Abstract
The treatment of locally advanced rectal cancer (LARC) is a challenging situation for radiation oncologists and colorectal surgeons. Most current approaches recommend neoadjuvant fluorouracil or capecitabine-based chemoradiotherapy followed by surgery as a standard of care. Intensification of concurrent chemotherapy by adding oxaliplatin to fluorouracil or capecitabine backbone to get better outcomes is the matter that has remained unresolved. In this review, we searched Medline and Google Scholar databases and selected 28 prospective phase II and III clinical trials that addressed this question. We discussed the potential advantages and drawbacks of incorporating oxaliplatin into concurrent chemoradiation therapy. We tried to define whether adding oxaliplatin to concurrent chemoradiation with excellent performance and high-risk features benefits some subpopulations. The available literature suggests that by adding oxaliplatin there are some benefits in enhancing response to neoadjuvant chemoradiotherapy, however, without any translated improvements in long-term outcomes including overall and disease-free survival.
Introduction
Rectal cancer is the ninth most common type of cancer worldwide, responsible for 7.6% of all cancers in 2020. 1 Rectal cancer presents as a locally advanced disease in more than 60% of patients, making multi-disciplinary management fundamental for achieving the best potential outcome. The current National Comprehensive Cancer Network (NCCN) guidelines suggest concurrent chemoradiotherapy followed by surgery with or without chemotherapy as the standard treatment of locally advanced rectal cancer (LARC). 2 Multiple concurrent chemotherapy agents have been proposed and tested, in adjunct to radiotherapy, to improve the outcomes over the past years.
The current NCCN and ESMO (European Society for Medical Oncology) guidelines recommend using 5-fluorouracil (5FU) or capecitabine together with long-course radiotherapy in the neoadjuvant setting for rectal cancer.2,3 This approach seems insufficient for some high-risk patients (eg, T4 primary tumors, those with large mesorectal nodes or extra-mesorectal nodes, and those with mucinous histology, involved mesorectal fascia or extra-mural venous invasion) who died to local or distant recurrence of the disease. Also, some patients especially with low-lying tumors may be candidates for organ preservation in the presence of a clinical complete response. Numerous investigations have been carried out to enhance the effectiveness of neoadjuvant treatments. These investigations can be broadly classified into the following categories: increasing the intensity of radiotherapy, simultaneous implementation of external beam radiotherapy (EBRT) boost, employing brachytherapy boost, total neoadjuvant treatment (TNT) approach, and administering combinational chemotherapies concurrently with radiation.
Among these, the role of adding oxaliplatin to fluoropyrimidine-based chemoradiotherapy to get better locoregional control and survival benefits has been investigated in numerous prospective and retrospective studies. However, there are still various issues that still need to be solved regarding its actual benefit. In summary, although most randomized phase III trials reported no actual benefit despite increased toxicity for adding oxaliplatin to radiotherapy,4-7 a few phase III and many phase II trials have mentioned some benefits regarding pathologic complete response (pCR), disease-free survival (DFS), and/or locoregional control (LRC).8-15 This study aims to review the recent studies evaluating the effects of adding oxaliplatin to neoadjuvant chemoradiation for LARC and examine the potential advantages and disadvantages.
Method
To find the relevant studies from the literature, we searched Medline and Google Scholar databases (Figure 1). The search keywords included rectal cancer, oxaliplatin, and radiotherapy. Using RT in the postoperative setting, short-course RT, lack of proper data, and nonrandomized trials were the reasons for exclusion. A total of 277 abstracts were screened, and those related to treatment intensification with oxaliplatin were selected. Finally, 41 full-text articles were carefully read, and finally, a total of 28 prospective phases II and III randomized clinical trials (RCTs) and 3 meta-analyses were selected between 2003 and 2022. Outcomes and complications were analyzed from each study.

Flowchart for included and excluded trial.
Type of Trials and Efficacy of Oxaliplatin
Phase III RCTs
Several randomized phase III trials addressed the potential benefit of adding oxaliplatin to neoadjuvant chemoradiation in resectable LARC (Table 1). These include FOWARC, 14 PETACC6, 4 STAR-01,7,16 CAO/ARO/AIO-04, 15 NSABP R-04, 6 and ACCORD125 trials.
The oncological short- and long-term outcomes of phase III randomized clinical trials.
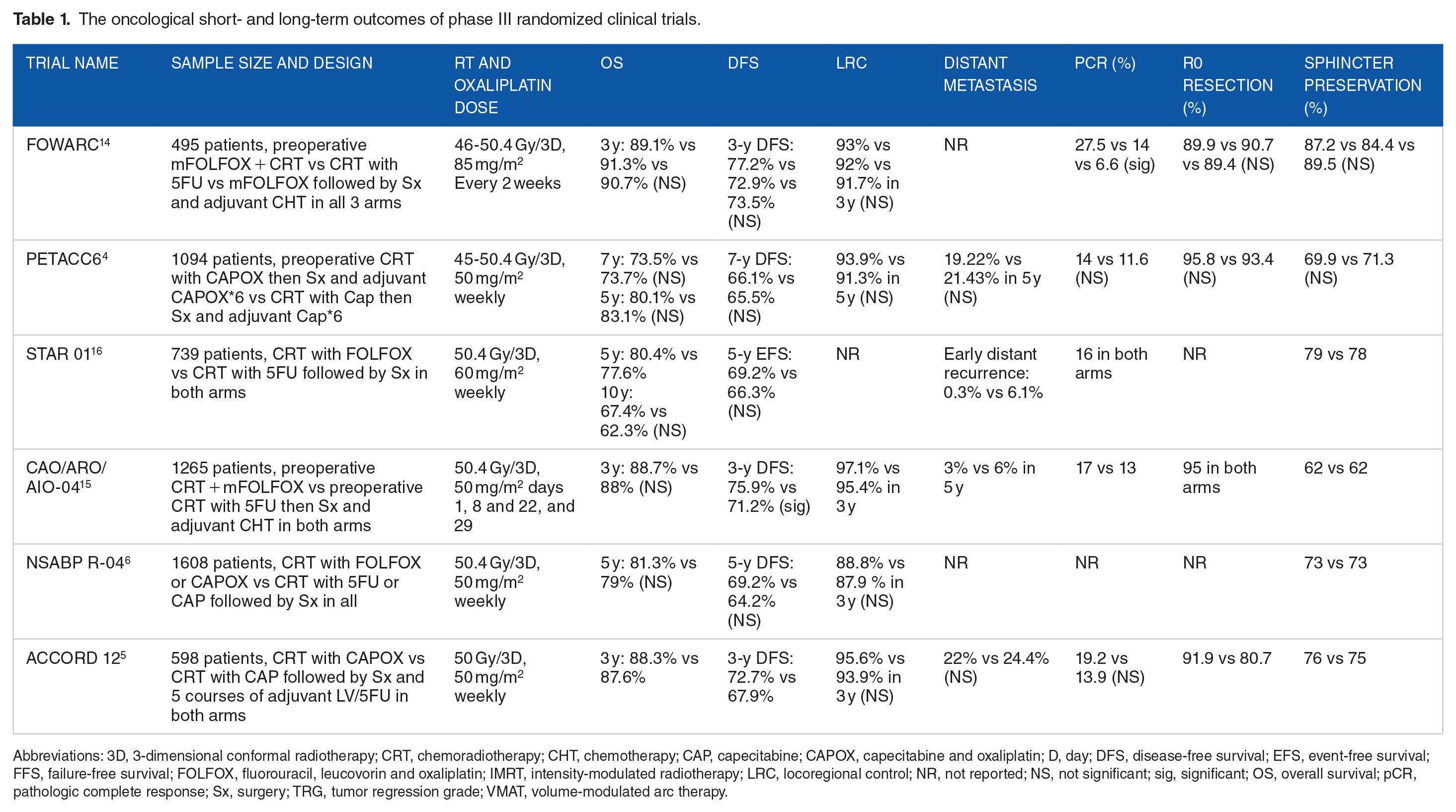
Abbreviations: 3D, 3-dimensional conformal radiotherapy; CRT, chemoradiotherapy; CHT, chemotherapy; CAP, capecitabine; CAPOX, capecitabine and oxaliplatin; D, day; DFS, disease-free survival; EFS, event-free survival; FFS, failure-free survival; FOLFOX, fluorouracil, leucovorin and oxaliplatin; IMRT, intensity-modulated radiotherapy; LRC, locoregional control; NR, not reported; NS, not significant; sig, significant; OS, overall survival; pCR, pathologic complete response; Sx, surgery; TRG, tumor regression grade; VMAT, volume-modulated arc therapy.
FOWARC was a 3-arm Chinese trial comparing mFOLFOX with neoadjuvant chemoradiotherapy (nCRT) vs nCRT with 5FU vs mFOLFOX chemotherapy alone, followed by surgery in all arms. The dose of oxaliplatin (85 mg/m2) was higher than in other studies but surprisingly did not result in significantly higher G3-4 complications. The authors reported inferior results and a lower pCR rate for chemotherapy alone rather than chemoradiotherapy. However, mFOLFOX chemoradiotherapy resulted in a higher pCR rate than 5FU-based treatment. 14 Nonetheless, in the long-term update, this improvement in pCR was not accompanied by any significant increase in survival. 17
The PETACC6 trial randomly assigned patients to perioperative nCRT with CAPOX or capecitabine followed by surgery and adjuvant CAPOX in both arms. The authors reported no benefit in 5-year DFS, overall survival (OS), and local control but significantly higher G3-4 toxicity in the CAPOX arm. Almost half of the patients experienced G2 toxicity and a higher rate of peripheral neuropathy, which was unacceptable considering the limited benefits of treatment. 4
In the multicenter Italian STAR-01 trial, patients received preoperative nCRT with oxaliplatin and 5FU vs 5FU alone, then surgery in both arms. After the interim analysis, the authors reported significantly higher toxicity without affecting the primary tumor response by adding oxaliplatin. 16 In the final survival analysis, the oxaliplatin arm showed a non-significant trend in favor of better OS and DFS. 7 An interesting finding was that the addition of oxaliplatin significantly improved the rate of early distant recurrence (pathological M1 during surgery) by affecting micrometastases. 18 Overall, this study did not meet its primary endpoint of a 30% reduction in mortality rates but reported a small nonsignificant OS benefit from the oxaliplatin arm that needs further investigation. 7
CAO/ARO/AIO-04 was the only phase III trial reporting significant DFS benefits for adding oxaliplatin to concurrent 5FU although without an OS benefit. The authors compared nCRT with oxaliplatin and 5FU vs nCRT with 5FU alone, followed by surgery and adjuvant chemotherapy in both arms. The oxaliplatin arm had superior results in pCR and 3-year DFS. The authors reported that adding oxaliplatin to fluorouracil-based nCRT significantly improved the DFS of patients with clinically staged cT3-4 or cN1-2 rectal cancer compared with the former fluorouracil-based combined modality regimen. 15
NSABP R-04 was the largest trial of its kind addressing the challenge of oxaliplatin addition to routine 5FU-based CRT. In this trial, the investigators randomized patients into CRT plus CAPOX/FOLFOX vs CRT plus CAP/5FU followed by a planned resection and postoperative chemotherapy in all patients. There was a slight improvement in 5-year OS and DFS that were not statistically significant. The authors stated that despite increasing toxicity, the addition of oxaliplatin did not improve any oncological outcome including LRC, DFS, or OS. 6
Patients were randomly assigned to 2 groups in the ACCORD12 trial. 5 Both groups received nCRT consisting of 45 Gy external beam radiotherapy (EBRT) with capecitabine in 1 group and 50 Gy EBRT with capecitabine and oxaliplatin (CAPOX) in the other. Surgery was performed 6 weeks after nCRT. Short-term results showed no significant differences in clinical outcomes between the 2 groups. Long-term results after 5 years showed no differences between groups regarding DFS or OS. 19 The authors reported no significant difference in clinical outcome with the intensified CAPOX regimen.
Phase II RCTs
Numerous phase II trials investigated adding oxaliplatin to concurrent chemoradiation (Table 2). These trials provided better results in both outcomes and toxicity compared with the phase III trials. In 2021 in the single-arm PROARCT trial, 10 opting for a split course method, patients were treated to a total dose of 50.4 Gy with concurrent CAPOX. The investigators reported the feasibility and safety of the protocol without increase in post-treatment complications. The INTERACT trial 9 which was published in 2018 compared an additional boost to a gross tumor to intensified concurrent chemotherapy. They randomized 534 patients to either 45 Gy EBRT and capecitabine plus 10 Gy boost to the bulky site or 45 Gy EBRT and concurrent oxaliplatin plus capecitabine. The dose of oxaliplatin in the intensified chemotherapy arm was 130 mg/m2 on days 1, 19, and 38. According to the final results, pCR, 5-year OS, and 5-year DFS were not different between groups, but the concomitant boost arm obtained significantly higher rates of tumor regression grades 1 to 2 with a low toxicity. Yu et al 8 performed a different method using simultaneous oxaliplatin and bevacizumab concurrently with radiotherapy and reached a promising pCR and 3-year survival rate. Complications were tolerable overall, but an increased number of anastomotic leaks has created a concern about the safety of this regimen, with further investigation needed to ensure its safety. Thus, this protocol is not recommended outside a clinical trial. Yaghobi Joybari et al 20 randomized 114 patients with cT3-4 N0 or T any N+ into nCRT with oxaliplatin and capecitabine followed by surgery and nCRT with capecitabine followed by surgery. The oxaliplatin dose was 50 mg/m2 weekly. Oxaliplatin did not improve pCR and DFS. Tang et al 21 used a high-dose oxaliplatin (130 mg/m2) regimen concurrent with CAP and RT followed by 1 course of CAPOX and surgery and postoperative chemotherapy with the same regimen. The LRC and distant metastasis (DM) control rates were excellent exceeding 95% at 51 months.
The oncological short- and long-term outcomes of phase II clinical trials.

Abbreviations: 3D, 3-dimension; CRT, chemoradiotherapy; CHT, chemotherapy; CAP, capecitabine; CAPOX, capecitabine and oxaliplatin; D, day; DFS, disease-free survival; EFS, event-free survival; FOLFOX, fluorouracil, leucovorin and oxaliplatin; IMRT, intensity-modulated radiotherapy; LN, lymph node; LRC, locoregional control; NR, not reported; NS, not significant; OS, overall survival; pCR, pathologic complete response; Sx, surgery; VMAT, volume-modulated arc therapy.
In another single-arm study in 2018, Aghili et al 22 assessed the efficacy and safety of concurrent and consolidation CAPOX during and following short-course RT followed by delayed surgery. The dose of oxaliplatin was 85 mg/m2 once on the first day of RT and 135 mg/m2 once as consolidation 3 to 4 weeks after completion of RT. The results were acceptable, and the authors reported a favorable pCR rate and feasibility and tolerability of their regimen.
In 2017, in a single-arm study by De Felice et al, 11 100 patients were treated by intensified nCRT with oxaliplatin and 5FU, followed by surgery and adjuvant chemotherapy. The authors reported favorable results and also the safety of this regimen. Haddad et al 23 included 36 patients with cT3, T4, or N+ adenocarcinoma of rectum within 15 cm from the anal verge (AV) to nCRT with capecitabine with or without oxaliplatin 60 mg/m2 weekly followed by surgery. They reported a higher rate of tumor downstaging and pCR at a final report and also higher but tolerable G3-4 complications. Hess et al 24 ran a single-arm trial in 2017 and treated 54 patients with 1 course of induction CAPOX, then nCRT with oxaliplatin (50 mg/m2 on days 1, 8, 22, and 29) and capecitabine followed by surgery. The authors reported high locoregional control but associated with increased toxicity. Larsen et al 12 treated 52 patients with T4 or circumferential radial margin (CRM)CRM-positive T3 rectal cancer by nCRT with oxaliplatin (50 mg/m2 weekly) and capecitabine followed by surgery. Five patients were metastatic at the study entry. Five courses of CAPOX were given to patients before surgery (before and after CRT). The trial achieved an impressive response rate in this high-risk group of patients and promising 5-year OS, and the final report declared this protocol safe and feasible.
Dueland et al 13 treated 97 patients with 2 courses of induction chemotherapy (FLOX regimen) followed by nCRT with oxaliplatin (50 mg/m2 weekly) and capecitabine followed by surgery in their single-arm trial in 2016. The authors reported high tumor response and OS with acceptable toxicity. Among other studies that were carried out in or before 2015, RTOG 0822 25 was another single-arm trial that treated 79 patients with cT3-4 N any and tumors less than 12 from the AV with nCRT, oxaliplatin (50 mg/m2 weekly), and capecitabine followed by surgery and adjuvant FOLFOX for 18 months. The study’s primary endpoint was acute grade 2 to 5 gastrointestinal (GI) toxicity, and the radiotherapy technique was intensity-modulated radiation therapy (IMRT) followed by a 5.4G boost with a 3D conformal technique. The final endpoints were promising, with acceptable G3-4 toxicity. Although the authors did not report the benefit of IMRT in GI toxicity reduction, overall toxicity was acceptable and tolerable, considering that oxaliplatin was added to chemoradiotherapy. The nCRT with S-1 and oxaliplatin followed by surgery was used in the Shogun trial 26 with 4 courses of 60 mg/m2 of oxaliplatin, and the results were promising, too. The authors reported a high pCR rate and favorable toxicity profile. Musio et al 27 conducted a single-arm trial including 80 patients with cT3-4 N any and tumors lower than 12 cm from the AV treated with nCRT, oxaliplatin (50 mg/m2 weekly), 5FU, and surgery followed by adjuvant chemotherapy if positive pathologic lymph nodes were found. The results of this trial were promising, with beneficial effects on OS and LRC. Greto et al, 28 XELOXART study group, 29 and Lee et al 30 conducted similar studies with promising findings regarding survival and pCR or R0 resection rates.
In 2011, Zhao et al 31 treated 25 patients with nCRT with CAPOX, followed by surgery and adjuvant CAPOX for 4 courses. The protocol was well tolerated, with improved pCR compared with the historical controls. Fernández-Martos et al, 32 in the Groupe Cancer de Recto 3 study, compared nCRT with oxaliplatin (50 mg/m2 weekly) and capecitabine followed by surgery and 4 cycles of CAPOX vs induction CAPOX, then nCRT with the same method followed by surgery in 108 patients. The inclusion criteria were cT3 Nany or resectable T4 and distal tumor border located less than 12 cm from the AV. The trial aimed to compare adjuvant and neoadjuvant chemotherapy and concurrent oxaliplatin was used in both arms, but overall toxicity was acceptable and tolerable in total.
Among the studies in the 2000s, Rödel et al 33 treated 110 patients with nCRT with oxaliplatin (50 mg/m2 weekly) and capecitabine, followed by surgery and 4 cycles of adjuvant CAPOX. Considering good final results regarding pCR and toxicity, the authors declared this protocol an active and feasible treatment. Preoperative nCRT with oxaliplatin and 5FU followed by surgery with oxaliplatin administration at 130 mg/m2 at weeks 1 and 5 was used in the Lyon R0-04 trial 34 in 2003. The regimen was well tolerated, with a favorable response rate.
Meta-analysis reports
In a meta-analysis published in 2019 by Hüttner et al, 35 after a review of 10 randomized trials and 5599 patients, there was no benefit for adding oxaliplatin in OS, DFS, or LRC. However, it led to a statistically significant increase in pathological complete response (OR = 1.31, 95% CI = 1.10-1.55, P = .002) and a statistically significant reduction in distant recurrence (OR = 0.78, 95% CI = 0.66-0.92, P = .004). The observed benefits were offset by higher rates of grade 3 or 4 toxicities.
Fu et al 36 released the results of another meta-analysis in 2017 on the same subject. A total of 8 RCTs with 6103 patients were analyzed. The authors declared in their final results that the oxaliplatin-based regimen group attained higher pathologic complete response (OR = 1.29, 95% CI = 1.12-1.49, P = .0005) and 3-year DFS (OR = 1.15, 95% CI = 0.93-1.42, P = .21), but suffered greater toxicity. There were no significant differences between the 2 regimens in sphincter-sparing surgery rates and OS.
Another meta-analysis of 4 randomized articles was published by De Felice et al 11 in 2017. According to the results, patients treated with oxaliplatin-based CRT had significantly decreased distant failure (OR = 0.76; 95% CI = 0.60-0.97; P = .03) compared with standard CRT. Overall survival, DFS, and LRC were not significantly different between groups.
Based on these meta-analysis data, oxaliplatin-based nCRT can improve pCR and distant metastasis outcomes but does not impact LRC and survival. Improvements in pCR and DF are at the cost of higher G3-4 toxicities.
Oxaliplatin dose and schedule
One of the main factors affecting the efficacy and safety of oxaliplatin as an agent in nCRT is its dose and administration schedule. The dominant pattern of oxaliplatin usage in most trials was 50 mg/m2 weekly as a concurrent agent with 5FU-based radiotherapy. However, some studies opted for a different dosage and schedule.
In the oxaliplatin arm of the FOWARC trial, 14 five 2-week cycles of infusional 5FU and oxaliplatin plus radiotherapy (during cycles 2 through 4) followed by surgery and 7 cycles of mFOLFOX chemotherapy were administered. The oxaliplatin dose was 85 mg/m2, and patients took 3 cycles of oxaliplatin concurrently with radiotherapy. Higher G3-4 toxicity and more postoperative complications were observed in patients who received mFOLFOX and radiotherapy, but the complications generally were tolerable. It should be noted that there were improvements in pCR by this protocol.
Tang et al 21 in 2018 treated 45 patients with nCRT with oxaliplatin and capecitabine followed by 1 cycle of CAPOX followed by surgery and then completion of CAPOX to 6 cycles. The authors reported excellent disease control and long-term survival with tolerable toxicity. Contrary to the dominant weekly schedule in phase III trials, oxaliplatin was administered at 130 mg/m2 on weeks 1 and 3. This different protocol deserves further investigation in future trials.
Neoadjuvant CRT with oxaliplatin and 5FU followed by surgery was tested in the Lyon R0-04 trial in 2003. 34 The oxaliplatin dose was 130 mg/m2 administered on weeks 1 and 5 of radiotherapy. The authors claimed good tolerability with a fair response rate.
Greto et al 28 in 2013 investigated a protocol of nCRT with oxaliplatin and 5FU followed by surgery. The oxaliplatin dose was 80 mg/m2 on weeks 1 and 5. Outcomes were acceptable, and the authors reported that this regimen would be well tolerated with good results for OS, pCR, and LRC.
Lee et al 30 performed nCRT with oxaliplatin and 5FU, followed by surgery with an oxaliplatin dose of 130 mg/m2 in weeks 1 and 5. Despite the high anastomotic leak rates, the total toxicity rate was acceptable, and the authors reported a sphincter preservation rate of 93.5%. However, there were no improvements in pCR contrary to the trials mentioned above.
In summary, it seems that modifications in oxaliplatin dose, especially used in the FOWARC trial, 14 could be safer than the standard dose of weekly 50 mg/m2 and could be an option when concurrent oxaliplatin is considered in patients with good performance status. Administering two 130 mg/m2 cycles of oxaliplatin is another option with good primary results needing further investigation.
Oxaliplatin in metastatic patients
One proposed role of oxaliplatin with RT would be in metastatic patients treated with curative intent. In these patients, controlling the metastatic foci during radiotherapy would benefit from adding oxaliplatin. However, there needs to be more evidence besides 2 studies.
Dueland et al 13 performed a single-arm trial investigating neoadjuvant chemotherapy followed by nCRT with oxaliplatin, capecitabine, and surgery. They included resectable lung or liver metastatic patients in their trial and added oxaliplatin to radiation to obtain a better response. The results were promising with good 5-year OS, 5-year DFS, pCR, and R0 resection rate. The percentage of R0 resection in metastatic patients was 70%, which is promising. This scheme could be an option for patients with resectable metastases in future trials. Eventually, the authors reported high tumor response and OS with acceptable toxicity for this method.
Larsen et al 12 performed a trial that included resectable M1 patients and reported that better responses could be obtained by adding oxaliplatin to nCRT. The RT dose was 54 Gy using the IMRT technique. The authors reported a safe and feasible response rate and promising 5-year OS. Considering these 2 trials, adding oxaliplatin to RT in patients with resectable metastases could be proposed to obtain a better response in metastatic sites while focusing on the local disease. This approach is especially encouraged in patients with good performance status who can tolerate this treatment.
Oxaliplatin-Related Toxicity and Safety
Acute complications
Acute complications are mainly limited to the RT treatment and usually fade soon after the completion of radiation. Therefore, acute complications are considered less relevant than late complications. The main concern about acute complications is the potential to interfere with maximal chemoradiation therapy in a reasonable period. In most trials, more than 90% of patients had received the total dose of nCRT without delay in treatment. Thus, adding oxaliplatin was not a limiting factor for completing nCRT or surgery and adjuvant chemotherapy after that. The major acute complications during RT included diarrhea, proctitis, neutropenia, fatigue, anal pain, and cystitis (Tables 3 and 4).
The treatment-related toxicity in the oxaliplatin-containing arm vs the control arm in phase III randomized clinical trials.

Abbreviations: CAP, capecitabine; CAPOX, capecitabine and oxaliplatin; FOLFOX, fluorouracil and oxaliplatin; NR, not reported.
Treatment-related toxicity in phase II clinical trials.

Abbreviations: BMS, bone marrow suppression; CAP, capecitabine; CAPOX, capecitabine and oxaliplatin; CHT, chemotherapy; FOLFOX, fluorouracil and oxaliplatin; G, grade of toxicity; LFT, liver function test; NR, not reported; NS, not significant; OX, oxaliplatin.
The PETACC64 study reported significantly higher acute complications such as diarrhea, proctitis, and renal injury in the oxaliplatin arm. Statistically significant overall and grade 3-4 diarrhea was reported in the NSABP R-046 in the oxaliplatin arm. In the STAR-01, 16 the total G3-4 adverse effect was 24% in the oxaliplatin arm vs 8% in the control arm without improving immediate results. On the contrary, adverse effects in ACCORD125 and German CAO/ARO/AIO-0415 trials are comparable and acceptable, considering the survival benefit reported in the CAO/ARO/AIO-04 trial. In addition, acute complications in phase II trials were lower, and most of the G3-4 complications were tolerable in most of the mentioned trials.
Peripheral neuropathy
Peripheral neuropathy is a known complication of oxaliplatin that, in severe cases, can be debilitating. Since this complication is not seen with fluoropyrimidine-based RT, adding oxaliplatin to RT requires attention. The highest peripheral neuropathy rate with oxaliplatin was reported in the PETACC trial (48% vs 1% G2-4). 4 In the CAO/ARO/AIO-04 trial, 15 the rate of neuropathy was significantly higher in the oxaliplatin arm vs the control arm (10% vs 1% G3-4). However, G3-4 sensory neuropathy in the oxaliplatin group decreased from 41 (10%) patients during treatment to 13 patients (3%) at 1-year follow-up. In the NSABP R-04 phase III trial, 6 the rate of G2-4 sensory neuropathy was 6.5% in oxaliplatin plus capecitabine and 5.6% in oxaliplatin plus 5FU groups.
Despite the results of PETACC6 and CAO/ARO/AIO-04 trials, peripheral neuropathy rates were surprisingly lower in most phase II trials with no evidence to be a limiting complication for treatment. Considering all these, most of the phase II trials mentioned above and listed in Table 3 reported acceptable early and late toxicity after adding oxaliplatin.
Adding oxaliplatin to RT could cause low- and high-grade peripheral neuropathy, but whether it is acceptable or not is related to the outcomes of treatment and is still a matter of debate.
Postoperative and late complications other than peripheral neuropathy
The toxicity of an extra agent for concurrent chemotherapy can be a limiting factor that can interfere with RT. It can also cause more perioperative complications, affecting the treatment protocol of the patients.
The most commonly reported postoperative complications included anastomotic leakage, wound infection, fistula formation, sexual dysfunction, fecal incontinence, and abscess. In the FOWARC study, 14 22 investigators reported no significant difference between the 2 arms on anastomotic leakage and fistula formation. The results of PETACC 4 for postoperative fistula or presacral sinus formation were similar, with no significant difference between the 2 arms (3.1% vs 2.6%). The ACCORD125 reported similar anal continence, erectile dysfunction, and social life quality comparing oxaliplatin and non-oxaliplatin groups. Yu et al 8 and Lee et al 30 reported a rate of 13% for anastomotic leakage among patients receiving oxaliplatin compared with 2% in the control arm. Hess et al 24 reported 33% fecal incontinence for adding oxaliplatin, the highest rate reported in the literature. Surprisingly, De Felice et al 11 found a rate of 11% for thromboembolism that seems unrelated to oxaliplatin and needs further investigation.
Late G3-4 adverse events in patients who received concurrent oxaliplatin in the CAO-ARO-AIO-04 trial 15 occurred in 25% and 21% of patients in the oxaliplatin and control groups, respectively. Late complications were tolerable overall, and chemotherapy-related deaths occurred in only 4 patients (1%). The final results reported 2 infection-related multi-organ failures and 2 cardiac failures.
Future directions
It should be noted that in addition to the concurrent oxaliplatin, other techniques can also be used to intensify the neoadjuvant therapy in rectal cancer. 37 Total neoadjuvant treatment is one of the most exciting approaches that has become a new standard of care. The TNT has improved the outcomes, especially for high-risk cases, including T4 primary, extra-mural venous invasion, involved mesorectal fascia or a high number of mesorectal positive lymph nodes or deposits, or the presence of extra-mesorectal pathologic lymph nodes. 38 Aside from pathologic response, TNT may lead to better chances of organ preservation and non-operative management in complete clinical responders, with modern studies achieving a 53% rate of total mesorectal excision-free survival. 39 However, no data exist for adding oxaliplatin to concurrent CRT patients treated with TNT or organ preservation protocols. Thus, the introduction of oxaliplatin-based CRT in these protocols can be tested in randomized controlled clinical trials. On the contrary, however, some believe that a subset of patients with a lower risk of failure are at risk of overtreatment by TNT. This notion warrants a head-to-head comparison between TNT and oxaliplatin-based CRT regarding efficacy and safety.
Improved response predictions by novel biomarkers like ctDNA are another area of active investigation that needs more clarification. 40 In the future, oxaliplatin-containing CRT can be revisited in this context for patients who are expected to be poor responders to conventional treatments.
One crucial issue that can affect the utility of oxaliplatin combined with chemoradiotherapy is the new management strategies for chemotherapy-induced neurotoxicity, which is the primary limiting toxicity for oxaliplatin. 41 Based on the evidence, prophylactic rather than therapeutic usage of duloxetine or gabapentin is more beneficial. 42 Re-examining the role of oxaliplatin together with CIN-preventing agents could be investigated in future studies.
Conclusions
The available literature suggests that by adding oxaliplatin there are some benefits in enhancing response to nCRT, however, without any translated improvements in long-term outcomes including overall and DFS. Thus, the challenge of oxaliplatin use has not been resolved completely in the literature. It needs further investigation, especially in some subgroups with rectal cancer. These may include patients with excellent to good performance status with low-lying tumors requiring sphincter preservation or synchronous resectable metastases. Oxaliplatin can also be considered for patients with excellent performance status who have tumors with a high risk of failure.
Footnotes
Author Contributions
RN and RG contributed to the concept or design of the work. RN, RG, SMAT, and BB contributed to acquisition, analysis, or interpretation of data. RN, RG, and GNP drafted the article. SMAT, BB, NM, MA, and MAG revised it critically for important intellectual content. All the authors approved this version to be published and took public responsibility for appropriate portions of the content.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
