Abstract
Background:
Although radioisotope (RI) combined with blue dye (BD) is the standard technique for sentinel lymph node (SLN) biopsy in breast cancer, the use of RI is limited at some institutions due to the specific equipment needed. Indocyanine green (ICG) fluorescence detection has been developed as a potential substitute for RI method. However, reports on the sensitivity of ICG and RI techniques in detecting SLN are inconsistent; hence, the present study was designed to compare the clinical efficacy between the combined method of ICG + BD (ICG-B) and RI + BD (RI-B).
Methods:
A prospective observational study was performed that identified 138 breast cancer patients who had undergone lymphatic mapping and SLN biopsy with ICG-B or RI-B. The SLN detection rate, positive SLN counts, and lymph node metastasis between the 2 groups were compared.
Results:
A total of 71 patients were recruited in the ICG-B group, while 67 were recruited in the RI-B group. The SLN detection rate was 100% in both the ICG-B and RI-B groups. Lymph node metastasis was found in 13 patients using ICG-B and in 12 patients using the RI-B technique (18.31% vs 17.91%, respectively; P = .61). No significant differences were observed in the positive SLN counts (3.12 ± 2.01 vs 3.33 ± 2.24, P = .37) between the 2 groups.
Conclusions:
Indocyanine green combined with BD has an equal efficacy compared with RI plus BD when performing an axillary SLN biopsy in breast cancer. The ICG plus BD procedure is a promising alternative to traditional standard mapping methods.
Introduction
Sentinel lymph node (SLN) biopsy has become a standard procedure for axillary staging in breast cancer as it is associated with less morbidity while providing accurate assessment of lymph node involvement.1,2 The recommended technique for an SLN biopsy is the combined use of radioisotope (RI) method and the blue dye (BD) method. The benefits of BD method include its simplicity, safety, and cost-effectiveness. Although the overall SLN detection rates using BD alone can reach up to 97.1% (ranging from 65.6% to 97.1%), 3 it still has several deficiencies, for example, this method cannot indicate the location of the SLN before the skin incision, and can cause semi-permanent skin staining, which may not fade even after several months. 4 On the contrary, RI method can identify the precise location of SLN, but special equipment is needed to localize the lymph node as the RI cannot be identified with the naked eye. Consequently, to improve the identification rate of SLN, the combined method of BD and RI has been suggested by the American Society of Clinical Oncology as a standard procedure for axillary lymph node detection in breast cancer. 5 However, the use of RI is not available at all treatment centers due to the relatively high cost, complicated surgical preparations, painful preoperative injections, and requirement of a special gamma detector device for detection. 4 Therefore, nonradioactive tracer methods need to be developed that can accurately provide SLN identification.
Over the last decades, many intraoperative fluorescence dyes have been developed for SLN biopsy.6-8 Among them, indocyanine green (ICG) is the most attractive substitute for RI method as it is the only near-infrared fluorescence agent authorized by both the United States and China for clinical usage. Kitai et al 9 first employed a new technique for lymphatic mapping using ICG in breast cancer. Although more studies have supported that the ICG technique is a safe and feasible method for SLN biopsy,10-12 the comparable results on SLN detection rate between ICG + BD and RI + BD were inconsistent, which need more probe. Therefore, the present study was designed to prospectively assess the concordance between the ICG + BD (ICG-B) method and the 99mTc-labeled radiotracer RI + BD (RI-B) method to determine the efficacy of ICG to identify the SLN.
Materials and Methods
Patients
This is an observational study conducted to compare the clinical efficacies between the combined method of ICG + BD and RI + BD on SLN detection in breast cancer patients. Between June 2019 and July 2022, a total of 138 patients with T1 to 2 (tumors staging of 1-2) primary invasive breast cancer confirmed by core- or fine-needle biopsy and a clinically negative axilla at the Department of Breast Surgery, the First Affiliated Hospital of Kunming Medical University, were enrolled in the present study. The exclusion criteria included bilateral breast cancer, inflammatory breast cancer, distant metastatic tumor, previous neoadjuvant chemotherapy or radiotherapy, previous axillary surgery, and pregnancy. All SLN biopsy procedures were performed by senior breast surgeons.
The study was approved by the Ethical Committee of the First Affiliated Hospital of Kunming Medical University. All patients provided a written informed consent.
Surgical procedure
The SLN biopsy using either ICG-B or RI-B was performed following a standard procedure. Methylene blue (Jichuan Co., Jiangshu, China) was used for both groups of patients after anesthesia administration. 0.2 mL of methylene blue (10 mg/mL) was subcutaneously injected in the 6-, 7-, and 9-o’clock directions of the breast near the areola and then the whole breast was gently massaged for approximately 5 minutes.
In the ICG-B group, 0.2 mL of 1.0 mg/mL ICG (Dandongyichuang Co., Liaoning, China) solution was subcutaneously injected in the same locations as the methylene blue. The whole breast was then gently massaged for 5 minutes. Next, the surgical lights were turned off, and a portable fluorescence imaging system (Mingde Biology Co., Hebei, China) was used to trace the lymphatic flow from the breast to the axilla in real time. The lymphatic vessels began to appear 1 to 2 minutes after injection. The average time to reach the SLNs was 10 to 15 minutes (Figure 1). Subsequently, the access incision for the SLN biopsy was made at the site where the fluorescence disappeared. The blue-stained lymph nodes were visualized (Figure 2A), and the fluorescence imaging confirmed to be in the same lymph node (Figure 2B). Blue-stained nodes, fluorescent-positive nodes, and palpable suspicious nodes were all excised.
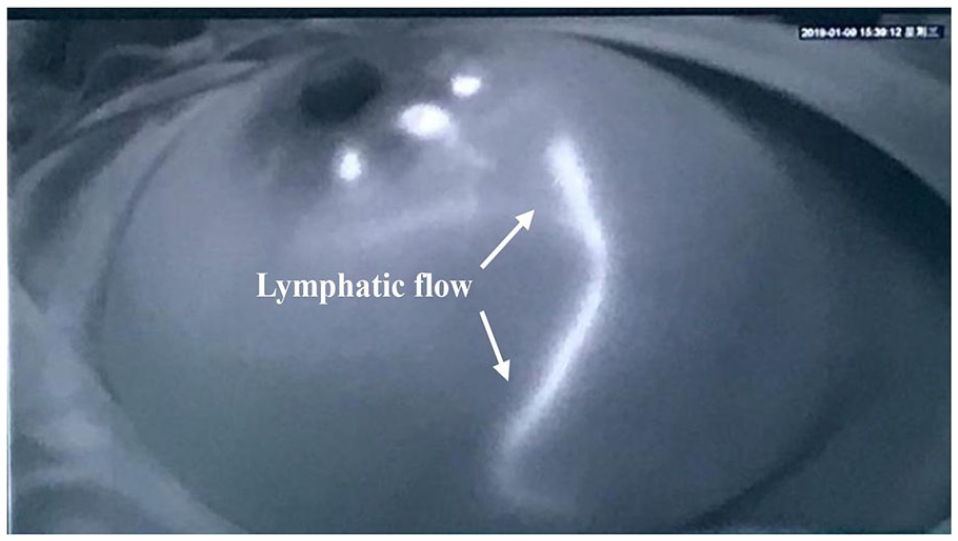
Lymphatic flow was clearly visualized using a fluorescence imaging system after injection of indocyanine green.

Sentinel lymph node detection was performed using methylene blue dye and fluorescence imaging. (A) Lymphatic vessels and the sentinel lymph nodes continue blue dye. (B) Fluorescence imaging of the same lymphatic vessels and the sentinel lymph nodes.
For RI-B group, 1 mL of 99Tcm (SK Pharmaceuticals, Seoul, Korea) was mixed with 1 mg of lyophilized human serum albumin (SK Pharmaceuticals). 0.2 mL of the solution was subcutaneously injected into the periareolar region 6 to 12 hours prior to BD injection. Under the guidance of a gamma probe (Neoprobe Gamma Detection system; Devicor Medical Products, Inc. part of Leica Biosystems, USA), RI-positive SLNs were dissected from the axilla and the lymphatic tissue.
All harvested nodes were sent for intraoperative frozen section staining with hematoxylin and eosin. When this staining was negative, further serial sectioning was performed on paraffin blocks, followed by immunohistochemistry analysis for further analysis. In patients with malignant cells of the SLN in the frozen sections, axillary lymph node dissection was performed as a standard axillary treatment.
Statistical analysis
We evaluated the identification rates and number of SLN biopsies between the 2 groups using the chi-square test with significance determined at P < .05. All statistical analyses were performed with SPSS Version 19.
Results
From June 2019 to July 2022, 138 patients had an SLN biopsy and were prospectively registered in our database: 71 in the ICG-B group and 67 in the RI-B group. Details of the patients’ tumor characteristics are shown in Table 1. The mean age of the ICG-B group and the RI-B group was 48.6 and 49.8 years (P > .05), respectively. There were no differences in the histopathologic factors, including tumor size, nodal stage, hormone receptor positivity, and human epidermal growth factor receptor-2 positivity, between the ICG-B group and the RI-B group. No adverse reactions or complications related to the ICG procedure, RI, or BD injection were recorded. All patients tolerated the SLN biopsy without complications. The rate of breast-conserving surgery for the ICG-B group and the RI-B group was 71.83% and 73.13%, respectively (P > .05).
Patient clinicopathologic characteristics.
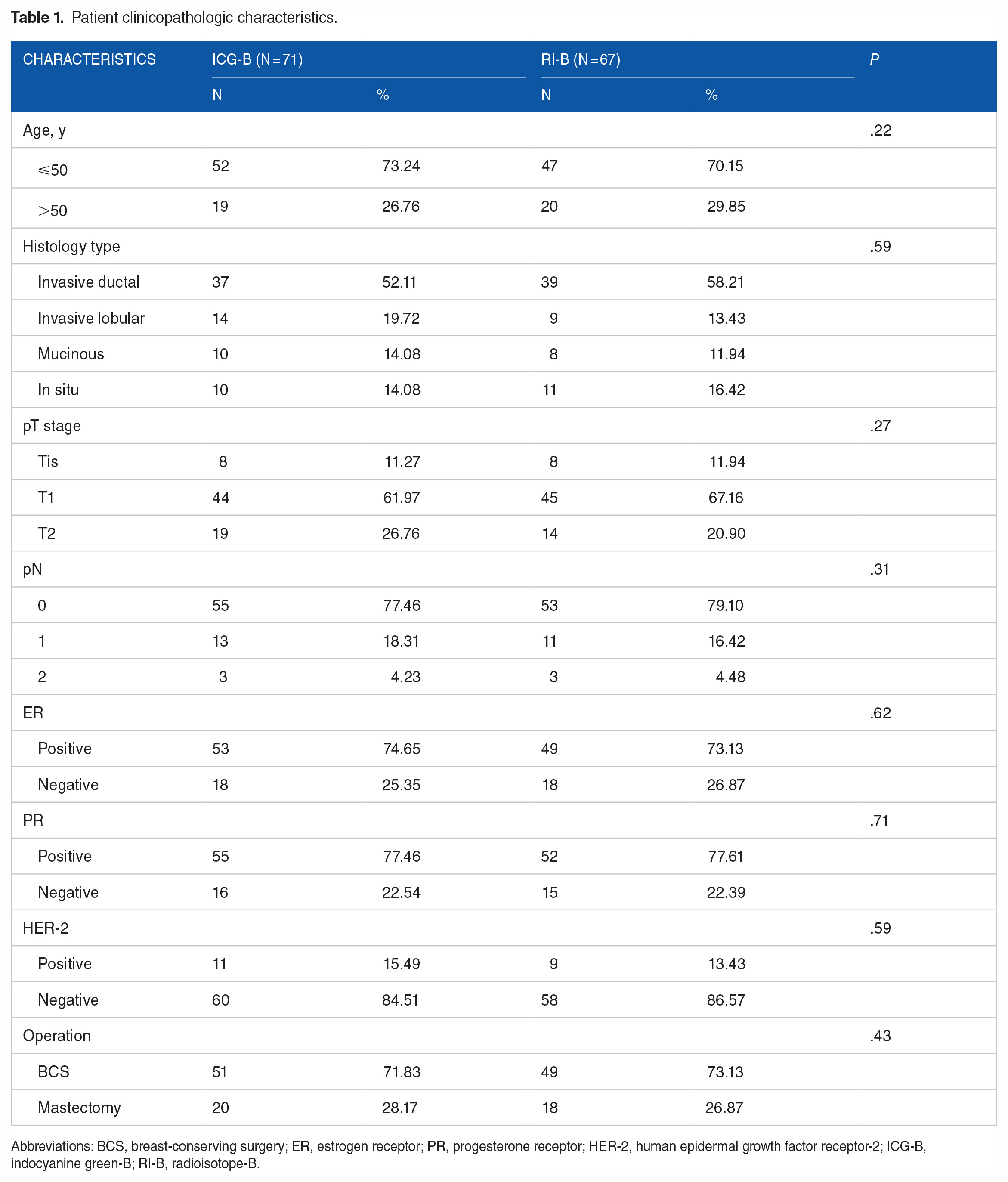
Abbreviations: BCS, breast-conserving surgery; ER, estrogen receptor; PR, progesterone receptor; HER-2, human epidermal growth factor receptor-2; ICG-B, indocyanine green-B; RI-B, radioisotope-B.
The detection rate of SLN, the number of SLNs detected per patient, and the extent of metastatic involvement for each group are shown in Table 2. Sentinel lymph nodes were detected in all patients in the ICG-B and RI-B groups. The number of SLNs ranged from 1 to 5 (average 3.12) in patients receiving ICG-B and from 1 to 6 (average 3.33) in those undergoing RI-B. There was no statistical difference in SLN metastasis rates between the 2 groups, with 13 patients (18.31%) in the ICG-B group and 12 patients (17.91%) in the RI-B group having a positive SLN on frozen section analysis. Findings were confirmed by the final immunohistochemistry analysis which showed positive SLNs in 13 patients (18.31%) and 13 patients (19.40%) in the ICG-B and RI-B groups, respectively (P > 0.05).
Comparison of sentinel lymph node biopsy results in the 2 groups.

Abbreviations: ICG-B, indocyanine green-B; RI-B, radioisotope-B; SLN, sentinel lymph node.
Discussion
The combination method of RI and BD is the standard method for SLN biopsy in breast cancer. 5 However, a survey conducted in 110 hospitals in China shows that the major method for SLN biopsy is BD (69 of 110, 62.73%); the RI + BD technique was used only in 16 of 110 (14.55%) hospitals, probably due to the limitation of radioactive substance disposal, relatively complicated surgical preparations, high cost, and requirements of special equipment. 13 Even in developed countries such as Japan, a nationwide survey for SLN biopsy practice showed that only 64% of eligible patients were using RI due to problems such as nuclear medicine setup, training requirements, and cost. 14 Therefore, a radioactive-free detection method, which can be used in conjunction with BD while maintaining the sensitivity of SLN detection rate, is needed. Indocyanine green, a nonallergenic and nonradioactive fluorescent dye, has been proposed as an effective tracer for SLN identification in breast cancer.10-12
It is well known that the SLN detection rates of ICG are superior to those of BD 15 ; however, the efficiency in SLN identification between ICG and RI method is controversial. Stoffels et al 16 found the use of ICG for SLN detection to be inferior compared with RI technique especially in patients in whom the lymph node basin cannot be predicted correctly. Interestingly, Sugie et al 17 believed that ICG was more sensitive than RI. In addition, a comprehensive systematic review supported that there was no significant difference between ICG and RI in SLN detection rate (odds ratio = 0.90; 95% CI = 0.40-2.03). 3 Whether the sensitivity of ICG is equal to or higher or even lower than that of RI especially when it is combined with BD is not known; therefore, there are much ongoing researches to compare the clinical efficiency between ICG + BD and RI + BD.
As previously reported, the detection efficiency for SLN can be increased when ICG is paired with BD. 18 In an aforementioned study, it was found that the SLN detection rates of BD and BD + ICG were 91.5% and 98.5%, respectively (P < .01). Their combination also resulted in more lymph node identification per patient (2.6 vs 3.1). However, the report on the clinical efficiency between ICG + BD and RI + BD was different. A meta-analysis showed a trend toward a superior efficiency in SLN identification rate using ICG + BD compared with RI + BD. 4 On the contrary, studies also supported that there were no difference in SLN detection rate between ICG + BD and RI + BD method. 19 In this study, the ICG + BD and RI + BD methods detected 100% of axillary SLNs. Our result is consistent with that of a study by Chirappapha et al 20 who reported that the relative detection rates by individual method (95.23% for ICG, 85.71% for BD, and 85.71% for RI) increased up to 100% when compared with the combined use of ICG and BD. In fact, the SLN detection rate of ICG combined with BD has been reported to be 96.4% to 99.2% and not 100%.21,22 The explanation may be that the tumors may block the lymphatic vessels, limiting the ability for the tracer to reach the SLNs; hence, if a research recruits patients with this kind of tumors, it cannot get 100% detection rate. Therefore, details of tumor location and lymphatic obstruction and large sample size will be included in our future study to explore this question. Moreover, the metastatic SLN detection rate in the present study was also close to that of the research conducted by Shen et al. 22 The direct comparison between ICG + BD method and RI + BD method showed that the efficacy of the ICG + BD method is no less than that of the RI + BD method; this finding is also supported by a randomized controlled trial, in which the authors compared the diagnostic performance of ICG + BD and RI + BD methods and showed no significant difference in the identification rate of SLNs, the identification rate of metastatic SLNs, SLN biopsy, and false-negative rate between the 2 groups. 23
Although the surgical time has not been monitored in this study, ICG showed high tissue penetration. This may help surgeons observe lymphatic drainage in real time and identify potential SLNs before an incision is made. Therefore, ICG combined with BD may improve incision placement and potentially reduce surgical time.
It is necessary to point out that the optimal dosage, volume, and concentration have not been standardized for ICG use.8,24 Mieog et al 25 reported the optimal injection dose of ICG to be between 400 and 800 μM, with other studies favoring 500 μM of ICG alone. In our study, 130 μM of ICG provided adequate fluorescence imaging of the lymph vessels and lymph nodes, which suggests that dosing is more flexible when used in SLN biopsy. Furthermore, it has been reported that a higher ICG concentration that is injected can lead to worse lymph node detection because of fluorophore dispersion 26 ; thus, the higher dose may not be more useful for SLN biopsy and reducing the dose of ICG can avoid halation after the first SLN is resected.
Limitations
There are limitations in this study. First, this is a single-center study only representing our local practice. In addition, the long-term follow-up is limited, making the recurrence of axillary disease unknown. Moreover, the number of patients included is small; hence, a multicentric and large-sample study is needed to further confirm the results.
Conclusions
Although there are shortcomings, our present results supported that the technique of combining ICG with BD is as sensitive as RI plus BD. Consequently, combined use of ICG with BD appears to be an effective method for detecting SLNs particularly in hospitals where no radiotracers are available.
Footnotes
Acknowledgements
The authors would like to thank Dr Irene Ma for language editing and Dr Guoqing Pan for pathology assistance.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Nature Science Foundation of China (No. 81860487) and Disciplinary leader of the high-level health technology talent project in Yunnan Province (No. D2018031). The funders did not participate in the experimental research or the preparation of the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
YW and YD designed the study. QP, PS, and XL collected and analyzed the data with YW. All the authors have read and approved the final submitted version.
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author on reasonable request.
Statement of Ethics
This study was conducted in accordance with the “Declaration of Helsinki,” and the written consents were obtained from the participants and approved by the Ethics Committee of the First Affiliated Hospital of Kunming Medical University (No. 4 Ethical review [2019] of the First Affiliated Hospital of Kunming Medical University, April 17, 2019). The choice of sentinel lymph node biopsy method is completely based on the comprehensive evaluation of patients. All the participants were notified that they have the right to refuse or terminate the study at any point of the interview.
