Abstract
Background:
To evaluate the efficacy and safety of cisplatin combined with alternating temozolomide (TMZ) for recurrent high-grade glioma, as current treatments lack standardized protocols and predictive markers.
Methods:
This study evaluated cisplatin (20 mg/m2 IV, days 1-3) and TMZ (125 mg/m2 orally, days 1-7 and 15-21) in 35 patients, using the RANO criteria with 6-month progression-free survival (PFS-6) as the primary endpoint. The Kaplan-Meier analysis was applied for survival, and tumor molecular profiles were retrospectively assessed.
Results:
A median follow-up time was 61.2 months. The PFS-6 rate was 45.2%, and the median time to progression was 5.07 months. Four patients showed partial response, 16 had stable disease, and 11 had disease progression, with predominantly grade I to II toxicities. Low CD8+ tumor-infiltrating lymphocytes (TILs) correlated with improved disease control (P = .031). Data from the CGGA showed that low CD8+ TILs were associated with better survival, while high CD8+ TILs indicated increased immune response and higher immune checkpoint expression, including programmed death 1 (PD-1).
Conclusions:
The cisplatin plus alternating TMZ regimen is feasible and safe for recurrent high-grade gliomas, with low CD8+ TILs potentially predicting favorable responses.
Keywords
Introduction
High-grade glioma (HGG) is the most common malignant brain tumor including anaplastic astrocytoma, anaplastic oligodendroglioma, and glioblastoma multiforme (GBM). Almost all primary HGG will relapse inevitably after radiotherapy and temozolomide (TMZ) adjuvant chemotherapy. However, there is still no standardized protocol for treating patients with recurrent HGG. The outcome of patients with recurrent GBM and anaplastic glioma is 25 to 30 weeks and 39 to 47 weeks, respectively. 1 Therefore, novel chemotherapeutic protocols, especially drug combination for these patients, require further exploration.
In addition, researchers have demonstrated that different dose intensities of TMZ, the most common chemotherapeutic drug in glioma, could have different efficacy in patients with brain glioma.2,3 The TMZ is an alkylating agent that primarily works by methylating DNA at the O6 position of guanine. This methylation can trigger apoptosis, especially in glioma cells with low O6-methylguanine-DNA methyltransferase (MGMT) activity, as these cells are less able to repair the DNA damage induced by TMZ. Patients treated with an alternating weekly TMZ regimen will be under longer periods of drug exposure compared with the standard 5/28-day regimen. The exposure may deplete MGMT and thus increase the cytotoxicity of TMZ toward glioma.3-5 Alternating 1 week-on/1 week-off TMZ regimen has been proved to be feasible, safe, and effective in patients with recurrent glioblastoma. Cisplatin (CDDP) functions by forming platinum-DNA adducts, leading to cross-linking of DNA strands, which disrupts DNA replication and transcription, eventually triggering cell death pathways. The CDDP has been reported in the treatment for patients with recurrent glioma combined with TMZ. Several phase II trials have shown this regimen is safe and efficacious.4,6 However, to our knowledge, most of the regimens in these trials are cisplatin plus standard 5-day TMZ administration. The safety and efficacy of cisplatin and alternating weekly TMZ combination are still unknown.
Tumor microenvironment and tumor-infiltrating lymphocytes (TILs) play an important role in improving clinical survival and affecting treatment response in many solid tumors such as breast cancer, lung cancer, and metastatic melanoma.7-9 Many tumors, especially glioma, are poorly immunogenic and resistant to cytotoxic T lymphocyte-mediated cell lysis. Cisplatin has been reported to enhance immune infiltration in ovarian cancer and lung cancer.10-12 The relationship between TILs and response in recurrent glioma treated with cisplatin and TMZ remains unclear.
We, therefore, performed a single-arm phase II trial for patients with recurrent HGG to assess its safety and efficacy. We retrospectively conducted molecular profile investigation and immune-biologic evaluation in the tumor microenvironment to perform further exploratory analyses for biomarkers.
Patients and Methods
Patients with recurrent malignant glioma were enrolled in the study. The primary tumor was required to be histologically diagnosed as GBM or WHO grade II to III astrocytoma/oligodendroglioma/oligoastrocytoma according to the pathological classification at the time of diagnosis. Multimodal imaging such as postcontrast T1-weighted and T2-weighted fluid-attenuated inversion recovery (T2/FLAIR) was used to identify tumor recurrence in patients with HGG or malignant progression in patients with WHO grade II glioma. Additional eligibility criteria included age between 18 and 75 years, Eastern Cooperative Oncology Group (ECOG) 13 score 0 to 2, neutrophil count ⩾1.5 × 109/L, platelet count ⩾100 × 109/L, hemoglobin ⩾8 g/dL, blood urea nitrogen, serum creatinine, and total bilirubin <1.5 times the upper limits of normal (ULN), alanine aminotransferase (ALT) and aspartate aminotransferase (AST) < 3 times the ULN, and alkaline phosphatase (ALP) < 2 times the ULN. All patients provided written informed consent before the initiation of treatment. The reporting of this study conforms to the CONSORT 2025 statement (see Supplementary Materials).
Drug administration
The CDDP was administered intravenously (IV) at 20 mg/m2, daily on days 1 to 3, with 500 mL of normal saline over a 2-hour infusion. The TMZ was administered orally at a starting dose of 125 mg/m2, daily on days 1 to 7 and days 15 to 21. The treatment regimen was repeated every 28 days. Ondansetron (40 mg orally) was prescribed as antiemetic prophylaxis 30 minutes prior to TMZ administration, and dexamethasone (5 mg IV) was prescribed 30 minutes prior to CDDP administration. Patients were monitored for toxicity according to CTCAE v4.0. Complete blood count (CBC) was assessed every week. Other hematological toxicity was assessed every 2 to 3 weeks. Each cycle was initiated only if the white blood cell count was >4 × 109/L and the platelet count was >100 × 109/L. Treatment should be discontinued if grade 4 non-hematological toxicity occurs.
Efficacy measures and statistics
Patients were evaluated radiologically with magnetic resonance imaging (MRI) scans (T1, T2, T2/FLAIR, postcontrast T1) to investigate response every other cycle. The investigators assessed response and disease progression according to the RANO criteria, including complete response (CR), partial response (PR), stable disease (SD), and progression disease (PD). 14 In this study, we defined patients with CR, PR, and SD as clinical responders, while those with PD were classified as non-responders. Time to progression (TTP) was defined as the time from treatment initiation to the date of progression. Duration of response was defined as the time from the date of the best radiological response to the date of progression. Overall survival (OS) time was calculated from the date of treatment initiation to the date of death from any cause. The Kaplan-Meier method and Cox regression were conducted to evaluate the prognostic value of clinical and pathological variables. Fisher’s exact test was conducted to detect the difference in the distribution of variables between responders and non-responders. P < .05 was considered statistically significant.
Posthoc analysis
According to our previous study, we performed the exploratory analysis including molecular profile investigation to examine the status of IDH1/2, TERT promoter, MGMT promoter and 1p/19q codeletion, and immunohistochemistry (IHC) to evaluate the expression status of CD3, CD4, CD8, and their relevance to our treatment regimen.
The mutational status of IDH1/2 and TERT promoter was assessed through Sanger sequencing as previously described. 15 Status of MGMT methylation and 1p/19q codeletion were detected by pyrosequencing and fluorescence in situ hybridization (FISH) as previously described. 15 The expression of CD3, CD4, and CD8 was evaluated as previously reported. 16 All IHC images were blindly evaluated by 2 experienced observers.
Bioinformatic analysis
We explored the associations between tumor microenvironment and survival outcomes in the Chinese Glioma Genome Atlas (CGGA) database (http://www.cgga.org.cn/). In the CGGA database (n = 693), patients with HGG (excluding the oligodendroglioma suggested by histology and molecular profiles) who underwent chemotherapy without radiotherapy after surgery were selected (n = 65) for analysis. The clinical materials and RNA sequencing data were downloaded from the CGGA website. The ESTIMATE and MCP-counter packages were applied to calculate immunescores reflecting tumor immunogenicity and abundance of CD8+ T lymphocytes in tumor tissues with gene expression, respectively.17,18 Patients were divided into low and high groups according to the infiltration level of CD8+ T cells by the median value. The sensitivity of each group to chemotherapy was compared, and the difference in immune status between the 2 groups was analyzed.
Results
Patient characteristics
Forty-five patients were enrolled in the study from August 2014 to October 2017. Ten patients quit the study before treatment for various reasons. Four patients quit after treatment without radiological assessment (Figure 1A and B). For the 31 measurable patients, the clinical characteristics are shown in Table 1. The histology at the baseline assessment consisted of 17 GBM, 6 astrocytomas, 4 oligodendrogliomas, and 4 oligoastrocytomas. The ratio of male to female is 1.38:1. The median age was 47 (range = 23-70). Before enrollment, 29 of 30 patients received radiotherapy. Twenty-six of 30 patients received chemotherapy. Sixteen patients received concomitant radio-chemotherapy, followed by adjuvant TMZ. Other adjuvant chemotherapy regimens included ACNU and VM-26. Median follow-up time was 61.2 (95% confidence interval [CI] = 56.45 to 65.95) months, with follow-up ending in June 2020.

Overview of enrollment, treatment, and follow-up. (A) Flow chart of patients’ inclusion and follow-up. (B) Schedule of cisplatin plus alternative temozolomide administration in this study. (C) Swim lane plot of all measurable patients.
Patient characteristics.

Survival analyses
According to the RANO criteria, 4 patients had a PR, which is the best radiographic response; 16 patients had SD and 11 patients had disease progression without evidence of radiographic response (Figure 1C). Outcome analyses are shown in Figure 2A. The PFS-6 was 45.2%, and the median TTP was 5.07 months. The median OS time was 18.8 months, with 61.3% and 37.5% survival rates at 1 year and 2 years, respectively. The objective response rate (ORR) was 12.9%, and the disease control rate (DCR) was 64.5%. The median duration of response was 8.38 months in the clinical respondents (n = 20). Of note, clinical responders had both significant PFS and OS benefits compared with non-responders (P < .0001 and P = .00016, respectively, Figure 2B and C).

Kaplan-Meier survival curves for 31 measurable patients. (A) Curves for progression-free survival, overall survival, overall survival from recurrence, and duration of response in the study cohort. (B, C) Curves for progression-free survival, overall survival, and overall survival from recurrence showed responders had significant survival benefit against non-responders.
Univariate analysis showed that age, tumor location, clinical response, and MGMT methylation status had a prognostic impact (P < .1) on survival benefit (Figure 3A and Supplemental Table S1). Multivariate Cox regression showed that clinical responders had significant PFS and OS benefit against non-responders. Also, patients with MGMT promoter un-methylation had a borderline increased risk with a shorter overall survival time. Patients over 40 years old had a significant OS benefit compared with those under 40 years old (Figure 3B and Supplemental Table S2). The distribution of radiological responses in different clinical variables showed that patients with frontal tumor location had a better DCR than those with non-frontal location (14/16 vs 6/15, P = .008). There are no significant different distributions of other clinical variables between responders and non-responders (Figure 3C and Table 2).

Prognostic value of clinicopathological and immunobiologic variables in patients treated with cisplatin plus alternative temozolomide. (A) Univariate analysis of clinicopathological variables for progression-free survival and overall survival probabilities. (B) Multivariate forest plot showed hazard ratio for progression-free survival and overall survival in different subgroups. (C) Distribution of response with molecular and immunobiologic variables.
Association of response with clinical, molecular, and immunobiological variables.

Note. Values in bold indicate statistically significant results (P < 0.05).
Toxicities
All toxicities were recorded and graded according to the CTCAE v4.0. A total of 104 treatment cycles given to 31 patients were recorded. Toxicities were mostly of grade I to II, including anemia (18 cycles), leukopenia (32 cycles), thrombocytopenia (14 cycles), nausea or vomiting (20 cycles), fatigue (8 cycles), hepatic toxicity (8 cycles), decreased fibrinogen (15 cycles), and electrolyte disturbance (25 cycles) (Table 3).
Overall toxicities in 31 patients.

Abbreviation: G, grade.
Posthoc analysis
In our cohort, 24 patients had molecular analyses for both IDH1/2 and TERT promoter by Sanger sequencing (8 IDH1 mutant, 1 IDH2 mutant, 15 IDH1/2 wild type, 7 TERT promoter mutant, and 17 wild type). The MGMT promoter methylation was assessed in 20 patients (9 MGMT methylated and 11 unmethylated). The 1p/19q codeletion was assessed in 11 patients (4 with 1p/19q codeletion and 7 non-codeleted) by FISH. The distribution of radiological response in different molecular subgroups and immunobiological subgroups was demonstrated in Table 2. Of note, patients with IDH MT or no CD8 expression had a better DCR than patients with IDH WT (9/9 vs 7/15, P = .009) and positive CD8 expression (13/17 vs 0/3, P = .031) (Table 2 and Figure 4).

The representative radiological and histological images of patients with partial response, stable disease, and progression disease directly. Patient no. 26 suffered from right frontal recurrent GBM showed partial response and had a duration time of 14.33 months (A). IHC demonstrated negative expression of CD8 (D). Patient no. 29 suffered from frontal recurrent GBM showed stable disease and had a duration time of 13.76 months (B). IHC demonstrated negative expression of CD8 (E). Patient no. 40 suffered from left temporal recurrent GBM showed progression disease directly after 2 cycles of treatment (C). IHC demonstrated high expression of CD8 (F). Scale bar = 20 μm.
Database verification
Sixty-five patients from the CGGA database were selected for further analyses, as mentioned in section “Methods.” Patients were divided into low CD8+ T group and high CD8+ T group by median MCP CD8 T cells. Patients with low CD8+ TILs could achieve significant overall survival benefit from chemotherapy (P = .023, Figure 5A), while those with high CD8+ TILs did not show significant survival benefit through chemotherapy (P = .64, Figure 5B). Of note, the Immune Score was significantly high in the high CD8+ T-cell group (P = .016, Figure 5C), accompanied by a significantly high mRNA expression level of certain immune checkpoint molecules (eg, PD-1, CTLA4, and TIM-3).
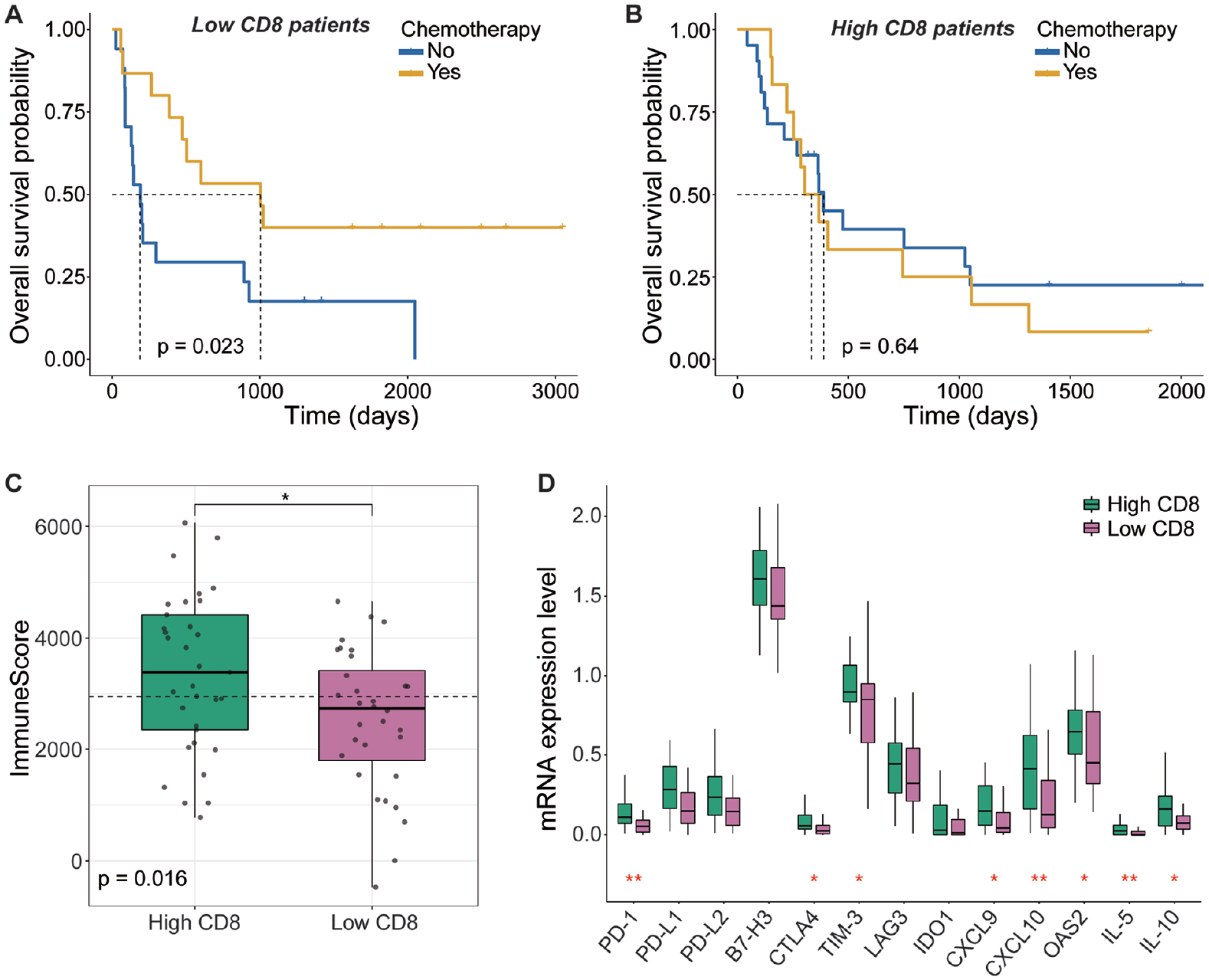
CGGA database verification. (A) Kaplan-Meier curves for overall survival probabilities in patients with low CD8+ TIL expression. (B) Kaplan-Meier curves for overall survival probabilities in patients with high CD8+ TIL expression. (C) Immune score of patients with different expression levels of CD8 TILs. (D) The mRNA expression level of tumor-infiltrating immunologic markers in patients with different expression levels of CD8 TILs.
Discussion
As we know, both cisplatin and TMZ are recommended by the National Comprehensive Cancer Network (NCCN) for patients with recurrent GBM; however, there is still no study treating patients with cisplatin plus alternating weekly TMZ. We performed a literature review and found 6 representative clinical studies using a cisplatin-based regimen to treat patients with recurrent HGGs (Table 4).4,6,19-22
Clinical studies in recurrent high-grade glioma.

Abbreviations: RR, response rate; TTP, time to progression; PFS-6, progression-free survival rate at 6 months; OS, overall survival; mo, months; GBM, glioblastoma multiforme; wk, weeks; HGG, high-grade glioma; AA, anaplastic astrocytoma.
Silvani performed cisplatin plus a standard TMZ regimen to treat patients with recurrent HGG. They obtained a PFS-6 of 52%, a response rate of 18.8%, and a median TTP of 33 weeks. Brandes et al reported a PFS-6 of 34%, a RR of 20.4%, and a median TTP of 18.4 weeks with a similar regimen. They administered TMZ in 10 fractions, but the cumulative dose was the same as which standard regimen. The response rate of our study was lower than the previous study, but our study had a comparable PFS-6 of 45.2% and median TTP of 5.07 months. Also, the high DCR (64.5%) suggested that the regimen of the chemotherapy was promising.
Temozolomide, an oral alkylator of the imidazotetrazine family, is used to treat malignant brain tumors such as glioblastoma and astrocytoma. Its active metabolite is 5-(3-methyltriazen-1-yl) imidazole-4-carboxamide (MTIC). The MTIC further methylates DNA purine residues, preferentially O6 guanine in guanine-rich regions. The MGMT can repair the cytotoxic lesions caused by MTIC, which is thought to be associated with TMZ resistance. 4 Cisplatin is a conventional chemotherapeutic agent to treat HGG, and the ex vivo experiment showed it can inactivate MGMT. 23 So, a combination of cisplatin with TMZ may have a theoretical basis.
In patients with accessible molecular and immunobiological profiles, our study showed that patients with IDH MT and low extent of infiltration with CD8+ T-cells might have relatively better survival probabilities and better DCR than patients with IDH WT and high CD8 infiltration, respectively. Per database investigation, we found the overall immune response level was significantly decreased in the low CD8+ T-cell group, which represented immunologically “cold” tumors. Hence, appropriate treatment for converting “cold” into “hot” tumors should be considered. Chemotherapy releases a large number of antigens while killing tumor cells. This process might enhance immunogenicity, induce immune cell infiltration to produce an immune response, and enhance clinical benefits. On the contrary, although there was more infiltration of immune cells in the immune microenvironment of the high CD8+ T group, the expression level of immune checkpoint molecules was also high. This phenomenon implied that the immune escape in the high CD8+ T group is not due to a lack of immune cells but the inhibitory effect of immune checkpoint molecules. In such a situation, the enhancement of immunogenicity by chemotherapy-induced antigen release may not be adequate, and the effect of immunosuppressive molecules should be inhibited at the same time.
The limitations of our study include the single-arm study design, the heterogeneous primary histology of enrolled patients, and the lack of enough molecular investigation and immunobiologic profiles. Therefore, we did not reclassify the pathological types of the participants in our cohort according to the 2021 WHO classification of tumors of the Central Nervous System. 24 As this was an exploratory single-arm phase II trial, no formal sample size calculation was conducted. The relatively small sample size may limit the statistical power and external validity of the study results. Moreover, tumor progression was evaluated using MRI. However, conventional MRI has limitations in distinguishing true progression from treatment-related changes. Advanced imaging modalities like amino acid positron emission tomography (PET) may improve diagnostic accuracy in glioblastoma recurrence. 25 It is important to note that similar studies investigating the combination of TMZ and cisplatin have been conducted in previous trials, which somewhat diminishes the novelty of our study. We chose an alternating dosing regimen of TMZ based on its potential to enhance drug exposure while minimizing toxicity. However, the gains from alternating dosing were not statistically significant when compared with conventional 5/28 dosing, suggesting that further research is needed to fully understand the implications of this treatment strategy. Also, salvage therapy such as bevacizumab or re-operation after disease progression will bias the effect of our study on overall survival. Furthermore, although our chemotherapy regimen showed a promising disease control rate and acceptable toxicity profile, the absence of a comparator group limits the strength of conclusions regarding its superiority over current standard treatments. However, our study introduced an alternative chemotherapeutic regimen without severe side effects, and a comparable treatment outcome was achieved contrary to the previous studies. These limitations restrict the broader clinical applicability of our results. Future randomized controlled trials are needed to validate the observed survival benefits and to further elucidate the mechanistic role of CD8+ TILs in modulating treatment response.
Conclusions
In conclusion, we suggest that the regimen of cisplatin plus alternating weekly TMZ is feasible and safe in the management of recurrent HGGs. The CD8 TILs may influence the survival benefit and treatment response of this regimen. Further investigation was warranted to explore the relevance between efficacy and status of the tumor microenvironment.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251350188 – Supplemental material for Cisplatin and Alternating Temozolomide in Recurrent High-Grade Gliomas: Efficacy and the Role of Tumor-Infiltrating Lymphocytes in a Phase II Clinical Trial
Supplemental material, sj-docx-1-onc-10.1177_11795549251350188 for Cisplatin and Alternating Temozolomide in Recurrent High-Grade Gliomas: Efficacy and the Role of Tumor-Infiltrating Lymphocytes in a Phase II Clinical Trial by Xiaojie Ding, Di Chen, Zhenyu Zhang, Ying Qi, Dikang Chen, Jianbo Wen, Yuyuan Wang, Haixia Cheng, Chunxia Ji, Lingchao Chen, Chao Tang and Yu Yao in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
None.
Ethical considerations
The study was approved by the Institutional Review Board of Huashan Hospital, Fudan University (KY 2014-162), registered on clinicaltrials.gov as “Efficacy and Tolerability of Cisplatin Plus Alternating Weekly Temozolomide in Recurrent High-grade Gliomas” in Aug 2014 (NCT02263105), and conducted in Huashan Hospital (Shanghai, China) in accordance with Good Clinical Practice guidelines and the Declaration of Helsinki.
Consent to participate
De-identified participant data, statistical code, and related materials will be made available upon reasonable request from the corresponding author.
Consent for publication
All participants (or their legal guardians, where applicable) provided written informed consent for the publication of anonymized clinical data included in this study.
Author contributions
X.D., Z.Z., and Y.Y. contributed to the conceptualization. X.D., Z.Z., and Y.Y. contributed to the methodology. J.W. contributed to the software. D.C., H.C., and Z.Z. contributed to the validation. X.D., D.C., and Z.Z. contributed to the formal analysis. X.D., D.C., Z.Z., Y.Q., D.C., and Y.Y. contributed to the investigation. Y.Q., Y.W., C.J., L.C., C.T., and Y.Y. contributed to the resources. X.D. and D.C. contributed to the data curation. X.D. and Z.Z. contributed to the writing – original draft preparation. D.C. and Y.Y. contributed to the writing – review and editing. D.C. and J.W. contributed to the visualization. Y.Y. contributed to the supervision. X.D. and Y.Y. contributed to the project administration. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
