Abstract
Background:
Euthyroid sick syndrome (ESS) is associated with cancer staging and influences prognosis in patients with lung and various other solid tumors. Although previous studies have examined the association between ESS and factors such as age, body mass index, Eastern Cooperative Oncology Group performance status (ECOG PS), hemoglobin (Hb), and acute-phase reactants in patients with solid cancers, its relationship with 25-hydroxyvitamin D [25(OH)D] remains unclear. In this study, we investigated the factors associated with ESS and explored the relationship between 25(OH)D levels and ESS in hospitalized patients with solid cancers (HSC).
Methods:
This retrospective study included 105 patients with HSC. Laboratory parameters were assessed using the initial blood samples collected upon hospitalization. Patients with low fT3, normal or low fT4, and normal thyrotropin (TSH) levels were classified as having ESS.
Results:
The overall prevalence of ESS was 37.1%. Among HSC receiving supportive care, the ESS rate was 40.3%, whereas it was lower—28.6%—in those hospitalized for chemotherapy. A statistically significant association was observed between ESS and ECOG PS, C-reactive protein (CRP), and Hb levels. Specifically, a poor ECOG PS (P < .001), low Hb levels (P = .001), and elevated CRP levels (P < .001) were significantly associated with ESS. However, no significant relationship was found between 25(OH)D levels and ESS (P = .118).
Conclusion:
Euthyroid sick syndrome may be relatively common among patients with HSC and is more frequently observed in those hospitalized for supportive care. In this patient population, ESS was significantly associated with ECOG PS, Hb, and CRP; however, no significant association was identified between ESS and 25(OH)D.
Introduction
Euthyroid sick syndrome (ESS) is a clinical condition characterized by low free triiodothyronine (fT3) levels in the absence of intrinsic thyroid disease and with an intact hypothalamic-pituitary axis, thought to be driven by oxidative stress and various cytokines.1-3 Euthyroid sick syndrome has been observed in cases of malnutrition, liver disease, renal failure, in elderly individuals, and in patients with malignancies. Regardless of the underlying disease, ESS appears to correlate more strongly with disease severity than with disease type. Its pathophysiology remains controversial, with ongoing debate as to whether it represents an adaptive response or a maladaptive process at the tissue level.4-6
Previous studies have demonstrated an association between ESS and poor prognosis in cancer patients. The relationship between ESS and 25-hydroxyvitamin D [25(OH)D] has also been explored in the context of acute myocardial infarction (AMI). It is known that 25(OH)D regulates the expression of the deiodinase enzyme responsible for converting thyroxine (T4) to triiodothyronine (T3). Reduced deiodinase activity in AMI, low 25(OH)D levels in patients with ESS, and the potential benefits of 25(OH)D supplementation have been subjects of investigation.7-9
In this study, we aimed to determine the prevalence of ESS in hospitalized patients with solid cancers (HSC), both in those receiving chemotherapy with partial disease control and those undergoing supportive care without disease control. In addition, we sought to examine the association between ESS and low 25(OH)D levels, as well as the patient’s Eastern Cooperative Oncology Group (ECOG) performance status, hemoglobin (Hb), albumin-corrected calcium (cCa), C-reactive protein (CRP), and body mass index (BMI). 10
Materials and methods
Between July 2022 and December 2022, patients diagnosed with solid tumors and hospitalized for either supportive care or chemotherapy at the Department of Oncology, Erzurum Atatürk University Research Hospital, were consecutively enrolled in this single-center study.
Inclusion criteria comprised patients with solid tumors—specifically brain, head and neck, lung, gastric, colorectal, pancreatic, biliary, prostate, ovarian, bladder, testicular, cervical, breast, sarcoma, and skin cancers—who were hospitalized for chemotherapy or supportive treatment. Exclusion criteria included patients with missing data, hematologic malignancies, or those receiving thyroid or antithyroid medications. Demographic and clinical data, including age, sex, height, weight, tumor type, tumor stage (metastatic or non-metastatic), ECOG performance status, 25(OH)D, cCa, parathormone (PTH), thyrotropin (TSH), free thyroxine (fT4), fT3, CRP, and Hb, were retrieved from patient records and the hospital information system. Albumin-corrected calcium was calculated using the formula: (4 – albumin) × 0.8 + albumin. All laboratory parameters were measured using the first blood samples collected at the time of hospitalization. Euthyroid sick syndrome was defined as low fT3, normal or low fT4, and normal TSH levels. The study was approved by the Atatürk University Research Hospital Ethics Committee (30 March 2023; B.30.2.ATA.0.01.00/181). The study was conducted in accordance with the principles of the Declaration of Helsinki (1975), as revised in 2024, and its reporting adheres to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 11
Statistical analysis
In the study, descriptive data are presented as frequencies and percentages, while continuous variables are expressed as either mean ± standard deviation or median (minimum–maximum), as appropriate. Pearson χ2 test and the χ2 test for trend were used to compare categorical variables where applicable. The Kolmogorov–Smirnov test and histogram plots were employed to assess the normality of data distribution. For normally distributed variables, comparisons between independent groups were conducted using the independent-samples t test, whereas the Mann-Whitney U test was applied for non-normally distributed variables. A P value of <.05 was considered statistically significant. All analyses were performed using SPSS version 20.
Results
In total, 110 hospitalized cancer patients were enrolled in the study. Of these, 1 was excluded due to hematologic malignancy, 2 were excluded due to thyroid medication use, and 2 were excluded due to missing data. A total of 105 patients were included in the study, comprising 38 women (36.2%) and 67 men (63.8%). The mean age of the patients was 61.9 ± 13.2, and the mean BMI was 24.2 ± 5.5. The study population comprised 37 patients (35.2%) with upper gastrointestinal (gastric and esophageal) cancer, 19 patients (18.1%) with lower gastrointestinal (pancreatic, biliary, and colorectal) cancer, 18 patients (17.1%) with respiratory (lung) cancer, 7 patients (6.7%) with head and neck cancer, 10 patients (9.5%) with genitourinary cancer (prostate, ovary, bladder, testis, and cervix), 6 patients (5.7%) with breast cancer, and 8 patients (7.6%) with other cancers (sarcoma, skin, and brain). Among the cohort, 85 patients (81%) had metastatic disease, and 20 patients (19%) were non-metastatic. Overall, 28 patients (26.7%) were hospitalized for chemotherapy, and 77 patients (73.3%) were hospitalized for supportive care. Within the metastatic patient group, 24 patients (22.9%) were hospitalized for chemotherapy, and 61 patients (58.1%) were hospitalized for supportive care. In the non-metastatic patient group, 4 patients (3.8%) were hospitalized for chemotherapy, and 16 patients (15.2%) were hospitalized for supportive care. The ECOG performance status distribution was as follows: 26 patients (24.8%) with ECOG score 1, 38 patients (36.2%) with ECOG score 2, 34 patients (32.4%) with ECOG score 3, and 7 patients (6.7%) with ECOG score 4. Among the entire study population, 39 patients (37.1%) had ESS, while 66 patients (62.9%) did not (Figure 1 and Table 1).

The flow chart of this study. Euthyroid sick syndrome (ESS).
Descriptive demographic and clinical data of the study group.
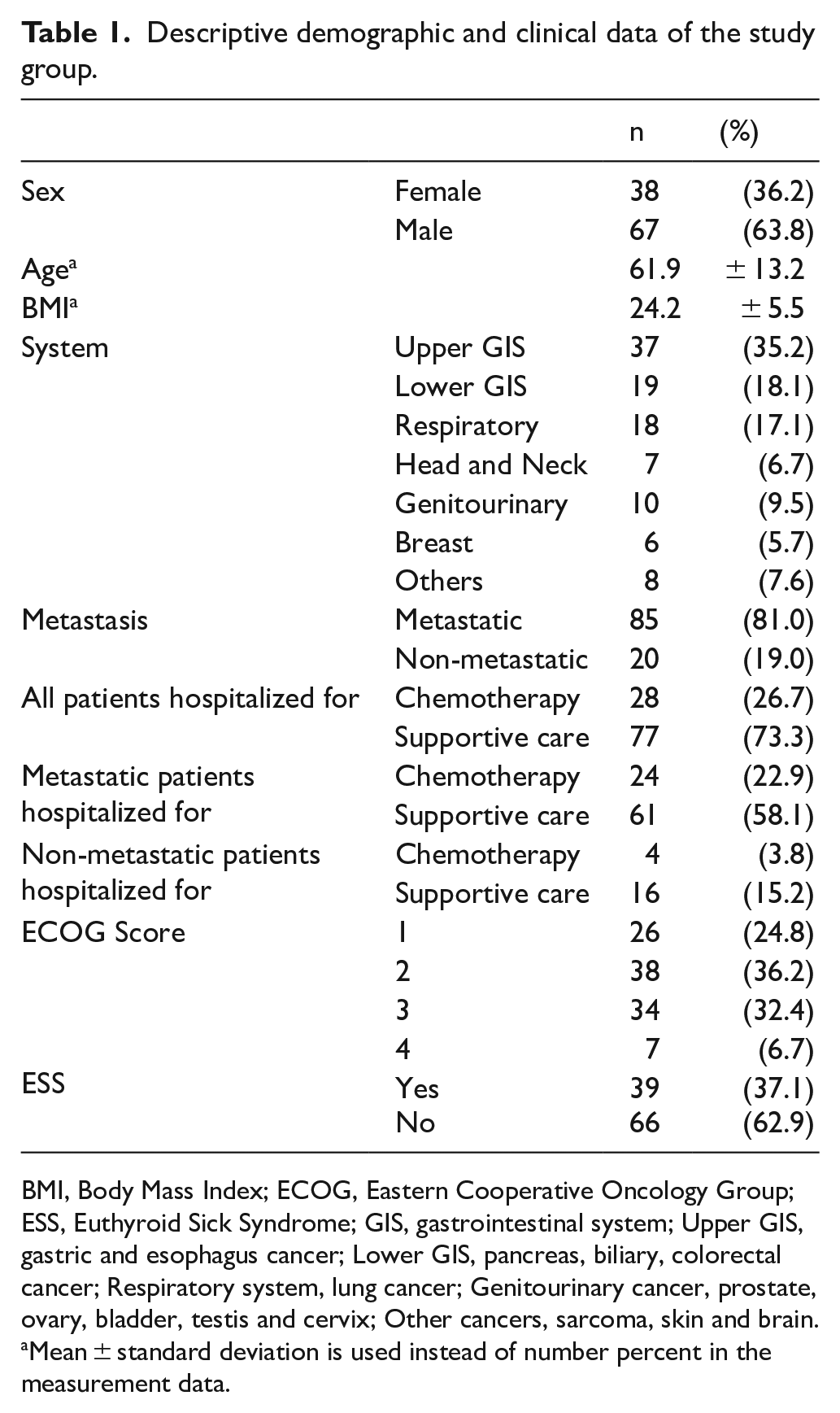
BMI, Body Mass Index; ECOG, Eastern Cooperative Oncology Group; ESS, Euthyroid Sick Syndrome; GIS, gastrointestinal system; Upper GIS, gastric and esophagus cancer; Lower GIS, pancreas, biliary, colorectal cancer; Respiratory system, lung cancer; Genitourinary cancer, prostate, ovary, bladder, testis and cervix; Other cancers, sarcoma, skin and brain.
Mean ± standard deviation is used instead of number percent in the measurement data.
A significant correlation was observed between the demographic characteristics of the study groups and ECOG performance scores. As the ECOG score increased, the proportion of patients with ESS also rose significantly (P < .001). However, no statistically significant association was found between ESS and sex, age, tumor type, tumor stage, reason for hospitalization, or BMI (P > .05) (Table 2).
Analysis of demographic characteristics, tumor type, tumor stage, and ECOG score characteristics by study group.

BMI, Body Mass Index; ECOG, Eastern Cooperative Oncology Group; ESS, Euthyroid Sick Syndrome; GIS, gastrointestinal system; Upper GIS, gastric and esophagus cancer; Lower GIS, pancreas, biliary, colorectal cancer; Respiratory system, lung cancer; Genitourinary cancer, prostate, ovary, bladder, testis and cervix; Other cancers, sarcoma, skin and brain.
Statistically significant P values in bold.
Pearson Kikare test.
Mean ± standard deviation is used instead of number percent in the measurement data.
In Independent Groups T test.
Trend Kikare test.
When the laboratory data were analyzed, ESS was significantly associated with CRP and Hb levels. Patients with ESS had significantly higher CRP levels than those without ESS (P < .001) (Figure 2), and significantly lower Hb levels (P < .001) (Figure 3). No statistically significant correlations were found between ESS and 25(OH)D, PTH, or cCa levels (P > .05) (Table 3).

Boxplot graph comparing C-reactive protein (CRP) levels in patients with and without euthyroid sick syndrome (ESS).

Mean hemoglobin levels (±1 SD) in patients with and without euthyroid sick syndrome (ESS).
Analyzing laboratory data based on study group.
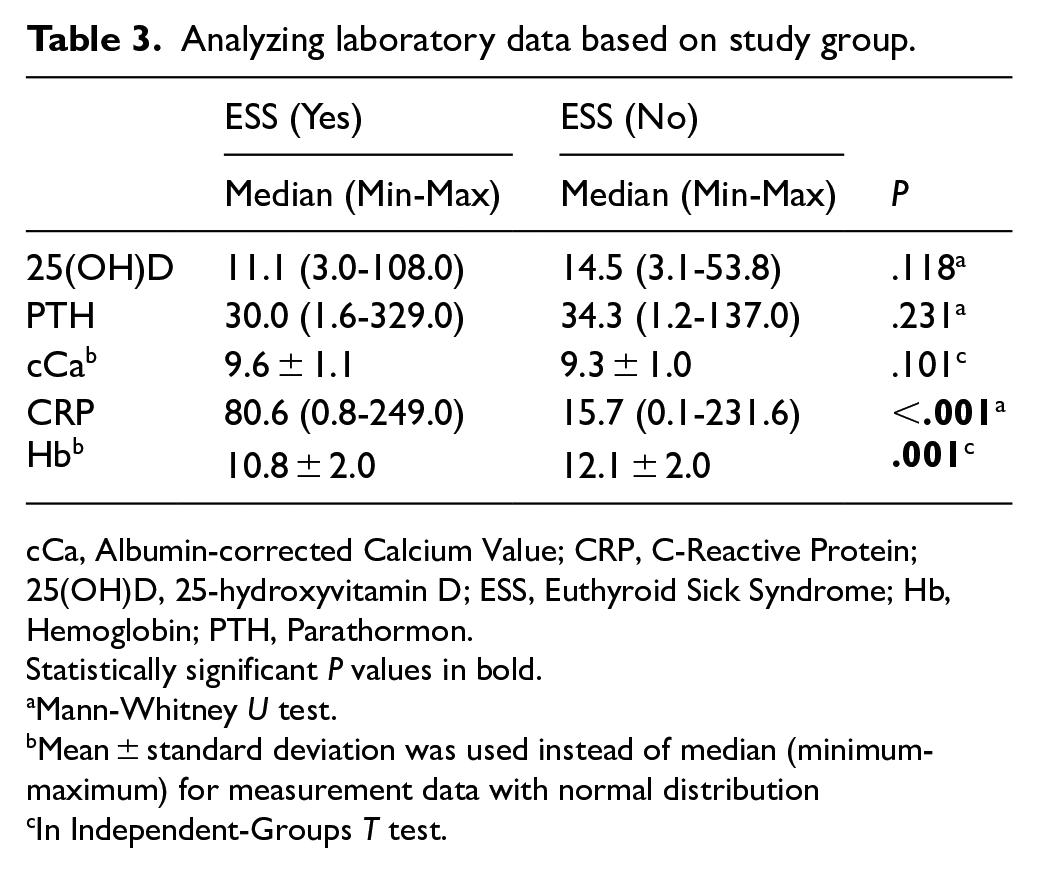
cCa, Albumin-corrected Calcium Value; CRP, C-Reactive Protein; 25(OH)D, 25-hydroxyvitamin D; ESS, Euthyroid Sick Syndrome; Hb, Hemoglobin; PTH, Parathormon.
Statistically significant P values in bold.
Mann-Whitney U test.
Mean ± standard deviation was used instead of median (minimum-maximum) for measurement data with normal distribution
In Independent-Groups T test.
Discussion
Cancer is associated with increasing rates of morbidity and mortality, making it essential to identify prognostic factors that influence clinical outcomes. One such factor is the presence of ESS, which is linked to disease severity and considered an indicator of poor prognosis.12-16
In the study of Tellini et al, 13 ESS was identified in 58% of 220 hospitalized elderly patients with malignancy. In our study, ESS was found to be 37.1%. While Tellini et al focused on an elderly patient population, our study included a broader, non-age-specific cohort. The higher ESS prevalence reported by Tellini et al may be attributable to differences in the age distribution of the study populations.
Cengiz et al 12 reported an ESS prevalence of 35% in 80 newly diagnosed patients with non-small cell lung cancer (NSCLC). In our study, the overall ESS rate was 37.1%, which aligns closely with their findings. However, different patient subgroups in our study exhibited varying ESS rates: 40.3% among patients hospitalized for supportive care and 28.6% among those hospitalized for chemotherapy. This difference may reflect the partial disease control achieved in patients undergoing chemotherapy.
In the study of Pingitore et al, 9 low 25(OH)D and ESS were investigated in patients with AMI. The presence of ESS in patients with AMI was found to be significantly associated with low levels of 25(OH)D, leading to suggestions for vitamin D supplementation due to its antioxidant effects in cardiac remodeling. However, our study found no significant relationship between ESS and 25(OH)D levels in hospitalized cancer patients. This discrepancy may be attributed to the acute nature of AMI versus the chronic course of cancer, as well as the still unclear pathophysiology of ESS.7,17,18
Bossoni et al 19 found a relationship between ESS and age in geriatric patients. In the study of Tellini et al, 13 no relationship was found between age and ESS in the oncological patient group. Similarly, our study did not reveal any relationship between age and ESS This may be attributable to the difference in the patient group and age distribution.
In a study by Disel et al 20 examining thyroid dysfunction and various parameters in 457 untreated cancer patients, no relationship was found between BMI and ESS. Similarly, our study found no relationship between BMI and ESS.
In the study conducted by Cengiz et al, a significant association was found between ESS and cancer stage, with ESS occurring more frequently in advanced stages. Notably, 53 of the 80 patients (58%) in their study were non-metastatic. In contrast, our study did not find a relationship between disease stage and ESS. This discrepancy may be explained by the predominance of metastatic patients in our cohort and the limited number of non-metastatic cases, as well as the absence of detailed staging. Furthermore, the higher prevalence of ESS observed in our non-metastatic patients may be related to the fact that most of this subgroup was hospitalized for supportive care.
In a study by Disel et al 20 on untreated cancer patients, the ECOG performance score was found to be associated with ESS. Similarly, in our study, ESS was found to be higher in patients with poor ECOG performance.
In contrast, Duyu et al, 3 in a study of 80 untreated pediatric cancer patients, reported no correlation between Hb levels and ESS. However, our findings revealed a significant association between Hb and ESS. This discrepancy may be due to differences in study populations, as Duyu et al focused on pediatric patients, whereas our cohort consisted of adult hospitalized patients.
In Cengiz et al 12 study of 80 newly diagnosed NSCLC patients, transferrin, an acute-phase reactant, was not found to be associated with ESS. In contrast, our study identified a significant association between ESS and CRP, another acute-phase reactant. The differences in findings may be attributed to variations in patient populations, as Cengiz et al included early-stage patients, while our cohort consisted of hospitalized patients without stage differentiation, and the acute-phase reactants studied were different.
There are some limitations of our study. A sample size calculation was not performed at the outset of our study, and the limited number of patients may have affected the statistical significance of the findings. In addition, including only hospitalized adult patients with solid tumors may have introduced selection bias, thereby limiting the generalizability of the results to outpatient cancer populations. Furthermore, survival data were not included. Factors known to influence vitamin D levels—such as seasonal variation, sunlight exposure, physical activity, nutritional status, and supplement use—were not evaluated. In diagnosing ESS, the hypothalamic-pituitary axis was assumed to be normal without performing the thyrotropin-releasing hormone (TRH) stimulation test; in addition, the study lacked a control group and was retrospective and single-center in design.
Conclusion
The association of ESS with disease stage and prognosis in patients with solid tumors underscores its clinical significance. The lack of disease control, poor ECOG performance status, low hemoglobin levels, and elevated CRP in HSC patients further highlight the relevance of these parameters. While 25(OH)D supplementation has been considered due to its association with ESS in AMI patients, our study found no correlation between ESS and 25(OH)D levels in HSC patients. Further research is warranted to elucidate the unresolved pathophysiology of ESS.
Footnotes
Acknowledgements
The authors would like to thank all staff and volunteers who contributed to this study.
Ethical Considerations
The study protocol was reviewed and approved by the Atatürk University Research Hospital Ethics Committee (March 30, 2023; approval number B.30.2.ATA.0.01.00/181).
Consent to Participate
Informed consent was waived by the Ethics Committee (Atatürk University Research Hospital) due to the retrospective nature of the study. The research was conducted in accordance with the ethical standards of the 1975 Helsinki Declaration, as revised in 2024.
Consent for Publication
Not applicable.
Author Contributions
Design of the work: MEB. Acquisition, analysis, or interpretation of data: ZH, AT, AAC, and PCE. Drafted the article: MEB. Revised it critically for important intellectual content: SBT, MB. Approved the version to be published: MEB. All authors reviewed and approved of the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability Statement
The datasets used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
