Abstract
Background:
The time interval between palliative thoracic radiotherapy and bevacizumab treatment strongly influences the frequency of adverse events (AEs) when both are concurrently applied to patients with advanced lung cancer. Herein, we aimed to elucidate the optimal time interval between the treatments in these patients.
Methods:
The medical records of patients with stage IV nonsquamous non–small-cell lung cancer (NSCLC) without epidermal growth factor receptor and anaplastic lymphoma kinase alteration who underwent palliative thoracic radiotherapy and bevacizumab treatment from January 2008 to January 2020 were collected and analyzed. Patients were divided into 2 groups based on the time interval between treatments: <3 weeks (⩽3W group) and >3 weeks (>3W group). The progression-free survival (PFS) and overall survival (OS) for the time intervals were evaluated using the Kaplan-Meier method and Cox proportional hazard models. Adverse events were assessed by the fifth version of the Common Terminology Criteria for Adverse Events.
Results:
In total, 72 patients with stage IV NSCLC (⩽3W group, 37 patients; >3W group, 35 patients) who concurrently or sequentially received palliative thoracic radiotherapy and bevacizumab treatment were included in this study. In the >3W and ⩽3W groups, the median PFS (8 vs 6 months, respectively) and OS (15 vs 12 months, respectively) differed significantly. Multivariate analyses findings revealed significantly shorter OS in the latter group. In addition, the frequency of most AEs was marginally higher in the latter group (P > .05).
Conclusions:
The time interval between palliative thoracic radiotherapy and bevacizumab treatment that offers optimal safety is >3 weeks.
Keywords
Introduction
Vascular endothelial growth factor (VEGF) is a key factor in tumor angiogenesis; therefore, it is an important therapeutic target for antiangiogenesis therapies. 1 Bevacizumab, an anti-VEGF recombinant humanized monoclonal antibody, has been approved in combination with chemotherapy for the treatment of advanced non–small-cell lung cancer (NSCLC) by the US Food and Drug Administration. 2 In a phase III clinical trial (Eastern Cooperative Oncology Group [ECOG] 4599), patients with nonsquamous NSCLC who received paclitaxel/carboplatin with bevacizumab showed a significantly longer progression-free survival (PFS, 6.2 vs 4.5 months; hazard ratio [HR], 0.66; 95% confidence interval [CI] 0.57-0.77; P < .001) and overall survival (OS, 12.3 vs 10.3 months; HR, 0.79; 95% CI, 0.67-0.92; P = .003) than those who received chemotherapy alone. 3
Vascular endothelial growth factor is the key mediator of angiogenesis, and VEGF-targeted therapy has shown synergistic effects with radiotherapy in vitro, wherein VEGF-targeted therapy overcomes VEGF-induced protection of endothelial cell resistance to radiotherapy.4,5 In addition, in vitro and in vivo experiments have confirmed the synergistic antitumor effect of bevacizumab + radiotherapy. 6 Therefore, bevacizumab should be incorporated with radiotherapy for the treatment of advanced NSCLC. However, several clinical trials have reported that concurrent or sequential use of radiotherapy and bevacizumab treatment may lead to adverse events (AEs).7 -9 In actual clinical practice, concurrent or sequential use of radiotherapy and bevacizumab treatment may be needed in some cases. For instance, patients with advanced nonsquamous NSCLC with superior vena cava syndrome and obstructive pneumonia have been treated with thoracic radiotherapy + bevacizumab in an attempt to reverse the airway obstruction and contraction of the superior vena cava lumen caused by the compressive effect of a tumor mass.10,11 Therefore, determining the optimal time interval between radiotherapy and bevacizumab remains a challenge.
Currently, there is no standard guideline or protocol that suggests the optimal time interval between thoracic radiotherapy and bevacizumab treatment. Therefore, this study aimed to determine this duration for the combined treatment of radiotherapy and bevacizumab.
Materials and Methods
Patients and study design
The medical records of 72 patients with stage IV NSCLC, who were recruited from West China Hospital and underwent palliative thoracic radiotherapy + bevacizumab treatment from January 2008 to January 2020, were collected and analyzed. The inclusion criteria were as follows: (1) cases of stage IV nonsquamous NSCLC without EGFR and ALK alterations in which combined treatment of palliative thoracic radiotherapy and bevacizumab treatment was administered and (2) cases with complete clinical and follow-up data. Patients were excluded based on the following criteria: (1) patients with a history of other types and stages of cancer; (2) patients with potentially fatal diseases, hemoptysis, arteriovenous thrombosis, hypertension, pulmonary disfunction, and tracheoesophageal fistula (TEF); and (3) patients with radical radiotherapy. This retrospective study was approved by the Ethics Committee of West China Hospital, Sichuan University.
Treatment
The National Comprehensive Cancer Network guidelines for palliative thoracic radiotherapy in patients with NSCLC recommend individualizing the principle of dose and fraction of palliative thoracic radiotherapy according to signs, symptoms, treatment goals, subsequent treatment planning, and ECOG scores. As shorter radiotherapy courses offer symptomatic relief comparable with longer radiotherapy courses, shorter courses are preferred in cases with shorter life expectancy or poor ECOG scores. However, there is a higher potential requirement for retreatment. To resolve thoracic symptoms, thoracic radiotherapy with lower doses and shorter courses (⩽30 Gy in 10 fractions) offers marginally improved survival and symptomatic relief, particularly in patients having poor ECOG scores. Therefore, all patients with stage IV NSCLC were divided into those who received ⩽30 Gy/10 fractions and >30 Gy/10 fractions. 12 Patients received bevacizumab (7.5 mg/kg) before or after undergoing thoracic radiotherapy.13,14 This was combined with paclitaxel + carboplatin or pemetrexed + carboplatin or docetaxel. The time interval between thoracic radiotherapy and bevacizumab administration was defined as the time between the last dose of thoracic radiotherapy and the first dose of bevacizumab (R-B group) or vice versa (B-R group). Based on this time interval, all patients were divided into a group with time interval ⩽3 weeks (⩽3W group) and a group with time interval >3 weeks (>3W group). In addition, the patients’ clinicopathological characteristics, which included age, sex, smoking history, performance status, and histology, were obtained.
All the patients in this study were PD-L1-negative. None of the patients received durvalumab, atezolizumab, nivolumab, or pembrolizumab because the Chinese Food and Drug Administration had not approved these immunotherapy drugs for PD-L1–negative NSCLC at the time of this study.
Follow-up
The OS was defined as the time from the initial diagnosis until the time of cancer-related death or until the last visit. It was recorded during a follow-up clinical visit or by telephone. The patients’ AEs were recorded based on follow-up clinical visits or follow-up phone calls. Patients were monitored for toxicity every week during the radiotherapy course, and then, the effectiveness was evaluated every 6 weeks. Analysis of toxicity was mainly based on the clinician’s judgment. Pulmonary toxicity and TEF were assessed for 6 months after the completion of radiotherapy within the radiotherapy field. In addition, hypertension, arteriovenous thrombosis, and hemoptysis were assessed for 6 months after treatment with a combination of palliative thoracic radiotherapy and bevacizumab. The AEs were assessed following the fifth edition of the Common Terminology Criteria for Adverse Events. All patients were followed up until death or January 2020.
Statistical analysis
Statistical analyses were performed using SPSS 22.0 (SPSS, Chicago, Illinois). Kaplan-Meier analysis was used to assess the clinical outcomes, and statistical significance was determined using the log-rank test. Cox proportional hazards regression models were used to study the relationship between PFS/OS and the combination of palliative thoracic radiotherapy and bevacizumab treatment in patients with nonsquamous NSCLC. The estimates derived from the Cox regressions were presented as HRs with 95% CI. In addition, to identify significant independent prognostic factors, multivariate analyses were used. Significance was defined as P values <.05 (2-sided).
Results
A total of 72 patients and 85 treatment-related AEs were included in the analyses. Table 1 summarizes the patients’ clinicopathological characteristics. The included patients were 46 men (63.9%) and 26 women (36.1%) aged 22 to 75 years (mean age, 54.9 years). Performance status assessment showed that most cases (n = 64; 88.9%) were grade 0-1 according to the ECOG scoring system. In addition, 59 (81.9%) cases were confirmed adenocarcinomas and 13 (18.1%) were large-cell carcinomas. The median follow-up time was 36 months (range, 22-50 months). According to the time interval between palliative thoracic radiotherapy and bevacizumab treatment, there were 37 (51.4%) patients in the ⩽3W group and 35 patients (48.6%) in the >3W group. Among the patients who were treated sequentially, there were 36 patients in the B-R group (median time interval, 8.99 weeks; range, 0.4-67.6 weeks) and 36 patients in the R-B group (median time interval, 13.16 weeks; range, 1.0-108.1 weeks). Although there were no statistical differences between the B-R group and the R-B group, most treatment-related high-grade AEs (grade 3 or 4) were observed in the B-R group. The characteristics of the treatments are summarized in Table 2. The fractionation schemes were as follows: ⩽30 Gy in 10 fractions (n = 56) and >30 Gy in 10 fractions (n = 16). The median dose delivered was 32 Gy. The median clinical target volume was 135 mL (70-840 mL), and the median planning target volume was 402 mL (50-1200 mL). In this study, 64 patients (88.9%) received bevacizumab-based chemotherapy as first-line chemotherapy. All patients received bevacizumab (7.5 mg/kg) before or after undergoing thoracic radiotherapy. It was combined with paclitaxel + carboplatin in 28 patients (38.9%), with pemetrexed + carboplatin in 36 patients (50.0%), and with docetaxel in 8 patients (11.1%).
Patients clinicopathological characteristics (n = 72).

Abbreviations: B-R, patients receiving radiotherapy after the last administration of bevacizumab; ECOG, Eastern Cooperative Oncology Group; R-B, patients receiving bevacizumab after the last irradiation; W, weeks.
Characteristics of the treatments.
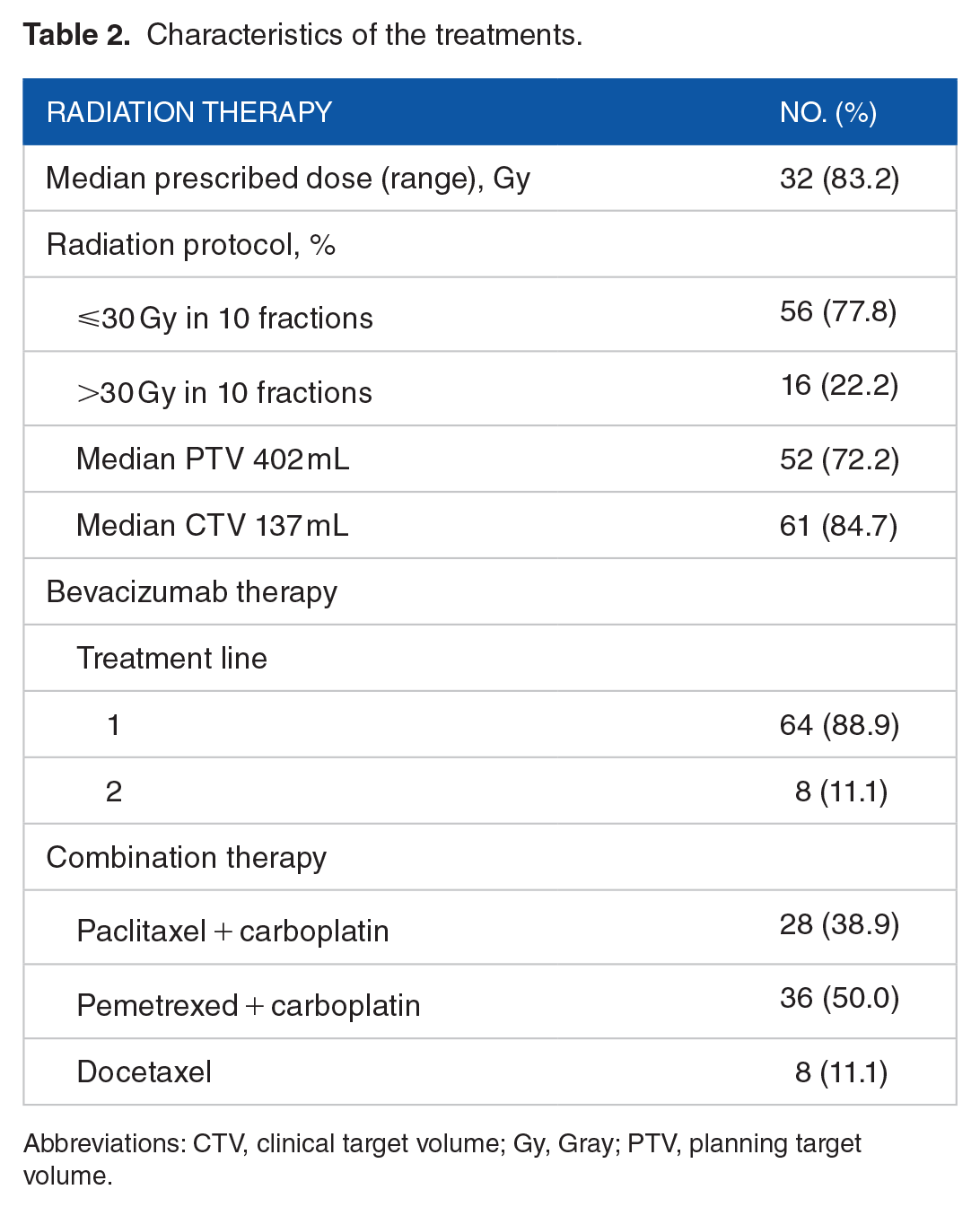
Abbreviations: CTV, clinical target volume; Gy, Gray; PTV, planning target volume.
As shown in Figure 1, the median PFS was 7 months (95% CI, 3-12 months) in all patients. The PFS was significantly shorter in the ⩽3W group (median PFS, 6 months; 95% CI, 4-8 months) than in the >3W group (median PFS, 8 months; 95% CI, 6-13 months; P < .0001; Figure 1A). As shown in Figure 1B, the median OS of the whole population was 14.5 months (95% CI, 8.2-20.8 months). The OS was significantly shorter in the ⩽3W group (median OS, 12 months; 95% CI, 8-16 months) than in the >3W group (median OS, 15 months; 95% CI, 8-22 months; P < .0001; Figure 1B).

Progression-free survival (PFS) and overall survival (OS) of patients with nonsquamous NSCLC who received palliative thoracic radiotherapy and bevacizumab treatment with a time interval of >3 weeks or ⩽3 weeks: (A) the median PFS was significantly longer with a time interval of >3 weeks than of ⩽3 weeks (8 vs 6 months; P < .0001); and (B) the median OS was significantly longer with a time interval of >3 weeks than of ⩽3 weeks (15 vs 12 months; P < .0001).
Univariate and multivariate analyses
First, univariate Cox regression analyses were performed to examine the association between each potential confounder and the clinical outcomes. The potential confounders for outcomes included age, sex, smoking index, performance status, histology, chemotherapy regimens, and the time interval between palliative thoracic radiotherapy and bevacizumab treatment. Second, separate univariate Cox regression models were fitted to evaluate the influence of each covariate on the strength of the association between the combination of palliative thoracic radiotherapy and bevacizumab treatment and the clinical outcomes in patients with nonsquamous NSCLC.
The univariate analysis indicated that the time interval of ⩽3 weeks was statistically significantly associated with increased mortality risk in patients who received a combination of palliative thoracic radiotherapy with bevacizumab (HR, 0.67; 95% CI, 0.46-1.01; P = .0054; Table 3). In addition, a multivariate analysis of OS revealed that the 12-month mortality risk of patients with NSCLC being administered a combination of palliative thoracic radiotherapy with bevacizumab was higher than that of patients not being administered a combination therapy by 20% (HR, 0.80; 95% CI, 0.50-1.28; P = .0001). In addition, univariate and multivariate analyses revealed no statistically significant association of age, sex, smoking history, performance status, histology, and different chemotherapy regimens with increased mortality risk; however, a statistically significant association of time interval with increased mortality risk was identified.
Univariate analysis of OS in patients with NSCLC administered combined treatment of palliative thoracic radiotherapy and bevacizumab.

Abbreviations: 95% CI, 95% confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; NSCLC, non–small-cell lung cancer; OS, overall survival; W, weeks.
Treatment-related toxicities are mentioned in Table 4. The most common AEs were hemoptysis (25 patients, 34.7%), pulmonary toxicity (19 patients, 26.4%), arteriovenous thrombosis (15 patients, 20.8%), TEF (9 patients, 12.5%), and hypertension (4 patients, 5.6%). Overall, the most common toxicities among 37 patients in the ⩽3W group included hemoptysis in 13 patients (35.1%), pulmonary toxicity in 11 patients (29.7%), TEF in 7 patients (18.9%), arteriovenous thrombosis in 4 patients (10.8%), and hypertension in 2 patients (5.5%). Most bevacizumab and radiotherapy combination treatment-related AEs (including high-grade AEs [grade 3 or 4]) were observed in the ⩽3W group, such as hemoptysis and TEF. In addition, although a trend of a higher mortality risk was noted in patients with NSCLC in the ⩽3W group (Figure 2), no AE was individually significantly associated with increased mortality risk during this time period (Figure 3).
Treatment toxicity.

Abbreviations: AE, adverse events; RT, radiotherapy.

Treatment-related AEs in the ⩽3W group and the >3W group. Forest map shows more treatment-related AEs among every subgroup in the ⩽3W group than in the >3W group.

Treatment-related AEs in the ⩽3W group and the >3W group. Forest map shows more treatment-related AEs in the ⩽3W group than in the >3W group.
Discussion
Due to the development of new radiotherapy technologies, precise, high-dose irradiation of a gross tumor with minimal irradiation of normal tissues has significantly reduced the adverse reactions. 15 However, in most situations, physicians still recommend exercising caution or avoiding a combination of radiotherapy and bevacizumab. This is because several clinical trials have reported AEs in patients with lung cancer who were treated with a combination of thoracic radiotherapy and bevacizumab administered concurrently.7,8,16 However, the abovementioned clinical trials did not suggest an optimal time interval between thoracic radiotherapy and bevacizumab treatment for patients with NSCLC. In cases wherein patients’ superior vena cava syndrome and obstructive pneumonia were relieved by thoracic radiotherapy, the subsequent systemic treatments may involve bevacizumab administration. Therefore, the present study was aimed at elucidating the impact of the time interval between bevacizumab and palliative thoracic radiotherapy on the prognosis of patients with stage IV nonsquamous NSCLC. Kaplan-Meier and multivariate analyses indicated that a time interval of ⩽3 weeks significantly decreased the PFS and OS in patients with nonsquamous NSCLC who were administered a combination of bevacizumab and palliative thoracic radiotherapy. In addition, this duration was found to be associated with more AEs, even high-grade AEs (grade 3 or 4), such as hemoptysis and TEF, in patients with stage IV NSCLC who were concurrently or sequentially treated with a combination of palliative thoracic radiotherapy and bevacizumab.
Bevacizumab inhibits the VEGF signaling pathway, which is essential for wound healing of normal tissue injury following radiotherapy, 17 indicating that caution should be exercised when treating patients with bevacizumab and radiotherapy, both sequentially and concurrently. Therefore, the methods to maximize the safety and efficiency of bevacizumab when administered in combination with radiotherapy require investigation. Optimal scheduling of the combined therapy of bevacizumab with radiotherapy requires knowledge of the optimal time interval required for the repair of blood vessels that are injured by radiotherapy. 18 Pharmacokinetic studies have reported the mean half-life of bevacizumab as approximately 20 days (range, 11-50 days). 19 A phase III trial suggested a washout period of ⩾3 weeks between starting bevacizumab therapy and the last dose of radiotherapy in patients with advanced cervical cancer to remove any potential adverse effects. 20 Bevacizumab has a delayed creatinine clearance rate and long half-life decay; sex and weight were the covariates that were mainly responsible for inter-individual factors in bevacizumab clearance. 21 According to the pharmacokinetic data analysis, the bevacizumab clearance rate was 0.207 and 0.262 L/d in women and men with cancer, respectively, and the median half-life of bevacizumab in the body was 20.6 and 19.5 days, respectively. 21 In addition, tumor burden impacted bevacizumab clearance, with a 7% faster drug clearance noted in patients with cancer having a large tumor burden. The pharmacokinetic aspects of bevacizumab were comparable between patients with colon cancer who received long-term bevacizumab adjuvant therapy for 1 year and those who received short-term bevacizumab therapy. 22 Considering the long elimination half-life of bevacizumab, the estimated median serum drug concentrations at 3 and 6 months since the last dose were 6.14 and 0.23 g/mL, respectively. 22 The plasma half-life of bevacizumab was comparable in Chinese and white patients with cancer. 23 Therefore, in this study, based on the half-life of bevacizumab and the repair of the injured blood vessels, the optimal time interval was set to 3 weeks. Then, further AEs were observed in the ⩽3W group, which included hemoptysis (even grade 3 or 4 hemoptysis) and TEF. Subgroup analysis included sex, age, ECOG scores, smoking history, and histology. Most treatment-related AEs were in the ⩽3W group. More serious AEs may lead to a situation wherein patients cannot tolerate further antitumor treatments, consequently affecting their PFS and OS.
Notably, TEF is an infrequent, life-threatening treatment complication from bevacizumab and thoracic radiotherapy treatment in patients with NSCLC. Rapid regression of the tumor caused by radiotherapy and damage to the tracheal fibrous membrane caused by bevacizumab may lead to TEF. Although the underlying mechanism of bevacizumab action on TEF remains unclear, it is hypothesized that the damaged angiogenesis by bevacizumab delays the wound healing process.24,25 The predisposing factors for TEF reported in the literature are tracheoesophageal injury due to instrumentation, tumor position, and thoracic radiotherapy history. 26 The time interval between TEF and radiotherapy in NSCLC is different in previous TEF-based reports, with the shortest time interval of 2 months after the completion of radiotherapy and the longest time interval of 21 months.7,27 In agreement with previous research, in this study, most TEF cases were in the ⩽3W group. These data indicate that the incidence of TEF was closely associated with the interval between thoracic radiotherapy and bevacizumab treatment. This could be due to bevacizumab’s special role in inhibiting angiogenesis and consequently delaying mucosa repair. In addition, no difference in baseline history of chronic mucosal inflammation (eg, tracheitis or esophagitis) was observed between the ⩽3W and >3W groups. Therefore, altered scheduling to ⩾3 weeks could help avoid potential toxicities associated with bevacizumab treatment. In addition, there were more TEF cases in the B-R group. This may be because the median time interval was longer in the R-B group (median, 13.16 weeks) than in the B-R group (median, 8.99 weeks), which may avoid potential toxicities associated with bevacizumab.
This study has several limitations. First, the retrospective nature of this study could not fully exclude selection bias. Second, all data were collected from a single institution. Therefore, the results should be interpreted by oncologists with caution. Third, only some types of treatment-related AEs (eg, hemoptysis, arteriovenous thrombosis, hypertension, pulmonary toxicity, and TEF) were observed. Finally, the sample size was very small, which affects the generalizability of our findings. Therefore, further multicenter studies aimed at identifying the optimal time interval between thoracic radiotherapy and bevacizumab treatment in patients with stage IV NSCLC are required to confirm our findings. To address these concerns, we will design a prospective, more rigorous, and multicenter clinical trial in the near future.
Conclusions
In summary, this study suggests that a time interval of >3 weeks is relatively safe for patients with stage IV nonsquamous NSCLC undergoing combined treatment with thoracic radiotherapy and bevacizumab.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
JZ and JJL designed the study. WGX, MY, YX and YYL conducted the experiments and analyzed the data. WGX wrote the manuscript. All authors contributed to the article and approved the submitted version.
Ethical Approval
This retrospective study was in compliance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Human Investigation Committee of West China Hospital, Sichuan University approved this study (no. 2022445).
