Abstract
Background:
Febrile neutropenia (FN) poses a significant challenge in cancer treatment, with a high incidence among patients undergoing standard therapies. Predicting FN complications and outcomes remains crucial for improving patient management strategies. Biomarkers, including procalcitonin and albumin, have garnered attention for their potential prognostic value in FN.
Methods:
We conducted a prospective observational study at a tertiary hospital, enrolling 185 adult cancer patients experiencing FN episodes. We assessed serum albumin levels and incorporated them into the Multinational Association for Supportive Care in Cancer (MASCC) risk index to enhance risk stratification.
Results:
Serum albumin levels displayed promising prognostic utility in febrile neutropenia (FN). They exhibited moderate specificity and sensitivity in predicting mortality during FN and 28-day mortality. Serum albumin levels were significantly associated with gastrointestinal infections, serving as an independent predictor. Integrating serum albumin into the MASCC risk index improved predictive accuracy for FN mortality by 50%, 28-day mortality by 66.67%, and respiratory tract infections by 62.50%, enhancing in this way risk stratification for FN-related complications.
Conclusion:
Serum albumin emerges as a promising biomarker for prognostication in FN, complementing existing risk assessment frameworks. Its incorporation into the MASCC risk index enhances predictive capabilities, aiding clinicians in identifying high-risk patients promptly. While albumin shows potential in predicting mortality and complications, further research is warranted to optimize sensitivity and specificity, ensuring its clinical utility.
Introduction
Febrile neutropenia (FN) represents a common complication resulting from standard therapies administered to cancer patients. 1 Its incidence varies significantly, affecting approximately 10% to 50% of patients with solid tumors and rising to as high as 80% among individuals with hematological malignancies.1,2 Classified as a medical emergency, FN is accompanied with complications and adverse outcomes that significantly impact patient survival. Studies have reported an elevated in-hospital mortality rate associated with FN. 3 Although FN contributes to increased mortality immediately following its occurrence, its influence tends to diminish over time. 3 FN predisposes individuals to a spectrum of infections, including bloodstream infections such as bacteremia and septicemia, respiratory tract infections (RTI), gastrointestinal infections (GI), urinary tract infections (UTI), as well as skin and soft tissue infections.4 -7 This condition can also facilitate the development of mucositis, which can create a pathway for bacteria to enter the bloodstream, potentially leading to secondary bloodstream infections.4,5
Given the profound impact of FN on the treatment and prognosis of cancer patients, predicting its complications and outcomes is of paramount interest. Numerous studies have investigated the potential of biomarkers, particularly those associated with inflammation, in predicting FN outcomes and complications.8-11 Given the prevalent issue of malnutrition among cancer patients and its detrimental impact on prognosis, 12 there is compelling interest in investigating whether biomarkers related to malnutrition can predict FN outcomes and complications. Albumin, traditionally recognized as a marker of nutritional status, 13 is routinely used to detect malnutrition in cancer patients. 14 In addition, albumin levels have been associated with predicting malnutrition in this patient population. 15 Notably, albumin levels serve as indicators of both malnutrition and inflammation, with low serum albumin levels indicating severe inflammation. 16 Given its dual role in reflecting these pivotal factors in cancer and FN, exploring albumin’s potential in predicting FN outcomes and complications is crucial. Further research in this area could provide valuable insights into improving FN management strategies and enhancing patient care.
Considering the previously mentioned fact this study was designed to investigate the potential of albumin in early prediction of complications and outcomes associated with FN. Furthermore, we sought to assess whether the inclusion of this biomarker in the Multinational Association for Supportive Care in Cancer (MASCC) risk index score could enhance the stratification of FN patients into low and high-risk groups.
Methods
The study was performed in accordance with guidelines from the Reporting of Observational Studies in Epidemiology (STROBE) statement.
Study design
This prospective observational study was conducted at the Institute for Oncology and Radiology of Serbia, a tertiary university hospital situated in Belgrade, Serbia, and serving as the National Cancer Research Center. The study spanned 20 months, commencing in May 2022 and concluding in December 2023.
Study population and sample size calculation
The sample size calculation determined that a total of 185 patients were required for the study, with a 5% margin of error and an 80% confidence level. The study enrolled male and female patients aged eighteen or older diagnosed with solid or hematological malignancies, and who experienced at least 1 episode of FN during the study period. Patients who developed FN while undergoing concurrent chemotherapy and radiotherapy or radiotherapy alone were excluded from the study.
Statistics
The data were initially collected using Microsoft Excel 2016 and subsequently transferred to GraphPad Prism 10 Software (GraphPad Software, San Diego, CA) for a comprehensive statistical analysis. Normality of the data distribution was assessed using the D’Agostino and Pearson omnibus normality test. Depending on the distribution, results were presented as mean and standard deviation (SD) or median and interquartile range (IQR).
We evaluated the prediction performance of albumin in predicting complications and outcomes associated with FN by constructing receiver operating characteristic (ROC) curves. The ROC curves were compared with those generated for the MASCC risk index. We used Youden’s J statistic to determine the optimal cut-off value for each ROC curve, enabling calculation of sensitivity, specificity, positive predictive value, negative predictive value, and likelihood ratio.
In addition, our methodology involved conducting a multivariate logistic regression analysis to identify the independent predictors of GI during FN episodes and mortality occurring within FN episodes. These factors included age, albumin level, MASCC index, and Eastern Cooperative Oncology Group (ECOG) performance status were included in this comprehensive analysis. While mortality during FN was assessed using albumin as a continuous variable, to identify predictors for GI, albumin levels were converted into categorical values based on a calculated cutoff” for clarity. This adjustment was essential due to observed linear dependencies among certain predictors. We set a significance threshold of P < .05 to determine statistical significance in our findings.
Results
We conducted an analysis of the data obtained from a cohort comprising 185 participants diagnosed with FN, as reported at the Institute for Oncology and Radiology of Serbia. Key demographic and clinical characteristics relevant to our study are presented in Figure 1 for reference. Notably, the average absolute neutrophil count was 0.1 (0.27) × 109/L, white blood cell count was 1.1 (0.80) × 109/L, and serum albumin level was 36.16 (4.99) g/L.

Infographic that demonstrates patient demographics.
The ROC curves for serum albumin levels revealed the biomarker’s accuracy in predicting mortality during FN, 28-day mortality, as well as RTI and GI (Table 1, Figure 2). However, serum albumin levels did not show predictive value for other FN complications, such as bacteremia, septicemia, mucositis, urinary tract infections, and skin and soft tissue infections.
The area under the curve and P value for different FN outcomes and complications.
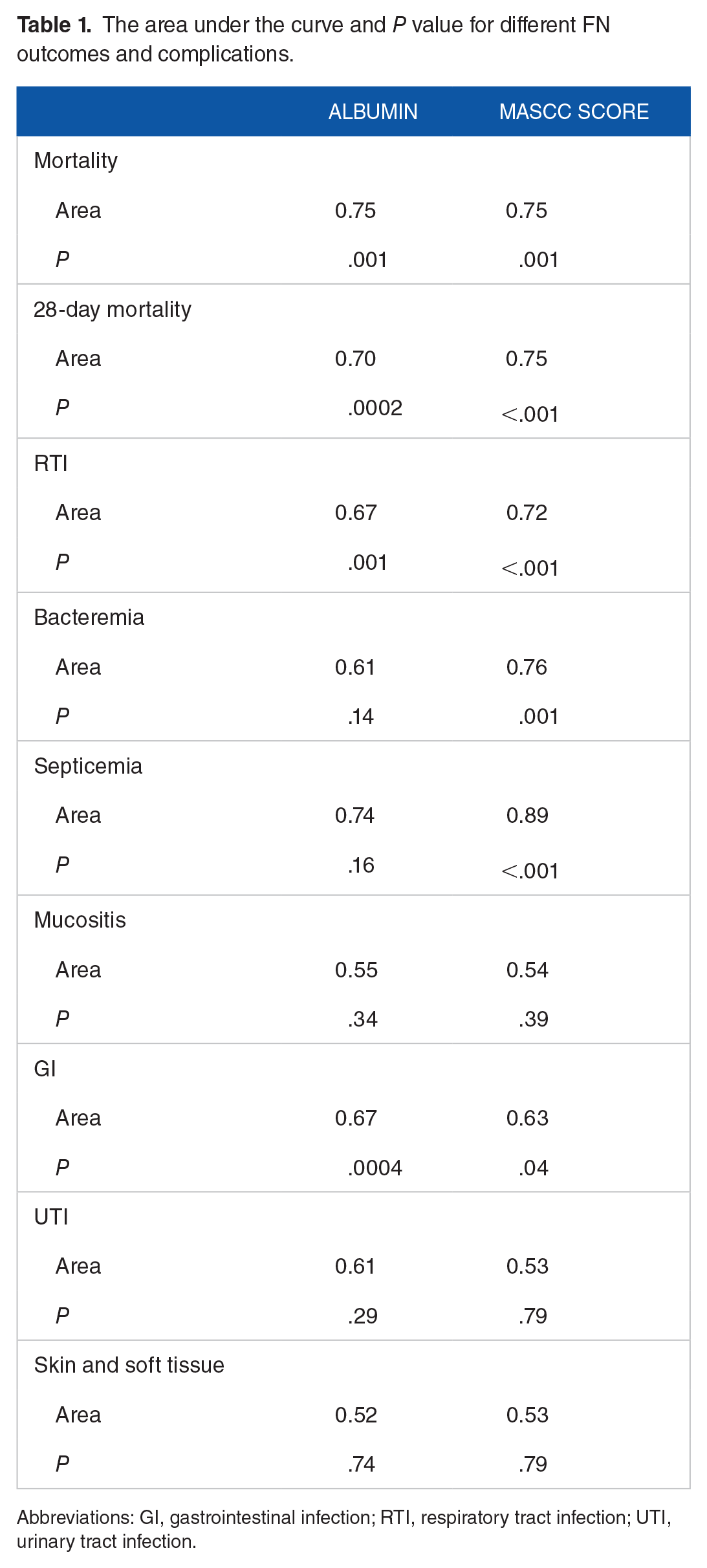
Abbreviations: GI, gastrointestinal infection; RTI, respiratory tract infection; UTI, urinary tract infection.

Receiver operating characteristic curves for prediction of mortality during FN (A), and gastrointestinal infections (B).
Conversely, the ROC curve analysis for the MASCC risk index demonstrated its utility in predicting mortality (both during FN and 28-day mortality), bloodstream infections (bacteremia and septicemia), RTI, and GI (Table 1, Figure 2). Similar to serum albumin, the MASCC risk index did not exhibit predictive capability for mucositis, urinary tract infections, and skin and soft tissue infections.
Interestingly, serum albumin levels exhibited similar area under the curve (AUC) values as the MASCC risk index for predicting mortality during FN. However, MASCC risk index showed slightly higher AUC values for 28-day mortality and RTI. Conversely, serum albumin levels had a higher AUC value for predicting GI compared with the MASCC risk index (Table 1). The performance of albumin in predicting complications and outcomes associated with FN is summarized in Table 2. Serum albumin levels exhibit varying degrees of predictive capability across different outcomes.
Diagnostic performance of albumin in predicting mortality during FN, and 28-day mortality, respiratory tract infections and gastrointestinal infections.
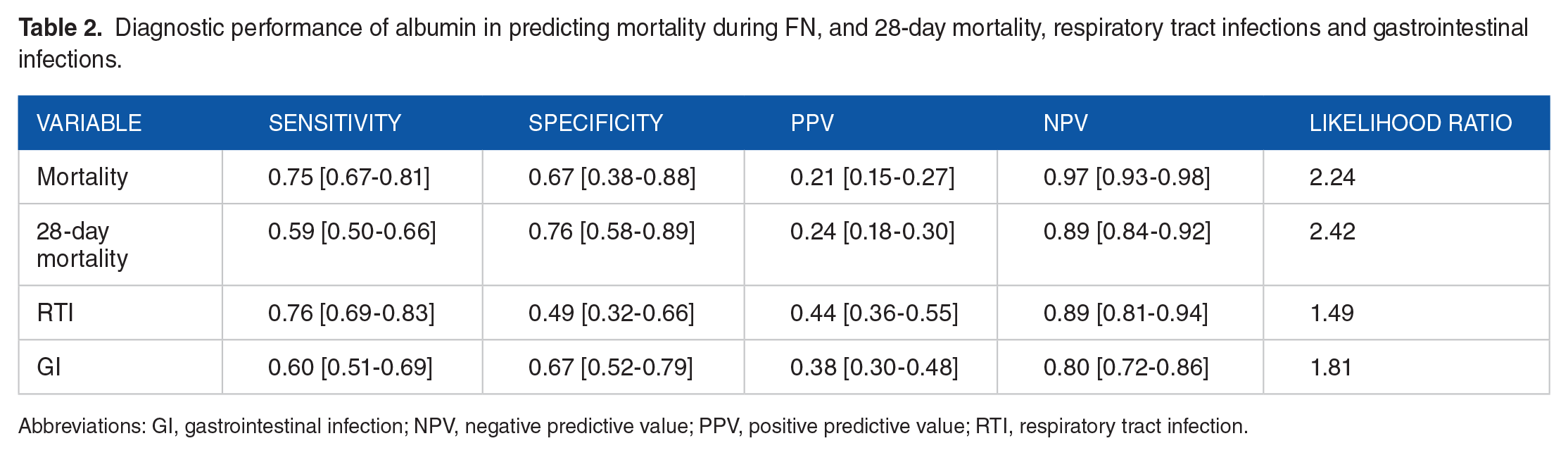
Abbreviations: GI, gastrointestinal infection; NPV, negative predictive value; PPV, positive predictive value; RTI, respiratory tract infection.
Serum albumin levels demonstrate varied predictive performance across different outcomes in FN. For the prediction of mortality during FN and 28-day mortality, serum albumin shows moderate specificity and sensitivity, low positive predictive value (PPV), high negative predictive value (NPV), and a moderate likelihood ratio. In the context of RTI, serum albumin exhibits moderate sensitivity, low specificity, moderate PPV, high NPV, and a low likelihood ratio (Table 2). For GI, serum albumin displays low specificity and sensitivity, along with low PPV, high NPV, and a low likelihood ratio (Table 2). These findings highlight the variable performance of serum albumin levels across different FN-related outcomes, emphasizing its potential utility as a prognostic marker while also indicating its limitations in certain predictive contexts.
Four individuals with a low MASCC score (⩾21) died during FN, highlighting the MASCC score’s inability to predict these outcomes alone. Adding albumin (cut-off: 33.5 g/L) to this group identified 9 patients at higher risk of mortality during FN (Figure 3A). Among these, 2 deaths were recorded, demonstrating that albumin predicted 50% of additional deaths in this group. Furthermore, integrating albumin into the low-risk MASCC score group improved the accuracy of predicting RTI (cut-off: 33.5 g/L) by 62.5% (Figure 3B), and 28-day mortality (cut-off: 35.5 g/L) by 66.67% (Figure 3C).

Incorporation of albumin to the MASCC risk-index score in the risk stratification of mortality during FN (A), respiratory tract infections (B), and 28-day mortality (C).
The results of the multivariate logistic regression analysis are depicted in Figure 4. Our analysis revealed that albumin level (OR = 1.17; 95% CI = 1.02-1.36; P = .028) and ECOG performance score (OR = 0.32; 95% CI = 0.12-0.75; P = .013) emerged as significant predictors of mortality during FN (Table 3). Conversely, an albumin level < 33.5 g/L stood out as an independent predictor of GI (OR = 2.86; 95% CI = 1.37-6.13; P = .006).

Albumins predicted probability for gastrointestinal infection (A), and mortality during FN (B). – represents median value; ----- represents interquartile range.
Results of the multivariate logistic regression analysis for predictors of outcomes.
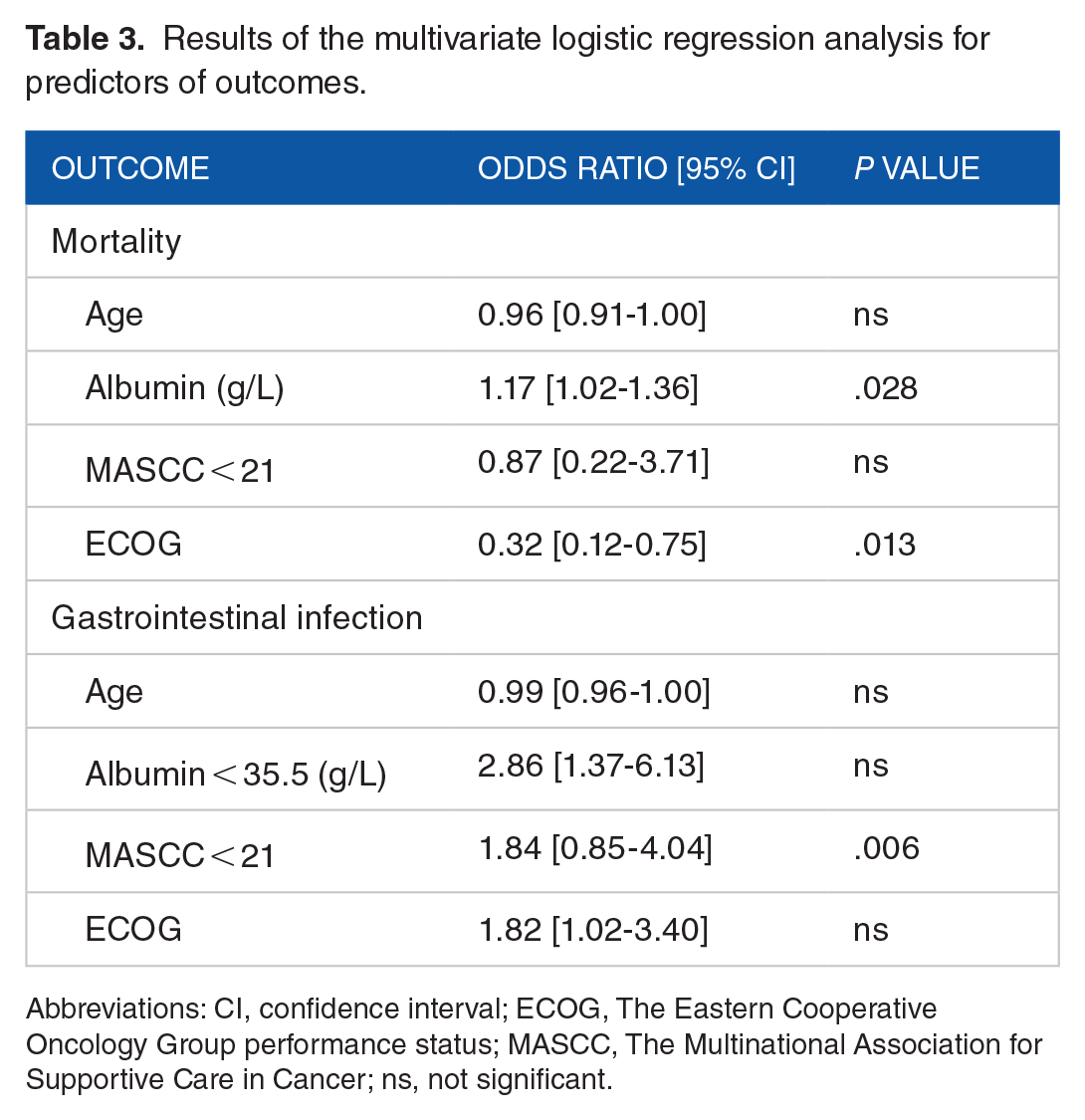
Abbreviations: CI, confidence interval; ECOG, The Eastern Cooperative Oncology Group performance status; MASCC, The Multinational Association for Supportive Care in Cancer; ns, not significant.
Discussion
This study highlights the significant prognostic value of serum albumin levels in predicting various complications and outcomes associated with FN. Our findings indicate that albumin serves as a reliable predictor of mortality risk among FN patients, both in the immediate and 28-day period following diagnosis. Remarkably, albumin demonstrates a predictive efficacy comparable with that of the widely used MASCC risk index. When serum albumin predicts mortality, including during FN and within 28 days, the tests excel in excluding the condition rather than confirming it. Positive test results in these scenarios moderately increase the odds of developing the condition compared with not developing it. Serum albumin levels also demonstrate potential in predicting RTI and GI complications, thereby supplementing the predictive capacity of the MASCC risk index for GI. Notably, the performance analysis indicates that serum albumin levels provide valuable insights, particularly in effectively ruling out the likelihood of GI development. Nevertheless, positive results should be interpreted cautiously, warranting further evaluation to confirm positive cases. Incorporating serum albumin levels into the low-risk category defined by the MASCC risk index significantly enhances its predictive capability. Specifically, the inclusion of serum albumin elevates the predictive accuracy for FN mortality by 50%, 28-day mortality by 66.67%, and RTI by 62.50%. The multivariate logistic regression analysis indicated that elevated albumin levels and lower ECOG performance scores were associated with decreased mortality risk during FN. Conversely, an albumin level below 33.5 g/L emerged as an independent predictor of GI complications.
The consequences of FN can vary based on individual patient characteristics and the underlying disease. 1 Several crucial factors influencing the severity of FN include age, the presence of comorbidities, cancer type, and cancer stage. Febrile neutropenia carries a high mortality rate ranging from 10% to 30%.17-20 However, mortality is not the only concern associated with FN. The emergence of comorbidities further complicates matters, potentially necessitating a reduction in cytostatic doses or treatment cycle delays. Both outcomes can adversely impact the effectiveness of cancer treatment and the overall survival rates. The mortality rate associated with FN, coupled with potential comorbidities, underscores the importance of prompt hospitalization and intensive treatment for these patients. In cancer supportive care, ongoing efforts to enhance prediction accuracy are crucial due to the complexity and severity of FN-related complications.
One commonly explored avenue in research for predicting the consequences and outcomes of FN involves biomarkers. Among these biomarkers, albumin stands out for ability to reflect both inflammatory and nutritional status, critical factors in FN. Previous studies have established albumin as a valuable predictor of mortality among hospitalized patients, 21 with lower levels correlating with increased mortality risk. 22 Our study supports these findings, demonstrating albumin’s predictive capacity for both mortality during FN and 28-day mortality. Specifically, albumin exhibited comparable predictive ability with the MASCC risk index for mortality during FN, with identical area under the curve values and P values (AUC = 0.75; P = .001). While albumin also predicted 28-day mortality (AUC = 0.70; P < .001), the AUC for the MASCC risk index showed a marginally higher (0.75; P < .001). Interestingly, our study suggests that serum albumin levels better predict survival rather than mortality, as indicated by a relatively low PPV and high NPV. The suboptimal sensitivity observed in our study contributed to the low PPV, 23 likely influenced by the relatively low incidence of mortality. For instance, among the 185 participants included, only 14 individuals (8.15%) died during FN, a figure lower than the expected incidence of up to 30%. 17
Complications arising from febrile neutropenia, such as bloodstream infections or nonsystemic infections, significantly impact on the overall survival of cancer patients. 24 Predicting the onset of these complications holds the potential to greatly enhance patient stratification and ensure timely, tailored care for those in need. In our study, serum albumin levels were found to predict the development of RTI and GI complications with AUC values exceeding 0.5. However, the AUC values for predicting RTIs using serum albumin were lower compared with those using the MASCC risk index, indicating the index’s superior predictive ability. Conversely, for predicting GI complications, the AUC value of serum albumin exceeded 0.5, suggesting better performance than random chance. Nonetheless, the serum albumin AUC value of 0.67 for predicting GI might be considered poor for a diagnostic test or prediction, 25 unless we acknowledge the absence of a superior option, especially given that the MASCC risk index AUC for GI complications is 0.63.
Patients classified as low risk by the MASCC risk index still face the possibility of experiencing adverse outcomes.26-28 To address this concern, we integrated serum albumin levels into the MASCC risk index, aiming to improve the prediction of mortality and complications. Our analysis demonstrated that the incorporating serum albumin levels significantly enhanced the predictive accuracy for mortality during febrile neutropenia, 28-day mortality, and RTIs.
Our study highlights that low serum albumin levels can help identify FN patients at higher risk of mortality and severe complications. This stratification can prompt more intensive monitoring and early interventions for these high-risk patients. These findings are in accordance with previous studies that show low albumin levels are associated with higher mortality rates in septic patients and that albumin is established as a valuable predictor of mortality among hospitalized medical patients.29,30
This study presents several limitations that warrant consideration. First, it is important to acknowledge that this is a single-center study, which inherently limits the generalizability of the findings to broader populations. Second, most the participants had solid tumors, thereby restricting the applicability of the results to patients with different cancer types or hematological malignancies. Third, the potential impact of varied antibiotic regimens and granulocyte colony-stimulating factor administration on patient outcomes cannot be discounted, as these factors could have influenced the observed results. In addition, while our tests demonstrated utility, it is clear that improvements in sensitivity and specificity could further enhance the validity and reliability of our findings.
Conclusion
In conclusion, serum albumin emerges as a promising biomarker for risk stratification and prognostication in FN patients, providing valuable insights into mortality and complications. Its incorporation into established risk assessment frameworks such as the MASCC index improves predictive accuracy. This enhancement facilitates more informed clinical decision-making and enables tailored patient management strategies.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
MS, JD, and DK made a substantial contribution to the concept or design of the work.
MS, JD, DK, MČ, and OĐ contributed to acquisition, analysis or interpretation of data.
MS, JD, DK, MČ, and OĐ drafted the article or revised it critically for important intellectual content.
MS, JD, DK, MČ, and OĐ approved the version to be published.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results received funding from the Ministry of Education, Science and Technological Development of Republic of Serbia under Grant Agreement No. 451-03-9/2021-14/200110 (old No. 175023).
Availability of Data and Materials
The data sets generated and analyzed during this study are available from the corresponding author upon reasonable request.
Ethics Approval and Consent to Participate
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. Ethical approval was granted by the following Institutional Review Boards:
1. Institutional Review Board of the Institute for Oncology and Radiology of Serbia, which serves as the National Cancer Research Center. Approval number for this study: No. 1184-01.
2. Institutional Review Board of the Medical Faculty, University of Belgrade. Approval number for this study: No. 1322/VII-15.
Written informed consent was obtained from all participants involved in this study. They were informed about the purpose of the study, procedures involved, potential risks and benefits, and their right to withdraw from the study at any time without consequence. Confidentiality of participants’ information was strictly maintained throughout the study. To ensure confidentiality and privacy of participants all study data were analyzed anonymously. Personally identifiable information was removed or replaced with unique identifiers before data were analyzed. Only authorized personnel had access to the coded data, and any identifying information was securely stored separately from the analyzed data set.
Consent for Publication
Not applicable.
