Abstract
Background:
Hepatocellular carcinoma (HCC) has insidious onset. Most HCC patients are in advanced stage at the time of initial diagnosis, and the treatment response is poor. The purpose of this study was to compare the clinical effectiveness of conventional transcatheter arterial chemoembolization (c-TACE) combined with sorafenib versus c-TACE monotherapy in the treatment of advanced HCC.
Methods:
A retrospective analysis was performed on patients with advanced HCC (stage C based on the Barcelona Clinic Liver Cancer staging system) admitted to the Affiliated Hospital of Southwest Medical University from December 9, 2013, to February 25, 2021. After screening for inclusion and exclusion criteria, 120 patients were finally included, including 60 patients in the c-TACE group and 60 patients in c-TACE + sorafenib group. There were no statistically significant differences in general data between the 2 groups before treatment. Overall survival (OS) and progression-free survival (PFS) were compared between the 2 groups, and prognostic factors were assessed by Cox proportional risk model.
Results:
The study found that median PFS was 7.37 months in the c-TACE + sorafenib group and 5.97 months in c-TACE group, a statistically significant difference (χ2 = 5.239, P = .022 < .05). The median OS was 22.9 months in the combination group and 12.1 months in c-TACE monotherapy group, also a statistically significant difference (χ2 = 5.848, P = .016 < .05). The Cox proportional risk model found that c-TACE number and presence of ascites were common risk factors among patients in both groups (P < .05).
Conclusion:
c-TACE + sorafenib was superior to c-TACE alone in the treatment of advanced HCC and yielded significant improvements in PFS and OS in our study. The number of c-TACE and presence of ascites were common risk factors affecting the survival of patients in the 2 groups.
Introduction
Hepatocellular carcinoma (HCC) is the leading cause of malignant tumor-related death worldwide.1,2 Surgical resection is still one of the most effective treatments. 3 However, due to insidious onset, most HCC patients are already in advanced stage at the time of diagnosis, and few patients are suitable for surgical treatment. The 5-year survival rate is only 20%. 4
Transcatheter arterial chemoembolization (TACE) is a treatment method to inject chemotherapeutic drugs and embolization agents into the blood-supplying arteries of tumors, causing vascular embolization and tumor ischemic necrosis. Transcatheter arterial chemoembolization is currently recognized as the preferred local treatment for intermediate and advanced HCC. 5 However, due to the existence of multiple blood supplies for liver tumors, the hypoxic environment formed after focal embolization can promote the establishment of angiogenesis and collateral circulation. As a result, TACE is not sufficiently effective as a monotherapy and may be more efficacious in combination with another treatment. Sorafenib, a multikinase vascular endothelial growth factor (VEGF) inhibitor, is the most widely used systemic chemotherapy approved as a first-line agent for unresectable or advanced HCC. It has been shown to improve the survival rate of patients with advanced HCC and effectively controls disease in about 50% of patients. 6 Studies have shown that sorafenib combined with TACE is superior to TACE alone in the treatment of HCC.7,8 However, there are relatively few studies on sorafenib + TACE combination therapy in the treatment of advanced HCC. This study retrospectively analyzed the clinical effectiveness of c-TACE + sorafenib combination therapy versus c-TACE alone in patients with advanced HCC, compared the overall survival (OS) and progression-free survival (PFS) of the 2 groups and evaluated the risk factors.
Materials and Methods
Study design and patient population
A retrospective analysis was performed on 145 patients with stage c HCC in the Barcelona Clinic Liver Cancer (BCLC) staging system who were hospitalized in the Affiliated Hospital of Southwest Medical University from December 9, 2013, to February 25, 2021. After screening for inclusion and exclusion criteria, 120 patients were finally included, including 60 patients in the c-TACE group and 60 patients in c-TACE + sorafenib group (Figure 1).

Flow diagram of the study population.
Inclusion and exclusion criteria
Inclusion criteria
Age ⩾ 18 years; histological or cytological diagnosis of HCC; stage C based on the BCLC staging system; Child-Pugh classification A or B; score of 0 to 1 according to Eastern Cooperative Oncology Group performance status (ECOG PS).
Exclusion criteria
Liver transplantation, history of esophageal variceal hemorrhage, massive ascites, hepatic encephalopathy, abdominal infection, severe liver and kidney dysfunction, moderate to severe bone marrow dysfunction, heart dysfunction, and inability to tolerate treatment due to serious side effects.
Research ethics and patient consent
All patients provided written informed consent prior to any study-related procedures in accordance with the principles of the World Medical Association Declaration of Helsinki. The study has been approved by the Clinical Trials Ethics Committee of the Affiliated Hospital of Southwest Medical University.
Therapeutic methods
c-TACE method
The Seldinger technique was used to puncture the femoral artery and intubate the hepatic artery for selective angiography. The tumor-supplying artery was identified.
A mixture of lobaplatin, 5-fluorouracil and poppy ethyl iodide oil was injected into the tumor-supplying artery via catheter, and the tumor-supplying artery was embolized with polyvinyl alcohol embolization microspheres. Angiography was performed after embolization, which showed a decrease in tumor-supplying arteries and the treatment ended.
Sorafenib treatment
Sorafenib treatment was started on the 4th to 7th day after c-TACE, at 400 mg twice a day, and continued until disease progression or intolerance.
Observation method
Follow-up began on the first day after c-TACE treatment. Overall survival was calculated as the time from the day after c-TACE treatment to death or the last follow-up. Progression-free survival was calculated as the time from the day after c-TACE treatment to disease progression or death. The final follow-up date was August 13, 2021.
Statistics analysis
Statistical analyses were performed using SPSS 20.0 software. The measurement data were represented by mean ± standard deviation and were analyzed by T test. The counting data were represented by case numbers and were analyzed by chi-square test. Cox risk regression model was used to analyze the risk factors affecting survival rate. Kaplan-Meier was used to calculate PFS and OS and their median time, and log-rank analysis was used to test significance. P < .05 was considered statistically significant in all cases.
Results
Patient demographics and clinical characteristics
A total of 120 patients met the inclusion criteria. Baseline characteristics of patients in the 2 groups (age, sex, child classification, primary tumor diameter, number of tumor, extrahepatic metastases, tumor invaded the hepatic vein, the level of AFP, the number of c-TACE, and portal vein invasion and glutamic-pyruvic transaminase) were not significantly different (P > .05, Table 1). The average number of c-TACE operations in the c-TACE + sorafenib group was 2.2 and 1.8 in the c-TACE group. The average AFP level was 5935.4 in the c-TACE + sorafenib group and 5099 in the c-TACE group. The average age of the c-TACE + sorafenib group was 53.9 years, while the average age of the c-TACE group was 54.1 years.
Patient demographics and clinical characteristics.

Abbreviation: TACE, transcatheter arterial chemoembolization; AFP, alpha-fetoprotein.
There was no statistical significance in age, sex, child classification, primary tumor diameter, number of tumor, extrahepatic metastases, and tumor invaded the hepatic vein, AFP level, TACE number, portal vein invasion and glutamic-pyruvic transaminase between the 2 groups (P > .05).
Comparison of PFS and OS between the 2 groups
Kaplan-Meier survival analysis showed that the median PFS was 7.37 (5.13-9.6) months in c-TACE + sorafenib group and 5.97 (2.96-8.98) months in c-TACE group, a statistically significant difference (χ2 = 5.239, P = .022 < .05, Figure 2). The median OS was 22.9 (14.55-31.25) months in c-TACE + sorafenib group and 12.1 (8.68-15.52) months in c-TACE group, a statistically significant difference (χ2 = 5.848, P = .016 < .05, Figure 3).
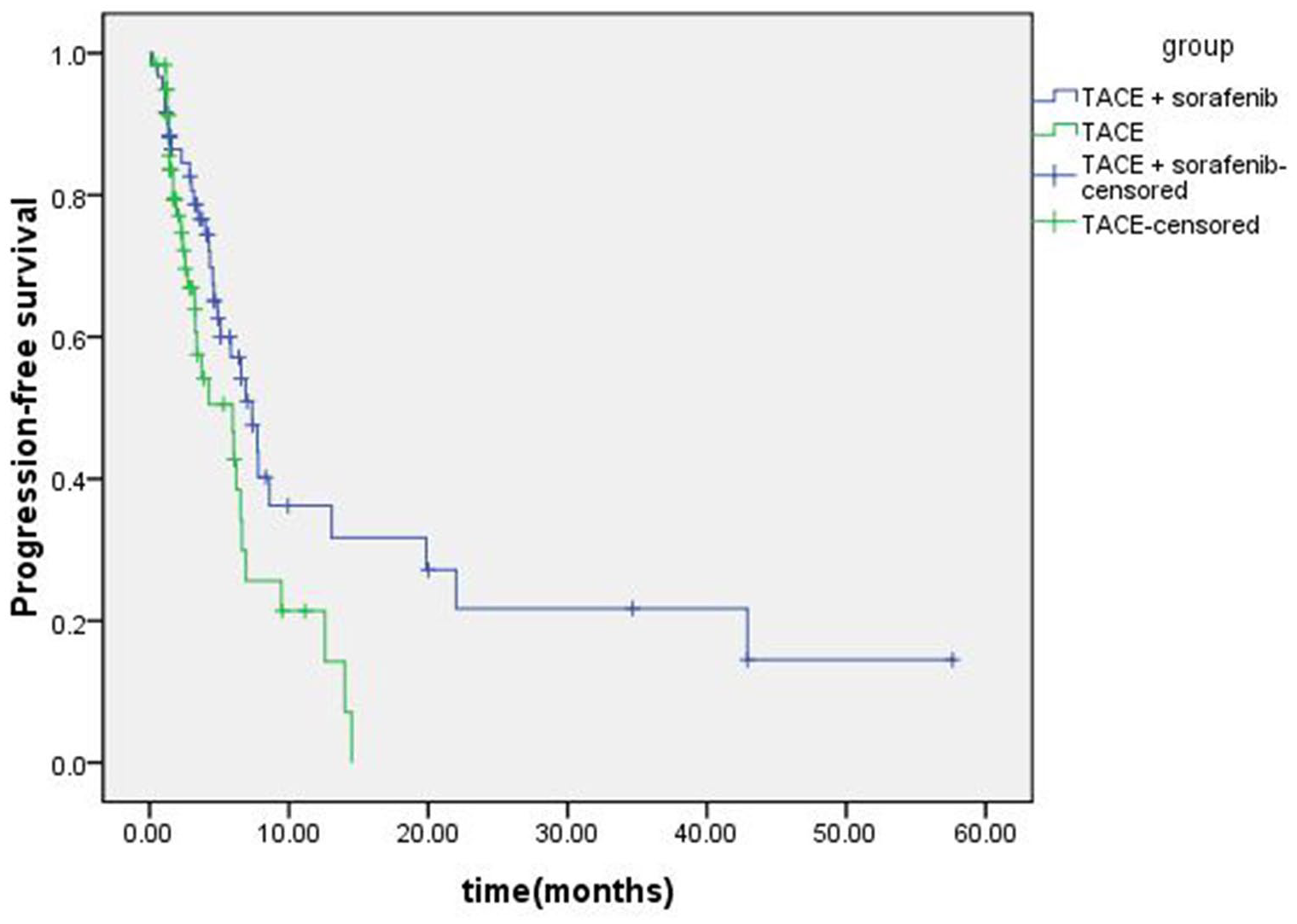
Kaplan-Meier survival analysis showed that the median PFS was 7.37 months in TACE + sorafenib group and 5.97 months in TACE group, a statistically significant difference (χ2 = 5.239, P = .022 < .05).

Kaplan-Meier survival analysis showed that the median OS was 22.9 months in TACE + sorafenib group and 12.1 months in TACE group, a statistically significant difference (χ2 = 5.848, P = .016 < .05).
Analysis of survival risk factors in 2 groups
The possible factors affecting the survival and prognosis of patients were analyzed by Cox risk regression model. The study found that portal vein invasion, c-TACE number, presence of liver cirrhosis, and the presence of ascites were found to be risk factors affecting survival in c-TACE + sorafenib group (P < .05). Conversely, tumor number (if > 3), c-TACE number, Child-Pugh classification status, and the presence of ascites were risk factors affecting survival of patients in the c-TACE group (P < .05). Notably, the number of c-TACE and the presence of ascites were common risk factors affecting the survival of patients in both groups (P < .05, Table 2).
Analysis of risk factors affecting survival of patients in the 2 groups.

Abbreviation: TACE, transcatheter arterial chemoembolization.
Portal vein invasion, the number of TACE, liver cirrhosis and ascites were found to be risk factors affecting survival in TACE + sorafenib group (P < .05). The number of tumors > 3, the number of TACE, child classification and ascites were risk factors affecting survival of patients in the TACE group (P < .05).
Discussion
Hepatocellular carcinoma is a common malignant tumor and the second leading cause of tumor death. 9 The early clinical symptoms of HCC are not obvious, and it is generally more advanced when it is found. 10 Transcatheter arterial chemoembolization is mainly used for the treatment of advanced HCC patients who do not meet the conditions for radical resection. 11 Transcatheter arterial chemoembolization includes c-TACE, drug-eluting microsphere transarterial chemoembolisation (DEM-TACE), degradable starch microsphere transarterial chemoembolisation (DSM-TACE), and balloon-occluded transarterial chemoembolisation (b-TACE). Transcatheter arterial chemoembolization can be combined with systemic therapies, such as targeted therapy and immunotherapy. 12 Sorafenib is an oral multikinase inhibitor that can affect the activity of neovascular-related receptors and the secretion of cytokines such as VEGF, thus blocking tumor angiogenesis and delaying tumor growth. Studies have shown that TACE in combination with sorafenib provides a greater therapeutic effect in the HCC compared with TACE or sorafenib alone.13,14
Kuang et al 15 found that the clinical efficacy of sorafenib combined with TACE in the treatment of advanced HCC was superior to that of TACE alone. Adverse reactions were tolerable, serum tumor marker levels were significantly reduced, and survival time of patients was prolonged. Addition of TACE to sorafenib improved survival and reduced mortality by 26%. 16 For patients with TACE refractory liver cancer, the therapeutic effect can also be increased with the addition of sorafenib. Lin et al 17 reported that the median OS of patients with TACE refractory liver cancer treated with sorafenib was significantly longer than those treated with TACE alone. In a retrospective study of TACE + sorafenib combination therapy in TACE refractory advanced HCC, 5-year OS and time to progression (TTP) were prolonged in the TACE + sorafenib group compared with TACE alone. Although adverse events (AEs) did not occur in the TACE group and severe AEs occurred in 2 patients in the TACE + sorafenib group, they were effectively alleviated by lowering the dose of sorafenib. 18 Survival outcomes (PFS and OS) of TACE combined with sorafenib were also superior to TACE alone in locally advanced HCC patients with macroscopic vascular invasion (MVI). Moreover, in this study, propensity score matching (PSM) was used to minimize the influence of selection bias and potential confounders, thus reducing the experimental error, which was worthy of recommendation. 19 Park et al 20 found that sorafenib combined with c-TACE significantly improved TTP, PFS, and tumor response rates in advanced HCC patients compared with sorafenib alone, although it did not improve OS. Patients in the trial who received ⩾2 c-TACE treatments had significantly longer survival than those who received sorafenib alone. In this study, the number of TACE treatments was also a common risk factor for TACE alone or in combination with sorafenib and affected survival. 20 These previous studies indicate that sorafenib combination therapy can indeed improve the prognosis of HCC compared with TACE treatment alone, and our study further supports this idea. We observed prolonged OS and PFS in advanced HCC patients who were treated with TACE + sorafenib combination therapy versus those who were treated with TACE alone.
This can also be supported by theory. From the theoretical background of the study, TACE therapy embolizes the tumor-supplying artery, resulting in increased HIF-1α level, which is associated with tumor recurrence, disease progression, and distant metastasis.21,22 Second, TACE stimulates the overexpression of VEGF in liver cancer, thereby promoting angiogenesis.23,24 However, sorafenib can effectively reduce the expression of HIF-1α and VEGF, inhibit VEGF receptors and platelet-derived growth factor receptors, and alleviate the adverse conditions induced by TACE.25-27 Therefore, TACE plus sorafenib treatment can improve the clinical efficacy, inhibit tumor recurrence and metastasis and prolong the survival of patients.
However, the results were inconsistent in the meta-analysis. Some meta-analyses showed that TACE combined with sorafenib significantly prolonged patients’ OS compared with TACE alone.13,28 However, some meta-analyses showed that TACE combined with sorafenib did not significantly prolong patients’ OS compared with TACE alone.29-31 There may be many reasons for the inconsistent meta-analysis results, including different research methods, different analysis methods, different ranges of included studies, limited number of included studies, different follow-up times, different timing of taking sorafenib, different treatment courses and dosages of sorafenib, and different TACE times. All of these may affect the results of meta-analysis. However, regardless of the meta-analysis results, all studies have shown that TACE plus sorafenib benefits unresectable HCC patients compared with TACE alone.
Ni et al 32 analyzed the prognostic factors of TACE combined with sorafenib for HCC. Multivariate analysis showed that BCLC stage, extrahepatic spread, duration of sorafenib treatment were independent prognostic factors associated with OS. Biolato et al 33 found that nontumor segmental portal vein thrombosis, serum sodium, maximum nodule diameter, nodule number, a-fetoprotein, and alkaline phosphatase were independent prognostic factors affecting OS. For patients with TACE refractory HCC, tumor size, baseline alpha-fetoprotein, increases in alpha-fetoprotein, and Child-Pugh score after initial TACE treatment were predictors of refractory TACE. 17 Different risk factor outcomes may still be related to different study methods, different analysis methods, different scope of inclusion studies, limited number of inclusion studies, and different duration of follow-up. Prognostic factors in the above studies were also inconsistent with the results of this study. In this study, it was found that the number of TACE and the presence of ascites were common risk factors in both groups. Portal vein invasion and the presence of liver cirrhosis were independent risk factors for survival in the c-TACE + sorafenib group. In contrast, the number of tumors (if >3) and Child-Pugh classification status were independent risk factor for survival in the c-TACE group. This suggests that the number of c-TACE and the presence of ascites affect the prognosis of patients with advanced HCC whether treated alone or in combination with sorafenib. This also serves as a reminder to clinicians to give attention to the number of c-TACE and avoid the formation of ascites in the treatment process, so as to prolong the survival of patients as much as possible.
Through the study of c-TACE alone or combined with sorafenib in the treatment of advanced HCC, it was found that compared with c-TACE alone, c-TACE combined with sorafenib could prolong OS and PFS. However, there are still some limitations in this study. It was designed as a retrospective study, which may have selectivity or reporting bias. In addition, the relatively small sample size may have some influence on data analysis. Therefore, further prospective studies with greater sample sizes would be helpful in validating the findings of this study.
Conclusions
Compared with c-TACE alone, c-TACE + sorafenib combination therapy in the treatment of advanced HCC has a better therapeutic effect and can better improve patients’ PFS and OS. Portal vein invasion, c-TACE number, presence of liver cirrhosis, and the presence of ascites were found to be risk factors affecting survival in c-TACE + sorafenib group. Conversely, tumor number (if >3), c-TACE number, Child-Pugh classification status, and the presence of ascites were risk factors affecting survival of patients in the c-TACE group. Number of c-TACE and the presence of ascites were common risk factors among both groups.
Footnotes
Acknowledgements
The authors thank the teachers of Statistics Department of Southwest Medical University for their help in data analysis of this study and the Oncology Department of Affiliated Hospital of Southwest Medical University for their support in data collection.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Key Laboratory of Medical Electrophysiology (Southwest Medical University), Open Fund(grant no. KeyME-KeyME-2020-011); Research Project of Southwest Medical University (grant no. 2021ZKQN068).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
FW made a substantial contribution to the design of the work; SL, YH, and ZZ made a substantial contribution to the acquisition and analysis of data; SL and YH drafted the article, ZZ and FW revised it critically for important intellectual content; FW approved the version to be published.
