Abstract
Cryoablation is gaining attention as a minimally invasive treatment option for prostate cancer (PCa), offering a balance between effective oncological control and preserving genitourinary functions and quality of life. Focal cryoablation is emerging as a viable option for patients with PCa, particularly those who prioritize functional outcomes such as erectile functions and urinary continence. Whole-gland cryoablation, on the contrary, may be more appropriate for intermediate- and high-risk PCa where complete ablation of the prostate is necessary to ensure oncological control. Despite promising results, there is considerable heterogeneity in the available data regarding the long-term oncological and functional outcomes of cryoablation techniques, making it premature to issue definitive treatment recommendations. Further studies, particularly randomized controlled trials, are needed to clarify the role of cryoablation in PCa treatment. This narrative review aims to present the most relevant and up-to-date evidence on both focal and whole-gland cryoablation in PCa, providing a comprehensive overview of their current clinical applications, outcomes, and future potential.
Keywords
Introduction
Globally, prostate cancer (PCa) is the most common cancer in men across 112 countries, with an estimated 1.4 million new cases reported in 2020. 1 A majority of PCa cases are classified as low to intermediate risk, often not requiring radical treatments such as radical prostatectomy (RP) or radiation therapy (RT), which are associated with significant complications, particularly affecting urinary and erectile functions. 2
Given the drawbacks of radical treatments, there is an increasing interest in less invasive therapies like cryoablation. 3 Along with other focal therapies (FTs) such as irreversible electroporation, photodynamic therapy, or high-intensity focused ultrasound (HIFU), cryoablation has emerged as a promising PCa treatment option, aiming to reduce the functional complications while preserving favorable oncological outcomes.4 -6 In addition, it is becoming increasingly used as a salvage treatment following RT failure. Cryoablation specifically targets the freezing of cancerous tissue, resulting in cell death through mechanisms including the induction of apoptosis and necrosis. 7 The rapid freezing induces intracellular ice crystal formation, leading to mechanical disruption of cellular membranes and organelles. In addition, the freeze-thaw cycles cause osmotic imbalances, further compromising cell integrity and function. Vascular damage within the tumor microenvironment contributes to ischemia, exacerbating cell death. 8 Beyond these physical effects, cryoablation can stimulate an antitumor immune response. 9 The release of tumor antigens from lysed cells may activate dendritic cells, promoting a systemic immune reaction against residual or metastatic cancer cells. 10 This immunological aspect suggests that combining cryoablation with immunotherapies could enhance overall treatment efficacy. 4
The purpose of this narrative review is to evaluate the current evidence on cryoablation as both focal and whole-gland treatment for PCa, assessing its effectiveness, safety, and potential for integration into clinical practice as an alternative to more radical methods. The search strategy involved using keywords such as “cryoablation,” “cryosurgery,” “cryotherapy,” “focal,” “whole-gland,” “local therapy,” and “prostate cancer” and included searches in PubMed, Embase, and Web of Science. A total of 5129 articles and abstracts were initially identified, from which 63 references were ultimately selected. We focused on studies that provide insights into the mechanisms, clinical outcomes, and comparative effectiveness of cryoablation in PCa management.
This study is a narrative review, which carries certain limitations. Unlike systematic reviews or meta-analyses, our search strategy was nonsystematic, meaning that relevant studies may not have been exhaustively captured. While we conducted a comprehensive literature search across multiple databases, the selection of references was subjective, based on relevance and contribution to the topic rather than predefined inclusion criteria. In addition, the heterogeneity in study designs, patient populations, and outcome measures across the included studies limits direct comparisons and generalizability.
Current Guidelines
According to the European Association of Urology (EAU) guidelines, 11 whole-gland or focal cryoablation may be offered as a primary treatment option for patients with low- or intermediate-risk localized PCa. As a salvage therapy, cryoablation should be considered for patients with biopsy-confirmed locally recurrent PCa following RT failure.
The American Urological Association (AUA) guidelines12,13 suggest that whole-gland or focal ablation could be considered in men with intermediate- or high-risk localized PCa, especially in patients not suitable for RP or RT. As a salvage therapy, cryoablation can be offered in biopsy-proven locally recurrent patients with PCa who experienced biochemical recurrence (BCR) after RT.
The European Society for Medical Oncology (ESMO) guidelines 14 recommend cryoablation as a salvage therapy, whereas the AUA and the EAU guidelines consider it for both primary and salvage treatment. According to the ESMO guidelines, cryotherapy may be offered as a salvage treatment for patients with locally recurrent PCa after radical treatment, particularly following RT failure.
All guidelines endorse cryoablation as an option for localized PCa, highlighting its use within clinical trials and prospective registries. However, it remains an investigational therapy, warranting careful consideration of individual patient needs. While cryoablation may be effective in selected cases, all guidelines highlight the need for further long-term studies to better understand its outcomes. Furthermore, the guidelines lack standardized protocols for monitoring patients who have undergone FT. 15 Table 1 presents current guidelines concerning cryoablation in PCa.
Current guidelines concerning cryoablation in prostate cancer.
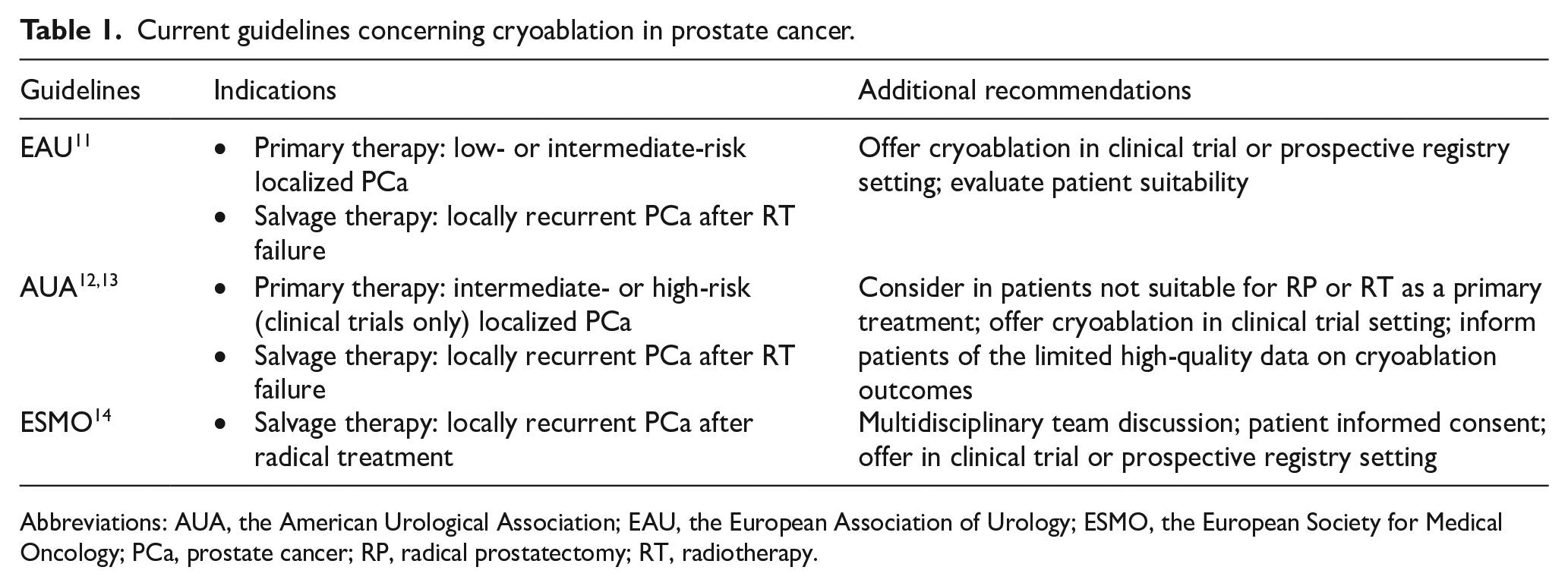
Abbreviations: AUA, the American Urological Association; EAU, the European Association of Urology; ESMO, the European Society for Medical Oncology; PCa, prostate cancer; RP, radical prostatectomy; RT, radiotherapy.
Cryoablation Techniques
Cryoablation is a minimally invasive procedure that uses extreme cold to destroy cancerous tissue through a controlled freeze-thaw cycle.7,16 The procedure involves inserting cryoprobes into the prostate under image guidance, often using transrectal ultrasound or magnetic resonance imaging (MRI) fusion. Liquid nitrogen or argon gas is commonly used to freeze the tissue, creating an “ice ball” around the targeted area. This process causes both immediate cell death and delayed apoptosis as blood flow to the region is disrupted. The most common prostate cryoablation systems involve the Visual ICE, SeedNet (Boston Scientific Corporation), or IceCure Medical (IceCure Medical Ltd).
There are 2 primary approaches to cryotherapy for PCa: focal cryotherapy, which targets specific areas of the prostate, and whole-gland cryotherapy, which freezes the entire gland. In addition, intermediate techniques such as hemigland and subtotal cryotherapy can be used to treat larger portions of the prostate while aiming to preserve some function. The debate between focal and whole-gland cryoablation in PCa centers on balancing functional outcomes with oncological control. 17
Whole-gland cryoablation
Whole-gland cryoablation involves the ablation of the entire prostate gland and may be considered for patients with more aggressive or recurrent PCa where comprehensive disease control is prioritized. While this approach aims to eradicate cancerous tissue effectively, it is associated with a higher incidence of complications, particularly erectile dysfunction (ED) and urinary incontinence (UI).
A 20-year retrospective study on 260 men (214 cT1, 43 cT2, 2 cT3, and 1 patient with missing data), evaluating primary whole-gland cryoablation, reported that 36% of patients experienced ED posttreatment, and 5% experienced UI. Despite these functional challenges, the study demonstrated favorable oncological outcomes, with a 10-year cancer-specific survival (CSS) rate of 80%. 18 In the study by Selvaggio et al, whole-gland cryoablation was evaluated as a treatment alternative to RP in 102 patients with localized PCa, including those at high risk for lymph node invasion (LNI). Recurrence-free survival (RFS) rates at 3 years were 93% for low-risk, 82% for intermediate-risk, and 72% for high-risk patients. At a median follow-up of 37 months, metastasis-free survival (MFS) was 97%, and additional treatment was required in 16% of cases. Importantly, oncological outcomes did not differ between patients with LNI probabilities above or below 5%, suggesting that a high preoperative risk of LNI may not be considered an exclusion criterion for whole-gland cryoablation. 19
In cases where whole-gland cryoablation is used as a salvage therapy following RT, the complication rates appear to be higher. Research indicates that 73% of patients experienced ED and 14% experienced UI post-salvage cryoablation. However, the procedure provided effective cancer control, with a 5-year BCR-free survival (BCR-FS) rate of 54%. 20
In the study by Deivasigamani et al, a systematic review was conducted to assess the mid- to long-term oncological and functional outcomes of whole-gland cryoablation for localized PCa. The analysis included 29 studies (14 on cryoablation and 15 on HIFU) with a median follow-up of 72 months. The findings revealed that BCR-FS at 10 years was 58%, whereas CSS and overall survival (OS) rates were 96% and 63%, respectively. RFS ranged between 71% and 79%, and MFS was 84%. From a functional standpoint, erectile function was preserved in 37% of cases, whereas 96% of patients achieved pad-free continence, with 1-year rates ranging between 97.4% and 98.8%. Reported complication rates included stricture formation (11%), urinary retention (9.5%), urinary tract infection (UTI; 8%), rectourethral fistula (0.7%), and sepsis (0.8%). 21
Focal cryoablation
Focal cryoablation may be a viable option for patients prioritizing preservation of function, especially those with localized or low-risk PCa. Whole-gland cryoablation remains appropriate for more aggressive or recurrent disease where disease control is prioritized. Advances in imaging, including multiparametric MRI (mpMRI) and prostate-specific membrane antigen–targeted positron emission tomography (PET) scans, have increased the precision of both focal and whole-gland therapies, optimizing patient outcomes in both approaches. 22 Borges et al 23 found that focal cryoablation significantly improves posttreatment sexual function and continence in comparison with the whole-gland procedure. At 12 months, patients undergoing FT had better International Index of Erectile Function (IIEF-5) scores (median 13 vs 9) and higher odds of preserving urinary continence (odds ratio [OR] 0.7). Sexual potency was also more frequently preserved with focal ablation (OR 7.7 at 3 months, OR 3.9 at 12 months).
In the recently published study by Ślusarczyk et al, systematic review and meta-analysis were conducted to assess the efficacy and safety of FTs for localized PCa based on data from prospective trials. The analysis included 54 studies with a total of 6040 patients, among whom 12 studies (n = 906 patients) specifically evaluated cryoablation. Cryotherapy and HIFU were identified as the 2 most extensively studied FTs. The primary endpoint, clinically significant PCa (csPCa) RFS, was 86% at 12 months and 81% at 24 months. In the intermediate-risk subgroup, the 12-month csPCa RFS was 79%. Five-year radical and systemic treatment-free survival was 80%, indicating that most patients did not require escalation to more aggressive treatments. The safety profile of FTs was favorable, with ⩾grade 3 adverse events occurring in only 3% of patients. UI requiring pad use increased by 3%, whereas new-onset ED was observed in 9% of cases. However, the study acknowledged a median follow-up of only 18 months, limiting the assessment of long-term oncological control. 24
Focal cryoablation for PCa relies on the accurate identification of the index lesion, typically using mpMRI. However, PCa is often multifocal, and mpMRI may not detect all cancer foci. 25 Studies have shown that while mpMRI has a high sensitivity for larger and higher-grade tumors, its overall sensitivity for tumor detection is around 47%, with increased sensitivity for larger (>1.0 cm) and higher-grade (Gleason ⩾ 7) tumors. 26 This limitation raises concerns about the potential for undetected high-grade lesions when relying solely on mpMRI for treatment planning. Therefore, while mpMRI is a valuable tool, its limitations necessitate a cautious approach, potentially incorporating additional diagnostic methods to ensure comprehensive assessment before focal cryoablation.
Overall, current evidence indicates that focal cryoablation offers better functional outcomes compared with radical therapies.27,28 However, the higher recurrence rates remain a significant concern, highlighting the need for long-term, randomized studies to establish its oncological efficacy. 29
Primary Versus Salvage Treatment
Primary treatment
The EAU guidelines indicate that primary cryoablation can be offered to localized patients with PCa within a clinical trial or a prospective registry setting. 30 Moreover, there are reports in the literature about the use of focal cryotherapy in intermediate-risk PCa, which up to now has not been recognized as a standard of care, but which produces noteworthy results, including a BCR rate of 16.7% and well preserved genitourinary function. 31 In addition, reports about the use of total prostate cryoablation for selected high-risk patients with PCa can be found. Among high-risk patients with PCa treated with primary whole-gland cryoablation the 5-year BCR-FS rate was 66.7% and the median time to BCR was 34.5 months. The authors emphasize the importance of using a preoperative nomogram to select patients with high-risk disease who will benefit from cryoablation and a perioperative nomogram to help identify patients who require early salvage treatment. 32
The 2023 meta-analysis demonstrated that the rates of BCR-FS, CSS, OS, RFS, and MFS for cryoablation and HIFU therapies are comparable to those of conventional treatment modalities in both intermediate- and long-term outcomes. 21
In summary, primary cryoablation may be considered for low-risk patients with PCa who prefer active treatment over active surveillance (AS), which remains the recommended management approach. It can also be offered to intermediate-risk patients who prioritize preserving functional outcomes over achieving optimal oncological control, as well as to selected high-risk patients where other treatment options may be less suitable.
Salvage treatment
Salvage cryoablation may be considered in patients with recurrent PCa after RT failure, as a strategy to delay the initiation of androgen deprivation therapy (ADT), avoid more invasive treatments such as salvage RP, and potentially defer the need for further systemic therapies.11,12,14 Volatility of the pooled rates of BCR-FS within 1 to 5 years of different local salvage therapies were compared. The results showed that the highest pooled rate of 5-year biochemical control was achieved by patients who underwent cryoablation (0.52; 95% confidence interval [CI]: 0.33-0.69) versus overall pooled rates for all salvage modalities 0.36 (95% CI: 0.27-0.47). In addition, cryoablation induced a quite low incidence of grade ⩾3 gastrointestinal toxicities (GI3) and was among the salvage treatment methods which presented low outcome (0.09; 95% CI: 0.04-0.14) of grade ⩾3 genitourinary toxicities (GU3). 33 Moreover, patients who underwent cryoablation presented the lowest (0%; 95% CI: 0%-1%) prevalence of urethral stricture among patients treated with different local salvage therapies 2% (95% CI: 1%-5%). It is worth noting that the prevalence of perineal pain was higher for cryoablation patients (8%; 95% CI: 3%-15%) than the overall pooled prevalence (3%; 95% CI: 1%-6%). Likewise, ED prevalence was higher among patients who undergo cryoablation (37%; 95% CI: 6%-76%) than the prevalence for all treatment subgroups. 27
The study by Tan et al 20 investigated oncological and functional outcomes in men undergoing salvage whole-gland cryoablation for radiation-resistant/recurrent PCa. It reported that the BCR-FS rates were 81% at 2 years and 71% at 5 years, with better outcomes in patients with lower prostate-specific antigen (PSA) nadir after the procedure. In terms of functional outcomes, 9% of patients experienced UI by 12 months, while erectile function remained significantly compromised, with median IIEF-5 scores dropping from 5 before the procedure to 1 after. This study suggests that salvage whole-gland cryoablation offers good oncological control with a relatively low rate of severe complications, making it a viable option for salvage treatment in selected patients.
The data from a systematic review of salvage FTs for radiorecurrent PCa revealed that BCR-FS rates at 3 years from 48.1% to 72.4% for salvage cryotherapy. Only salvage cryotherapy studies reported 5-year BCR-FS, with rates between 46.5% and 54.4%. This indicates that salvage focal ablation provides acceptable oncological control. However, the review highlighted significant heterogeneity in study design, patient selection, and reporting. 34
Cryoablation of metastatic lymph nodes
In a study by Mushtaq et al, 35 evaluating MRI-guided cryoablation for metastatic LNs in PCa, 95% of ablated LNs showed no significant activity on PET scans at a 4-month follow-up. In addition, 30.4% of patients had PSA levels drop to undetectable levels. Adverse events occurred in 10.9% of patients and resolved without long-term effects. The authors indicate that MRI-guided cryoablation is a safe and effective option for managing LNI in PCa.
In another study evaluating the feasibility, safety, and efficacy of computed tomography–guided percutaneous cryoablation for LNI, 15 patients with PCa were treated, accounting for 20 metastatic LNs. The study reported a technical success rate of 98% and local tumor control in 82% of treated LNs, with an 80% success rate specifically for PCa-related LNI. However, local progression occurred in 18% of cases, with a median time to recurrence of 11 months. Major complications were rare, with only 2 cases (3%) reported, including pneumothorax and bleeding. 36
These findings suggest that cryoablation is a feasible and well-tolerated treatment for metastatic LNs in patients with PCa, though further studies are needed to assess its long-term efficacy and role in oncologic treatment.
Oncological Outcomes
Numerous studies highlight the potential benefits of cryoablation as an FT for localized PCa. For instance, advancements in mpMRI have led to improved detection and localization of tumors, enabling more accurate focal cryoablation treatments with promising oncological outcomes and reduced risk of complications.37 -39 MRI–guided cryoablation, in particular, allows for better precision in targeting tumors, which reduces the likelihood of side effects compared with traditional ultrasound-guided methods. 39
The growing interest in cryoablation also stems from RP limitations, a more invasive procedure that carries a high risk of UI and ED. Studies on FT emphasize its appeal as a less invasive alternative, offering fewer complications and better functional outcomes.2,40 As such, cryoablation presents a viable option for patients seeking effective cancer control with a lower risk of morbidity.
Cryoablation is especially attractive for patients who may not tolerate more invasive treatments due to age or comorbidities. 41 Although longer follow-up is needed to make definitive conclusions, early evidence suggests that cryoablation provides oncological outcomes comparable to traditional treatments in terms of BCR-FS and overall cancer control.16,19,32 Moreover, clinical studies reinforce that cryoablation is effective in managing localized PCa, offering the advantages of repeatability, shorter recovery times, and reduced damage to healthy tissues. 16
The data from a systematic review included 24 papers that examined oncological outcomes of the focal cryotherapy. BCR rate after focal cryotherapy ranged from 0% to 67.5%, reflecting variability in patient selection and treatment protocols. The need for salvage treatment after focal cryotherapy was reported in up to 54% of cases when considering re-treatment with FT, and up to 66.7% when considering radical treatments. 42
In a retrospective study comparing partial gland cryoablation (PGC) to RP for intermediate-risk PCa, it was found that patients undergoing PGC had significantly higher rates of treatment failure. At 24 months, treatment failure rates for PGC were 33%, compared with 11% for RP. By 48 months, these rates increased to 43% for PGC and 14% for RP. The study also revealed that while metastasis development was low and similar between both groups (2.1% for PGC and 0.7% for RP), adjusted analyses showed that PGC was associated with an almost fivefold higher risk of treatment failure (hazard ratio 4.6, 95% CI: 2.7-7.9). These findings highlight the higher recurrence risk and need for salvage treatment following PGC during short-term follow-up compared with RP. 43
One of the major challenges in assessing oncological outcomes after focal cryoablation is the lack of a standardized BCR definition. Unlike radical treatments such as RP or RT, where BCR criteria are well established, FTs do not have universally accepted PSA-based recurrence thresholds. 11 This remains a key disadvantage of FT, making direct comparisons with conventional therapies difficult. Due to the absence of a standardized BCR protocol, most studies do not rely on BCR as the primary follow-up strategy after focal cryotherapy. Instead, mpMRI and scheduled systematic and targeted biopsies are utilized.44,45 In studies where BCR has been used as a follow-up measure, the Phoenix criteria (nadir + 2.0 ng/mL) or the ASTRO criteria (3 consecutive PSA rises) are the most commonly applied definitions.46-48 However, their applicability to FTs remains debated, as PSA kinetics after FT differ from those observed after whole-gland treatments. The continued reliance on imaging and biopsy for post-cryoablation monitoring highlights the need for future research to establish more precise and standardized BCR criteria for FT in PCa.
Table 2 presents an overview of ongoing clinical trials involving cryoablation in patients with PCa.
Ongoing clinical trials involving cryoablation procedures in patients with prostate cancer.

Abbreviations: mPCa, metastatic prostate cancer; n, number of patients; NCT, the National Clinical Trial; PCa, prostate cancer; US, United States.
Functional Outcomes
Several studies report results regarding ED and UI in patients with PCa following cryoablation. The differences in results are partly due to the timing of the functional assessments. Typically, improvements in both urinary continence and erectile function are observed over time after treatment. However, long-term follow-up data can be challenging to interpret, as new cases of ED and UI often arise independently of the procedure. This complicates the assessment of treatment-related outcomes versus naturally occurring age-related changes.
The rates of UI range from 0.0% to 25.3%.49-54 The rates of ED vary from 3.7% to 88.0%. 54 The wide range of results correlates with the extent of the procedure. In the case of focal procedures, there are lower rates of ED, with 3.7% and 46.0% reported in studies on hemigland cryoablation,50,51 and 75% reported in the study on whole-gland cryoablation. 50
Chinenov et al 49 compared urinary continence in whole-gland cryoablation and laparoscopic RP. In the cryoablation group, all patients maintained urinary continence, whereas 21% of the patients who had undergone RP experienced stress-induced UI after 6 months, and 15% after 12 months. In the study by Bossier et al, 50 postoperative and 1-year urinary continence rates were 60% and 83% for whole-gland cryoablation, respectively, and 72% and 83% for hemigland cryoablation, respectively. ED occurring de novo after cryotherapy was reported at 75% for whole-gland and 46% for hemigland cryoablation. In the study by Chuang et al, 51 patients underwent primary hemigland cryoablation. One patient experienced new-onset ED, which was successfully treated with an oral phosphodiesterase type 5 inhibitor. No patients experienced UI. In the study by Mercader et al, 52 after the procedure of whole-gland cryoablation, 95% of patients maintained full continence, with only 1.7% reporting UI after a year. However, all patients experienced some degree of dry ejaculation. Gregg et al 53 investigated the subtotal prostate cryoablation (approximately 85% of the gland). After 6 months, no patients experienced UI. In terms of ED, the results were monitored using the Erectile Function Questionnaire, the AUA Symptom Score (AUAss), and IIEF surveys. The baseline score, which was at 80%, deteriorated to about 40% at 3 months, then improved significantly to 60% at 24 months. Despite relevant improvements in subsequent evaluations, preprocedural levels were never fully regained by the cohort as a whole.
Tan et al 20 analyzed the outcomes of salvage whole-gland cryoablation for radiation-resistant PCa, assessing post-procedure UI and obtaining a result of 5% after 3 months and 9% after 12 months. In the study by Khan et al, 55 conducted on a group of 163 patients with localized PCa who underwent primary focal cryoablation, most men below the age of 65 maintained erectile function with or without the use of 5-phosphodiesterase inhibitors. Most cases of ED showed resolution naturally, without the need for pharmacologic intervention, within 9 to 12 months. In the focal cohort, only 3.1% of patients reported ED at any point during follow-up, and UI was observed in 1.8% of these patients.
In the meta-analysis examining ED after cryotherapy for PCa, the overall pooled incidence rate was 0.27 (95% CI: 0.26-0.28). 28 The incidence of ED was lower in patients who underwent cryotherapy as a primary treatment for localized PCa, with a rate of 0.49 (95% CI: 0.30-0.68). In patients who received salvage cryotherapy after RT failure, the incidence was significantly higher, at 0.61 (95% CI: 0.43-0.79). This highlights the greater risk of ED in salvage cryotherapy settings. Table 3 presents an overview of functional outcomes data in patients with PCa following cryoablation, as reported in selected studies.
An overview of data on functional outcomes in prostate cancer patients after cryoablation in selected studies.

Abbreviations: ED, erectile dysfunction; FU, follow-up; n, number of patients; UI, urinary incontinence.
One patient reported new-onset ED, which responded to oral phosphodiesterase type 5 inhibitor therapy.
Approximately 85% of the gland.
Complications
Complications following cryoablation for PCa include ED (3.7%-75%), UI (0%-17.5%), UTIs (17.5%), lower urinary tract symptoms (LUTS) (13.5%), including urinary retention (8.5%), perineal hematoma (11.0%), hematuria (9.6%), postoperative pain (4.5%), and epididymitis.49-53
In a study by Chinenov et al, 49 the authors compared the results of cryoablation and laparoscopic RP in the treatment of localized PCa. The average operation time was shorter with cryoablation – 127 minutes (ranging from 95 to 165 minutes) – compared with 140 minutes (ranging from 70 to 210 minutes) for RP. Hemorrhage was not observed when cryoablation was performed, whereas the average hemorrhage during RP was 216 mL. The average duration for catheter removal was 8 days after RP, with a maximum of 11 days. In the cryoablation group, the catheter was typically removed on the fifth postoperative day, ranging from 3 to 10 days. In addition, the pain scale score was lower for cryoablation compared with RP (0.7 vs 1).
In another study, 51 postoperative complications related to treatment were generally mild and short-lived. Two patients developed epididymitis, which resolved with oral antibiotics. Two others suffered from urinary retention, but this problem resolved after several weeks.
In the study by Mercader et al, 52 postoperative complications (apart from UI and some degree of ED) included UTIs (17.5%), hematuria (9.6%), perineal hematoma (11.0%), and postoperative pain (4.5%). Some degree of LUTS was reported (13.5%) and was treated conservatively; acute urinary retention occurred in 8.5% of cases.
Data from another study, 64 using the SEER-Medicare database, which included 21 344 men diagnosed with localized PCa, showed that men receiving cryotherapy were generally older and had more comorbidities compared with those treated with RP or RT. Complication rates associated with cryotherapy increased from 3 to 12 months, with UI at 9.8%, lower urinary tract obstruction at 28.7%, ED at 20.1%, and bowel bleeding at 3.3% by the 12th month. However, serious complications such as hydronephrosis, urinary, or bowel fistulas were not observed. Corrective invasive procedures for lower urinary tract obstruction and ED remained below 2.9%, indicating that while some complications were common, they were generally modest and manageable.
Conclusions
Cryoablation, especially in the focal setting, may offer a promising balance between oncological control and functional preservation in selected patients with PCa. Focal cryoablation is an interesting option for patients with low to intermediate-risk disease, whereas whole-gland cryoablation remains a viable option for those with intermediate-risk disease. Current data highlight cryoablation’s potential to minimize complications such as UI and ED, making it an appealing alternative to more invasive treatments. In addition, it can be used as salvage treatment after RT failure to postpone ADT. However, due to the heterogeneity in available data and study designs, further research is needed before making strong, evidence-based recommendations. One of key limitations is unclear patient selection for primary treatment. Thus, cryoablation should continue to be offered in clinical trials or specialized centers with careful selection of patients.
Footnotes
Ethics considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Jakub Karwacki, Bartosz Małkiewicz: conceptualization and design.
Justyna Kiełbasa, Zuzanna Szczepaniak, Jakub Karwacki: acquisition of data.
Karol Zagórski, Jakub Karwacki, Maximilian Kobylański, Adam Gurwin, Wojciech Krajewski: analysis and interpretation of data.
Jakub Karwacki, Justyna Kiełbasa, Zuzanna Szczepaniak, Karol Zagórski, Patryk Patrzałek: drafting of the article.
Bartosz Małkiewicz, Tomasz Szydełko, Maximilian Kobylański, Adam Gurwin, Dawid Janczak, Wojciech Krajewski: revising the article.
Bartosz Małkiewicz, Tomasz Szydełko: obtaining funding.
Tomasz Szydełko, Adam Gurwin: administrative, technical or material support.
Bartosz Małkiewicz: supervision.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant support: SUBZ.C090.24.089 Wroclaw Medical University and SUBZ.C090.25.095 Wroclaw Medical University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availabilty statement
Not applicable.
