risk group was statistically significantly longer than that of patients in the high-risk group (51.5 ± 1.3 months (95% CI: 49.0-54.0), 42.8 ± 1.4 months (95% CI: 40.1-45.5), P < .001) (Figure 2).

BCR cut-off value according to ROC analysis (nonmetastatic group).

Median overall survival according to BCR (Kaplan-Meier Analysis-Logrank Test).
The BCR cut-off value was determined as 3.5 according to ROC analysis in the metastatic group (Figure 3). The risk of death was statistically significantly 2.1 times higher in those with a BCR value < 3.5 (95% CI: 1.4-3.2; P < .001).

BCR cut-off value according to ROC analysis (advanced stage group).
The BCR value of surviving patients was statistically significantly higher than that of deceased patients (BCR 4.4 (0.1-125), BCR 2.6 (0.1-88.2), P = .001, respectively).
When evaluated according to cancer groups, the BCR cut-off value was calculated as 3.6 by ROC analysis in those with only GIS malignancy (n = 92) and the mortality risk was found to be statistically significantly 2.7 times higher in the group with BCR value < 3.6 (95% CI: 1.1-6.3, P < .049). The median OS in the group with all GIS malignancy was 44.6 ± 2.0 months (95% CI: 40.7-48.7). Median OS of patients with gastrointestinal (GI) malignancy in the low-risk group (n = 35) was statistically significantly longer than that of patients in the high-risk group (n = 57) (51.3 ± 2.8 (95% CI: 45.9-56.8) months vs 40.5 ± 2.6 (95% CI: 35.4-45.6) months; P < .014).
Univariate and multivariate analyses
In the univariate analysis evaluating the effects of gender, BCR, LDH/alb ratio (LAR), MPV, NLR, and PLR on mortality, it was shown that all these factors, in addition to BCR, were among the risk factors that had a statistically significant effect on mortality (Table 2).
Univariate analysis of factors affecting mortality.
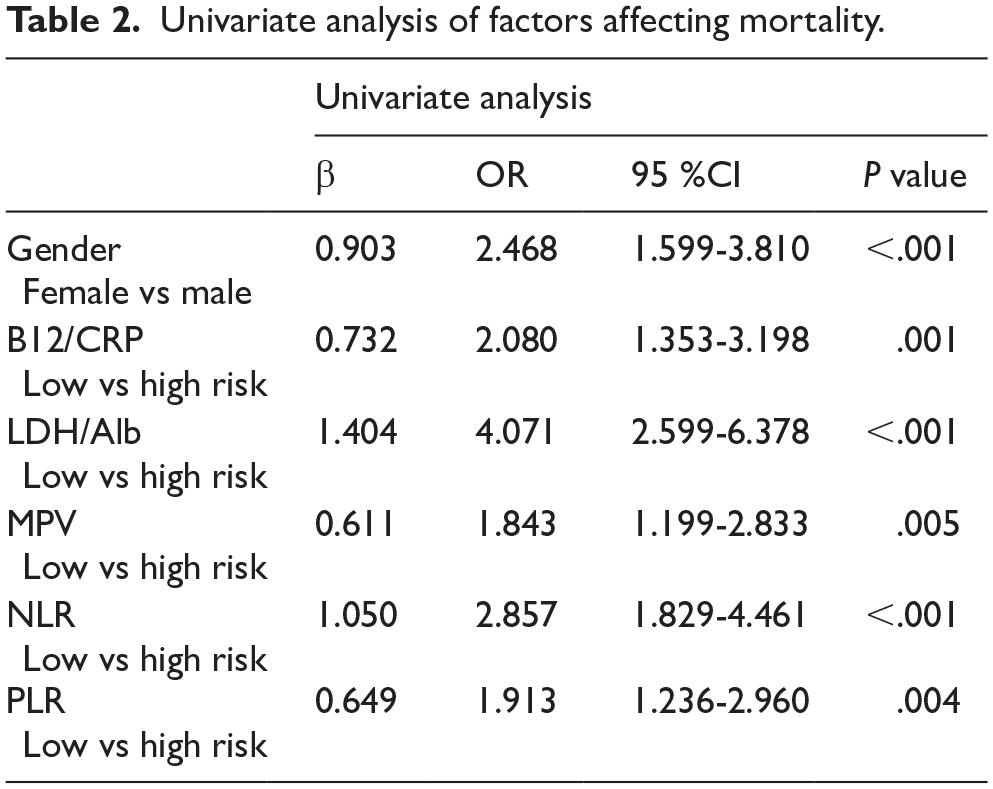
In multivariate analyses, it was seen that BCR value was not a statistically significant risk factor on mortality, while LAR and gender were shown to be the most statistically significant independent risk factors for mortality, being in the high-risk group (Table 3).
Multivariate analysis of factors affecting mortality.

Discussion
This observational retrospective study investigated the prognostic value of BCR at the time of diagnosis in patients with solid cancer. And cut-off values were determined for BCR for both the nonmetastatic group (BCR: 3.8) and metastatic group (BCR: 3.5). Patients below these values were grouped as high risk, and those above were grouped as low risk. In the group with low BCR, OS was shown to be shorter and mortality rate was higher in both nonmetastatic and metastatic disease. In the analysis performed according to cancer subgroups, it was seen that OS was longer in the group with high BCR in patients with GI malignancy. To our knowledge, this is the first study in the literature showing the prognostic value of BCR in patients with solid cancer.
Although there is no study in the literature on the prognostic value of BCR in solid cancers, there are studies on the prognostic value of vitamin B12, CRP, and B12×CRP Index (BCI) in patients with cancer diagnosis.1,3,11
When we evaluate the literature on the prognostic value of vitamin B12, CRP, and BCI in cancer, starting from vitamin B12, there have been some studies on the relationship between serum vitamin B12 values and cancer, including population-based cohort studies and studies demonstrating the relationship between cancer risk and mortality.5,7,4 In 2 population-based cohort studies investigating the relationship between vitamin B12 levels and cancer in people without a cancer diagnosis, both in the United Kingdom and in Denmark based on medical records, it was shown that the risk of cancer was higher in the group with higher vitamin B12 levels.5,7 A systematic review including 7 studies evaluating cancer mortality and 6 studies evaluating the risk of developing cancer also showed that there was a relationship between high vitamin B12 levels and the risk of developing cancer, but the relationship with mortality was less consistent. 4
C-reactive protein, another cancer-related biomarker other than vitamin B12, is a systemic inflammation marker and a measure of the inflammatory response, and high CRP values have been shown to be associated with advanced cancer stage, metastasis, and poor prognosis in many types of cancer. 15 , 16 In a meta-analysis evaluating the relationship between tumor recurrence and treatment response in solid cancers, including retrospective and prospective studies, it was shown that high CRP values were associated with higher mortality in 90% of those diagnosed with solid tumors, especially in GI malignancies and kidney malignancies, but also in other solid tumors (lung, pancreas, hepatocellular cancer, and bladder). 17 As seen when we reviewed the literature on the prognostic value of vitamin B12 and CRP in cancer, in our study, similar to the literature, the median CRP and vitamin B12 levels were higher in patients who died, and the mortality rate was also higher in cancer patients with high CRP and vitamin B12 levels.
As mentioned before, although there is no study in the literature on the prognostic value of BCI in cancer that we know of, there are several studies on the prognostic value of BCI by Geissbühler et al 1 and Kelly et al.3,11 Both the original study by Geissbühler et al and the validation evaluation by Kelly et al investigated the prognostic value of BCI on mortality, and it was shown that mortality was higher in the group with high BCI (>40 000). In addition, this study also showed that OS was shorter in the group with high vitamin B12 levels (>600 pmol/l), which is an indicator of liver involvement and inflammation. In our study, it is seen that the statistically significant higher vitamin B12 levels in the deceased patients are consistent with the literatüre.1,11 Apart from these studies, in the study conducted by Montegut et al, 3 where the prognostic value of BCI was evaluated in cancer patients in the geriatric age group, OS was longer in the group with low BCI levels (<10 000), and unlike other studies, it was also shown that high BCI levels were associated with fragility and impaired functional status in this study.
As aforementioned, studies have evaluated the using of vitamin B12, CRP, and BCI as prognostic biomarkers in cancer patients. As a result of these studies, it has been concluded that vitamin B12, due to the relationship between high levels of transcobalamins or haptocorrin in circulation and neoplastic or inflammatory diseases, these proteins can act as an “acute phase reactant,” and it has been shown that they can be used as a cancer prognostic biomarker.1,4,5,7 Another biomarker candidate, CRP, is secreted from hepatocytes with the effect of cytokines such as interleukin -6, interleukin-1, and tumor necrosis factor-α, and its value has been shown to increase in malignancy, and studies have concluded that it is an acute phase protein with prognostic value in cancer.1,15,16 After the prognostic value of both vitamin B12 and CRP in cancer was revealed, it was shown that BCI, which is obtained by multiplying vitamin B12 and CRP, has prognostic value in cancer patients, including in the geriatric cancer population.1,3,11 Based on all the information, we predicted that BCR, a new biomarker consisting of vitamin B12 and CRP, may have prognostic value in patients diagnosed with both metastatic and nonmetastatic solid cancer. In our study, similar to studies conducted with vitamin B12, CRP, and BCI in the literature, it was shown that BCR value has prognostic value in patients diagnosed with solid cancer.
This study had some limitations. First, it was a retrospective study, second, it reflected the results of data obtained from a single center, third, since no similar study has been conducted in the literature, an effective comparison cannot be made, and fourth, there is no standard cut-off value for BCR. We believe that if a multicenter and prospective study is conducted on this subject, it will contribute to BCR becoming a more valuable and meaningful prognostic marker and strengthen our results.
Conclusion
In conclusion, this study has shown that BCR can be an easy, accessible, and simple biomarker in patients with solid cancer diagnosis, and it has been concluded that BCR can be used in addition to traditional clinicopathological prognostic features in cancer. It can be used in daily practice, but to use a standard cut-off, validation studies are needed in addition to prospective and multicenter studies.
Footnotes
Acknowledgements
None.
Ethical Considerations
This study was designed retrospectively in accordance with the principles of the Declaration of Helsinki, and the use of participant data was permitted without obtaining informed consent with the permission of the hospital administration. The requirement for written consent was waived by the Institutional Review Board/Ethics Committee because permission was obtained from the hospital administration to use patient data. Based on this, the study was approved by the Dokuz Eylül University Faculty of Medicine Non-Interventional Research Ethics Committee (Date: March 1, 2021/No: 2021/07-31).
Author Contributions
MK, ECB, and TY: Conceptualization; ECB: Acquisition of data; MK: Methodology, Software, Data analysis; MK and TY: Interpretation of data; MK, TY, and ECB: Writing—Original draft preparation, Writing—Reviewing and Editing, Critical Review. All authors have participated in the review, approved the version to be published, and have participated in the work to the extent that they can take public responsibility for appropriate portions of the content. And also the final manuscript was reviewed and approved by all authors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed in this study are not publicly available but are available from the corresponding author on reasonable request.
Data Availability Statement included at the end of the article
