Abstract
The most frequent malignant tumor of the head and neck is head and neck squamous cell carcinoma (HNSCC), which has a high frequency, a poor prognosis in the late stages, and subpar therapeutic results. As a result, early HNSCC diagnosis and treatment are urgently needed; however, there are no good diagnostic biomarkers or efficient therapeutic targets at this time. The long-stranded non-coding RNA HOTAIR may be important in the pathogenesis of cancer, according to recent research. By interactions with DNA, RNA, and proteins, it has been demonstrated that HOTAIR, a >200 nucleotide RNA transcript, plays a role in the biological processes of many types of tumor cells, including proliferation, metastasis, and prognosis of HNSCC. Hence, this review discusses HOTAIR’s function and molecular mechanisms in HNSCC.
Introduction
Head and neck squamous cell carcinoma (HNSCC) is a group of malignant tumors originating from the nasal cavity, paranasal sinuses, oral cavity, oropharynx, larynx, and hypopharynx and covered by squamous epithelial tissue. 1 In recent years, the incidence of HNSCC has been increasing year by year, with more than 600 000 new cases worldwide each year, ranking sixth among all cancers in terms of incidence. 2 Although great progress has been made in the treatment of HNSCC, the 5-year survival rate of patients has not been significantly improved. 3 Therefore, it is of great significance to elucidate the molecular mechanisms underlying the occurrence, progression, and metastasis of HNSCC and to discover effective early diagnostic and prognostic biomarkers as well as therapeutic targets for early screening and improving patient survival.
Long non-coding RNAs (LncRNAs) are non-protein-coding RNAs consisting of about 200 nucleotides. LncRNAs have been found to be overexpressed in tumor tissues and cells from breast, lung, liver, bladder, prostate, nasopharynx, and larynx cancers. LncRNAs can act as pro-oncogenes and oncogenes to regulate tumor cell growth, invasion, and migration, as well as participate in HNSCC pathophysiology at multiple levels, including transcriptional and epigenetic modifications. 4 The HOX anti-sense intergenic RNA (HOTAIR) was the first LncRNA discovered to have a retrotranspositional function and is found in a variety of tumors. 5 The expression abundance of HOTAIR is closely related to tumor size, stage, and poor prognosis and can be used to assess the clinicopathological characteristics and prognosis of patients. Second, HOTAIR can also influence the biological characteristics of tumor cell proliferation, apoptosis, invasion, and metastasis. 6 This article reviews the research progress, clinical significance, and molecular mechanism of HOTAIR, aiming to elucidate the important role of HOTAIR in the diagnosis, prognosis, and treatment of HNSCC and provide a basis for future research.
Materials and Methods
As a review of published papers, this work was exempt from institutional ethics committee approval. Our inclusion criteria were met, and this review includes all publications examining the function, molecular processes, and clinical importance of HOTAIR in HNSCC. Using a mix of terms from each of the 3 categories, a thorough search of 3 electronic databases—PubMed, Medline, and Google—was carried out.
“Long non-coding RNA HOTAIR,” “LncRNA HOTAIR,” “HOTAIR.”
“head and neck squamous cell carcinoma,” “HNSCC,” “hypopharyngeal squamous cell carcinoma,” “HSCC,” “nasopharyngeal carcinoma,” “NPC,” “oral squamous cell carcinoma,” “OSCC,” “ tongue squamous cell carcinoma,” “TSCC,” “Laryngeal squamous cell carcinoma,” “LSCC.”
“Tumorigenesis,” “Progression,” “Recurrence,” “Metastasis,” “Prognosis,” “Survival.”
To find studies that the computerized database search missed, we additionally looked at the reference lists of the studies that were included. Studies that satisfied all of the following criteria were included in this review after being independently reviewed by 2 researchers based on the title and abstract of the retrieved articles: (1) published in English and (2) to assess the functional importance, molecular processes, and/or clinical significance of LncRNA HOTAIR in HNSCC.
The Structure and Function of HOTAIR
HOTAIR is the first LncRNA with a trans-transcriptional regulatory role, first identified by Howard Chang et al in 2007. 7 HOTAIR is transcribed from the HOXC gene on chromosome 2, which has a total length of 2158 nucleotides, 6 exons, and is highly conserved evolutionary. 7 Studies have reported that HOTAIR can regulate gene transcription through protein complexes and also regulate mRNA transcription and translation through chromosomal remodeling, epigenetic regulation, repression of microRNA (miRNA), or altered mRNA modification, affecting the occurrence, metastasis, recurrence, prognosis, and treatment response of HNSCC from multiple dimensions of regulatory patterns.8,9
Regulatory Factors and Mechanisms of HOTAIR in HNSCC
The development of HNSCC is a complex process with multi-factorial, multi-stage, and multi-gene mutation accumulation, involving a complex mechanism of permanent cell proliferation, including oncogene overexpression, oncogene inactivation, cell cycle control failure, and tolerance to apoptosis. By interacting with numerous key factors, HOTAIR regulates biological functions such as proliferation, invasion, metastasis, and drug resistance. As a result, a better understanding of HOTAIR’s mechanisms of action and related signaling networks can help guide the development of targeted therapies for HNSCC. Table 1 exhibited the mechanisms of HOTAIR in HNSCC.
HOTAIR target molecules and consequences of their targeting in HNSCC.

Abbreviations: EMT, epithelial mesenchymal transition; HNSCC, head and neck squamous cell carcinoma; HOTAIR, HOX anti-sense intergenic RNA; LSCC, laryngeal squamous cell carcinoma; OSCC, oral squamous cell carcinoma.
The ceRNA network of HOTAIR in HNSCC cancer
The mechanism of interaction between LncRNAs and miRNA-competing endogenous RNAs (ceRNAs) has recently become a hot topic of research. Target mRNAs bind to miRNAs via microRNA response elements (MREs), which can inhibit or mediate their translation, thereby regulating gene expression at the post-transcriptional level. Researchers discovered that lncRNAs, in addition to mRNAs, have MREs as the study progressed. lncRNAs function as a molecular sponge, binding to microRNAs via MREs and “adsorbing” miRNAs from target mRNAs, resulting in abnormal regulation of target gene expression. 23 There is substantial evidence that lncRNAs can function as important members of the ceRNA regulatory network in the progression of HNSCC.
SRY-box transcription factor 4 (SOX4) is involved in regulating a variety of biological processes such as tissue differentiation, growth and development, and stress response. MiR-106a-5p is aberrantly expressed in many types of cancers, including nasopharyngeal carcinoma (NPC), 24 hepatocellular, 25 and colon cancers. 26 In cisplatin-resistant NPC cell lines, HOTAIR improved the resistance of NPC cells to cisplatin by mediating HOTAIR/miR-106a-5p/SOX4 activity.
Cyclooxygenase (COX) is a key enzyme for the conversion of arachidonic acid to prostaglandin H2 (PGH2), 27 and there are 2 isoforms of COX: COX-1 and COX-2. 22 According to research, high COX-2 expression is associated with lymph node metastasis in NPC. 28 HOTAIR promotes cell proliferation and migration in NPC by regulating the miR-101/COX-2 axis. Specifically, HOTAIR downregulates miR-101 expression before targeting and regulating the 3′-untranslated region (UTR) of COX-2.
GRP78, an immunoglobulin heavy chain binding protein (BIP), maintains cellular homeostasis by responding to unfolded proteins. 11 Overexpression of GRP78 is usually associated with malignant tumorigenesis or poor prognosis. 29 A targeting relationship between miR-30a-5p and GRP78 was found. 10 Coincidentally, GRP78 has also been identified as a functional target of HOTAIR and is involved in HOTAIR-mediated angiogenesis in NPC. 30
Stanniocalcin-2 (STC2) has been implicated in the metastasis and progression of malignant tumors. 12 Notably, increased STC2 expression promotes HNSCC cell proliferation, invasion, and migration. 31 MiR-206 inhibits STC2 expression by targeting its binding. 32 HOTAIR binds to miR-206 competitively and inhibits STC2 expression, thereby blocking the PI3K/AKT signaling pathway and inhibiting HNSCC cell proliferation, migration, and invasion. 19
HuR is a specific RNA-binding protein (RBP) that interacts with a large number of RNA transcription products to mediate post-transcriptional regulation. 33 HOTAIR actively regulates HuR expression by acting as a sponge for miR-7 and thus controls HNSCC metastasis and progression. Furthermore, in addition to being a member of the ceRNA regulatory loop, HuR can directly bind to HOTAIR, enhancing HOTAIR’s “sponge attraction” to miR-7. 13
The zinc finger transcription factor SNAI2 is a member of the SNAI/SLUG superfamily. SNAI2 binds to the E-box of the target gene promoter, repressing gene transcription and inducing epithelial mesenchymal transition (EMT), and has been linked to cancer invasion and metastasis. 34 Downregulation of HOTAIR suppresses EMT and drug resistance in laryngeal squamous cell carcinoma (LSCC) cells by sponging and increasing the expression of miR-613, resulting in a decrease in SNAI2 expression. 35 Figure 1 illustrates the regulatory mechanism model of HOTAIR.

The ceRNA mechanism and potential downstream regulatory mechanisms of HOTAIR.
The HOTAIR-related signaling pathways
There was a significant inverse relationship between the abundance of HOTAIR expression in oral squamous cell carcinoma (OSCC) tissues and cell lines and the level of EMT marker E-cadherin expression. HOTAIR helps to bind EZH2 and H3K27me3 to the E-cadherin promoter. HOTAIR promotes cancer progression in OSCC cell lines by recruiting EZH2 and H3K27me3 to regulate E-cadherin, inducing proliferation and invasion, and decreasing apoptosis. 36 Furthermore, HOTAIR recruits EZH2 to catalyze triple methylation of H3K27, which reduces the expression of tumor suppressor genes. In addition, inhibiting the growth of HNSCC cell lines by targeting HOTAIR and EZH2 induces mitochondria-associated apoptosis. 15
Changing the chemical modification of EZH2 causes changes in its affinity for HOTAIR. Among the chemical modifications of EZH2, phosphorylation of the T345 site could increase its affinity for binding to HOTAIR and affect the expression of downstream target genes. HOTAIR and EZH2 were found to be overexpressed in LSCC tissues and significantly correlated with T stage, pathological grade, and lymphatic metastasis. Depletion of HOTAIR expression stimulated EZH2 expression, blocked the proliferation of LSCC cell line AMC-HN8 cells, and increased sensitivity to cisplatin. 18 Furthermore, crosstalk between the STAT3/HOTAIR/EZH2 and PI3K/AKT pathways influences the therapeutic sensitivity of cisplatin and cetuximab to HNSCC. Overexpression of PTCSC3 inhibited HOTAIR expression, and in addition, PTCSC3 overexpression mediated the downregulation of STAT3, which in turn mediated the upregulation of HOTAIR. Thus, PTCSC3 may interact negatively with HOTAIR through STAT3 to inhibit the proliferation of LSCC cell lines. 21
Relationship Between HOTAIR and HNSCC
In this review, we summarize and briefly review 5 widely reported HNSCC associated with HOTAIR dysregulation, focusing on hypopharyngeal squamous cell carcinoma (HSCC), NPC, OSCC, tongue squamous cell carcinoma (TSCC), and LSCC.
Abnormal expression of HOTAIR in HNSCC
It is clear that dysregulated expression of HOTAIR is strongly linked to proliferation, metastasis, and invasion of HNSCC, and abnormally expressed LncRNA may be a significant inducer and accelerator of tumor development and metastasis. 37 When compared with healthy oral mucosa tissues and oral keratin-forming cells, OSCC tissues and Hep-2/R and TU686/R cell lines showed increased expression of HOTAIR. The level of this expression was also positively correlated with the tumor lymph node metastasis (TNM) stage, histological grade, and regional lymph node metastasis.14,17,35 In addition, patients with LSCC had considerably greater plasma levels of HOTAIR than did healthy controls. 21 Patients with vocal cord polyps had considerably lower levels of HOTAIR in cancer tissue exosomes compared with patients with LSCC. 20 The abundance of HOTAIR was also significantly increased in NPC tissues and cell lines (CNE1, CNE2) compared with chronic nasopharyngeal mucosal inflammatory tissues and the normal nasopharyngeal cell line NP460.21,38
Further in vitro experiments revealed that HOTAIR plays a role in tumorigenesis, invasion, and metastasis. Downregulation of HOTAIR expression inhibited the proliferation of LSCC cells and promoted apoptosis in NPC cells.37,39 Specific HOTAIR shRNA inhibited the migration, invasion, and epithelial cell mesenchymal transition of OSCC and LSCC cells.35,40
Furthermore, HOTAIR expression was linked to drug resistance in HNSCC. After chemotherapy for LSCC, HOTAIR expression decreased significantly with increasing cisplatin and paclitaxel concentrations and treatment duration. 41 Reduced HOTAIR levels significantly limited drug resistance in LSCC cells, and this inhibitory effect was significantly eliminated by miR-613 inhibitors. 35
Correlation of HOTAIR expression and single-nucleotide polymorphisms with prognosis and clinicopathological features in HNSCC
HOTAIR has also been reported to be strongly associated with overall survival, disease-free survival, and prognosis of patients with HNSCC. HOTAIR can be an independent prognostic factor for HNSCC, and we have showed the latest data on the prognostic value and clinicopathological features of HOTAIR in HNSCC.
Patients with LSCC, 42 NPC, 43 and OSCC36,44 have been discovered to have high expression of HOTAIR, which generally indicates poor overall, disease-free survival. In addition, there was a strong correlation between the level of HOTAIR expression and the clinicopathological features of patients with HNSCC, such as T classification, pathological grade, clinical stage, and lymph node metastasis. High HOTAIR expression was associated with lymph node metastases and poor overall survival in patients. 45 HOTAIR expression was upregulated in large volume, advanced tumor stage, and NPC with increased lymph node tumor load. The level of HOTAIR expression gradually increased with the progression of clinical stage. 43
Squamous cell carcinomas of the larynx in the T3-T4 stage have considerably greater HOTAIR expression levels than those in the T1-T2 stage. Individuals with G2 pathological grading had considerably higher levels of HOTAIR expression than patients with G1 grade. 18 Furthermore, patients with T3/T4 classification, III/IV clinical stage, and lymph node metastases of LSCC displayed considerably higher levels of exosomal HOTAIR expression than those with early stage and no lymph node metastasis. 20 A strong correlation between high HOTAIR expression and a family history of LSCC was discovered. 46
In addition, single-nucleotide polymorphisms (SNPs) of HOTAIR have been reported to help in the prediction of HNSCC susceptibility and the effectiveness of targeted therapy. The rs1899663 polymorphism of HOTAIR was significantly associated with the risk of developing OSCC. Non-betel nut users bearing the T allele for the HOTAIR rs920778 polymorphism were more likely to develop OSCC. Moreover, the G allele of rs4759314 polymorphism and the T allele of rs920778 polymorphism were linked to lymph node metastasis and large volume tumors, respectively. 33 In the Chinese population, the HOTAIR rs4759314 polymorphism has been associated with a considerably higher incidence of HNSCC and may serve as a diagnostic biomarker. 43 HOTAIR expression abundance and SNPs are strongly associated with the prognosis, clinicopathological features, and risk of developing HNSCC. As a result, HOTAIR expression and genotype may be useful as complementary indicators for prognosis, clinicopathological staging, and disease risk assessment to classify high-risk patients and assist clinicians in selecting the best personalized treatment strategy. We have plotted the relationship between HOTAIR and SNPs in Table 2.
The effects of SNPs in HOTAIR in HNSCC.
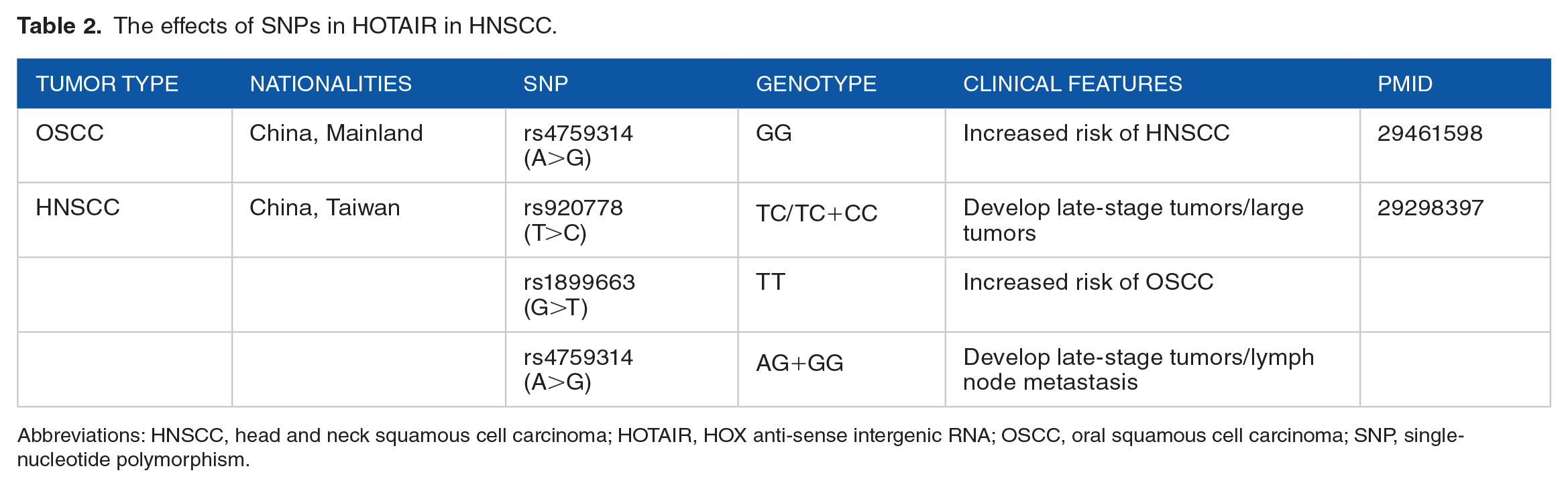
Abbreviations: HNSCC, head and neck squamous cell carcinoma; HOTAIR, HOX anti-sense intergenic RNA; OSCC, oral squamous cell carcinoma; SNP, single-nucleotide polymorphism.
The role of HOTAIR in the treatment of HNSCC
Despite the fact that many studies have shown that HOTAIR has a significant negative impact on the occurrence, progression, metastasis, and prognosis of HNSCC, targeting HOTAIR leads to inhibition of its expression or function, and thus targeting HOTAIR may be a promising strategy for cancer treatment.
HOTAIR knockdown induced apoptosis, mitochondrial membrane potential changes, mitochondrial calcium uptake 1 (MICU1)-dependent cell death, and mitochondria-associated cell death pathways in the OSCC line Tca8113 and the TSCC cell line Tscca. Furthermore, a Tscca xenograft tumor model validated that HOTAIR depletion inhibits tumor growth. 46 Depletion of HOTAIR expression significantly triggers cell cycle arrest and apoptosis in the LSCC Tu212 and Hep-2. 37 When compared with control cell lines, HOTAIR knockdown in NPC cell lines CNE1 and CNE2 exhibited impaired independent growth as well as reduced migration capacity. 38 HOTAIR, p21, and p53 mRNA expression were increased in doxorubicin (DOX)-treated or γ-irradiated Tca8113 cells. DNA damage-induced high expression of HOTAIR promoted Tca8113 cell proliferation while decreasing the incidence of DOX-induced apoptosis. When HOTAIR expression was suppressed with specific siRNA, the proportion of cells in the G2/M and S phases increased and decreased, respectively, while the rate of DOX-induced apoptosis increased. 47
Epithelial mesenchymal transition is a process by which epithelial cells acquire mesenchymal features under specific conditions. 37 Epithelial mesenchymal transition causes epithelial cells to lose characteristics such as apical-basal polarity and E-cadherin expression while gaining mesenchymal biomarkers such as Twist, Slug, Snail, Vimentin, and N-cadherin. Epithelial mesenchymal transition provides cancer cells with enhanced migratory, invasive, proliferative, and anti-apoptotic capabilities and has received widespread attention as a major cause of cancer metastasis during cancer development. 48
HOTAIR shRNA reverses increased N-cadherin levels and decreased E-cadherin in Hep-2/R, TU686, and TU686/R of LSCC cells and the EMT process is inhibited by HOTAIR shRNA. 35 HOTAIR siRNA significantly increased E-cadherin gene and protein expression levels in OSCC cells TSCCA and Tca8113, reduced colony formation, invasion and migration ability of cancer cells, and induced apoptosis in vitro. 36
When HOTAIR was silenced, the expression of cystathione 3, cystathione 7, and bim was upregulated in KB and CAL-27 of OSCC cells, while Mcl-1, survivin, and bcl-2 were downregulated, the number of apoptotic cells was dramatically increased, and cisplatin sensitivity was improved. HOTAIR silencing reduced the wound healing ability of KB and CAL-27 cells as well as the migratory and invasive properties of cancer cells. 49
HOTAIR was found to be highly expressed in tongue phalloid carcinoma cell line SCC25, laryngeal squamous carcinoma cell line HN4, oral squamous cell line HN13, and pharyngeal squamous carcinoma cell line FaDu when compared with non-tumorigenic cell lines. HOTAIR inhibition suppressed the proliferation, migration, and invasion of all of the above tumorigenic cell lines while increasing apoptosis and cell cycle arrest. 13
Downregulation of HOTAIR expression inhibited the proliferation, invasion, and migration of Tu686, TSCCA, and Cal27 cell lines by blocking the PI3K/AKT signaling pathway. Silencing HOTAIR also inhibited the growth of HNSCC tumors in nude mice. 19 Knockdown of HOTAIR resulted in inhibition of FASN expression, ab initio synthesis of free fatty acids, and expression of MMP-9 and p21 in the NPC cell lines CNE2 and 5-8F, and therefore, cell proliferation and invasion were impaired. 50 HOTAIR depletion stimulated EZH2 expression and increased the sensitivity of the LSCC cell line AMC-HN to cisplatin. 18
In cisplatin-resistant NPC cells, including C666-1/DDP and CNE2/DDP, knockdown of HOTAIR reduced cell viability as well as the expression of chemoresistance-associated proteins, including multidrug resistance mutation 1 (MDR1), multidrug resistance-associated protein 5 (MDR5), LDL receptor-associated protein-1 (LRP1), and ATP-binding cassette subfamily B member 1 (ABCB1), thereby increasing cellular sensitivity to cisplatin. 16 HOTAIR shRNA largely reversed the upregulation of MDR1 protein expression and reduced the dose of cisplatin in LSCC cell lines Hep-2/R and TU686/R cells, indicating that HOTAIR shRNA could effectively inhibit drug resistance in LSCC. 35
In summary, because of the important role of HOTAIR in HNSCC, targeting HOTAIR via antisense oligonucleotide, RNAi inhibition of HOTAIR expression, and small molecule inhibitors blocking HOTAIR function, resulting in HOTAIR expression or function inhibition, could be a potential strategy for cancer therapy.
Future Perspectives
Understanding the function of HOTAIR in tumor cell biology has advanced significantly in recent years. However, further investigation is required to solve the larger practical puzzles of HOTAIR. The investigation of HOTAIR expression and the use of SNPs as prognostic and diagnostic biomarkers are 2 areas of clinical application. Several studies have demonstrated a correlation between HOTAIR expression and SNPs and tumor stage, aggressiveness, poor prognosis, and recurrence. Second, there is mounting evidence that HOTAIR may play a role in the initiation and progression of oral, laryngeal, and HNSCC, with higher expression in these tissues and cell lines. Uncertainty still exists regarding the transcriptional and upstream signaling networks that support enhanced HOTAIR expression in malignancies. Finding the regulatory networks that cause tumor cells to overexpress HOTAIR and decrease its action may reveal new therapeutic targets. Also, it is now recognized that HOTAIR expression is linked to the development of chemoresistance and affects the effectiveness of chemotherapy drugs like cisplatin. As a result, it can be used in conjunction with currently available medications to increase the sensitivity of malignant tumors to chemotherapeutic treatments. Finding a high-efficiency lncRNA target and a reliable in vivo drug delivery mechanism are necessary for the aforementioned idea to be applied clinically. Today’s genetic modification techniques, siRNA and shRNA, can be used as an efficient method to suppress HOTAIR and play a part in the therapy of HNSCC because they can successfully reduce the production of lncRNA in cells. Further research examining the precise mechanisms of HOTAIR activity and function will help to develop new treatment targets and biomarkers, as well as aid in the early detection of HNSCC. These innovations will aid in the advancement of individualized precision medicine and enhance the prognosis for patients with HNSCC.
Conclusions
The role and mechanism of HOTAIR on HNSCC have been documented as a result of the advancement of biological science and technology as well as additional investigation of the lncRNA’s molecular mechanism. HNSCC have high levels of HOTAIR, which suggests that it could be used as a biomarker for cancer diagnosis, prognosis, or treatment. As an oncogene, HOTAIR can accelerate tumor growth and contribute to a variety of malignancies. Several mechanisms underlie HOTAIR effects. Understanding the mechanism of HOTAIR can aid in the investigation of the connection between lncRNA and cancers, which is important for elucidating the pathophysiology of malignancies.
Footnotes
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
Yuandi Xiang searched Literatures and wrote the original manuscript.
Qingquan Hua designed theme and revised the manuscript.
