Abstract
Background:
With the update of equipment, the hospital base of this study began to adopt craniospinal irradiation (CSI) intensity-modulated radiotherapy (IMRT) in May 2018 to replace the traditional CSI conventional radiotherapy (CRT) technology. The purpose of this study was designed to compare the differences in acute hematological adverse reactions induced by CSI-IMRT and CSI-CRT.
Methods:
The clinical data and hematological data of 102 patients with central nervous system malignant tumors who underwent CSI treatment at the 900th Hospital of Joint Logistics Support Force of PLA from January 2008 to August 2022 were analyzed retrospectively. The patients included 63 men and 39 women, aged 3 to 56 years old. On the basis of the radiotherapy technique used, the patients were divided into the CSI-IMRT group (38 cases) and CSI-CRT group (64 cases). Acute hematological adverse reactions during radiotherapy were compared between the two groups according to the Common Terminology Criteria for Adverse Events version 4.0. The Mann-Whitney U test was used to compare the measurement data, and the χ2 test was used to compare the count data.
Results:
No significant difference was found between the CSI-IMRT group and the CSI-CRT group in terms of sex, histopathological type, tumor location, spinal cord invasion, surgery, and the Eastern Cooperative Oncology Group score (χ2 = 0.004 to 6.213; all P > .05). No significant difference was found in onset time of myelosuppression (11 days (interquartile range [IQR]: 7 to 14; minimum [min] to maximum [max]: 0 to 26) vs 8 days (IQR: 7 to 15; min to max: 3 to 29)) and nadir time of myelosuppression (21 days (IQR: 18 to 25; min to max: 12 to 35) vs 22 days (IQR: 15 to 25; min to max: 12 to 36)) between the CSI-IMRT group and the CSI-CRT group (Z = −0.856, −0.248; all P > .05). There were no significant differences in the incidence of decreased white blood cell counts (WBC), platelet counts, and hemoglobin concentration between the CSI-IMRT group and the CSI-CRT group, 86.8% (33/38) vs 78.1% (50/64), 57.9% (22/38) vs 42.2% (27/64), 57.9% (22/38) vs 53.1% (34/64); χ2 = 1.195, 2.357, 0.219; all P > .05. There were no significant differences in the incidence of decreased WBC, platelet counts, and hemoglobin concentration (severe myelosuppression) in grades III and IV, 23.7% (9/38) vs 21.9% (14/64), 7.9% (3/38) vs 3.1% (2/64), 5.3% (2/38) vs 9.4% (6/64); χ2 = 0.045, 1.164, 0.558; all P > .05.
Conclusions:
There was no significant difference in the incidence of myelosuppression and severe myelosuppression (grade III or above) induced by CSI-IMRT and CSI-CRT. CSI-IMRT is worthy of further clinical application.
Keywords
Introduction
Craniospinal irradiation (CSI) is often used to treat medulloblastoma, germ cell tumors, malignant ependymoma, central nervous system malignant lymphoma, and childhood central nervous system leukemia with overt or tendency to cerebrospinal fluid metastasis. 1 CSI conventional radiotherapy (CRT) produces cold and hot dosage spots at the junctions of radiation fields. It was used to move the junction of the two fields regularly to reduce the influence of dose non-uniformity. 2 As a result of having a complex treatment process, CRT has been gradually replaced with emerging radiotherapy techniques, such as intensity-modulated radiation therapy (IMRT), volumetric modulated arc therapy (VMAT), and helical tomotherapy (HT). These new radiotherapy techniques produce significant improvements in dose distribution in the target volume and the junction layer.3,4 However, they also expand the low dose range of irradiation, which may generate severe myelosuppression when the prescribed dose is consistent with that of CSI-CRT. 5 Severe myelosuppression in patients often affects the smooth progress of CSI treatment 6 and also reduces the tumor cure rate, 7 so it has become a hot issue of concern to clinicians. The purpose of this study was designed to compare the differences in acute hematological adverse reactions induced by CSI-IMRT and CSI-CRT.
Materials and Methods
Case data
The clinical data and hematological data of 102 patients with central nervous system malignant tumors who underwent CSI treatment at the 900th Hospital of Joint Logistics Support Force of PLA from January 2008 to August 2022 were collected. The patients included 63 men and 39 women, aged 3 to 56 years old. On the basis of the radiotherapy technique used, the patients were divided into the CSI-IMRT group (38 cases) and CSI-CRT group (64 cases). Inclusion criteria were as follows: (1) patients with diagnosis of central nervous system malignant tumors based on histology or imaging with human chorionic gonadotropin (HCG) and alpha fetoprotein (AFP), (2) patients undergoing CSI after maximum safe resection or CSI alone treatment, (3) patients with an Eastern Cooperative Oncology Group (ECOG) score 8 of 0 to 1. Exclusion criteria were as follows: (1) patients undergoing adjuvant chemotherapy prior to CSI or (2) patients who completed whole-brain radiotherapy and whole-spinal cord radiotherapy sequentially.
Implementation of CSI-CRT
First stage: CSI was conducted with 6 MV X-rays using a linear accelerator (2100C, Varian, USA) under a prescribed dose: 23.4 to 36 Gy/13 to 18 F (median dose: 36 Gy/18 F), with 5 fractions a week. The prescribed dose was normalized with the geometric center of target volume as the reference point (100% of prescribed dose was targeted to the geometric center of target volume). The radiation field was designed with reference to the patient’s fixed position in the X-ray simulator (LX-40A, Toshiba, Japan) prior to treatment. Radiation field design: isocenter techniques across the whole brain field were used to irradiate the left and right parallel fields with the lower boundary at the level of the fourth cervical vertebra. Posterior single-field fixed source skin distance vertical irradiation was adopted in the whole spinal cord field with the top of the whole brain field as the upper boundary. The spinal cord field was divided into 2 to 3 fields, depending on the patient’s height. The interval at the junction of the radiation fields was 1 cm. The interval was adjusted once a week and the radiation field was redesigned to reduce underdose and overdose at the junction.
Second stage: Three-dimensional conformal radiotherapy (3DCRT) with 6 MV X-rays was used on a linear accelerator (600C/D, Varian, USA). The patients adopted a supine position with head-neck-shoulder thermoplastic films and CT localization scanning (LightSpeed VCT, GE, USA) was performed in 5 mm slices from the top of the skull to the lower edge of the second cervical vertebra. CT images were transmitted to the Oncentra Master Plan (v3.3, Elekta, Sweden) treatment-planning system (TPS) via the local area network. Clinical target volume (CTV) was delineated on the TPS for the primary tumor bed or posterior cranial fossa and planning target volume (PTV) was generated with 3 mm margins. Organs at risk (OARs), such as lens, eyeball, optic nerve, optic chiasm, pituitary, brain stem, and spinal cord were delineated. The PTV was assigned a prescribed dose: 20 to 22 Gy/10 to 11 F (median dose: 20 Gy/10 F), with 5 fractions a week.
Three to five radiation fields were incorporated into a three-dimensional CRT (3D-CRT) plan. The PTV dose was designed to be 95% to 110% of the prescribed dose and to limit the dose to OARs.
Implementation of CSI-IMRT
CSI-IMRT was performed with 6 MV X-rays using a linear accelerator (TrueBeam, Varian, USA). The patient adopted a supine position with arms at the sides of the body and a headrest under the head. The head was fixed with head-neck-shoulder thermoplastic films and the body with body films and a negative pressure vacuum pad. CT simulator (Brilliance CT Big Bore, Philips) performed localization scanning in 5 mm slices from the top of the skull to the ischial tuberosity.
The clinical target volume CTVcsi, craniospinal target volume, was delineated on the Eclipse (v13.6, Varian, USA) TPS and the planning target volume PTVcsi was generated with 3 mm margins. CTVtb (irradiation volume for the local boost) was delineated and the PTVtb was generated with 3 mm margins. OARs (lens, eyeball, optic nerve, optic chiasm, pituitary, brain stem, inner ear, parotid gland, thyroid gland, oral cavity, spinal cord, lungs, heart, liver, kidneys, and bladder) were delineated. PTVcsi involved a prescribed dose: 23.4 to 36 Gy/13 to 18 F (median dose: 36 Gy/18 F), with 5 fractions a week. PTVtb involved a prescribed dose: 20 to 22 Gy/10 to 11 F (median dose: 20 Gy/10 F), with 5 fractions a week.
First stage
The PTVcsi IMRT plan was devised including 2 to 3 isocenters depending on the length of PTVcsi. The Y-axis values were changed but those of the X- and Z-axes were not (to enable changing of the center during treatment) to generate different isocenters. Five coplanar fields were designed (0°, 72°, 144°, 216°, and 288°) and the collimator angle was set to 0°. Radiation fields with different centers were added manually, Plansum from all radiation fields was automatically optimized and parameters of each part of PTVcsi were subject to continual optimization, so that PTVcsi was a dose of 98% to 105% of the prescribed dose. Following optimization, Plansum was divided into 2 to 3 treatment plans. Second stage: The PTVtb IMRT plan was devised with 5 to 7 fields and a dose of 98% to 105% of the prescribed dose. The total dose received over the two stages should not exceed dose limitations for OARs.
Evaluation of acute hematologic adverse reactions
During radiotherapy, the changes of WBC, platelet count, and hemoglobin were observed every 3 to 4 days. When grade III to IV myelosuppression occurred, radiotherapy was suspended, and symptomatic treatments such as raising WBC, platelets or blood transfusion were given, and blood picture was rechecked every day. Acute hematological adverse reactions, according to the Common Terminology Criteria for Adverse Events version 4.0 (CTCAE v4.0), 9 were assessed (Grade 0: normal hemogram; grades I to II: mild myelosuppression; grades III to IV: severe myelosuppression).
Statistical analysis
SPSS25.0 software was used for statistical analysis. Measurement data were expressed as median (interquartile range [IQR] and range, as appropriate) and compared between two groups by Mann-Whitney U test. Count data were compared using χ2 test. A value of P < .05 was considered to indicate a statistically significant difference.
Results
Comparison of general data
No significant difference was found between the CSI-IMRT group and the CSI-CRT group in terms of sex, histopathological type, tumor location, spinal cord invasion, surgery, and the ECOG score (χ2 = 0.004 to 6.213; all P > .05; see Table 1).
Comparison of general data of patients between CSI-IMRT group and CSI-CRT group.

Abbreviations: CRT, conventional radiotherapy; CSI, craniospinal irradiation; ECOG, Eastern Cooperative Oncology Group; IMRT, intensity-modulated radiotherapy; PNET, primitive neuroectodermal tumor.
Onset of hematological adverse reactions
From the start of radiotherapy, median onset time of myelosuppression was at 11 days (IQR: 7 to 14; minimum (min) to maximum (max): 0 to 26) during CSI-IMRT and 8 days (IQR: 7 to 15; min to max: 3 to 29) during CSI-CRT (see Figure 1) with median nadir time of myelosuppression occurring at 21 days (IQR: 18 to 25; min to max: 12 to 35) and 22 days (IQR: 15 to 25; min to max: 12 to 36; see Figure 2), respectively. There were no statistically significant differences between the two (Z = –0.856, –0.248; all P > .05).

Onset time of myelosuppression between CSI-IMRT group and CSI-CRT group.

Nadir time of myelosuppression between CSI-IMRT group and CSI-CRT group.
Severity of hematological adverse reactions
No significant difference was observed in the incidence of decreased WBC and platelet counts and hemoglobin concentration between the CSI-IMRT group and CSI-CRT group (86.8% [33/38] vs 78.1% [50/64], 57.9% [22/38] vs 42.2% [27/64], 57.9% [22/38] vs. 53.1% [34/64]; χ2 = 1.195, 2.357, 0.219; all P > .05; see Table 2). No significant difference was observed in the incidence of decreased WBC and platelet counts and decreased hemoglobin concentration (severe myelosuppression) in grades III to IV, (23.7% [9/38] vs 21.9% [14/64], 7.9% [3/38] vs 3.1% [2/64], 5.3% [2/38] vs 9.4% [6/64]; χ2 = 0.045, 1.164, 0.558; all P > .05; see Table 3).
Comparison of hematological indexes of myelosuppression between CSI-IMRT group and CSI-CRT group (cases [%]).

Abbreviations: 0, normal hemogram; I-IV, myelosuppression; CRT, conventional radiotherapy; CSI, craniospinal irradiation; IMRT, intensity-modulated radiotherapy; WBC, white blood cell.
Comparison of hematological indexes at the most severe degree of myelosuppression between CSI-IMRT group and CSI-CRT group (cases [%]).

Abbreviations: < III, normal hemogram and mild myelosuppression; III-IV, severe myelosuppression; CRT, conventional radiotherapy; CSI, craniospinal irradiation; IMRT, intensity-modulated radiotherapy; WBC, white blood cell.
Discussion
This study showed that most of 102 patients with central nervous system tumors who received CSI-CRT and CSI-IMRT had hematologic adverse reactions. Among the 64 CSI-CRT patients, 50 (78.1%) developed grade I to IV myelosuppression and 14 (21.9%) developed grade III to IV myelosuppression. Among the 38 patients treated with CSI-IMRT, 33 patients (86.8%) developed grade I to IV myelosuppression, and 9 patients (23.7%) developed grade III to IV myelosuppression. At the same time, there was no significant difference in acute hematological adverse reactions between the two groups.
Central nervous system tumors are rare.10,11 CSI-CRT has played an important role in the treatment of central nervous system tumors.12,13 Despite the importance of CSI-CRT as a therapy, severe hematological toxicity is one of the side effects. Teng et al 14 reported that among 31 Chinese CSI-CRT patients, 26 (83.9%) had varying degrees of leukopenia with grade III to IV myelosuppression occurring in 11 (35.5%) cases. Similarly, Yang et al 7 reported an incidence of leukopenia of 91.1% (51/56 patients) and a grade III to IV myelosuppression rate of 35.7% (20/56 patients). Similar results have been reported elsewhere, with Jeffries et al 15 finding that 33% of CSI-CRT patients had grade III to IV hematological toxicity. This study found that 50 out of 64 (78.1%) CSI-CRT patients had leukopenia, of whom 14 (21.9%) had grade III to IV myelosuppression. The incidence of hematological adverse reactions caused by CSI-CRT was slightly lower than reported above, which may be related to the exclusion of patients receiving adjuvant chemotherapy in this study.
Radiotherapy techniques have developed considerably and therapies, such as IMRT, VMAT, and HT, have gradually replaced CRT. Indeed, IMRT has become the radiotherapy technique of choice due to its modest equipment and low treatment costs. 16 CSI-IMRT overcomes the problem of dose non-uniformity in the target volume (especially at field junctions) experienced during CSI-CRT but the range of low-dose irradiation is extended with potential adverse effects on hematological toxicity. Zhang 17 reported that 13 out of 50 (26%) CSI-IMRT patients suffered from myelosuppression greater than grade III. Robinson et al 10 reported that the incidence of grade III to IV myelosuppression was 19%. This study found that 86.8% (33 cases) of CSI-IMRT patients suffered varying degrees of leukopenia and 23.7% (9 cases) had grade III to IV leukopenia. These findings are consistent with those of previous studies.
Previous literature reported that the incidence of grade III to IV hematological adverse reactions caused by CSI-CRT and CSI-IMRT were 33.0%~35.7% and 19.0%~26.0%, respectively. This study found the incidence of grade III to IV hematological adverse reactions to be 21.9% for CSI-CRT and 23.7% for CSI-IMRT with no significant difference between the two groups (P > .05). Previous reported values for overall incidence of hematological toxicity (grade I to IV) are 78.1% to 91.1% for CSI-CRT5,8,14,17 and this study found an incidence of 86.8% for CSI-IMRT and 78.1% for CSI-CRT. These values are not significantly different. Therefore, it can be inferred that CSI-IMRT and CSI-CRT result in similar rates of acute hematological adverse reactions, as shown in Table 4.
Comparison of hematologic toxicity rates between CSI-CRT and CSI-IMRT in different studies (%).
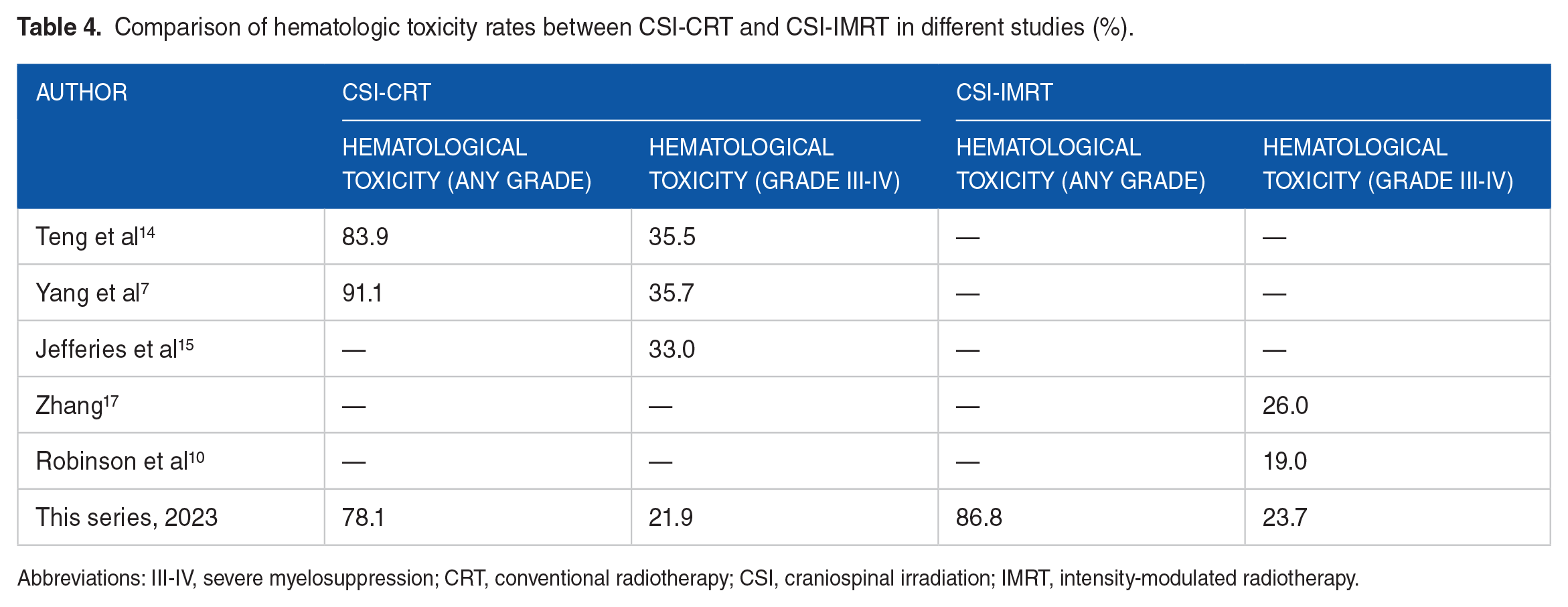
Abbreviations: III-IV, severe myelosuppression; CRT, conventional radiotherapy; CSI, craniospinal irradiation; IMRT, intensity-modulated radiotherapy.
Previous studies have shown low-dose irradiation of the bone marrow to be an important factor causing acute myelosuppression.18-20 Therefore, the author infers that radiation-induced damage of the bone marrow inhibits proliferation, maturation, and release of cells causing severe hematological toxicity in both CSI-CRT and CSI-IMRT. Yang et al 7 found that CSI-HT caused more severe hematological toxicity (grade III to IV leukopenia) than CSI-CRT (85.7% vs 35.8%; P < .05) despite there being only a slight (15.8 vs 14.9 Gy) and non-significant increase in radiation dose to the bone marrow from CSI-HT. 2 Therefore, further bone marrow dosimetric studies are required to ascertain whether the hematological adverse reactions of CSI is related to bone marrow irradiation.
This study’s shortcoming was no in-depth dosimetric study on the causes of hematological adverse reactions caused by CSI. In this study, due to the limitation of technical conditions at that time, the dose of OARs in the first stage of CSI-CRT was missing, which hindered the dosimetric comparison between CSI-CRT and CSI-IMRT. In addition, the relatively small sample size is another limitation. Therefore, prospective clinical trials with large sample sizes are still needed to confirm the safety of CSI-IMRT for hematological adverse reactions.
Conclusions
In conclusion, there are comparable rates of acute hematological adverse reactions following the two radiotherapies. We consider that CSI-IMRT is worthy of greater clinical use and application.
Footnotes
Author Contributions
ZF contributed to the conceptualization and design; YZ contributed to the collection and assembly of data; JC contributed to the data analysis and interpretation; JC and YZ contributed to article writing; and final approval of the article was done by all authors.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The study was conducted in accordance with the Declaration of Helsinki and the Harmonized Tripartite Guideline for Good Clinical Practice from the International Conference on Harmonization. This study was reviewed and approved by the Ethics Committee of 900th Hospital of Joint Logistics Support Force of PLA (approval number: 2022-031). All patients enrolled completed the informed consent form.
Consent for Publication
All patients enrolled completed the informed consent form.
Availability of Data and Materials
The data that support the findings of this study are contained in the published article.
