Abstract
Background:
It is estimated that 75% of urothelial bladder cancers are non–muscle-invasive cancers (NMIBCs). The development of more effective methods for optimizing the management of this subset of patients is of paramount importance. This study aimed to evaluate the effectiveness and side effects of modified maintenance Bacillus Calmette-Guérin (BCG) therapy in patients with high-risk NMIBC.
Methods:
A total of 84 patients with NMIBC who met the inclusion criteria were randomly divided into 2 groups of 42 patients after receiving intravesical BCG weekly, 1 month after transurethral resection of the bladder tumor (TURT) for 6 weeks as the induction. In group I, patients continued monthly intravesical instillation of BCG for 6 months as maintenance, whereas group II patients did not. All patients were followed up for recurrence and progression for 2 years.
Results:
Although the recurrence rate was lower in group I (16.7% vs 31%), there was no significant difference among groups (P = .124). Pathology progression was also lower in group I (7.1% vs 11.9%) with no significant difference among groups (P = .713). Complications were not statistically different among groups (P = .651). A statistically significant difference was not observed between the groups in the acceptance rate of patients (97.6% in group I vs 100% in group II).
Conclusions:
The recurrence rate and progression rate in NMIBC patients with maintenance-free induction therapy after TURT were almost twice as high as those with 6-month maintenance therapy; however, it was not statistically significant. Modified BCG maintenance protocol made favorable compliance for patients.
Trial registration:
This study was retrospectively registered at Iranian Registery of Clinical Trials with the code IRCT20220302054165N1.
Introduction
Non–muscle-invasive bladder cancer (NMIBC) accounts for 75% of urothelial bladder cancers. 1 The development of new methods to optimize the management of this subset of patients is essential. It is the fourth most common cancer in men after prostate, lung, and colorectal cancer, and the ninth most common cancer in women.2,3 It occurs 3 to 4 times more often in men than in women. 3 In the United States, 100,000 new cases of bladder cancer are detected annually. 4
In stage Ta and T1, transurethral resection of the bladder tumor (TURBT) should be performed to diagnose and remove all visible lesions. 5 For low-grade Ta tumors, a single dose of intravesical mitomycin is considered. 6 One of the standard treatments to prevent recurrence and progression of the disease is the intravesical injection of Bacillus Calmette-Guérin (BCG). 7 Bacillus Calmette-Guérin intravesical instillation has been accepted as a maintenance method to prevent recurrence and progression of the disease. 8 In cases of multiple low-grade, large, or recurrent Ta tumors, a single dose of intravesical chemotherapy with mitomycin is recommended in addition to the induction course of intravesical BCG injection. 9 Maintenance treatment is optional in these cases. For high-grade T1 tumors, a single dose of intravesical chemotherapy plus an induction BCG injection is recommended, followed by maintenance therapy with intravesical BCG. 9
One of the problems in the treatment of NMIBC is recurrence and tumor progression. Various studies have been conducted on the effectiveness of maintenance BCG therapy, but the exact protocol has not yet been established.
Although previous studies had a small number of patients who completed their 3-year maintenance courses, and most of them discontinued their treatments, patients who received 6 months of modified BCG maintenance therapy had better results than those who received only induction BCG therapy, with acceptable compliance for treatment.10,11 This study aimed to evaluate the effect of modified 6-month BCG maintenance therapy on disease progression and recurrence in patients with high-risk NMIBC. The primary outcome was to compare patients receiving maintenance doses with those not receiving maintenance treatment. Our secondary outcome was to compare BCG side effects between the 2 groups.
Materials and Methods
Trial design and patient selection
In this clinical trial, a total of 84 patients with NMIBC were referred to Labbafinejad Hospital, Shahid Beheshti University of Medical Sciences from 2018 to 2019. These patients met the inclusion criteria and were divided into 2 groups using random blocks. The inclusion criteria were patients with high-risk NMIBC (including high grade or multiple or larger than 3 cm Ta or T1 lesions) and those who had a history of TURBT 1 month before BCG therapy. Patients with a previous history of intravesical BCG instillation and those who were contraindicated for BCG instillation were excluded from the study. Departmental and Ethical Board Review approval was obtained before the start of this academic work (ethic code: IR.SBMU.UNRC.9112.2). Informed consent was acquired from all patients. The study was registered at http://www.irct.ir (identifier: IRCT20220302054165N1).
Taking into consideration the reported prevalence of NMIBC cases in the Iranian population as per available data, 12 we used the formula for calculating the sample size for a randomized controlled trial, n = (Z2 × [p1 × q1 + p2 × q2]/d2). Employing this method, it was determined that a sample size of 64 (a minimum of 32 participants per group) was required for the study.
All patients in the study underwent intravesical BCG instillation for 6 weeks 1 month after TURBT as the induction therapy. Intravesical BCG instillation continued for up to 6 months in the maintenance group (group I, n = 42). In the other group, which served as the control group, no maintenance therapy was performed, and only induction of intravesical BCG instillation was done (group II, n = 42). These 2 groups were followed up every 3 months for 2 years to investigate the recurrence and progression of the disease with cystoscopy and urine cytology. In the following 2 years, cystoscopy was performed every 6 months. The results were collected and analyzed.
Statistical analysis
The Statistical Package for Social Sciences IBM (SPSS-IBM), version 26 (SPSS Inc, Chicago, IL, USA), was used to perform the analysis after the data had been double-checked. All statistical analyses were carried out using this software. The significance of differences in patient background factors between the 2 groups was examined by the chi-square test. Recurrence-free survival (RFS) and progression-free survival were calculated by the Kaplan-Meier method, and the statistical significance was determined by the log-rank test. A P value < .05 was considered statistically significant.
Results
The CONSORT diagram in Figure 1 shows the flow of participants. A total of 54 men (64.3%) and 30 women (35.7%) were included in this study, and there was no significant difference among groups. The mean ± standard deviation (SD) age of the participants was 61.69 ± 9.26 years, and there was no significant difference between the 2 groups (P = .761).
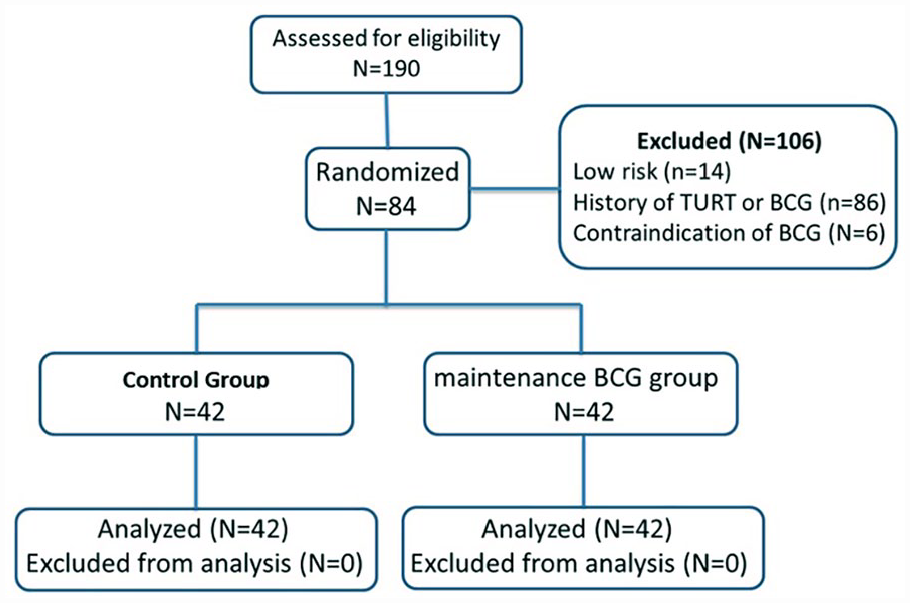
CONSORT flow diagram.
The recurrence rate was 16.7% (n = 7) in group I, whereas the disease has recurred in 13 cases in group II (31%) during the 3 years follow-up. Despite about twice the recurrence rate in the induction group, the difference was not statistically significant (P = .124). Table 1 shows the relationship between both groups and different variables. There was no significant difference in RFS and progression-free survival between the groups (Figure 2).
Relationship between both groups and different variables.

Abbreviation: SD, standard deviation.

(A) Recurrence-free survival and (B) progression-free survival of maintenance BCG group and control group. A lower recurrence rate was observed in the maintenance group than in the control group, but the difference between the 2 groups was not significant.
The pathology progression was 7.1% (n = 3) in group I, whereas the disease progressed in 5 cases in group II (11.9%) during the study follow-up. Similar to the recurrence rate, there was no statistically significant difference among groups (P = .713). Table 2 depicts the relationship between the progression rate and different variables.
Comparing the progression rate with different variables.

Abbreviation: BCG, Bacillus Calmette-Guérin.
Regarding complications, 12 cases (28.5%) showed a grade 1 toxicity rate and 2 individuals (4.7%) had a grade 2 toxicity rate in group I. However, 15 cases (35.7%) showed grade 1 toxicity, and 2 patients (4.7%) suffered from grade 2 toxicity in group II. The difference was not significant among groups (P = .651). There was no case of grade 3 toxicity groups. Table 3 depicts the relationship between toxicity grade and different variables. The acceptance rate was not significantly different among groups (97.6% in group I vs 100% in group II). All patients completed their scheduled BCG therapies in 2 groups.
Comparing the toxicity grade with different variables.
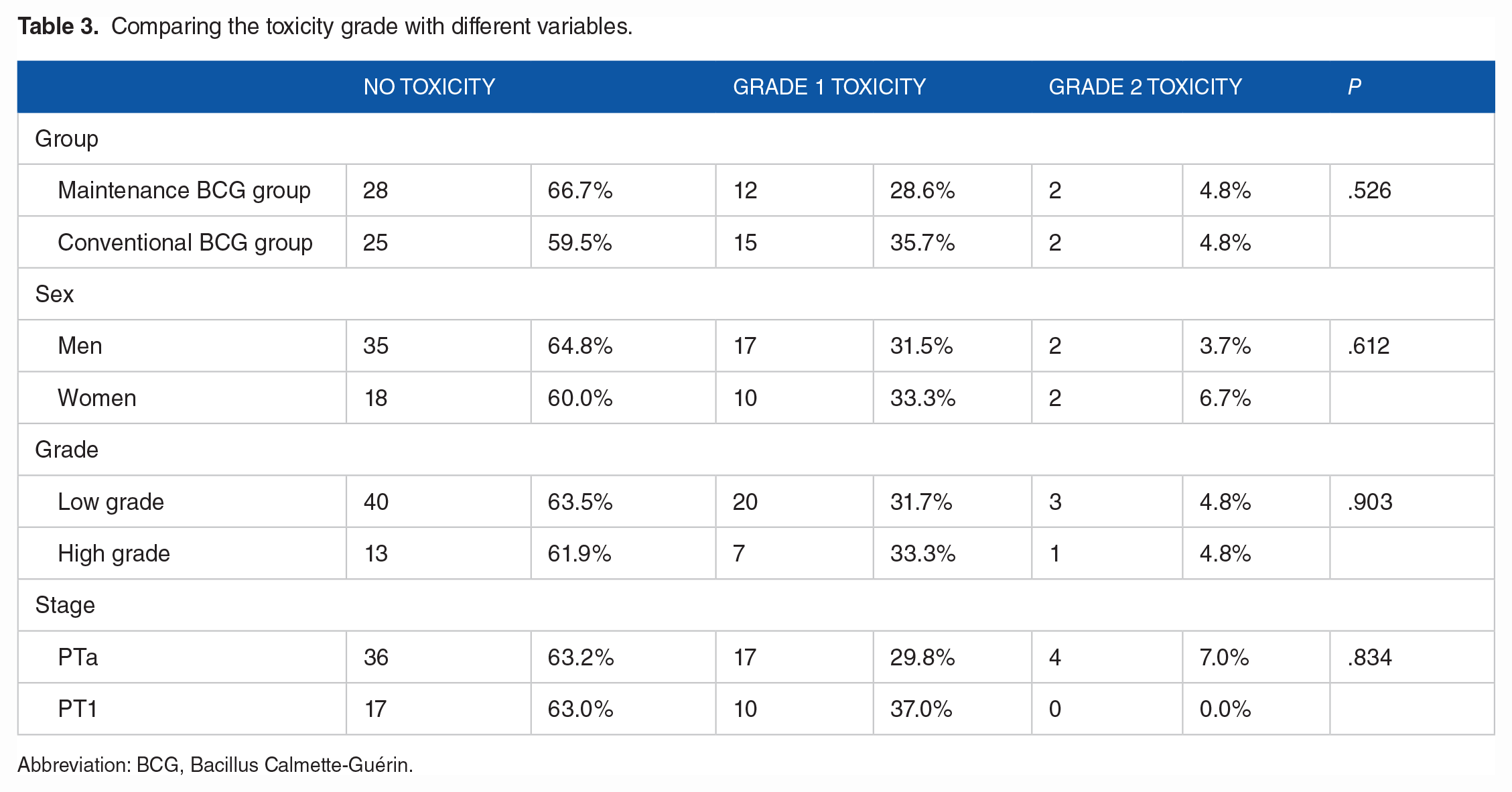
Abbreviation: BCG, Bacillus Calmette-Guérin.
Discussion
This study suggests that the BCG maintenance regimen does not increase admission rate and complications. In addition, patients on a maintenance diet ultimately performed better than other patients in adherence to treatment and regular cystoscopy referral. The great problem in prolonged maintenance therapy is patients’ low compliance with continuing the treatment. There was no patient who discontinued their BCG courses in this modified 6-month maintenance therapy.
Numerous guidelines have recommended BCG for intermediate-risk or high-risk NMIBC patients13,14; however, not every patient will benefit from this therapy. 15 The BCG treatment has been estimated to be failed in long-term follow-up in more than 40 to 50 patients, 16 and the respective 1-year and the 5-year recurrence rate is approximately 25% and 40% in patients with NMIBC receiving BCG. 17 Clinicopathological and molecular factors used to predict the effects of BCG have no application in predicting BCG response.18,19
Bacillus Calmette-Guérin has been developed in different laboratories to generate numerous genetic strains as a live organism grown in culture. Patients may obtain other responses when different organisms are applied, leading to variable efficacy and toxicity. 20 As recombinant forms of BCG may improve efficacy and reduce adverse effects, the developed BCG is created by resistance to antimicrobial peptides, higher internalization, and increased release of antitumor cytokines. 21
The RFS rate is lower in NMIBC patients who received general anesthesia during TURBT + BCG therapy than in those who received spinal anesthesia, which may be due to the inhibition of the immune response system throughout the surgery. 22 A recent study suggests that to improve the medical management of NMIBC, the importance of diversity and specificity of human leukocyte antigen (HLA)-B loci due to the antitumor effect of BCG injection should be considered. 7 Another in vitro study revealed that MyD88, an innate immune molecule, is essential for an efficient immune response in MB49 cells infected with BCG and co-cultured with macrophages. 23 Muto et al 24 suggested that Mycobacterium tuberculosis complex polymerase chain reaction positive patients are less likely to exhibit recurrence and progression after BCG therapy. Endothelin-1 has been suggested to worsen the prognosis of patients treated with adjuvant BCG installations for primary high-grade non–muscle-invasive urothelial bladder cancer. 25
According to the recent study, patients with high-risk NMIBC who underwent partial and complete induction of BCG experienced similar outcomes. 26 These results are comparable with our findings in continuing or discontinuing the maintenance therapy. Lamm et al 10 evaluated 550 patients with transitional cell carcinoma of the bladder with carcinoma in situ or an increased risk of recurrence after TURT. The criteria for increased risk were described as more than 1 episode of tumor recurrence within 1 year and/or more than 2 tumors more than 6 months. The efficacy and toxicity of maintenance were studied in comparison with induction therapy. There were no toxicities above grade 3 in the 243 maintenance arm patients. Median RFS time was twice as long as in the maintenance arm compared with the no maintenance arm, and patients had significantly longer worsening-free survival. Despite the significant effect of maintenance therapy, only 16% of maintenance cases completed all 8 scheduled maintenance courses within 3 years. They suggested that the 6-month maintenance therapy may be biologically important. In a systematic review for assessing the intermediate-risk or high-risk NMIBC, induction BCG followed by maintenance BCG instillation after TURBT in comparison with non-maintenance BCG could reduce the risk of tumor progression and recurrence. 27 Lawler et al 28 found no significant difference in bladder cancer outcomes after BCG treatment due to the participants’ race. Similarly, we found no significant difference in sex and bladder cancer outcomes.
There were some limitations to this study. The sample size was relatively small, which may have impacted the results of the analysis. To validate the findings of this study, further prospective studies with larger sample sizes are recommended. In addition, this study was limited by the short follow-up period of the patients, which should be extended in future studies.
Conclusions
Although the observed differences between the 2 groups regarding recurrence and progression were not statistically significant, the results of this study continue to support the need for further studies on BCG therapy maintenance regimens with larger sample sizes.
Footnotes
Acknowledgements
The authors acknowledge their colleagues at Labbafinejad Medical Center, Shahid Beheshti University of Medical Sciences, for their leading suggestions on this manuscript.
Author Contributions
NS, FS, and AB contributed to the project development and supervision, manuscript drafting, and editing. MD, BN, SAK, ES, and RB contributed to the project development, data collection, manuscript drafting, and editing. NB and HR contributed to the data analysis and manuscript editing.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of Data and Materials
All data analyzed during this study are included in this manuscript.
Ethical Approval and Consent to Participate
The study group adheres to the principles of medical ethics introduced by the Health Ministry and the Declaration of Helsinki. Departmental and Ethical Board Review approval was taken before the start of this academic work (ethic code: IR.SBMU.UNRC.9112.2). Informed consent was acquired from all patients. The study was registered at ![]() (identifer: IRCT20220302054165N1).
(identifer: IRCT20220302054165N1).
