Abstract
Background:
Detoxification enzymes of the glutathione S-transferase (GST) family are cytosolic phase II detoxification enzymes and play an important role in the normal functioning of the human antioxidant system. When the normal function of GST is disturbed or absent, there can be disturbances in cell metabolism, proliferation, and apoptosis. Deletions in the GSTM1 and GSTT1 genes have been observed in several different diseases as well as in the development of cancer. There is a need to analyze the relationship between glioma and GSTM1 and GSTT1 gene deletion to better understand the relationship between brain tumors and GST polymorphisms, which is crucial for adopting a multidisciplinary approach to prognosis and treatment of brain tumors.
Methods:
In a cross-sectional clinical-laboratory study, gene deletions were examined in 34 patients with brain tumors originating from glial cells—gliomas and 88 healthy individuals. All participants were of Polish nationality and were not related.
Results:
An increase in GSTM1 and GSTT1 gene deletions was observed in glioma patients compared with the control group. The greatest increase showing a marked rise of 10 times (11.8% vs 1.14%, P < .05) is in the null genotype of both genes (GSTM1−/GSTT1) [odds ratio [OR] = 0.86; 95% confidence interval [CI] = 0.09-0.802] but less in the genotype with deletion of 1 GST gene (GSTM1−/GSTT1+ and GSTM1+/GSTT1−). In addition, the findings indicated a decrease in the non-deletion genotype of both genes (GSTM1+/GSTT1+) in healthy individuals. This study showed a higher frequency of GST gene deletion in glioma patients in the studied population.
Conclusions:
Based on the obtained findings, it can be said that the examination of the selected detoxification enzymes can be a useful marker in the diagnosis of glioblastoma.
Introduction
Detoxification enzymes from the glutathione S-transferase (GST) family play an important role in the normal functioning of the human antioxidant system. 1 Glutathione S-transferase M1 (GSTM1) and Glutathione S-transferase T1 (GSTT1) are cytosolic enzymes consisting of 2 subunits and a family of phase II detoxification enzymes.2,3 These enzymes protect living cells by helping to attach glutathione (GSH) to various electrophilic molecules, which can come from both within the body and from external sources. 4
Glutathione S-transferase along with other enzymes is represented differently in the tissues of the human body. This variability accounts for the diverse reactions of organs to the effects of natural or synthetic toxic substances and stress. 1
When the normal function of GST is disturbed or absent, various processes can occur that lead to disturbances in cell metabolism, affect cell proliferation and apoptosis, lead to errors in post-translational modifications, and develop drug resistance. 5 Mutations in GSTM1 have been observed in several different diseases and conditions, contributing to the development of adenomas and adenocarcinomas, melanoma, osteosarcoma, and glioma. Previous research has shown that the GSTT1 null genotype is associated with an increased risk of lung cancer and urinary system cancer.6,7 In the population of individuals with deletions in both GST genes (GSTM1 and GSTT1), research has shown a close association between GST polymorphisms, smoking, and lung cancer, hepatocellular carcinoma, and bladder cancer risk.8-10
Benign gliomas are described as slow-growing glial tumors with low proliferation rates, typically referred to as “low-grade” gliomas (grade I or II), and are most often found in children and young people.11,12 The prognosis of the 2 basic types of benign gliomas differs. Non-infiltrative tumor subtypes have a good prognosis, whereas diffusely infiltrative tumors grow slowly but are still fatal in the end.11,13 Malignant gliomas are primary malignant brain tumors with diffuse brain infiltration; they are the most common and have a high recurrence rate. 14 Traditional histopathological classification of malignant gliomas has been improved by insight into genetic changes, dividing patients into groups based on their molecular biomarkers, which leads to better delineation of malignant tumors and thus enables more effective treatment.14-16
The results of genetic research on glioma are isolated molecular biomarkers that are guidelines for the classification and therapeutic intervention of patients and are also important for the prognosis of treatment. In the previous genetic studies of glioma, significant mutations have been shown such as Isocitrate Dehydrogenase (IDH1, IDH2), Cyclin-dependent kinase inhibitor (CDKN2A, CDKN2B), Epidermal growth factor receptor (EGFR), Telomerase reverse transcriptase (TERT) promoter, Alpha thalassemia/mental retardation syndrome X-linked (ATRX), Capicua (CIC), Far upstream element-binding protein 1 (FUBP1), Tumor protein 53 (TP53), and Phosphatase and tensin homolog (PTEN).17-20
Glioma therapy involves a multidisciplinary approach, considering tumor histological and molecular features, and includes surgical removal, radiotherapy, and chemotherapy. 16 The association between brain tumors and deletions in GST genes has produced inconsistent results in the scientific literature. 21 Therefore, investigating the link between glioma therapy and GST polymorphism, which influences drug metabolism and multidrug resistance, is very important.22-25
Material and methods
Frequencies of genotypes for deletions of GSTM1 and GSTT1 genes and their combined genotypes investigated in a population of glioma patients and healthy individuals from Poland.
Sample collection
In the cross-sectional clinical-laboratory study, deletions of genes were determined in a total of 34 samples from patients with brain tumors originating from glial cells—gliomas. Blood samples were collected from 22 men and 12 women, with an average age of 51.47 years among the patients (the oldest patient was 80 years old, the youngest 22 years old). Blood samples for control were taken from 88 healthy individuals (58 men and 30 women), average age 40.22 years (the oldest patient was 59 years old, the youngest 20 years old). All participants were of Polish ethnicity and unrelated. Exclusion criteria were the absence of other tumors, the simultaneous appearance of 2 tumors, subjects younger than 18 years. The material was taken on admission to the Department of Radiotherapy, Medical University in Lublin, Poland, before receiving any therapy in period between 2017 and 2019. Samples were obtained from peripheral blood and stored at −18°C until tested. Molecular analyzes of investigated polymorphisms were conducted at the Institute of Molecular Genetics and Genetic Engineering (IMGGE), University of Belgrade, Serbia.
Histopathological diagnosis
Patients from whom blood samples were obtained presented diverse histopathological diagnoses of brain tumors, specifically classified as gliomas. The types of gliomas of the patients involved in the study are: Astrocytomas: Diffuse astrocytoma (DA), Anaplastic astrocytoma (AA), Glioblastoma multiforme (GBM), Giant cell glioblastoma (GBM GC), Glioblastomas with primitive neuronal components (GBM-PNC), Astrocytoma policyticum (AP), Astrocytoma gemistocyticum (AG); Oligodendrioglioma: Oligodendroglioma (OD), Anaplastic oligodendroglioma (AOD); and Mixed gliomas: Oligoastrocytoma (OA).
Polymerase chain reaction and genotyping
Genomic DNA was isolated from peripheral blood samples of patients with glioma and healthy individuals using the Genomic DNA Mini Kit (Qiagen, Germany). Detection of homozygous deletions of the GSTM1 and GSTT1 genes was conducted using the multiplex polymerase chain reaction (multiplex PCR) method, following the previously described procedure. 26 The gene β-globin was used as a positive control for the PCR reaction, and analysis of PCR products was performed on a 2% agarose gel. The primers were used for the PCR reaction for the genes GSTM1 (GSTM1_F: 5'-GAGATGAAGTCCTTCAGA-3', GSTM1_R: 5'-GCTTCACGTGTTATGGAGGTT-3'), GSTT1 (GSTT1_F: 5'-ATGTGACCCTGCAGTTGC-3', GSTT1_R: 5'-GAGATGTGAGCACCAGTAAGGAA-3'), and β-globin (β-globin_F: 5'-CAACTTCATCCACGTTCACC-3', β-globin_R: 5'-GAAGAGCCAAGGACAGGTAC-3').
Statistical analysis
Chi-square (χ2) and Student’s t-tests were used in the analysis of demographic and clinical data of glioma patients and control. Frequencies of GST polymorphisms were assessed using the allele counting method, and genotype frequencies were examined through Pearson’s chi-square test and Fisher’s exact probability test. All correlations were determined by Spearman’s correlation analysis. A P-value < .05 was considered statistically significant. Statistical analysis of the obtained results was performed using IBM SPSS Statistics (version 25.0). The statistical power analysis was performed according to the 2 proportions formula for sample size and the odds ratio (OR) formula 27 with the alpha value of 0.05 and power 0.80, using the proportions according to a study by Schwartzbaum et al, 28 whereas the confidence intervals were calculated using the formula from the book by Hsiao et al. 29
Results
The results of molecular analysis of GST polymorphisms were obtained in the form of digital images showing multiplex PCR products separated by electrophoresis (Figure 1).

A representative image of multiplex PCR analysis of GSTM1 and GSTT1 gene polymorphisms. The presence of a 268 bp (base pair) band indicated the presence of the β-globin gene and represented an internal positive control. (A) The absence of 151 and 70 bp bands indicates a homozygous deletion of the GSTM1 gene (GSTM1 null genotype) and the GSTT1 gene (GSTT1 null genotype). (B) The absence of 70 bp band indicates a homozygous deletion of the GSTM1 gene (GSTM1 null genotype). (C) The absence of 151 bp band indicates a homozygous deletion of the GSTT1 gene (GSTT1 null genotype). (D) The presence of 70 and 151 bp bands representing both gene GSTM1 and the GSTT1 genes.
All blood samples were obtained from patients with a confirmed brain tumor of glial cell origin. The most prevalent gliomas belonged to the group of astrocytic tumors, constituting 85.3% of cases (Table 1). Among astrocytomas, the most common is GBM, which is also the most aggressive brain tumor.
Histopathological brain tumor diagnosis.

The relationship between glioma stage and GST genotype does not exhibit clear statistical differentiation. As expected, the highest number of glioma patients with GST gene deletions had World Health Organization (WHO) grade IV glioma (69.6%), whereas the lowest number of patients with GST gene deletions had stage I glioma (4.3%) (data not shown). There is no statistically significant Spearman’s rank correlation between mutations and grade (ρ = −0.230; P = .191 > .05).
The results showed that there is a difference in the combined GSTM1/GSTT1 genotype in glioma patients and healthy controls (Figure 2).
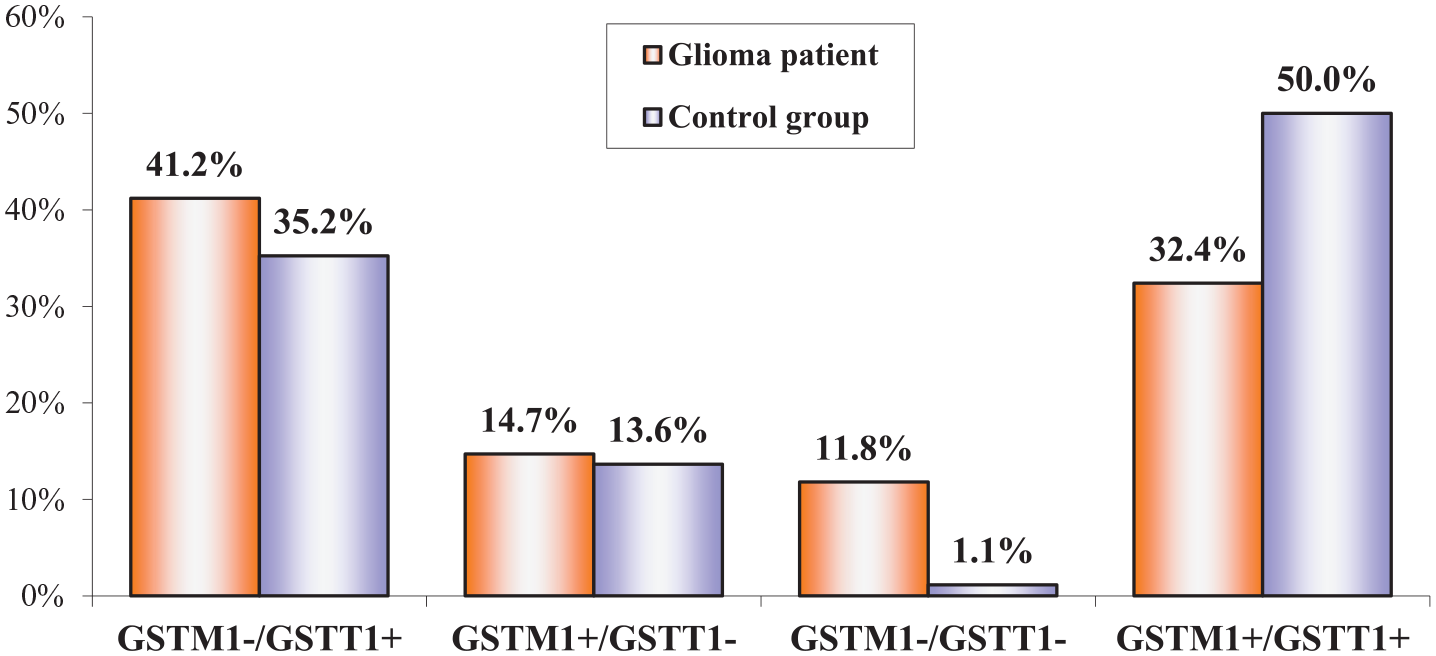
Combined GSTM1/GSTT1 genotype in glioma patients and healthy controls.
Analysis of the results showed a significant difference in the GST genotype between glioma patients (Glioma) and the genotype distribution within the healthy population (control) (Table 2).
Distribution of GSTM1 and GSTT1 genotypes according to gender and age among glioma patients and healthy controls from Poland.

Increase/decrease (−) – higher/lower (−) frequency of GSTM1/T1 genotype in (%) glioma patients compared with the control group.
Compared with the control group, in the glioma patients group, the most substantial increase is evident in the GSTM1–/GSTT1– genotype, showing a marked rise of 10 times (11.8% compared with 1.14%, P < .05) (OR = 0.86; 95% CI = 0.09-0.802). In addition, the GSTM1–/GSTT1+ genotype demonstrates an increase of 16.9% (from 41.2% to 35.23%) (OR = 0.916; 95% CI = 0.297-2.828), whereas the increase in the GSTM1+/GSTT1– genotype is the least pronounced, with a marginal rise of 7.8% (14.7% compared with 13.64%) (OR = 1.081; 95% CI = 0.3450-3.3899). Consistent with expectations, the percentage of the GSTM1+/GSTT1+ genotype among glioma patients was lower by 35.2% compared with healthy controls (32.4% compared with 50%) (OR = 2.091; 95% CI = 0.911-4.801).
The false positive rate for the GSTM1−/GSTT1+ genotype is 0.0818, whereas the false positive rate for the GSTM1+/GSTT1− genotype is 0.2661. The false positive rate for the GSTM1−/GSTT1− genotype is 0.3184, whereas the false positive rate for the GSTM1+/GSTT1+ genotype is 0.1153.
Based on the previous papers, to validate the foundations of this study with an OR of 1.5, a sample of 109 participants would be required.
The research results showed that all 3 GST genotypes with one or both deletions (GSTM1−/GSTT1+, GSTM1+/GSTT1−, and GSTM1−/GSTT1−) are more common in glioma patients compared with healthy control. As expected, the genotype without deletion in GSTM1 and GSTT1 genes (GSTM1+/GSTT1+) is more frequent in the control group in both men and women.
The work also studied the relationship between glioma occurrence and smoking using the chi-square test. Our results showed that no statistically significant correlation was found between smoking and the occurrence of brain tumors of glial origin (P > .05).
Discussion
Glutathione transferase enzymes are detoxification enzymes that remove harmful substances from the body, so deleting the genes encoding these enzymes led to metabolic disorders due to the accumulation of toxic compounds in higher concentrations. 30 In studies on the human population, the detoxification enzymes GSTM1 (OMIM: 138350) and GSTT1 (OMIM: 600436) and their polymorphisms were highlighted as very important against various toxic agents. 30
Gene GSTM1 is expressed at low levels in all tissues, with the lowest expression observed in the urinary bladder, lymph nodes, bone marrow, and brain, and the highest expression in the liver and smooth muscle tissue. 31 Mutations in GSTM1 have been observed in several different diseases and conditions. They lead to the development of adenomas and adenocarcinomas, melanoma, osteosarcoma, and glioma. 32 Equally important is the GSTT1 enzyme, the null genotype of which increases the risk of thyroid cancer, prostate tumors, gallbladder cancer, or breast cancer.33-36 One more example of why GST enzymes are important is a study that linked the null GSTT1 genotype and higher mortality in SARS-CoV-2 infection, with the fact that this enzyme is directly involved in the cellular detoxification process. 37
In previous studies, an analysis of the relationship between the GST gene and the risk of glioma formation was initiated, but with inconsistent results.38,39 In one of the studies, no association between brain tumor risk and GSTM1 and GSTT1 polymorphisms was demonstrated. 38 However, there are reports indicating that GSTT1 null/present variants are associated with an increased risk of glioma in Caucasians, in contrast to Asian populations. 39 In addition to the confirmed association between GST gene deletion and increased risk of glioma in Caucasians, the results of studies also highlighted the role of GST enzymes in the processes of chemoresistance in brain tumors. 39
According to scientific articles published so far, this study is the first to examine the association of homozygous deletions of the GST gene in glioma patients from Poland (Table 1). In order to better explain the potential influence of the GST gene, we examined the genotype combining deletions of 2 genes (GSTM1 and GSTT1), in healthy Poles and Poles with developed glioma using the multiplex PCR method (Figure 1, Table 2). The results of the study revealed that various genotypes with deletions of the GSTM1 and GSTT1 genes exhibit a higher prevalence among glioma patients from Poland compared with healthy Poles. When we analyze the frequency of the genotype with GST gene deletions in glioma patients of both sexes, the most prevalent is the GSTM1−/GSTT1+ genotype (Table 2).
Worldwide systematic reviews have noted a difference in GST gene distribution between ethnic groups, but a problem with these studies has been the lack of reports of GSTM1/GSTT1 genotype in non-European populations.30,40 Population studies across various countries have revealed distinct patterns in GST deletions. For instance, higher frequencies of GSTM1 deletions were observed among Caucasians in Great Britain and Portugal, contrasting with lower frequencies in Scandinavian nations like Finland, Denmark, and Sweden. 41 A lower frequency of the GSTT1 null genotype is observed in European populations compared with East Asian populations. Meanwhile, in Asian countries, Japan’s population exhibited lower deletion frequencies for both genes. Notably, African populations showed a notable difference in GSTM1 homozygous deletion frequencies between genders, with women and men displaying varying rates. 41 The findings from population studies examining GST variants in healthy individuals from the African continent revealed that the prevalence of homozygosity for GSTM1 deletion was 54.7% and for GSTT1 it was 42.1%. 42 In addition, 24.6% of individuals from the African continents in good health exhibit deletions in both genes. In control studies on healthy Caucasians, the GSTM1 null genotype is most commonly observed (40.6%), followed by the GSTT1 null genotype (10.4%), whereas the deletion of both GST genes is least commonly observed in healthy Caucasians (8.9%). 32 The mentioned variation of GST gene deletion frequency was also confirmed in our study.
The average age of patients diagnosed with a brain tumor is relatively high, with astrocytoma being diagnosed at 57 years and oligodendroglioma at 45 years. 43 Glioblastoma, on the contrary, is typically diagnosed later, with most patients being around 65 years old. It is difficult to examine which carcinogenic and mutagenic compounds patients were exposed to before the appearance of glioma, but it is possible to determine whether and which genotypic changes occurred. The significance of this research is analysis of the association of more than one detoxification enzyme and their gene mutations in investigated population contrary to previous study. Also, research additionally investigated whether the frequencies of GSTM1 and GSTT1 deletions differ in the population of healthy people and those suffering from glioma. Regardless of age, a relationship between gender and tumor biology has been observed in a large number of patients, which is why clinical and basic research provide important guidelines in a personalized approach to patients with brain tumors. 44
This study did not confirm a statistically significant difference between smoking, GST gene deletion, and the prevalence of glioma in the Polish population. Although smoking is not a significant risk factor for glioma, it is possible that bad habits such as smoking and drinking alcohol increase the risk. Men are more likely to engage in bad lifestyle habits, are more susceptible to developing brain tumors, and have a worse prognosis compared with women. 45 It should be noted that in this study there were more male patients, but that in both sexes, the most common tumor was stage IV glioblastoma. Addressing the limitation of the study, in the form of a small sample size, must be performed. Although the study has shown statistically significant results, the size of the sample does limit the possibility of declaring the conclusions as definite or highly probable. However, it should be noted that the results and conclusions concerning the genotypes GSTM1–/GSTT1+ and GSTM1−/GSTT1− show promise and should be investigated more thoroughly in a larger cohort to confirm the results of this article, but it must be emphasized that this may still be significant pilot study.
In addition, it should be noted that collecting samples of more than 100 participants would require sufficient effort that go beyond the scope of the capabilities of the research efforts for this article at the time of its publication.
Conclusions
Based on the findings, it can be said that the evaluation of genetic changes in detoxification enzymes may be an additional and useful molecular marker in the diagnosis of glioblastoma. In addition to those markers that are regularly used in everyday clinical practice, these should also be paid attention to.
Footnotes
Ethical considerations
This study adhered to the Helsinki Declaration and Rules of Good Clinical Practice. Bioethics Committee at the Medical University of Lublin approved the research included in this study (numbers KE-0254/171/2017 and KE-0254/74/2019).
Author contributions
LGS, VJ, SP, and LGS participated in the study’s design. LGS collected clinical data and selected patients. JT and VG performed experiments, investigation, and statistical analysis. IG and BŽ performed investigation. All authors contributed significantly and authors approved the final form of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article is supported by projects Ministry of Education, Science and Technological Development of the Republic of Serbia and partially by contract number: 451-03-66/2024-03/200378.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Informed consent
All patients provided written informed consent before collect and analyze blood samples per our institutional standards.
Data availability statement
All obtained data relevant to the study are included in the article. Other personal information about patients is protected by ethics.
