Abstract
Background:
HER2-targeting therapies may increase the risk of decreased left ventricular ejection fraction (LVEF), potentially leading to heart failure. The growing number of breast cancer survivors due to HER2-targeted treatments necessitates long-term cardiotoxicity management.
Method:
This retrospective study included HER2-positive breast cancer patients aged 18 or older who received at least 1 dose of HER2-targeting treatment between 2016 and 2020. The primary endpoint was the incidence of cardiotoxicity, defined as LVEF <50% with a 10% decline, LVEF drop by >15%, or onset of symptomatic heart failure. Secondary endpoints included the proportion of patients with baseline LVEF 50% to 55% developing cardiotoxicity, those discontinuing HER2 therapy due to heart failure, those treated with heart failure medications, and those continuing HER2 therapy while on heart failure medications. Another secondary outcome was the development of a hospital protocol for monitoring cardiotoxicity in these patients.
Results:
A total of 212 patients were included, with a median age of 56.5 years (interquartile range: 43-58 years). Twenty-two patients (10.37%) experienced cardiotoxicity from HER2-targeted treatment. Thirteen patients (6.13%) had asymptomatic heart failure with LVEF decrease of more than 10% to less than 50%. Five patients (2.35%) with LVEF less than 40% had asymptomatic heart failure, while 4 patients (1.88%) had symptomatic heart failure regardless of LVEF decline. HER2-targeted treatment was temporarily discontinued in 3 (13.63%) patients and permanently in 4 (18.18%) patients due to cardiotoxicity. The remaining 15 patients resumed treatment without interruption. Only 13 out of 22 patients were referred to cardiologists and prescribed heart failure medications.
Conclusion:
Close monitoring of LVEF in patients receiving HER2-targeting therapy can help health care providers initiate anti-heart failure medications to prevent LVEF deterioration and maintain HER2-targeting therapy.
Introduction
There is an increasing trend in cancer cases globally and in Saudi Arabia.1-3 Breast cancer is the most prevalent form of cancer among women. It has been reported that 15% to 25% of breast cancer patients overexpress the Human Epidermal growth factor Receptor-2 (HER2), which is linked with rapid growth of tumor tissues and bad prognosis. 4 HER-2 inhibitors (HER2-I), also known as HER-2 targeting therapies, such as trastuzumab, pertuzumab, lapatinib, and ado-trastuzumab emtansine (T-DM1), have been proven to increase the overall survival of breast cancer patients and have become the standard of care. 5 Nevertheless, they cause cardiotoxicity manifested as a reduction in the left ventricular ejection fraction (LVEF). 6
The incidence of decreased LVEF in individuals receiving HER2-I varies between 4% and 18.6%. 7 Additional confounding factors that increase the tendency of HER2-I to induce cardiotoxicity are advanced age, pre-existing cardiovascular diseases, diabetes mellitus, smoking, a baseline LVEF less than 50%, and radiation therapy to the cardiovascular structure. 4 Early recognition of HER2-I induced cardiotoxicity may avoid further adverse effects and assist in the early implementation of cardioprotective therapies. 8 And as survival rates for early-stage or metastatic breast cancer improve due to the use of HER2-I, the necessity for monitoring and care for their cardiotoxicity grows exponentially. 9
The package insert for trastuzumab underscores the critical need for consistent monitoring of left ventricular ejection fraction (LVEF) to manage the risk of cardiotoxicity effectively. According to the package insert, LVEF should be assessed at baseline before initiating therapy, every 3 months during treatment, and every 6 months for 2 years following the completion of adjuvant therapy. If LVEF decreases by more than 16% from baseline or falls below 50% with a 10% absolute decrease, trastuzumab administration should be paused. In such cases, LVEF should continue to be monitored every 3 months, and therapy should only resume once LVEF has recovered to 50% or higher. 10 In the context of combination therapy with trastuzumab and pertuzumab for metastatic breast cancer, the pertuzumab package insert advises initiating treatment only in patients with a baseline LVEF of 50% or higher, with cardiac surveillance conducted every 3 months during therapy. Treatment should be interrupted if LVEF drops below 40% or falls to 40% to 45% with a 10% absolute decrease from baseline. Resumption of therapy is recommended once LVEF improves to above 45% or stabilizes between 40% and 45% with less than a 10% absolute decrease from baseline. For early breast cancer treatment, trastuzumab combined with pertuzumab is recommended for patients with a baseline LVEF of 55% or higher, with LVEF monitoring at 3-month intervals during therapy. If LVEF decreases to less than 50% with a 10% or greater drop from baseline, treatment should be paused and only resumed when LVEF returns to 50% or higher. 11 Similarly, the package insert for T-DM1 (trastuzumab emtansine) recommends adherence to the same monitoring protocol as the trastuzumab and pertuzumab combination therapy in the metastatic setting. For early breast cancer, T-DM1 can be initiated in patients with an LVEF of 50% or higher, with routine LVEF assessments every 3 months. If LVEF drops below 45% or to 45% to 49% with a 10% or greater decrease, treatment should be temporarily halted, and more frequent LVEF monitoring every 3 weeks should be implemented. 12 Moreover, the European Society of Medical Oncology (ESMO), European Society of Cardiology (ESC), American Heart Association (AHA), and National Institute for Health and Care Excellence (NICE) guidelines recommend that patients who develop cardiotoxicity should be monitored every 3 weeks prior to resuming or discontinuing HER2-I permanently.13-16 Yet, to the best of our knowledge, no study has determined the optimal monitoring interval for the HER2-I in low-risk versus high-risk patients. The characterization of high-risk patients exhibits significant variability across different clinical guidelines, influenced by factors such as age, previous exposure to anthracycline or mediastinal or chest radiation therapy, existing cardiovascular conditions, and baseline left ventricular ejection fraction (LVEF) below 55%.13-16
The referral to cardio-oncology experts of patients who underwent anthracycline treatment and suffered cardiotoxicity has been examined in retrospective research. Compared with patients without cardiology consultation, those who were referred in this study had increased survival and adherence to cardioprotective medications. Despite a little decrease in LVEF, the continuation of HER-2-targeted treatment was boosted by cardioprotective therapies due to their ability to protect the heart. Troponin elevation may be used to diagnose cardiotoxicity in chemotherapy patients at an early stage. The administration of cardioprotective medication at the onset of elevated troponin levels has averted the development of cardiomyopathy and heart failure. 8 A study evaluated the changes in cardiac biomarkers and cancer therapy–related cardiac dysfunction (CTRCD), showed a significant association between N-terminal pro-B-type natriuretic peptide (NT-proBNP) and (CTRCD). An increase of NT-proBNP by 2 times was associated with an increase of CTRCD risk by 56% over 24 months of follow-up (hazard ratio, 1.56; 95% CI, 1.32-1.84, P < .001). 17
Our study intended to assess and quantify the incidence of cardiotoxicity induced by HER-2-targeting therapies in our center. This will aid in the development of a hospital protocol for monitoring and managing cardiac toxicity induced by HER-2-targeting medications.
Method
This retrospective study, conducted at King Abdullah Medical City in Jeddah, included all HER-2-positive breast cancer patients aged 18 or older who received at least 1 dose of HER-2-targeted therapy between June 2016 and June 2020. The primary endpoint was to determine the incidence of cardiotoxicity induced by HER-2 targeted treatment via echocardiogram which was defined as LVEF <50% with LVEF decline by 10%, LVEF drop by >15%, or the onset of symptomatic heart failure. The secondary endpoints were the proportion of patients with LVEF between 50% and 55% at baseline who developed cardiotoxicity, the proportion of patients who temporarily or permanently discontinued HER-2 targeting therapy due to HF, the proportion of patients who developed HF and were treated with HF medications, and the proportion of patients who received HF medications for cardiotoxicity and continued their HER-2 targeting therapy. The development of a hospital protocol for monitoring and managing cardiotoxicity in patients receiving HER-2-targeted treatments was another secondary outcome. Patients with chronic renal disease and Cockcroft-Gault creatinine clearance less than 30 mL/min, double primary tumors, decompensated liver disease with Child-Pugh level B or C, and bilateral breast tumors with distinct HER-2 expressions were excluded.
Statistical analysis
For categorical variables, frequencies and percentages are used to present descriptive statistics. Numerical variables are presented with median and interquartile range (IQR). Univariate and multivariate logistic regression models were used to evaluate the associations between potential risk factors and cardiotoxicity in breast cancer patients receiving HER-2 targeted therapies. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated for each potential risk factor. The statistical analysis was conducted using SPSS version 24.
Results
Baseline characteristics
Initially, a total of 215 patients were screened for inclusion and exclusion. We eliminated 2 individuals with double primary tumors and 1 patient with decompensated liver disease with Child-Pugh grade B. A total of 212 patients were included in the research, with a median age of 56.5 years and an interquartile range of 43 to 58 years. Of 212 patients, 105 patients had early breast cancer (EBC), and 107 patients had metastatic breast cancer (MBC).
Angiotensin-converting enzyme inhibitors (ACEIs) or angiotensin II receptor blockers (ARBs), beta-blockers, spironolactone, and either ACEIs or ARBs plus beta-blockers were used in 23 (10.85%), 8 (3.77%), 1 (0.5%), and 5 (2.35%) patients, respectively. These drugs were given to treat hypertension, arrhythmias, tachycardia, and cardiovascular disorders. In our analysis, the risk factors for developing heart failure were observed in previous anthracycline use; hypertension; diabetes mellitus; chronic artery disease; congestive heart failure; dyslipidemia; chronic kidney disease; a body mass index (BMI) greater than 30; a baseline LVEF less than 55%; and age over 50. In the study population, the percentages of those with a history of anthracycline use, hypertension, diabetes, age greater than 50, and a BMI greater than 30 were the highest. Specifically, 110 patients (51.9%) were at least 50 years old, 90 (42%) patients had previously received anthracycline; 77 (36.32%) patients had a BMI > 30; 44 (20.75%) patients had hypertension; and 38 (17.9%) patients had diabetes. Considering the risk of cardiotoxicity, 34 patients (16.03%) had no risk factors, whereas 114 patients (53.79%) had 2 or more risk factors. The median number of HER-2 targeted therapies received was 43 cycles (IQR: 21-69) for metastatic breast cancer and 17 cycles (IQR: 11-17) for early breast cancer (Table 1).
Baseline characteristics.
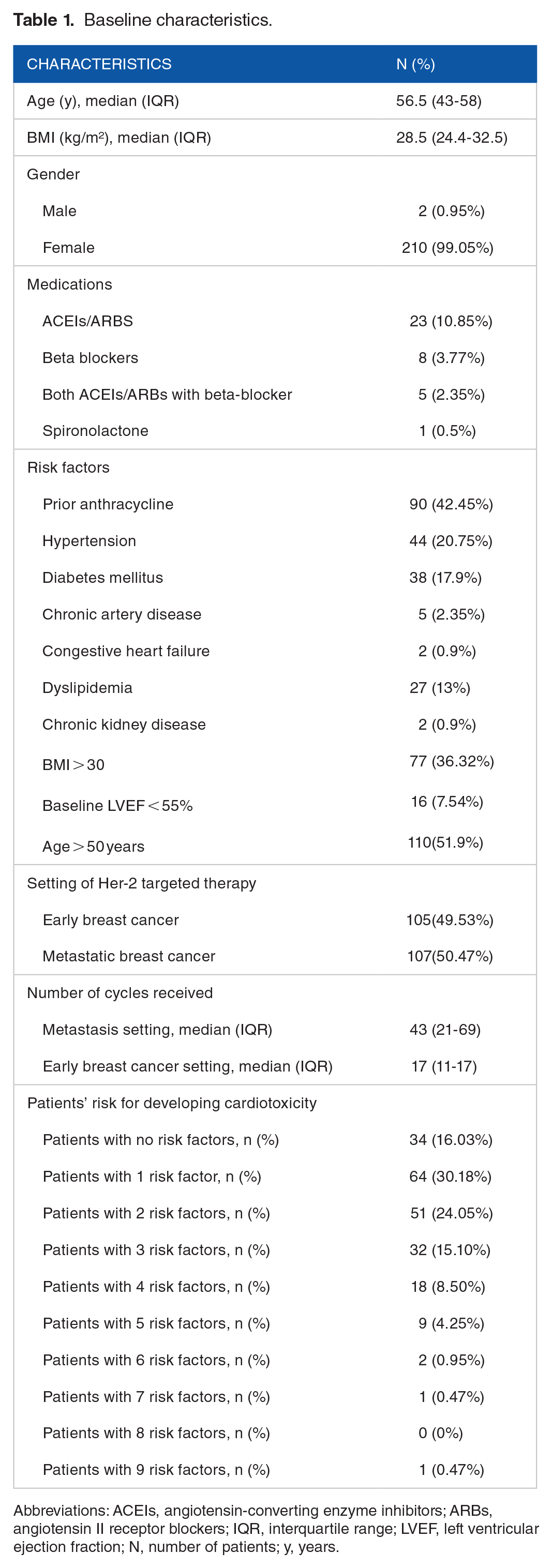
Abbreviations: ACEIs, angiotensin-converting enzyme inhibitors; ARBs, angiotensin II receptor blockers; IQR, interquartile range; LVEF, left ventricular ejection fraction; N, number of patients; y, years.
Cardiovascular outcomes
Twenty-two patients (10.37%) suffered cardiotoxicity with HER-2-specific treatment. Thirteen of the 22 patients (6.13%) had asymptomatic heart failure with a decrease in LVEF from baseline of more than 10% to less than 50%. Five patients (2.35%) with LVEF less than 40% had asymptomatic heart failure, while 4 patients (1.88%) presented with symptomatic heart failure regardless of the LVEF decline (Table 2). HER-2 targeted treatment was discontinued temporarily in 3 (13.63%) patients and permanently in 4 (18.18%) patients due to cardiotoxicity. The remaining 15 patients resumed their treatment without interruption. Only 13 out of 22 patients were referred to cardiologists and prescribed medicines for heart failure. This intervention enabled 9 out of 13 patients to effectively continue their treatment regimen and maintain an LVEF greater than 50%. Due to persistently low LVEF, 4 patients who received anti-heart failure medications were unable to continue HER-2-targeted treatment. Five (31.25%) of the 16 patients in our study with borderline LVEF (LVEF ⩾50% to <55%) who received HER-2 targeted treatment suffered cardiotoxicity (Table 2). See Table 3 for further information on patients who had cardiotoxicity. We devised a monitoring strategy based on the baseline LVEF and number of risk variables presented at the time of HER-2 targeted medication commencement, based on our findings (Figure 1).
Primary and secondary outcomes.

Abbreviations: HF, heart failure; LVEF, left ventricular ejection fraction.
Patients who developed cardiotoxicity.
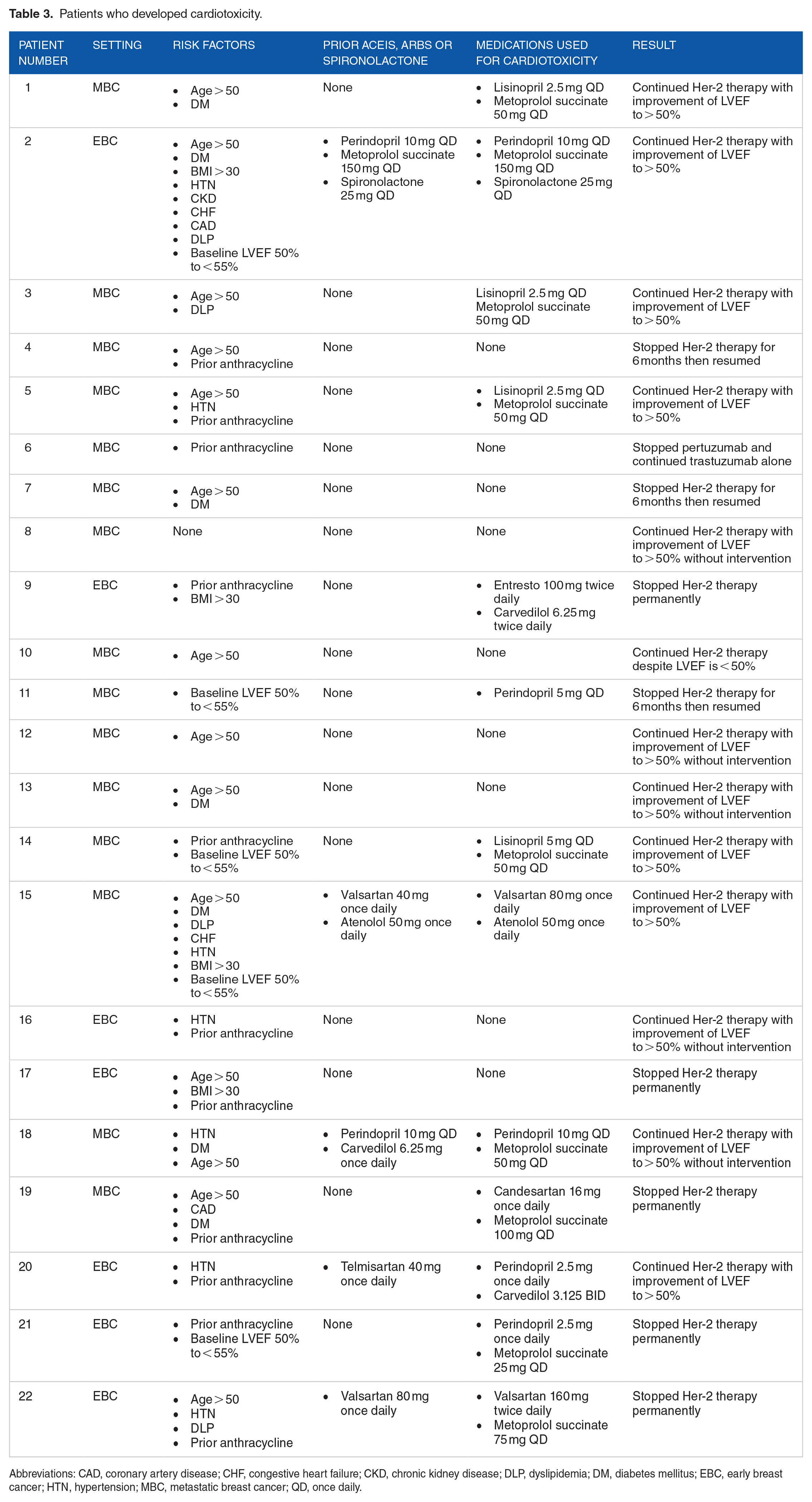
Abbreviations: CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; DLP, dyslipidemia; DM, diabetes mellitus; EBC, early breast cancer; HTN, hypertension; MBC, metastatic breast cancer; QD, once daily.

Monitoring LVEF in EBC/MBC.
For the adjusted model, prior anthracycline has the highest OR of 6.93 (95% CI of 1.37-34.97, P-value: .0190) indicating a significant association with the increased risk of cardiotoxicity. Baseline LVEF < 55% also has a high OR of 9.33 (95% CI of 2.16-40.39, P-value: .0028), which suggests a significant association with cardiotoxicity. Other factors, such as hypertension, diabetes mellitus, and dyslipidemia, did not provide significant results (Table 4).
Unadjusted and adjusted odd ratio estimates of cardiotoxicity.
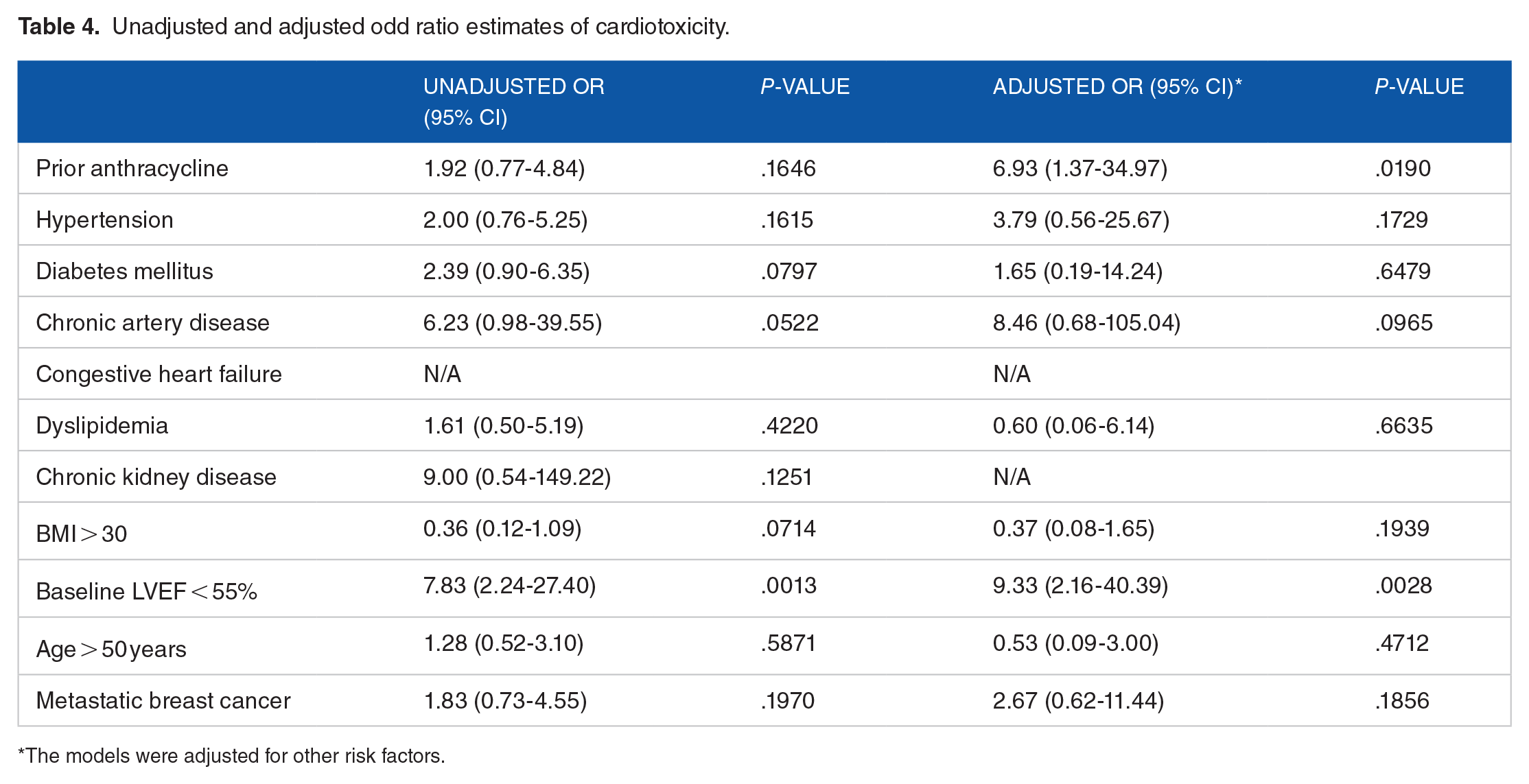
The models were adjusted for other risk factors.
Discussion
According to our analysis, the incidence of trastuzumab-induced heart failure was around 10.37% for the total number of patients with early and metastatic breast cancer, which is greater than what has been seen in previous studies. We believe that the differences in cardiotoxicity incidence could be attributed to demographic population differences, environmental factors, genetic variations, and the absence of a standardized hospital protocol for preventing cardiac dysfunction induced by HER-2 targeted therapy. In EBC patients, LV dysfunction occurred in 7 out of 105 (6.66%) patients, whereas it occurred in 15 out of 107 (14%) patients with MBC. These rates are much greater than those reported in randomized clinical studies.9,18 In the CLEOPATRA study, which compared the efficacy and safety of pertuzumab with trastuzumab and docetaxel versus trastuzumab and docetaxel in MBC patients, the cardiotoxicity rates were 6.6% for pertuzumab with trastuzumab and docetaxel and 8.6% for trastuzumab and docetaxel. 9 In the EMILIA study, randomized controlled research that assessed the safety of ado-trastuzumab emtansine against lapatinib with capecitabine in patients with MBC, the incidence of LV dysfunction was < 1%. 19 As documented in the HERA study, trastuzumab as adjuvant treatment in EBC for 1 year was responsible for 7.08% of the LVEF decline. 20 Thus, it is crucial to develop and implement a hospital-wide policy and guidelines for monitoring cardiotoxicity in patients receiving HER-2-targeted therapies. Establishing these protocols will help ensure early detection and intervention, thereby improving patient outcomes and minimizing the risks associated with these treatments.
According to the European definition of cardiotoxicity, most of the patients had heart failure without symptoms and a decrease in LVEF between 10% and 50%. Only 4 people had heart failure with symptoms. In the Paula et al study, 9.8% of participants had cardiotoxicity. Nevertheless, no instances of symptomatic heart failure were identified. Just 18% of 22 individuals discontinued trastuzumab permanently, while 13% ceased treatment temporarily. This percentage is lower compared with previous research, in which around 28% of patients discontinued taking trastuzumab entirely. 21
Similar to the findings of Perone et al, 59% of patients who developed heart failure started using medication to manage their heart failure. It is essential for patients with this illness to take medication to address heart failure. With the use of angiotensin-converting enzyme (ACE) inhibitors and/or beta-blockers, the LVEF may be improved. 22
The study’s risk variables include coronary artery disease, congestive heart failure, dyslipidemia, chronic renal disease, a baseline LVEF less than 55%, and the highest included participants had prior anthracycline, hypertension, diabetes, age above 50 years, and a BMI greater than 30 kg/m2. More than 53% of patients with various risk factors had heart failure due to the use of HER-2-targeted therapies (2 or more risk factors). But still, only 16% of individuals without risk factors experienced heart failure. Multiple studies have struggled to consistently identify risk factors significantly influencing the development of heart failure. While some research has pointed to factors such as age, diabetes, hypertension, prior anthracycline therapy, the number of treatment cycles, concurrent taxane therapy, breast surgery, and unilateral tumor site as potential risks, the findings have often been inconsistent. 22 - 25 Moreover, The concurrent administration of doxorubicin and cyclophosphamide with trastuzumab may increase the risk of cardiotoxicity by up to 27%, compared with an estimated 8% risk when doxorubicin and cyclophosphamide are administered without trastuzumab.6,26
As seen in our results, borderline LVEF is thought to be one of the most important factors that can lead to cardiotoxicity and heart failure. Several clinical studies have shown that the addition of HER-2-targeted therapies after the anthracycline based chemotherapy was related to an increased incidence of cardiac dysfunction, especially in patients with preexisting cardiac disease or a lower LVEF.7,27,28 HER-2-targeted therapies are linked with an increased incidence of cardiac dysfunction in individuals with borderline EF; thus, it is advised that HER-2-targeted therapies be used with caution in this situation and that the benefits should be evaluated against the risk of cardiotoxicity.7,28
Based on the study’s risk variables and several other studies, we determined that prior use of anthracyclines was associated with an increased risk of heart failure in individuals with normal LVEF. So, we recommend checking high-sensitive cardiac troponin T (hs-cTnT), and NT-proBNP, at baseline and every 2 to 3 cycles of trastuzumab, in patients with normal LVEF who have 1 risk factor of having previously used anthracyclines. This is because it’s likely that trastuzumab therapy will cause myocardial damage, which can result in higher troponin levels. 6 Patients with 2 or more risk factors, other than the use of anthracyclines in the past, should have their LVEF checked more frequently than 3 months and their (hs-cTnT), and NT-proBNP levels checked at baseline and every 2 to 3 cycles of trastuzumab. If the patient has no risk factors, LVEF should be monitored every 3 months and there is no need to obtain (hs-cTnT), and NT-proBNP. Patients with borderline LVEF need to start taking anti-heart failure medication and have their (hs-cTnT), NT-proBNP, and LVEF checked as high-risk patients. Finally, if LVEF is less than 50%, alternative treatment should be tried, if it is available, and a cardiologist should be consulted (Figure 1).
Our recommendations are grounded in several prospective studies that evaluate cardiac biomarkers in patients treated with anthracyclines followed by trastuzumab. In one study involving 43 patients, a significant change in high-sensitivity troponin-1 levels from baseline at 3 months was observed, with an odds ratio of 9 (95% CI: 1.8-50, P = .006), and negative and positive predictive values of 93% and 50%, respectively. 29 Another study demonstrated that patients who developed cardiotoxicity after receiving anthracyclines or trastuzumab had significantly higher increases in baseline troponin levels compared with those who did not (0.039 ± 0.0096 vs 0.007 ± 0.0013 ng/mL; P = .046). 30 Similarly, findings from the HERA trial indicated that elevated baseline NT-proBNP prior to trastuzumab administration was associated with an increased risk of cardiac dysfunction, although a precise threshold could not be determined. Baseline troponin I and T levels exceeding 40 and 14 ng/L, respectively, were also linked to a higher risk of cardiotoxicity. In addition, an incremental increase of 10 ng/L in NT-proBNP was associated with a 30% increased risk of trastuzumab-induced cardiac dysfunction. 31 A retrospective study of 66 patients further reinforced the association between NT-proBNP levels and the development of trastuzumab-induced cardiac dysfunction, reporting an estimated odds ratio of 22 (95% CI: 5.7-85.4, P < .0001). 32 The timing of cardiac biomarker measurement followed the American-European Cardio-Oncology guidelines, which recommend assessing troponin levels after each cycle of trastuzumab in patients with 2 or more risk factors—such as smoking, hypertension, diabetes, chronic renal insufficiency, and obesity. The guidelines also advise monitoring in patients with a single risk factor, including age over 60, prior mediastinal radiotherapy, pre-existing heart disease, elevated baseline cardiac biomarkers, or previous exposure to cardiotoxic drugs. 13 The European Society of Cardiology (ESC) guidelines on cardio-oncology, developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO), and the International Cardio-Oncology Society (IC-OS), recommend the measurement of cardiac troponins and natriuretic peptides at baseline, every 2 to 3 cycles during HER-2 targeted therapy, and at 3 and 12 months post-therapy in high-risk patients. The guidelines further advise discontinuing HER-2 targeted treatment in symptomatic patients or asymptomatic individuals with a left ventricular ejection fraction (LVEF) below 40%. These guidelines aim to monitor and mitigate the risk of cardiotoxicity associated with HER-2 targeted treatments. 33
This study represents the first research conducted in Saudi Arabia aimed at identifying the risk factors most strongly associated with HER-2 targeted therapies-induced cardiotoxicities. Our study’s limitation includes a relatively small sample size of eligible patients.
Conclusions
The rate of cardiotoxicity induced by HER-2 targeted therapies was greater than the rates reported in many randomized clinical trials. We suggest considering the risk factors for developing cardiotoxicity to customize an appropriate LVEF monitoring plan for breast cancer patients. Initiation of anti-heart failure medications in patients with cardiotoxicity can potentially reverse the cardiotoxicity and assist the patients to continue their treatment plan.
Footnotes
Author Contributions
AS contributed to data collection, data analysis, proposal, and manuscript writing. A Alhifany contributed to manuscript writing and editing. M Alnuhait contributed to manuscript writing and editing. A Alshammari contributed to manuscript writing and editing. YA contributed to data analysis and manuscript editing. M Alshamrani contributed to proposal and manuscript editing. A Alnatsheh contributed to proposal and manuscript editing. M Alharbi contributed to proposal and manuscript editing. AK contributed to proposal and manuscript editing and supervision. M Alfoheidi contributed to proposal and manuscript editing. A AlQubbany contributed to proposal and manuscript editing.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article.
Ethical Approval
The study was approved by King Abdullah International Medical Research Center, IRB approval (NRJ21J/174/07).
Written Informed Consent from the Subjects
Waived by the IRB.
