Abstract
Hepatocellular carcinoma (HCC) is one of the most common and deadly malignancies worldwide. Approximately, 80% of patients are initially diagnosed at intermediate or advanced stages, which means that curative therapies are unable to be performed. In most cases, systemic treatment is ineffective, especially when conventional cytotoxic agents are used. Sorafenib has been the only systemic agent proven to be effective in treating advanced HCC for over a decade. The rapid development of immunotherapy has remarkably revolutionized the management of advanced HCC. Besides, the combination of immunotherapy with molecular targeted agents or locoregional treatments is emerging as an effective tool for enhancing immunity. In the review, an overview of immunotherapy and its combination therapies for HCC is presented.
Keywords
Introduction
Hepatocellular carcinoma (HCC), accounting for 75% to 85% of primary liver cancer, is one of the most common and fatal cancers worldwide.1-3 It mainly occurs in the background of long-term liver disease. Approximately, 80% of HCCs are at intermediate or advanced stage at the time of initial diagnosis.3,4 Molecular targeted therapies have been developed as the preferred treatment recommendation for advanced HCC during the past decade.
Until 2016, sorafenib, the multityrosine kinase inhibitor (multi-TKI), was the only systemic agent for the treatment of advanced HCC. Subsequently, three new multi-TKIs have been approved worldwide since 2017.5-7 The first-line study found that lenvatinib was not inferior to sorafenib, and the second-line studies found that regorafenib, cabozantinib, and ramucirumab showed clear survival benefits over placebo.5-9 Recently, donafenib showed superiority over sorafenib in improving overall survival (OS) for advanced HCC and has been recommended as the first-line recommendation for advanced HCC in China. 10
Metronomic capecitabine seemed a treatment option for Child-Pugh B HCC patients, especially for these TKIs-intolerant patients. 11 In addition, the immune system is crucial in controlling cancer progression.12,13 Cancer immunotherapy has revolutionized the management of solid malignancies. The aim of immunotherapy is to selectively target and kill tumor cells by boosting an individual’s immune system. Based on the results of IMbrave 150 trial, atezolizumab combined with bevacizumab has been recommended as the preferred first-line choice for advanced HCC.14,15 This review summarizes the current application of immunotherapy and its combination therapies for the treatment of HCC.
Mechanism of Immunotherapy
Immune checkpoint molecules expressed by tumor cells, T-cells, and antigen-presenting cells (APCs) are pivotal modulators of antitumor T-cell responses. The main inhibitory immune checkpoint molecules, including programmed cell death l (PD-1), programmed cell death ligand 1 (PD-L1) as well as cytotoxic T lymphocyte-associated antigen 4 (CTLA-4), restrain T-cell activity; however, co-stimulatory immune checkpoint proteins, such as CD28, GITR, and OX40, can potentiate T-cell activity. 16
The therapeutic targets of PD-1/PD-L1/ CTLA-4 currently form the mainstay of immunotherapy of HCC. Studies have revealed that the interplay of PD-1 and PD-L1 triggers extensive dephosphorylation of T-cell activating kinases, 17 contributing to the loss of T-cell activity; therefore, PD-1 or PD-L1 inhibitor can restore potent CD8+ T-cell activity. 18 The inhibitory effect of CTLA-4 acts at the immune synapse that controls interaction forces between T-cells and APCs by facilitating the interplay between B7 co-stimulatory ligands and CD28, resulting in increased activation of CD4+ and CD8+ T-cells. 19 Anti-CTLA-4 treatment can activate and increase the richness of both CD4+ and CD8+ T-cells, and reduce the proliferation and expansion of peripheral T-cells in HCC patients. 20 Immune checkpoint inhibitors (ICIs) induce immune cell infiltration into “cold” tumors, followed by conversion to “hot” tumors and increased response rates.
Immunotherapeutic Options
ICI monotherapy
The first ICI-related trial focusing on the treatment of HCC was a phase II clinical trial investigating tremelimumab (anti-CTLA-4 antibody, Table 1) as monotherapy for hepatitis C virus (HCV)-related advanced HCC. The results demonstrated an objective response rate (ORR) of up to 17.6%. 21 Afterwards, an anti-PD-1 antibody, nivolumab, received Food and Drug Administration (FDA) accelerated authorization for treating advanced HCC after sorafenib based on the results of an ORR of 14.3% and an acceptable safety profile observed in the CheckMate 040 trial.22,23 The KEYNOTE-224 trial investigating the effectiveness and safety of an anti-PD-1 antibody, pembrolizumab, as the second-line choice for advanced HCC who failed from sorafenib. 24 The results showed that the median OS and progression-free survival (PFS) of was 12.9 and 4.9 months, respectively. In 2018, pembrolizumab was also approved as a second-line choice for advanced HCC.
ICIs and their targets in HCC.

Abbreviations: CTLA-4, cytotoxic T lymphocyte-associated antigen 4; HCC, hepatocellular carcinoma; ICI, immune checkpoint inhibitor; PD-1, programmed death-1; PD-L1, programmed cell death l ligand 1; VEGF, vascular endothelial growth factor.
Key trials involving immunotherapy for advanced HCC are shown in Table 2. In the CheckMate 459 phase III trial studying nivolumab against sorafenib in patients newly exposed to systemic agents as a first-line treatment, a longer median OS was observed with nivolumab versus sorafenib (16.4 vs 14.7 months; P > .05). Nevertheless, OS improvement in this study failed to reach the predetermined statistical significance criteria. 25 As a result, the indication for nivolumab as a single agent therapy was retracted from the US market. An additional phase III KEYNOTE-240 trial, studying pembrolizumab against placebo after sorafenib as a second-line treatment, revealed a statistically significant prolongation of median OS (13.9 vs 10.6 months; P < .05), whereas, it also failed to reach the predetermined threshold of statistical significance. 26 Potential explanations for the lack of success in these two trials are as follows: (1) diversity in post-progression therapies of included patients; (2) statistical designs; and (3) a limited number of patients benefit from ICIs clinically and ICIs have unique characteristics that encourage antitumor activity. In CheckMate 459, greater than or equal to 31% of patients treated with sorafenib then achieved an ICI-based treatment response, whereas, the proportion of patients receiving a TKI was relatively similar in both groups (36% and 23%). In KEYNOTE-240, the administration of ICIs after disease progression in patients treated with placebo might also have an effect on survival. The dual primary endpoints of OS and PFS possibly influenced the statistically negative results of KEYNOTE-240.
Results from key clinical trials involving immunotherapy for advanced HCC.
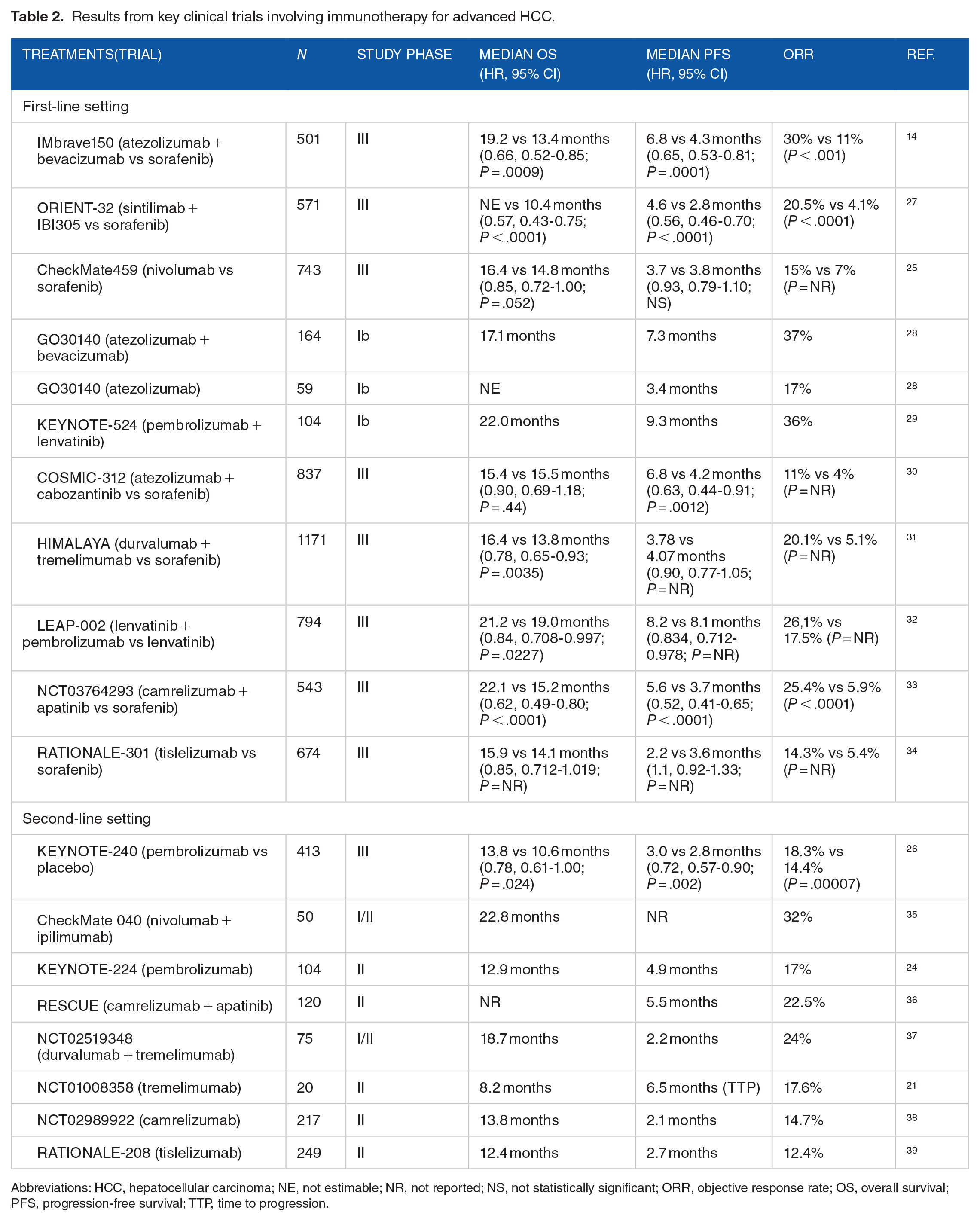
Abbreviations: HCC, hepatocellular carcinoma; NE, not estimable; NR, not reported; NS, not statistically significant; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; TTP, time to progression.
Recently, camrelizumab (anti-PD-1 antibody) revealed obvious antitumor effect in pre-treated patients with unresectable HCC and might represent a new treatment option for these patients. 38 Meanwhile, tislelizumab (anti-PD-1 antibody) revealed sustained responses and was well tolerated in patients with unresectable HCC who had received prior systemic treatment. 39 Based on these results, camrelizumab and tislelizumab have been approved as the second-line treatment for advanced HCC patients who previously failed first-line treatment in China. More recently, the RATIONALE-301 phase III trial exploring tislelizumab against sorafenib in the first-line setting for unresectable HCC patients showed clinically meaningful OS benefit that was non-inferior to sorafenib with a favorable safety profile for advance HCC patients. 34
Other ICIs
In addition to PD-1, PD-L1, and CTLA-4, other immune checkpoint molecules also have potential to stimulate antitumor immune responses, including T-cell immunoglobulin mucin-3 (TIM-3), 40 lymphocyte activation gene 3 (LAG-3), 41 and T-cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain (TIGHT). 42 Studies have shown that the upregulation of PD-L1+ tumor cells as well as PD-1+ CD8+ T-cells in the immune infiltrate is associated with unfavorable outcomes in HCC.43,44 TIM-3, which is most common in less differentiated HCC, 45 is reported to be expressed on CD4+ and CD8+ tumor-infiltrating lymphocytes (TILs) 46 as well as tumor-associated macrophages. 47 TIM-3 can negatively regulate T-cell effector activity, 48 while when expression on Treg cells generates boosted inhibitory activity. In HCC patients, LAG-3 expression is considerably increased on tumor-specific CD4+ as well as CD8+ TILs compared with other immune compartments and provides a negative signal to T-cells.41,49 The combination of PD-L1 inhibitor and other ICIs (TIM-3/LAG-3/CTLA-4) can further enhance its activity than PD-L1 monotherapy. 41 Future ICIs targeting TIM-3, LAG-3, and TIGHT have potential to achieve satisfied prognosis for advanced HCC.
Combination Therapy
Given the recent unsuccessful phase III trials of ICIs monotherapy,25,26 ICIs monotherapy is unable to achieve satisfactory treatment efficacy for advanced HCC. Several trials have demonstrated the synergistic advantage of combining PD-1/PD-L1 inhibitors plus antiangiogenic TKIs or antibodies.29,36,50-54 A number of new combination treatments are currently underway, including PD-1/PD-L1 inhibitors and anti-VEGF antibodies plus/or CTLA-4 inhibitors, which may represent a future focus of immunotherapy for HCC.
ICIs and anti-VEGF therapy
The combination of atezolizumab (anti-PD-L1 antibody) and bevacizumab, the first ICI-based treatment, achieved an improvement on OS as the first-line treatment for advanced HCC.14,55 In the IMbrave150 clinical trial that was carried out in 2020, 501 advanced HCC patients were randomized to receive either atezolizumab and bevacizumab or sorafenib. The study had the co-primary endpoints of OS and PFS, and showed remarkable improvement in OS and PFS at the first interim analysis (median follow-up of 8.6 months). Subsequently, with a median follow-up of 15.6 months, the median OS was 19.2 months in the atezolizumab plus bevacizumab group and 13.4 months in the sorafenib group (P < .001). The combination group also had better median PFS than the sorafenib group (6.8 vs 4.3 months; P < .001). Based on the findings of the IMbrave 150 trial, atezolizumab combined with bevacizumab became the preferred recommendation of first-line choice for advanced HCC.
The ORIENT-32 trial also demonstrated that sintilimab (anti-PD-1 antibody) combined with bevacizumab biosimilar (IBI305) was superior to first-line sorafenib in advanced HCC patients who had not received prior systemic therapy. 27 The median OS was not reached in the combination treatment and was 10.4 months in the sorafenib treatment after a median follow-up of 10 months. However, the COSMIC-312 trial revealed an obvious improvement in PFS with atezolizumab and cabozantinib than sorafenib (6.8 vs 4.2 months; P = .0012), whereas, a nonsignificant improvement in OS (15.4 vs 15.5 months P = .44) was observed. 30
In addition, a phase Ib trial exploring treatment efficacy and safety of pembrolizumab plus lenvatinib as first-line therapy for advanced HCC showed satisfied antitumor activity, with a median OS of 22.0 months and a median PFS of 9.3 months as well as an ORR of 46%. 29 Unfortunately, the LEAP-002 trial studying this combination against lenvatinib alone as the first-line treatment in advanced HCC patients who had not previously undergone systemic treatment did not meet the primary endpoints of OS and PFS with pre-specified statistical significance. 32 By contrast, a phase III trial revealed that camrelizumab plus apatinib as first-line therapy significantly prolonged OS and PFS versus sorafenib with comparable treatment safety. 33 Currently, the combination of ICIs and molecular targeted therapy is also being compared with transarterial chemoembolization (TACE) in the context of intermediate stage HCC, ie, the phase III RENOTACE (NCT04777851) trial as well as the phase III ABC-HCC (NCT04803994) trial.
In second-line therapy, atezolizumab is being incorporated into sorafenib/lenvatinib with the IMbrave251 trial, which will evaluate the effects on disease progression of atezolizumab-bevacizumab therapy (NCT04770896). A plausible explanation for this trial is that both sorafenib and lenvatinib can target tyrosine kinases capable of modulating immune activity and hence have the potential to act synergistically with atezolizumab.51,52
Dual immunotherapy
Combinations of different ICIs may provide additional desired effects. The CheckMate 040 study revealed that the ORR of ipilimumab (anti-CTLA-4 antibody) and nivolumab was 32%. 35 Meanwhile, this study demonstrated the best median OS of 22.8 months was achieved with 3 mg/kg ipilimumab once every 6 weeks plus 1 mg/kg nivolumab once every 2 weeks. This promising result has prompted the FDA to expedite approval of the combination to treat patients with advanced HCC following sorafenib. A CheckMate 9DW phase III trial (NCT04039607) is in progress to compare this combination with sorafenib/lenvatinib as the first-line treatment in advanced HCC patients.
A phase I/II trial using durvalumab (anti-PD-1 antibody) combined with tremelimumab was carried out in advanced HCC patients after sorafenib treatment. The trial yielded the most clinically significant benefit with 300 mg tremelimumab plus 1500 mg durvalumab once every 4 weeks and a median OS of 18.7 months and an ORR of 24% were observed. 37 In addition, in the HIMALAYA trial comparing durvalumab plus tremelimumab with sorafenib as the first-line treatment for advanced HCC, this combination produced superior efficacy compared with sorafenib (16.4 vs 13.8 months; P = .0035). 31
ICIs and locoregional therapy
Locoregional therapies, mainly including radiofrequency ablation (RFA), TACE, and radiotherapy (RT), lead to extensive local necrosis of the tumor and subsequently elicit antitumor immune responses that may be further enhanced by ICIs. 56 Moreover, targeted therapy promotes normalization of vessel formation and inhibits vascular endothelial growth factor (VEGF), which improves the efficacy of TACE/RFA and increases the levels of cytotoxic cells in the tumor microenvironment (TME). 56 Therefore, a synergistic combination with triple therapy may further enhance antitumor immune responses.
Mizukoshi et al 57 revealed that RFA could enhance T-cell infiltration and immune responses and that immunomodulatory agents might improve the immune effect of RFA against HCC. Zhang et al 58 reported that TACE combined with ICI might be an effective treatment approach for advanced HCC. According to a phase I/II trial evaluating tremelimumab combined with RFA/TACE to treat advanced HCC, 26% of patients responded, 89% of patients showed disease control, and OS was 12.3 months. 59 These results confirmed that the combination of tremelimumab with locoregional therapy was a potential new treatment. Meanwhile, a study revealed that RT combined with nivolumab was linked to prolonged PFS and OS in advanced HCC. 60
In addition, when antiangiogenic agents are added to locoregional therapies and ICIs, the efficacy could be greatly enhanced. 56 Some studies have shown the effectiveness and safety of TACE combined with antiangiogenic therapy and immunotherapy in advanced HCC.61-64 In addition, some studies have also demonstrated that TACE plus antiangiogenic therapy plus immunotherapy remarkably improved OS and PFS over antiangiogenic therapy plus immunotherapy in unresectable HCC patients.65,66 Several randomized controlled trials focusing on TACE combined with ICIs and anti-VEGF therapies are underway (Figure 1).

First treatment option, expected survival and ongoing key randomized trials involving immunotherapy for hepatocellular carcinoma. AFP indicates α-fetoprotein; BSC, best supportive care; D-TACE, drug-eluting bead transarterial chemoembolization; PS, performance status; TACE, transarterial chemoembolization.
Predictive Biomarkers of Immunotherapy Efficacy
Immunotherapy has been proven effective for treating advanced HCC in numerous studies. Nevertheless, not all patients will obtain clinical benefits from immunotherapy. This finding indicated that further translational studies should be performed to identify biomarkers predictive of response, which can help to identify patients who will achieve the greatest therapeutic benefit. 67 Identifying those with intrinsic resistance or who do not respond to immunotherapy will enable other treatment methods to be attempted, decrease the number of patients who may not receive clinical survival benefits from immunotherapy, and save a considerable amount of health resources. Some studies68,69 have shown that PD-L1 expression, tumor mutational burden (TMB), high microsatellite instability, TILs, and specific alterations can predict the efficacy of ICIs.
The phase II KEYNOTE-224 study 24 demonstrated that the combined positive score (PD-L1 expression on tumor cells plus immune cells) was related to the response to pembrolizumab and PFS in advanced HCC. However, tumor cells’ PD-L1 expression (cutoff ⩾ 1%) did not predict response to nivolumab or pembrolizumab.22,24,25 As in some patients with ICI-treated cancer, high TMB has been associated with increased ICI response. 53 In addition, the presence of high microsatellite instability is also associated with a higher tumor mutational burden, making ICIs more effective on tumors with that characteristic. Nevertheless, the utility of TMB and microsatellite instability as biomarkers to predict response to ICI is restricted by the low incidence of microsatellite TMB-high HCC70,71 and high instability status.72,73
Studies in several malignant tumors, including melanoma and non-small cell lung cancer (NSCLC), revealed that TILs were associated with OS.74,75 Therefore, TILs might act as a predictive biomarker for ICIs. WNT/β-catenin signaling that is activated has been related to immune rejection in HCC and has been considered a potentially useful biomarker of immunotherapy resistance.76,77 Nevertheless, several studies have questioned the prediction power of these alterations.78,79 Thus, this observation requires further prospective validation in a larger group of samples. Currently, gene expression profiling is gaining resurgence as a method for predicting response to ICIs. Studies indicated that interferon signaling pathways and genes associated with inflammation were abundant in pretreatment HCC patients who responded to ICIs.79,80 According to the present clinical data, a predictive model that involves several factors may offer a better reliable estimate of the probability of responding to immunotherapy compared with a single biomarker. 53
Management of Immune-Related Adverse Events
The skin, gastrointestinal (GI) tract, liver, lungs, and endocrine system are all susceptible to ICI toxicity. 81 Although these events are usually acceptable, they may also pose a life-threatening threat. 82 The most common immune-related adverse events (irAEs) that arise from impaired self-tolerance include fatigue, skin toxicities, and hepatotoxicity. However, a coexisting long-term liver disease, which is usually diagnosed at the cirrhotic stage, leads to the diagnosis of irAEs. Therefore, this diagnosis might be difficult given the mixed effects of organ dysfunction in this condition. 83
Severe irAEs were observed in approximately 10% to 20% of patients treated with PD-1/PD-L1 inhibitors and in approximately 25% of patients treated with CTLA-4 inhibitors.24-26,28 Combining PD-1/PD-L1 and CTLA-4 inhibitors may result in synergistic immunotoxicity, with approximately 50% of advanced HCC patients involved in the CheckMate 040 trial assessing nivolumab plus ipilimumab requiring corticosteroids. 35 The toxicities of PD-1/PD-L1 inhibitors and antiangiogenic agent combinations are additive and mostly nonoverlapping. Severe adverse events occurred in 67% of patients with pembrolizumab combined with lenvatinib. 29
The diagnosis and grading of irAEs was based on the National Cancer Institute Common Toxicity Criteria. 84 Grade 1 events are usually monitored only, while grade 2 toxicities can usually be mitigated with supportive treatments, with some patients able to continue ICI therapy. Grade ⩾ 3 toxicities need treatment interruption or discontinuation and prompt corticosteroid treatment for HCC patients with escalation to immunosuppressants if refractory to corticosteroids.85,86 Given that skin toxicity is associated with an increased chance of benefit from sorafenib, 87 a study showed that irAEs originating from ICIs were associated with more significant clinical benefit. 88 However, continuation of ICI therapy after severe toxicity ought to be based on an individual basis given the ~30% incidence of recurrent irAEs. 89
Other HCC Immunotherapies for HCC
In the context of passive therapy, adoptive cell therapy (ACT) with effector cells is considered immunotherapy. For ACT, the lymphocytes are sensitized and/or expanded in the laboratory and reinfused back into the patient. 90 These cells mainly include natural killer (NK) cells, lymphokine-activated killer (LAK) cells, TILs, cytokine-induced killer (CIK) cells, and redirected peripheral blood T-cells. Adjuvant therapy with LAK cells delayed HCC recurrences but did not lengthen life expectancy. 91 According to a multicenter phase III study conducted in 2015, adjuvant immunotherapy with CIK cells improved the OS and PFS of HCC patients undergoing surgery or ablation for curative purposes. 92 Patients with HCC were also shown to be amenable to adjuvant TILs based on a phase I trial. 93 Allogeneic NK cells are being studied in phase II clinical trials to treat advanced HCC patients who have a high incidence of recurrence following resection (NCT02008929) and patients undergoing TACE (NCT02854839).
Using chimeric antigen receptor T-cell (CAR-T) to treat hematological malignancies has proved to be an effective strategy,94-96 whereas, the application of CAR-T in treating HCC is still in development. Some preclinical studies have demonstrated the clinical potential of glypican-3 (GPC3)-CAR T-cells to treat HCC.97-99 Currently, a clinical trial is being carried out to evaluate the effectiveness and safety of GPC3-CAR T-cells (NCT04121273). An ongoing clinical trial investigating α-fetoprotein (AFP)-specific T-cell receptor T-cells for the treatment of HCC (NCT03132792) has demonstrated objective remissions. In addition, TP53 hotspot mutations commonly observed in HCC 100 as well as hepatitis B virus (HBV) antigens (NCT03899415) may also be targets for T-cell receptor-engineered T-cells.
To generate tumor-specific immunity, therapeutic vaccines are used against cancer. HCC peptide vaccines typically target tumor-associated antigens (TAAs), such as telomerase, GPC-3, and AFP. 101 However, there has been little progress on approaches targeting telomerase and GPC-3,102,103 and none are being developed into drugs. In HCC, many clinical trials of vaccines based on tumor lysates have failed to achieve consistent results. 104 Furthermore, clinical studies of vaccines using neoantigens are lacking, and the presence of mutations is not clearly related to the immune response. 83
Oncolytic virus therapies using genetically engineered or naturally occurring deficient viruses that can only replicate and kill cancer cells have the potential to be the next major advancement in cancer treatment after immunotherapy using ICIs. 105 Intralesional injection of the oncolytic virus JX-594 (pexastimogene devacirepvec, Pexa-Vec) into primary or metastatic liver tumors was generally well tolerated in a phase I trial. 106 Unfortunately, JX-594 did not significantly improve OS as second-line monotherapy in advanced HCC in the phase IIb trial (TRAVERSE). 107 Moreover, the PHOCUS phase III trial comparing JX-594 plus sorafenib as first-line treatment versus sorafenib monotherapy in advanced HCC was prematurely terminated after a planned interim futility analysis revealed no benefit. 108 Nevertheless, clinical trials using different oncolytic viruses are worth exploring. In addition, as demonstrated by Zou et al, 109 adenoviral vectors encoding antibody fragments were feasible for using to treat HCC, offering a new option for HCC immunotherapy.
Future Perspectives
Immunotherapy has started a new era to treat HCC. 2 In the future, there is potential for immunotherapy to enhance locoregional and radical treatments for HCC, and neoadjuvant therapy for HCC are likely to achieve unprecedented therapeutic outcomes. In addition, novel immunotherapies, including new target antibodies, bispecific antibodies, combination regimens, engineered cytokines, adoptive T-cell therapy, tumor vaccines, and oncolytic viruses, might be available to treat all stages of HCC in the near future. Moreover, studies must be carried out to determine whether new ICI-based combination treatments can provide advanced HCC patients with clinical survival benefits following resistance to atezolizumab and bevacizumab combination therapy in the first-line setting. Few studies have associated immune classes, 76 gene signatures, 80 and even specific mutations77,110 of HCC with therapy response or primary resistance. However, the initial nature of these studies means that the creation of new regimens will be developed empirically. All of these issues highlight the importance of identifying the molecular mechanisms that determine susceptibility and resistance to individual agents or combinations to develop valuable biomarkers that can assist in advancing personalized treatments. Based on the current reported evidence and ongoing trials, immunotherapy especially combination with other therapies has potential to act as a significant approach for the treatment of HCC.
Conclusion
The combination of atezolizumab and bevacizumab takes the treatment of advanced HCC into an era of immunotherapy. Immunotherapy with synergistic combination is likely to be the key in exploring effective immunotherapeutic breakthroughs in the future. Meanwhile, one of the focuses for the future work could be an exploration of prognostic biomarkers that could accurately predict survival and stratify beneficial patients from immunotherapy.
Footnotes
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (81901847), the Natural Science Foundation of Jiangsu Province (BK20190177), the Key R&D Program (Social Development) Project of Jiangsu Province (BE2021648).
Author Contributions
Huang JT, Zhang S, and Yang YH contributed equally to drafting the; Zhang ZC, Jiang N, Li WC, and Shen J polished up the vocabulary and the grammar; Zhong BY and Zhu XL contributed equally to the study concept and design; all authors contributed to reviewing and criticizing revision of the and approving the final version of the .
