Abstract
Background:
EGFR tyrosine kinase inhibitor (TKI) is recommended as the first-line therapy for patients with EGFR-mutant metastatic non-small cell lung cancer (NSCLC). Yet, resistance often occurs in 1 year after therapy and most progressions occur at the initial sites of disease. Addition of local therapy to the first-line TKI therapy may delay the progression and provide survival benefit to the patients.
Methods:
From 2010 to 2017, metastatic NSCLC patients with EGFR activating mutations who received first-line TKI and relatively radical local therapy (RRLT) were reviewed. RRLT was defined as local curative therapy to the main site or any intensity of local therapy to all sites of disease. The Kaplan-Meier method and log-rank test were used for survival estimation and comparison.
Results:
A total of 45 patients were included in this retrospective study with a median follow-up of 48.0 months. The median progression-free survival (PFS) and overall survival (OS) was 17.0 months (95% confidence interval [CI]: 14.6-19.3) and 55.0 months (95% CI: 49.3-60.6), respectively. Univariate analysis indicated that age ⩽ 60 years (P = .019), first-line TKI duration ⩾ 10 months (P = .028), and accumulated TKI duration ⩾ 20 months (P = .016) were significantly associated with favorable OS. Among the 36 patients who progressed during the follow-up, 55.8% of the progressions occurred at the new sites. RRLT combined with TKI did not show any severe toxicity to the patients.
Conclusions:
Combined application of RRLT and first-line TKI may improve the survival and alter the pattern of failure for metastatic NSCLC patients with EGFR activating mutations.
Keywords
Introduction
Lung cancer is the leading cause of cancer death, with 1.76 million deaths worldwide in 2018. 1 Non-small cell lung cancer (NSCLC) accounts for approximately 85% of lung cancers and more than half of the patients are diagnosed with metastatic disease. 2 According to the current National Comprehensive Cancer Network (NCCN) guideline, metastatic NSCLC patients are considered incurable and systemic strategies are recommended as the first-line therapy. 3 Local therapies, although recommended for well-selected patients with limited metastases, are mostly utilized as palliative therapy for the purpose of symptom relief or salvage treatment in clinical practice.
Traditional platinum-based doublet chemotherapy for metastatic NSCLC showed unsatisfactory results, with the median progression-free survival (PFS) of 4.0 to 6.0 months and overall survival (OS) of 8.0 to 11.0 months.4,5 In recent years, EGFR tyrosine kinase inhibitor (TKI) has emerged as a promising regimen for EGFR-mutant metastatic NSCLC patients. Since the approval by China Food and Drug Administration in 2011, the first-generation TKIs have remained as the first-line option for these patients. The median PFS and OS of first-generation EGFR TKI reached to 8.0 to 13.1 months and 19.3 to 30.9 months, respectively, much longer than those of traditional chemotherapy.6-8 Despite the significant improvement in survivals, most patients developed inevitably acquired resistance after 9 to 14 months of TKI treatment. 9 Pattern of failure (POF) studies suggested that progressions of TKI-treated NSCLC patients occurred predominantly in the initial site of disease, which accounted for 67% to 82% of the POF.10,11 The recognition of this clinical scenario has spurred the development of algorithms that combine local therapy with first-line TKI, which may allow patients to continue with the TKI treatment for an extended period and delay the need to switch to other systemic therapies.
Previous studies have revealed the benefits of local therapy and EGFR TKI for EGFR-unselected metastatic NSCLC patients.12,13 A multicenter phase II randomized trial indicated that the addition of local consolidation therapy significantly prolonged the PFS in oligometastatic NSCLC patients (⩽ 3 sites) when compared with those who received systemic therapy alone. 14 Yet, the results were insufficient for EGFR-mutant patients since only 12% (6/49) of the patients in the study harbored EGFR activating mutations. Data regarding the effects of local therapy combined with first-line TKI for metastatic NSCLC patients with EGFR activating mutations remain sparse.15-19 Most of them were retrospective studies with small sample size and various treatment intensities of local therapy. Since the first-line TKI already achieved promising survivals in EGFR-mutant patients, it is possible that more radical local therapies may add additional survival benefits to the population.
In this study, we first put forward a concept of “relatively radical local therapy (RRLT),” and performed a retrospective analysis to investigate the effects of RRLT combined with standard first-line EGFR TKI for EGFR-mutant metastatic NSCLC patients.
Methods
Definition of RRLT
Traditional local treatment for metastatic NSCLC is mostly palliative therapy for the purpose of symptom relief with short expected-survival. In this study, we put forward a novel definition of RRLT to refer an intermediate status between the radical and palliative local therapy. RRLT takes 2 important aspects into consideration: the treatment intensity and treatment extent. In synchronous metastatic patients without previous treatment, local therapies to the primary site with curative intent, or to all sites of disease with any intensity were considered RRLT. In metachronous metastatic patients regardless of previous therapy, local therapies to at least one progression site with curative intent, or to all progression sites with any intensity were also considered RRLT. R0 resection or radiotherapy with biologically equivalent dose (α/β = 10, BED10) ⩾ 53 Gy was considered local therapy with curative intent.20,21
Patients and interventions
We retrospectively reviewed patients who were diagnosed with synchronous or metachronous metastatic NSCLC from January 2010 to December 2017 at the Cancer Hospital, the Chinese Academy of Medical Sciences, Beijing, China. Patients were checked for eligibility according to the criteria as follows: (1) 18 years of age or older; (2) Eastern Cooperative Oncology Group performance status (ECOG PS) score ⩽ 2; (3) pathologically confirmed NSCLC with EGFR activating mutations; (4) synchronous or metachronous metastatic diseases according to the seventh edition of the American Joint Committee on Cancer staging system; (5) received RRLT and TKI as the first-line treatment; (6) no TKI treatment before the diagnosis of metastatic disease; (7) without progression within the first 3 months of TKI treatment or during RRLT; (8) without extensive pleural metastases, extensive meningeal metastases, or other tumors. The TKI prescribed in this study included Gefitinib 250 mg QD, Erlotinib 150 mg QD, and Icotinib 125 mg TID. The sequence of TKI administration and RRLT was not limited in this study. TKI and RRLT were regarded as concurrent when the interval between them was less than 1 month.
Baseline characteristics were collected from electronic records, including age at diagnosis, sex, ECOG PS, smoking status, tumor histology, initial TNM stage, EGFR mutations, metastatic sites, number of metastases, and treatment records. Tumor stage was assessed by systemic imaging (contrast-enhanced computed tomography, bone scan, or positron-emission tomography) and brain magnetic resonance imaging. Oligometastatic disease was defined as 1 to 5 lesions in one to multiple organs excluding the primary tumor. Tumor response was assessed every 3 months by experienced oncologists according to Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1). POFs were recorded based on available clinical or imaging reports of progression (local or distant). Toxicity was graded using the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. The study was approved by the Research Ethics Committee of Cancer Hospital, Chinese Academy of Medical Sciences (Approval No. 18-076/1654).
Statistical analysis
Descriptive statistics were used to summarize the characteristics of patient, tumor, and treatment. PFS was defined as the duration between the commencement of first-line treatment and the occurrence of first progression, last follow-up, or death of any cause. OS was defined as the time elapsing from the commencement of first-line treatment to the last follow-up or death of any cause. The Kaplan-Meier method was used for survival estimation and the log-rank test was used for univariate analyses of survival. Cox proportional hazard regression was used for the hazard ratio calculation. All analyses were performed using SPSS 24.0 and P < .05 was considered statistically significant.
Results
Patients and characteristics
The records from 1352 synchronous or metachronous metastatic NSCLC patients who received TKI therapy at our institution were reviewed and 45 patients were ultimately enrolled according to the criteria (Figure 1). Baseline demographics and clinical characteristics of these patients are listed in Table 1. The median age of these 45 patients was 58 years (range: 36-80). Among them, 18 (40%) had synchronous metastatic NSCLC and 27 (60%) had metachronous diseases. All patients were diagnosed with adenocarcinoma by tissue biopsy. Twenty-five (55.6%) patients harbored exon 19 deletion and 20 (44.4%) patients had exon 21 L858R mutation. The most frequent site of metastases was brain (68.9%), followed by bone (28.9%) and lung/pleural nodes (26.6%). Thirty-two (71.1%) patients had oligometastatic disease. All patients received first-line TKI treatment for more than 6 months, with a median duration of 13.7 months (range: 6.2-44.6). The median interval between the commencement of TKI and local therapy was 0.7 months (range: 0-11.4).

Flowchart of patient enrollment. ALK indicates anaplastic lymphoma kinase; NSCLC, non-small cell lung cancer; RRLT, relatively radical local therapy; TKI, tyrosine kinase inhibitor.
Patient demographics and clinical characteristics.

Abbreviations: ECOG PS, Eastern Cooperative Oncology Group performance status; TKI, tyrosine kinase inhibitor.
Range.
The characteristics of RRLT for these patients are listed in Table 2. Most of these patients received radiotherapy as local treatment approach, including 30 (66.7%) patients with radiotherapy and 4 (8.9%) patients with both radiotherapy and surgery. Eleven (24.4%) patients received surgery alone as local therapy. For the 18 patients with synchronous disease, 14 (77.8%) of them received RRLT to the primary site, and the median BED10 of radiotherapy was 67.5 Gy (range: 58.5-72.7). The rest 4 synchronous metastatic patients received RRLT to all sites of disease. For the 27 patients with metachronous disease, 23 (85.2%) of them received RRLT to all progression sites. The rest 4 patients received RRLT with curative intent to at least one progression site and the median BED10 of radiotherapy was 134.3 Gy (range: 80.52-144.42). A total of 208 lesions were identified in 45 patients and 139 (66.8%) of them received local treatment, including 95 (68.3%) lesions with curative intent and 44 (31.7%) lesions with palliative intent. The specific methods of RRLT for different lesions are listed in Table S1.
Characteristics of RRLT.

Abbreviations: BED10, biologically equivalent dose; Gy, gray; RRLT, relatively radical local therapy; RT, radiation therapy; S, surgery.
α/β = 10.
Survival and risk factor analysis
At a median follow-up period of 48.0 months (range: 33.1-62.9), 17 (37.8%) patients died. Figure 2 depicted the survival curves and corresponding 95% confidence interval (CI) for PFS and OS. The median PFS was 17.0 months (95% CI: 14.6-19.3), with 1-, 2-, and 3-year PFS rates of 64.1%, 31.9%, and 6.9%, respectively. The median OS was 55.0 months (95% CI: 49.3-60.6), with 1-, 3-, and 5-year OS rates of 95.4%, 64.4%, and 33.5%, respectively. Five patients achieved long-term survival (greater than 5 years). Univariate analyses showed that non-smoker (log rank: P = .012), first-line TKI duration ⩾ 10 months (log rank: P < .0001) were associated significantly with longer PFS (Figure 3). Age ⩽ 60 years (log rank: P = .019), first-line TKI duration ⩾ 10 months (log rank: P = .028), and accumulated TKI duration ⩾ 20 months (log rank: P = .016) were significantly associated with better OS. Patients with brain metastases seemed to have worse OS than those without brain metastases according to the Kaplan-Meier survival curve although their differences did not reach statistical significance (P = .167; Figure 4). TNM status, ECOG PS, EGFR mutation types, synchronous or metachronous metastases, metastatic sites, oligometastases, or types of RRLT did not present any significant associations with PFS or OS in our analyses (Table S2). In patients with brain metastases, we also found no significant differences in PFS or OS whether patients received local therapy to cranial lesions or not.

Survival curves of all patients received first-line TKI and RRLT. (A) Progression-free survival (PFS). (B) Overall survival (OS). Solid lines represent the estimated survival curves and dashed lines indicate the 95% confidence intervals of survival. CI indicates confidence interval; RRLT, relatively radical local therapy; TKI, tyrosine kinase inhibitor.

Progression-free survival stratified by variable factors. (A) Smoking status; (B) First-line TKI duration. TKI indicates tyrosine kinase inhibitor.

Overall survival stratified by variable factors. (A) Age; (B) first-line TKI duration; (C) accumulated TKI duration; (D) brain metastases. TKI indicates tyrosine kinase inhibitor.
POF and toxicity
Thirty-six (80%) patients developed disease progressions and 15 (41.7%) of them died. Three patients died without any progression, with a median OS of 16.7 months (range: 7.6-25.5). The progression sites included brain (N = 19, 52.8%), thorax (N = 9, 25%), bone (N = 7, 19.4%), and liver (N = 1, 2.8%). Nineteen (55.8%) patients progressed in new metastatic sites only, 14 (38.9%) progressed in the initial sites only, and 3 (8.3%) progressed in both. Most of the patients (N = 26, 72.2%) developed progressions in the new sites without local therapy, and only 10 (27.8%) patients progressed in the sites that received RRLT (Figure 5).

Pattern of failure classification in per-patient level. (A) Initial sites or new sites progressions. (B) Local therapy sites or non-local therapy sites progressions.
With the regard to toxicity, the most common toxicities included neutropenia, fatigue, skin rash, diarrhea, and hypohepatia during TKI therapy. Majority of the toxicities were Grade 1-2. Grade 3 hypohepatia was observed in only one patient (2.2%). Toxicities due to the addition of RRLT included headache, vomiting, nausea, radiation pneumonitis, radiation esophagitis, and radiation dermatitis. All RRLT-related toxicities were Grade 1-2. No Grade 4-5 TKI-related or RRLT-related toxicities were recorded.
Discussion
In this study, we report the benefits of combined application of RRLT and first-generation TKI in the first-line management of EGFR-mutant metastatic NSCLC patients. Prolonged PFS and OS from this combined therapy were observed when compared with prior survival data from the TKI monotherapy. Besides, the addition of RRLT altered the POF of TKI monotherapy. New sites metastases, rather than initial sites of progressions, became the major POF of first-line treatment. The toxicities of the combination therapy were acceptable. This study puts forward the definition of RRLT. As an intermediate status between radical local therapy and palliative local therapy, RRLT might be a much better choice than other local therapies for patients with EGFR mutations, a hypothesis that merits more clinical investigations.
Previous studies suggested that addition of local therapy to systemic treatment prolonged the survival of EGFR-unselected metastatic NSCLC patients, especially in those who had oligometastases.14,22,23 However, studies regarding the effects of local therapy combined with first-line TKI for metastatic NSCLC patients with EGFR activating mutations are rare. As listed in Table 3, very few publications suggested that the first-line TKI combined with local therapy, with the median PFS of 13.0-36.0 months and the median OS of 23.1-40.9 months, seemed beneficial for EGFR-mutant metastatic NSCLC patients when compared with previous survival data from the TKI monotherapy.16-19,24 Yet, most of them were retrospective studies with small sample size, and varied in local therapy intensity.
Studies of EGFR-mutant metastatic NSCLC treated with first-line TKI and local therapy.
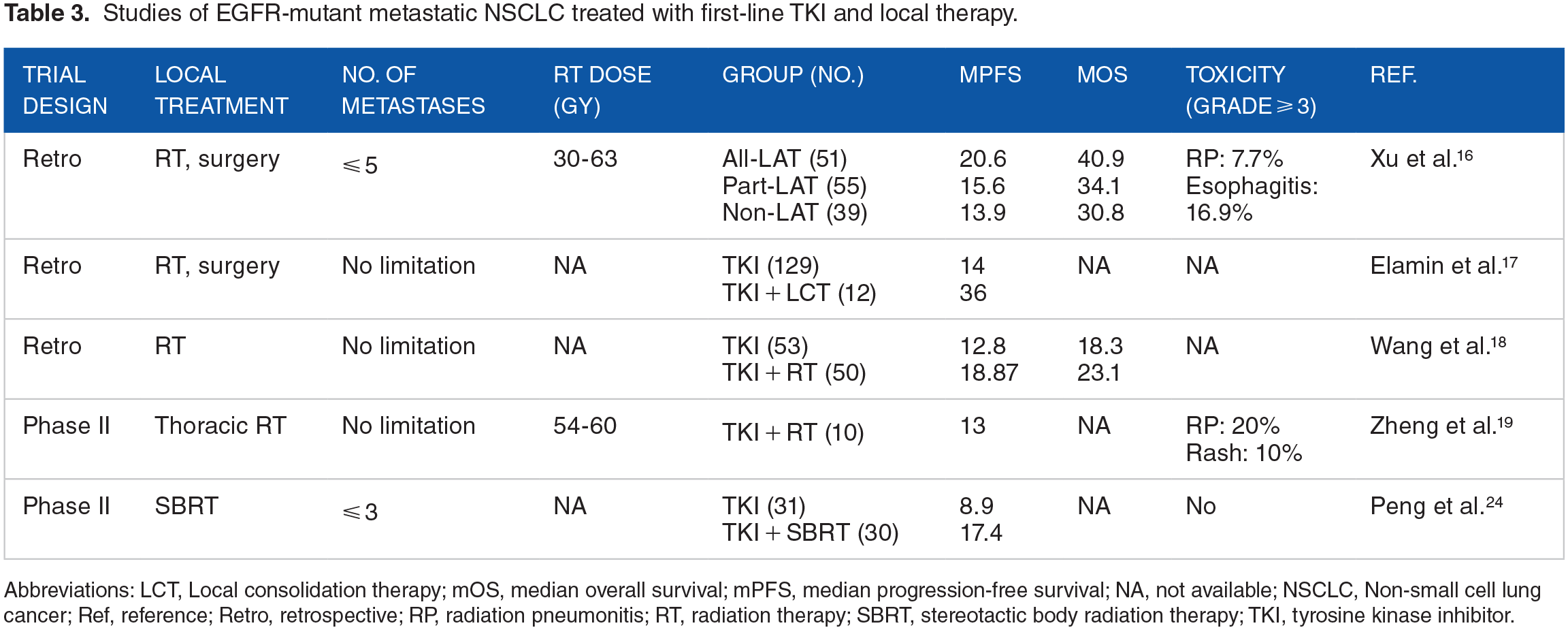
Abbreviations: LCT, Local consolidation therapy; mOS, median overall survival; mPFS, median progression-free survival; NA, not available; NSCLC, Non-small cell lung cancer; Ref, reference; Retro, retrospective; RP, radiation pneumonitis; RT, radiation therapy; SBRT, stereotactic body radiation therapy; TKI, tyrosine kinase inhibitor.
Xu et al. 16 conducted a retrospective study to investigate whether consolidative local ablative therapy (LAT) could improve the survival of EGFR-mutant oligometastatic patients during the first-line TKI therapy. The results showed that the all-LAT group had better PFS and OS than the part-LAT or non-LAT group (median PFS: 20.6, 15.6, and 13.9 months, respectively, P < .001; median OS: 40.9, 34.1, and 30.8 months, respectively, P < .001), but no survival difference was observed between the part-LAT and non-LAT groups. These results indicated that a more radical local therapy with larger extent and more curative intensity may be a proper choice for patients with more favorable survival such as those with EGFR activating mutations. Our study put forward a concept of RRLT for the first time and reported encouraging PFS and OS from the combination of RRLT and first-line TKI. The median OS (55 months) in our cohort was much better than that of the locally advanced NSCLC patients from our early report. 25 It is possible that the addition of RRLT improved the local control of initial sites and allowed patients to extend their existing TKI treatment, which was confirmed beneficial for the survival. 26 The proper extent and intensity of RRLT may also balance the disease control and treatment tolerance, which allow patients to receive further treatments after disease progressions. Together, RRLT may be a feasible option for metastatic NSCLC patients with EGFR activating mutations during first-line EGFR-TKI treatment, a hypothesis that is worth to test further.
The optimal timing of local therapy is another vital issue in clinical practice. In our cohort, local therapy combined with TKI as first-line management achieved improved survival. A few retrospective studies also reported that local therapy to oligoprogressive diseases after first-line TKI provided survival benefit with mild toxicities in EGFR-mutant NSCLC patients.27-29 Local therapy may be beneficial for EGFR-mutant NSCLC patients regardless of the timing of local therapy administration. One potential advantage of early administration of local therapy in non-progressing patients, instead of oligoprogressive patients, is that it may delay or prevent the emergence of resistant clones before additional metastatic spread occurs, as suggested by the observation that local therapy delays the time to new metastases. 17 Yet, the optimal timing of local therapy remains to be determined.
Regarding the eligible candidates for the combination therapy of local therapy and TKI in EGFR-mutant metastatic NSCLC patients, limited retrospective studies suggested that female, ECOG PS score of 0-1, non-smokers, stage N0-1, exon 19 deletion, and longer duration of TKI therapy may yield better survival.15,16 Our study implicated that non-smokers, younger than 60 years, first-line TKI duration longer than 10 months, and accumulated TKI duration ⩾ 20 months were potential predictors for better survival, which were in compliance with the favorable factors for NSCLC. Such information may help us identify candidate patients who can benefit from the combination therapy in clinical practice. As for patients with brain metastases, controversial results have been reported about the effects of local therapy incorporation into TKI therapy, and stereotactic radiosurgery seemed to be associated with better OS than whole-brain radiotherapy.16,30,31 However, we were unable to find any benefit of local therapy to cranial lesions in our study. Prospective, large scale, and randomized clinical trial may be warranted.
More study limitations exist in our analysis. First, as a retrospective study in a single institution, selection bias is difficult to avoid. For metastatic NSCLC patients, generally those who had favorable status, such as good performance score, no weight loss, few comorbidities, and limited numbers and volumes of metastases, were more apt to be selected to receive RRLT. Second, limited number of eligible patients makes it hard to identify a specific subgroup of patients who will benefit from RRLT. The main reason for the small sample size is that the combination of local therapy and TKI is not commonly adopted as the first-line treatment of metastatic NSCLC with EGFR activating mutations in the “real-world.” Several phase II studies were terminated early due to the slow enrollment. Furthermore, it is possible that our patient selection criteria were restricted to RRLT. Therefore, many patients who received palliative local therapy were excluded from the analysis. Third, this is a single-arm study. The results were only compared with previously reported data.
Conclusions
In conclusion, RRLT combined with first-line TKI improved the survival and altered the pattern of failure for metastatic NSCLC patients with EGFR activating mutations. Our study provides additional information to the limited evidence on the efficacy of local therapy combined with EGFR TKI as the first-line treatment for EGFR-mutant metastatic NSCLC patients. Well-designed, large-population prospective studies are warranted to evaluate further the benefit of RRLT combination with first-line TKI in similar candidate patients to our study and to optimize the timing and intensity of local therapy for selected patients.
Supplemental Material
sj-docx-1-onc-10.1177_11795549221080347 – Supplemental material for Local Therapy Combined With First-Line EGFR Tyrosine Kinase Inhibitor Achieves Favorable Survival in Patients With EGFR-Mutant Metastatic Non-Small Cell Lung Cancer
Supplemental material, sj-docx-1-onc-10.1177_11795549221080347 for Local Therapy Combined With First-Line EGFR Tyrosine Kinase Inhibitor Achieves Favorable Survival in Patients With EGFR-Mutant Metastatic Non-Small Cell Lung Cancer by Xiaotong Lu, Jingbo Wang, Chunyu Wang, Jun Liang, Zongmei Zhou, Dongfu Chen, Qinfu Feng, Zefen Xiao, Zhouguang Hui, Jima Lu, Tao Zhang, Wenyang Liu, Jianyang Wang, Xin Wang, Lei Deng, Yirui Zhai, Nan Bi and Luhua Wang in Clinical Medicine Insights: Oncology
Footnotes
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the CAMS Innovation Fund for Medical Sciences (2017-I2M-1-009 to L.W.), the Beijing Hope Run Special Fund of Cancer Foundation of China (LC2018A04 to J.W.), and the Sanming Project of Medicine in Shenzhen (SZSM201612063 to L.W.).
Author Contributions
Xiaotong Lu and Jingbo Wang reviewed the patients’ records, built the database and performed relevant analysis. Chunyu Wang, Jun Liang, Qinfu Feng, Zhouguang Hui, Zongmei Zhou and Dongfu Chen assisted the records review and patient selection. Zefen Xiao, Jima Lu, Lei Deng, Yirui Zhai, Nan Bi and Luhua Wang participated in experimental design. Tao Zhang, Wenyang Liu, Jianyang Wang and Xin Wang assisted the statistical analysis. Xiaotong Lu and Jingbo Wang wrote the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
