Abstract
Background:
Cancer of unknown primary (CUP) is difficult to diagnose and classify clinically, and the disease develops rapidly. Therefore, the primary tumor detected in patients with CUP plays a profound role in the diagnosis and treatment of patients. The search for the primary tumor of CUP is also one of the indications for 18F-fluoro-2-deoxyglucose-positron emission tomography and computed tomography (FDG-PET/CT). Our objective was to evaluate the role of 18F-FDG PET/CT imaging in primary tumor detection and treatment formulation in patients with CUP.
Methods:
Sixty-two patients with CUP were selected from a database consisting of 18 802 cases in the Jiangsu Cancer Hospital PET/CT center from May 18, 2016 to November 18, 2022. Clinical data and changes in treatment strategies before and after PET/CT were collected.
Results:
A total of 42 primary tumors (42/62, 67.7%) were identified by PET/CT examination. The tumor staging of patients before conventional PET/CT imaging (such as CT/magnetic resonance imaging [MRI]/US) and after PET/CT did not change in 28 patients (28/62, 45.2%), whereas for 34 patients (34/62, 54.8%), tumor staging changed. Forty-five patients (45/62, 72.6%) had not developed treatment plans before PET/CT examination, but treatment plans were clarified after PET/CT examination. Thirteen patients (13/62, 21.0%) underwent changes in treatments before and after PET/CT examination. Among the 20 patients (20/62, 32.3%) whose primary tumors were not detected, 16 patients (16/20, 80.0%) had no treatment plans before PET/CT and the treatment plans were defined after PET/CT, 3 patients (3/20, 15.0%) changed the treatment plans before and after PET/CT, and 1 patient (1/20, 5.0%) did not change the treatment plan.
Conclusions:
The 18F-FDG PET/CT plays an important role in the detection and staging of primary tumors in patients with CUP. The PET/CT findings can not only help clinicians develop appropriate treatment plans for patients with CUP but also serve as an effective approach to improve real-life treatment strategies for these patients.
Introduction
A tumor is a disease characterized by the disordered malignant proliferation of cells. According to the latest epidemiological data, there were approximately 19.3 million new cancer cases and almost 10.0 million cancer deaths every year, ranking second (cardiovascular and cerebrovascular diseases are the first with 18.6 million deaths per year).1,2 With the continuous progress of medical science and technology, the overall 5 year survival rate of malignant tumors continues to improve. Among all malignancies, cancer of unknown primary (CUP) refers to a series of comprehensive diagnostic methods, including clinical data, laboratory examination, traditional imaging examination (including computed tomography [CT], magnetic resonance imaging [MRI], X-ray, ultrasound [US]) and histopathologic examination, and the clinician considered metastatic cancer first when excluding other primary cancers, which accounts for approximately 1% to 2% of cancers.3,4 The 5 year overall survival rate of CUP has not improved significantly in the last 20 years. 5
Cancer of unknown primary is difficult to diagnose and classify clinically, and the disease develops rapidly. Most tumors are not sensitive to systematic treatment, bringing great difficulties to identifying appropriate clinical treatment. Therefore, the primary tumor detected in patients with CUP plays a profound role in the diagnosis and treatment of patients. 6 The key to effective treatment of CUP is to find the primary tumor so that targeted treatment may be developed. Traditional methods for finding primary lesions include clinical physical examination, laboratory examination, and traditional imaging studies, including CT, MRI, and US. Patients with CUP undergo a series of multidisciplinary diagnostic evaluations, and imaging plays an irreplaceable role. 7 During the past 30 years, the accuracy of detecting primary tumors by CT or magnetic resonance imaging has increased from 11%-26% to 33%-55%. 8 In a study with 879 participants,8,9 CT scans provided an overall diagnostic accuracy of 55%. More than 50% of patients did not have their primary tumor detected by CT or MRI. 10
In the past 20 years, a new medical imaging device, 18F-fluoro-2-deoxyglucose PET/CT (FDG-PET/CT), has been widely used in the clinic. Especially in cancer staging, prognosis, and patient management, 18F-FDG PET/CT plays an important role. The search for the primary tumor of CUP is also one of the indications for 18F-FDG PET/CT. However, compared with MRI or CT, the diagnostic value of 18F-FDG-PET/CT to identify the location of the primary tumor in CUP, the extent of the lesion, the metastatic lymph nodes, and distant metastases are still controversial in the literature. In some guidelines, 18F-FDG-PET/CT is used only as an additional supplement to routine tests.11-20 In a large study involving 449 patients, Yu et al 21 found that PET/CT scans accurately detected the primary tumor in 115 patients, which inspired the use of PET/CT studies in patients at an early stage of diagnosis. However, it has also been argued that despite statistical evidence that PET/CT is a better tool for identifying the origin of cancer than MRI (22%-44% vs 20%-27%). 22 The main reason for this difference may be that there are some differences in the CUP inclusion criteria. Generally, the more stringent the inclusion requirements, the better the PET/CT results.
Furthermore, patients with CUP can still change their treatment regimen even if no primary lesion is found after the 18F-FDG PET/CT examination. One third of the patients changed treatment due to the results of FDG-PET or PET/CT, a finding consistent between many subgroups.21,23
We aimed to provide evidence supporting the role of 18F-FDG PET/CT imaging in detecting primary lesions in patients with CUP and in establishing treatment plans.
Materials and Methods
Patients
Sixty-two patients with CUP were selected from a database consisting of 18 802 cases at the Jiangsu Cancer Hospital PET/CT center from May 18, 2016 to November 18, 2022.
As there were no uniform specific inclusion criteria for CUP in the previous literature, in this study, we adopted stricter inclusion criteria. The inclusion criteria were as follows: (1) histopathology-confirmed malignant tumor; (2) absence of primary tumor detected by CT/MRI; (3) clinician confirmation of CUP; (4) no history of malignant tumor treatment before PET/CT; and (5) clinician-developed or will soon develop a treatment plan. Exclusion criteria were as follows: (1) primary tumor detected by CT/MRI; (2) previous history of cancer; or (3) history of malignant tumor treatment before PET/CT (Figure 1).

Patient grouping flow chart of the study.
Positron emission tomography and computed tomography imaging
All patient images were captured using GE Medical Systems, Discovery 710 PET/CT, Waukesha, Wisconsin, USA. Each patient received an intravenous infusion of 18F-FDG at 0.1 to 0.2 mCi/kg after fasting for at least 6 hours. Fasting blood glucose levels were maintained below 11 mmol/L before intravenous radiopharmaceutical. The patient was then arranged to lie down in a quiet and comfortable room while receiving 1000 mL of water. Order scans began immediately after emptying the bladder. The patient was instructed to drink rink 300 to 500 mL of water to fill the gastrointestinal tract before entering the examination room.
Combined image acquisition was performed approximately 50 to 70 minutes after 18F-FDG injection. The first step was to perform a body scan. The scan covered 6 to 7 positions on the bed from the skull base to the middle of the thigh. During examination, the patients lay supine on the examination bed and held their hands above the head. Positron emission tomography and CT images were collected. Computed tomography scanning parameters: layer thickness 3.75 mm, pitch 0.984, tube voltage 140 kVp, automatic tube current mA, frame rotation speed 0.8 s/rot, 512 × 512 reconstructed matrix; PET scan parameters: 2 min/bed. Computed tomography images were reconstructed using the iterative image reconstruction algorithm, and the reconstruction function was Stnd Wide View. The second step was to perform a thin-slice CT scan of the chest, only CT images were collected. Thin-slice chest CT scanning: layer thickness of 1.25 mm, pitch of 1.375, tube voltage of 120 kVp, tube current of automatic adjustment mA, frame rotation speed of 0.6 s/rot, and 512 × 512 reconstructed matrix. An iterative image reconstruction algorithm was used for reconstruction. The mediastinal window reconstruction function was Stnd and Lung window reconstruction function. The third step was a head scan. The patients placed their hands at each side or on the abdomen of the body. Positron emission tomography and CT images were collected. Computed tomography scanning parameters: layer thickness 3.75 mm, pitch 0.516, tube voltage 120 kVp, tube current 300 mA, frame rotation speed 1.0 s/rot, 192 × 192 reconstructed matrix; PET scan parameters: 5 min/bed, 1 bed covered the scan area. The expected maximization algorithm of ordered subset based on CT attenuation correction was used to reconstruct the PET image data set iteratively. Trans axial, sagittal, coronal, and fused images were analyzed on an Advanced Workstation AW4.6 (GE HealthCare Biosciences, Piscataway, NJ, USA).
Positron emission tomography and computed tomography image analysis
All images were independently evaluated by 2 senior nuclear medicine physicians (RH and YZ) who have been practicing for more than 10 years and have reviewed more than 1000 cases of patient images every year. If the results were inconsistent, a third senior nuclear medicine physician (YH) was added to assess. The third senior nuclear medicine physician has been practicing for more than 20 years and has reviewed more than 1500 patient images every year.
Evaluation of patient treatment options
Progress notes from all patients were reviewed and recorded, including imaging data, biochemical results, expected treatment plans before PET/CT, and the treatment plans finalized and implemented after PET/CT.
Data analysis
Identification of the primary tumor with positron emission tomography and computed tomography
The detection of primary tumor, lymph node metastasis, and distant metastasis was evaluated on the basis of PET/CT reports. Puncture or biopsy of suspected primary tumors on PET/CT images showed clear histopathology, all of which indicated malignant tumors. None of patients had received therapy before PET/CT examination. Management before and after PET/CT were analyzed.
Determination of utility of positron emission tomography and computed tomography to assist in the formulation of the treatment plan or change the treatment plan
For other lesions found on PET/CT, such as metastatic lymph nodes and distant metastases, conclusions were drawn based on comprehensive clinical data. Some patients did not have a detailed treatment plan prior to PET/CT examination. After PET/CT examination, the clinicians prepared a detailed treatment plan for the patients according to the results and the relevant clinical data obtained. We identified this situation as PET/CT assisted in the formulation of a treatment plan.
For other patients, the clinician made a detailed treatment plan based on the clinical data obtained before PET/CT examination, but it had not been implemented. However, after PET/CT examination, the clinician changed the patient’s treatment plan according to the PET/CT results, including from surgery to chemotherapy or immunotherapy, chemotherapy to radiotherapy or surgery combined with chemotherapy, radiotherapy to surgery or chemoradiotherapy, chemoembolization to chemotherapy, all of which are considered to have changed the treatment plan following PET/CT.
Statistics
Continuous variables are expressed by mean ± SD, and categorical variables are expressed by frequency with percentage. The detection rate was calculated with 95% confidence intervals (CIs) for statistical hypothesis testing, the 95% CI was calculated by the exact probability method.
Results
Detection of primary tumors by positron emission tomography and computed tomography
Sixty-two patients with CUP syndrome were included (16 women, 46 men; mean age 60.26 ± 11.82 years) in this study.
Fifty-six patients (56/62, 90.3%) underwent traditional imaging examinations, such as MRI (13/62, 21.0%) or CT (36/62, 58.1%), or US (7/62, 11.3%), before PET/CT examination. The time interval between these examinations and PET/CT was 15.36 ±11.90 days. Six patients (6/62, 9.7%) did not undergo imaging prior to PET/CT examination.
A total of 62 primary tumors were confirmed by histopathology in 62 patients. The highest proportion was adenocarcinoma (29/62, 46.8% [95% CI = 34.0%-59.9%]), followed by squamous cell carcinoma (22/62, 35.5% [95% CI = 23.7%-48.7%]), poorly differentiated/undifferentiated carcinoma (7/62, 11.3% [95% CI = 4.7%-21.9%]), malignant melanoma (1/62, 1.6% [95%CI 0.0%-8.7%]), small cell carcinoma (1/62, 1.6% [95% CI = 0.0%-8.7%]), lymphoepithelial carcinoma (1/62, 1.6% [95% CI = 0.0%-8.7%]), and sarcoma (1/62, 1.6% [95% CI = 0.0%-8.7%]).
Fifty-seven patients (57/62, 91.9% [95% CI = 82.2%-97.3%]) had lymph node metastases, including 34 patients (34/62, 54.8% [95% CI = 41.7%-67.5%]) with cervical lymph node metastasis and 5 patients (5/62, 8.1% [95% CI = 2.7%-17.8%]) did not have lymph node metastases. Distant metastasis of different organs was detected in 30 patients (30/62, 48.4% [95% CI = 35.5%-61.4%]) and was not detected in 32 patients (32/62, 51.6% [95% CI = 38.6%-64.5%]).
All clinical details of patients with CUP are shown in Table 1. A total of 42 primary tumors (42/62, 67.7% [95% CI = 54.7%-79.1%]) were identified by PET/CT examination. Fifteen lesions (15/42, 35.7% [95% CI = 21.6%-52.0%]) were detected in the head and neck, 12 in the lungs (12/42, 28.6% [95% CI = 15.7%-44.6%]), 4 in the ovaries (4/42, 9.5% [95% CI = 2.7%-22.6%]), 3 in the stomach (3/42, 7.1% [95% CI = 1.5%-19.5%]), 3 in the colon (3/42, 7.1% [95% CI = 1.5%-19.5%]), 2 in the esophagus (2/42, 4.8% [95% CI = 0.6%-16.2%]), 1 in the gallbladder (1/42, 2.4% [95% CI = 0.1%-12.6%]), 1 in the kidneys (1/42, 2.4% [95% CI = 0.1%-12.6%]), and 1 in bone (1/42, 2.4% [95% CI = 0.1%-12.6%]). No primary tumor was identified in 20 patients (20/62, 32.3% [95% CI = 20.9%-45.3%]).
Clinical details of CUP patients.

The influence of positron emission tomography and computed tomography on the clinical treatment of patients
The tumor staging of patients before conventional PET/CT imaging (such as CT/MRI/US) and after PET/CT did not change in 28 patients (28/62, 45.2% [95% CI = 32.5%-58.3%]), 34 patients (34/62, 54.8% [95% CI = 41.7%-67.5%]) changed tumor staging (Figure 2). Of the 34 patients with changed tumor stage, 16 patients (16/34, 47.1% [95% CI = 29.8%-64.9%]) were upgraded from stage II/III to stage IV due to additional metastases identified on PET/CT; 18 patients (18/34, 52.9% [95% CI = 35.1%-70.2%]) had T stage diagnosed due to PET/CT detection of the primary tumor.
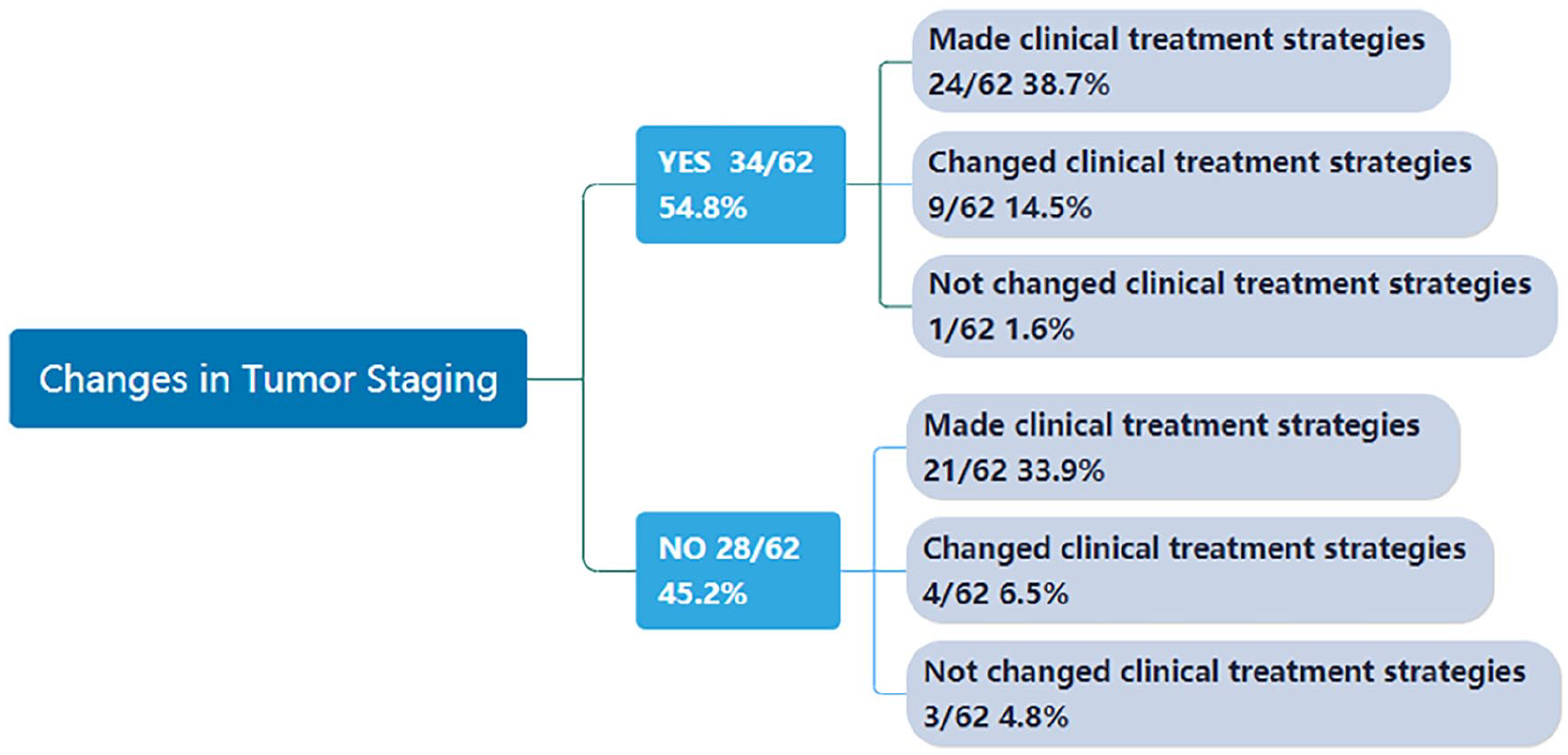
The tumor staging of the CUP changed or did not after PET/CT examination, resulting in a change in treatment strategy.
Among the 62 patients, 45 patients (45/62, 72.6% [95% CI = 59.8%-83.1%]) had not received a treatment plan prior to PET/CT examination, but for 44 patients (44/62, 71.0% [95% CI = 58.1%-81.8%]) plans were clarified after PET/CT examination. The treatment of 1 patient (1/62, 1.6% [95% CI = 0.0%-8.7%]) was not mentioned in the progress notes.
Thirteen patients (13/62, 21.0% [95% CI = 11.7%-33.2%]) underwent changes in their treatments before and after PET/CT examination (Figure 3). Details of changes in treatments for the patients are documented in Figure 4. The treatment regimens of 4 patients (4/62, 6.5% [95% CI = 1.8%-15.7%]) did not change after PET/CT.

Male patient, 54 years old. Pain in the chest area for more than 3 months. CT examination revealed a sternal mass. Pathological biopsy revealed high-grade sarcoma with necrosis. The clinician made the surgical plan. PET/CT examination was performed prior to surgery. The above figure shows 3DMIP, PET, CT, and fusion of PET and CT. There was a primary sternal malignancy (thin black arrow) with multiple metastases in the left lobe of the liver (thick black arrow). Then, the treatment was changed to chemotherapy.
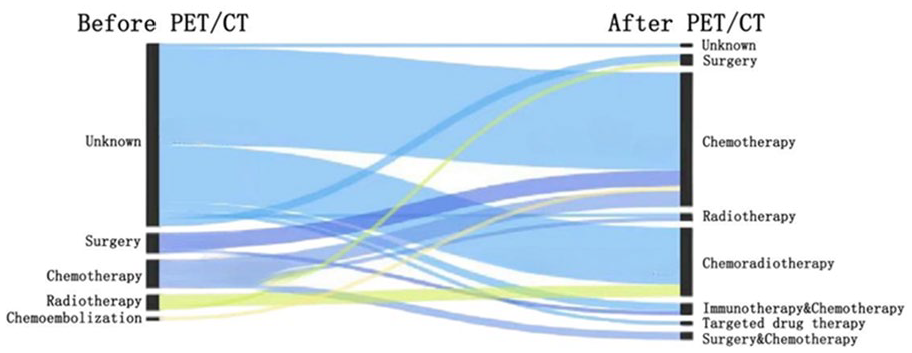
The Sankey chart shows the treatment strategies (left) before and (right) after 18F-FDG-PET/CT in a patient with unknown primary cancer. The left box indicates the pre-PET treatment plans and the right box indicates the post-PET treatments, annotated with the respective types of administration (such as surgery, radiotherapy, chemotherapy, chemoradiotherapy, chemoembolization, targeted drug therapy and immunotherapy). “Unknown” indicates that no definitive treatment plan was formulated.
Among the 20 patients (20/62, 32.3% [95% CI = 20.9%-45.3%]) whose primary tumor was not detected, 16 patients (16/20, 80.0% [95% CI = 56.3%-94.3%]) did not have a treatment plan before PET/CT, and the treatment plan was defined following the PET/CT, 3 patients (3/20, 15.0% [95% CI = 3.2%-37.9%]) had their treatment plan changed before and after PET/CT, and 1 patient (1/20, 5.0% [95% CI = 0.1%-24.9%]) did not receive any change in treatment plan.
Discussion
We selected 62 patients with CUP from a total of 18 802 cases visiting our center over the past 6 years for this study and the following findings were obtained: (1) primary tumors could be detected by PET/CT, (2) PET/CT could be used to assist clinicians in developing or changing treatment strategies, and (3) even if the primary tumor could not be found by PET/CT, it was helpful for the formulation (or modification) of the patient’s treatment plan.
In our cohort study, the detection rate of primary lesions in patients with CUP after PET/CT was 67.7%, which was at the high end of the detection rate reported in the relevant literature. According to the definition of CUP, the study design, the clinical manifestations and indications, the detection rate of primary tumors in patients with CUP was 14.6% to 73%.24,25 However, the estimated tumor detection rate for PET/CT was 39% for Soni et al. 26 In a 155-patient cohort study by Bicakci, 27 18 F-FDG PET/CT correctly diagnosed the primary tumor with a 41% (true positive) detection rate. We included a cohort of more stringent requirements which benefited the performance of FDG PET/CT. All of our patients were histopathologically confirmed with CUP, but in these studies, only some of the patients were histopathologically indicated, which have resulted in the relatively low rates of primary tumor detection on PET/CT. Most patients had tumors in the head and neck and lung, which is consistent with other reports in the literature;28,29 nonetheless, the histopathologic results showed that adenocarcinoma was the most frequent tumor, followed by squamous cell carcinoma. This result may be attributed to the following reason: head and neck tumors include nasopharyngeal cancer, oropharyngeal cancer, laryngeal cancer, and hypopharyngeal cancer, which can contribute to the highest incidence of head and neck cancer in statistics. There were also 20 patients with no primary tumor, most of which were histopathologically diagnosed as metastatic lymph nodes in the neck, perhaps related to latent nasopharyngeal carcinoma.
Cancer of unknown primary is a heterogeneous tumor, which is difficult to diagnose and classify clinically. Most tumors are not sensitive to systematic treatment, which results in great difficulty in clinical treatment. For isolated nodules, surgical excision or local radiotherapy with or without chemotherapy may be considered. Most patients with CUP are diagnosed at an advanced stage and lose the opportunity for surgical treatment. Clinically, chemotherapy, radiotherapy, immunotherapy, targeted therapy, or a combination of several therapies is used according to histopathologic classification.
Our study mainly analyzed changes in clinical treatment strategies in CUP patients before and after PET/CT examination. We found that 72.6% (45/62) of the patients did not have treatment plans prior to PET/CT examination, and treatment strategies were determined following the PET/CT examination, indicating that PET/CT is a powerful method for clinicians to develop treatment plans in CUP patients.
To date, most of the literature has focused on the ability of PET/CT to detect primary tumors in CUP patients. However, our study focused more on the impact of PET/CT on clinical treatment strategies for patients with CUP and showed a great clinical relevance. We found that 21.0% (13/62) of the patients changed their treatment regimen after PET/CT examination. The reasons for the changes either involved the discovery of primary tumor lesions or of metastatic lesions. Some patients received changes in tumor staging, although some did not; nonetheless, the original treatment plan was ultimately changed. Of these patients, 9 (9/13, 69.2%) received changes in tumor staging, and in 5, the original treatment regimen was adjusted due to the identification of the primary lesion. However, in this study, this subset of patients had initially been targeted for palliative management; although once the primary lesion was identified, the physician could adjust it a more aggressive or even more potentially curative treatment, from which the patient could benefit. However, this study did not evaluate whether the survival time was prolonged in the patient cohort, which could be addressed in future direction or as a prospective research study. Distant metastasis was identified in 3 patients; the treatment regimen was adjusted accordingly from curative treatment to palliative treatment, saving patients from unnecessary invasive injury.
Interestingly, not all patients with CUP who changed tumor staging changed their treatment strategy. In our study, 54.8% of patients changed tumor staging after PET/CT and only 14.5% changed the original treatment plan, with 38.7% of patients changing from an uncertain treatment plan to a more definite treatment. This was similar to the results of previous studies. 23 These differences may be attributed to chemotherapy originally being considered for some patients ineligible for surgery. Although the tumor staging was changed by the discovery of new metastases after PET/CT in some cases, the original chemotherapy treatment strategy was not affected. We may speculate that PET/CT will be more helpful for patients in earlier staging of treatment than for patients in staging III/IV. Conversely, PET/CT could be avoided over treatment of patients.
We found that in some patients with CUP, even though no primary tumor was found, PET/CT could still change their treatment strategies, and this was where the novelty of this study lied. In this study, for 20 patients, the primary tumor was not identified, but for most of these patients (16/20, 80.0%), PET/CT resulted in a change in treatment strategy (ie, chemotherapy or chemoradiotherapy). Following PET/CT examinations, 3 patients (3/20, 15.0%) had their original treatment strategies changed. In 1 patient, radiotherapy was decided prior to PET/CT screening, but following PET/CT, only 1 lymph node on the left-side of the neck was found to be swollen; thus, the treatment plan was changed to surgery and the patient was changed from palliative treatment to curative treatment. Only 1 patient (1/20, 5.0%) maintained the original treatment strategy.
Although PET/CT is increasingly used in patients with CUP, many guidelines currently do not include PET/CT as a routine test in the diagnosis of CUP. This may also depended on how rigid a health care system on reimbursement of FDG PET/CT was. However, in some Scandinavian countries, earlier accounts have pointed to “unlimited access.” 30 The clinician can decide whether to perform PET/CT according to the patient’s condition.4,31 Our results support the inclusion of PET/CT in the routine screening workup of patients with CUP as an effective approach to improve treatment regimens.
There are some limitations to this study. First, although we intentionally broadened the screening criteria, the number of eligible cases was still not sufficiently large. Second, this study was a retrospective study. Thus, even in cases with slight changes in the chemotherapy regimen in some patients or missing information from the medical history, no further collection may be performed, which may have led to selection bias.
Conclusions
Positron emission tomography and computed tomography plays an important role in the detection and staging of primary tumors in patients with CUP. Positron emission tomography and computed tomography can not only help clinicians develop treatment plans for patients with CUP but also serves as an effective approach to improve real-life treatment strategies for these patients. In addition, for a subset of patients with CUP whose primary tumor was not found, PET/CT could still be helpful in their treatment strategies.
Footnotes
Acknowledgements
None.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by RH, YH, and YZ. The first draft of the manuscript was written by RH, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the key project of Jiangsu Commission of Health [No. K2023021] and the talents program of Jiangsu Cancer Hospital [No. YC201801].
Availability of Data and Materials
The data and materials presented in this study are available on request from the corresponding author.
Consent for Publication
All patients signed consent forms for publication.
Ethics Approval and Consent to Participate
This study was approved by the Jiangsu Cancer Hospital Ethics Committee (No. 2022-K026). All patients signed informed consent forms.
