Abstract
Background:
Existing evidence demonstrates that radiotherapy and antiangiogenic drugs have synergistic antitumour effects and may be a promising treatment option for patients with solid tumour. Thus, we performed a phase II trial to evaluate the efficacy and safety of whole-brain radiotherapy (WBRT) combined with anlotinib for multiple brain metastases (BMs) from non-small cell lung cancer (NSCLC) without targetable driver mutations.
Methods:
Patients with multiple BMs (⩾3) from NSCLC without targetable driver mutations who failed to respond to at least first-line chemotherapy were enrolled. Eligible patients received WBRT (30 Gy/10 f, 5 f/week) and anlotinib (12 mg/day, day 1-14 of 21 days per cycle, 2 cycles) until disease progression or treatment intolerance. The primary endpoint was intracranial objective response rate (iORR) and secondary endpoints included intracranial progression-free survival (iPFS), disease control rate (DCR), overall survival (OS), and safety.
Results:
Between April 2019 and March 2021, 21 patients were enrolled in the trial, of which 12 were aged ⩾60 years (57.1%), 13 were men (61.9%), 7 had an Eastern Cooperative Oncology Group Performance Status score of 0 to 1 (81.0%), 18 had adenocarcinoma (85.7%), and 11 had ⩾6 BMs (52.4%). Of the 21 evaluable patients, the iORR was 66.7% (1 complete response + 13 partial response [PR]), and 28.6% (7PR) had extracerebral lesions. The DCRs for intracerebral and extracerebral lesions were 90.5% and 81.0%, respectively. The iPFS and OS were 10.3 months (95% confidence interval [CI]: 0-24.8 months) and 13.4 months (95% CI: 0-27.9 months), respectively. The most frequently observed toxicities were loss of appetite (61.9%), hypertension (52.4%), fatigue (47.6%), diarrhoea (28.6%), vomiting (19.0%), dizziness (42.9%), and headache (33.3%). None of the patients developed grade 4 or higher grade adverse reactions.
Conclusions:
Anlotinib combined with WBRT is effective and well tolerated in patients with multiple BMs (⩾3) from NSCLC without targetable driver mutations. Therefore, further validation studies are required.
Clinical trial registration number: ChiCTR 1900027769
Keywords
Introduction
Lung cancer is the leading cause of cancer death and is the cancer with the second highest incidence in China.1,2 Non-small cell lung cancer (NSCLC) accounts for approximately 85% of lung cancer cases, and the incidence of brain metastases (BMs) for NSCLC is as high as 10% at the time of first diagnosis, and 25% to 50% of patients with NSCLC will develop BM during the course of their disease. 3 BM from NSCLC is associated with poor prognosis, and the natural median overall survival (OS) is 2 to 3 months. 4 Patients with symptomatic or a limited number of BMs (⩽3) undergo local therapy with neurosurgical resection or stereotactic radiosurgery (SRS). 5 Since the 1970s, WBRT has gradually become the standard of care in patients with multiple BMs. The disease remission rate after WBRT is approximately 24% to 55%, while the median OS is extended to 3 to 6 months. 6 WBRT has better distant intracranial tumour control, but is associated with relatively low control of existing metastases.
Chemotherapy alone has a limited role in the treatment of BM because of the assumed inability of these drugs to cross the blood-brain barrier (BBB). The emergence of targeted drugs, such as tyrosine kinase inhibitors (TKIs), has improved the intracranial progression-free survival (iPFS) and OS of NSCLC with epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), and c-rosoncogene 1-receptor tyrosine kinase (ROS-1) mutations and BMs. 7 However, about 50% of patients with NSCLC in China are negative for targetable driver mutations, 8 and there is currently no effective systemic therapy for these patients with BM. Improving the response rates and prolonging the survival rates of patients with BM from NSCLC without targetable driver mutations is the current focus of treatment, and tumour growth is closely related to the growth of new blood vessels. Recently, some studies have shown that antiangiogenic drugs can improve the oxygenation of tumour cells and enhance the sensitivity to radiotherapy by normalising the tumour blood vessels and reducing the proportion of hypoxic cells. 9 Anlotinib is a new multitarget antitumour drug that is widely used in NSCLC, 10 - 12 soft tissue sarcoma, oesophageal cancer, and so on.13,14 There is a strong radiobiological rationale for harnessing the radiosensitising effects of anlotinib. This clinical trial aimed to evaluate the efficacy and safety of anlotinib combined with WBRT in patients with multiple BMs from NSCLC without targetable driver mutations and provide a new treatment paradigm for them.
Patients and Methods
Study design
This prospective, single-arm, phase II clinical trial was registered with the Chinese Clinical Trial Registry (number: ChiCTR 1900027769). Eligible patients with multiple BMs from NSCLC without targetable driver mutations who failed to respond to at least first-line chemotherapy received anlotinib (12 mg/day, day 1-14 of 21 days a cycle, 2 cycles) combined with WBRT. The BM of the enrolled patients were either advanced or newly identified after receiving cytotoxic systemic therapy regimens. The cytotoxic regimen for nonsquamous NSCLC was cisplatin or carboplatin combined with pemetrexed or paclitaxel, while that for squamous cell NSCLC was gemcitabine combined with cisplatin or carboplatin. All patients completed 4 to 6 cycles of chemotherapy.
The whole brain was defined as the clinical tumour target volume (CTV), and CTV expansion of 3 mm was defined as the planning target volume (PTV). The prescription dose for the PTV was 30 Gy/10 f (5 f/week). A linear accelerator (Siemens, Germany) was used to deliver the radiotherapy.
The primary endpoint was the intracranial objective response rate (iORR). The secondary endpoints were iPFS, disease control rate (DCR), OS, and safety. The iORR was defined as the proportion of patients achieving intracranial tumour complete response (CR) or partial response (PR) according to the Response Evaluation Criteria in Solid Tumours (RECIST). The iPFS was assessed from the time of WBRT with anlotinib administration to the time when the earliest sign of BM progression appeared based on RECIST or death from any cause. Disease control rate was defined as the proportion of patients with CR, PR, or stable disease (SD). OS was assessed from the time of WBRT until the time of death from any cause. Tumour response was assessed every 8 weeks using magnetic resonance imaging (MRI) until disease progression. Intracranial tumour progression was defined as enhanced MRI showing enlarged lesions with or without peritumoural oedema or new intracranial metastases, evaluated based on the RECIST. Toxic effects were assessed according to the National Cancer Institute Common Terminology Criteria (version 3.0).
All patients were recruited from the Shaoxing People’s Hospital, Zhejiang Province. This study was approved by the ethics committee of our hospital (approval no: 2019K-Y-090-02), and all patients provided informed consent.
Patients
Non-small cell lung cancer patients with multiple BMs admitted in our hospital were recruited from April 2019 to March 2021 (Figure 1). Inclusion criteria were as follows: (1) Patients who volunteered to participate in the study and provided informed consent; (2) patients aged 18 to 75 years, with ⩾3 BMs, and with an expected survival time of >3 months; (3) diagnosis of advanced NSCLC was confirmed by pathological or cytological evaluation with radiographic evidence of BM on contrast-enhanced MRI of the brain and measurable lesions; no EGFR, ALK, and ROS-1 gene mutations were noted. Epidermal growth factor receptor was detected by the amplification block mutation system, and ALK and ROS-1 fusion genes were detected by fluorescence in situ hybridisation; (4) patients with normal heart, liver, and kidney function; no contraindications to drug therapy, and an Eastern Cooperative Oncology Group Performance Status score of 0 to 2; and (5) patients who failed to respond to at least first-line chemotherapy. Exclusion criteria were as follows: patients with (1) intractable hypertension; (2) active bleeding, including haemoptysis and gastrointestinal bleeding; and (3) a history of craniocerebral radiotherapy.

Study flowchart. BM indicates brain metastasis; iPFS, intracranial progression-free survival; OS, overall survival.
Statistical analysis
Statistical analysis was performed using SPSS (version 22.0; SPSS Inc, Chicago, IL). Survival curves were estimated using the Kaplan-Meier method. Two-sided tests were used to analyse the significant differences. A P value of <.05 was considered significant.
Results
Patients’ characteristics
We recruited 30 patients between April 2019 and March 2021. Finally, only 21 patients were included in the study. All patients were treated with the allocated regimens and underwent follow-up evaluations. The follow-up rate was 100%. The median follow-up time was 10.37 (3.27-26.13) months. All patients tested negative for targetable driver mutations. Ten patients had 3 to 5 BMs, while 11 patients had >6 BMs. Eighteen patients were diagnosed with adenocarcinoma, while 3 patients were diagnosed with squamous cell carcinoma. Thirteen patients developed symptoms of BM, while 8 patients were asymptomatic. None of the patients developed leptomeningeal spread during the course of treatment. See Table 1 for additional information.
Patients’ characteristics (n = 21).

Abbreviation: ECOG PS, Eastern Cooperative Oncology Group performance status.
Response of brain metastases
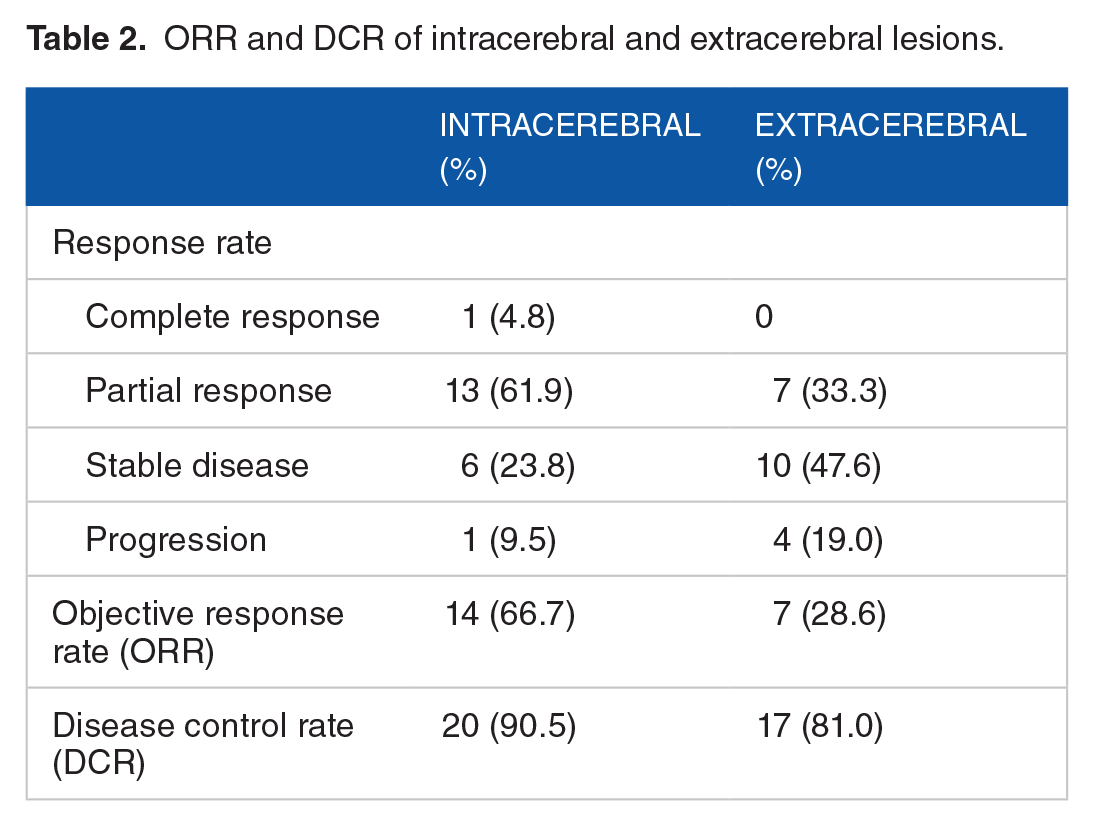
At the cut-off date of July 31, 2021, the 21 patients were evaluated to determine their treatment response (Table 2). Central nervous system symptoms improved in 11 patients. Patients with intracerebral disease had an ORR of 66.7% (1CR + 13PR), while those with extracerebral lesions had an ORR of 28.6% (0CR + 7PR). The DCRs of patients with intracerebral and extracerebral lesions were 90.5% and 81.0%, respectively. Figure 2 shows the percentage change in intracerebral and extracerebral tumour burdens.
ORR and DCR of intracerebral and extracerebral lesions.

Assessment of the best percentage change from baseline in target lesion size for all patients (n = 21). BOR indicates best objective response; CR, complete response; PD, progressive disease; PR, partial response; SD, stable disease.
Survival
At the time of analysis, 11 patients had died due to tumour progression. The 10 patients who survived were followed up. The median iPFS and OS were 10.3 months (range: 0-24.8 months) and 13.4 months (range: 0-27.9 months), respectively. The 1-year iPFS and OS rates were 46.2% and 50.5%, respectively (Figure 3).
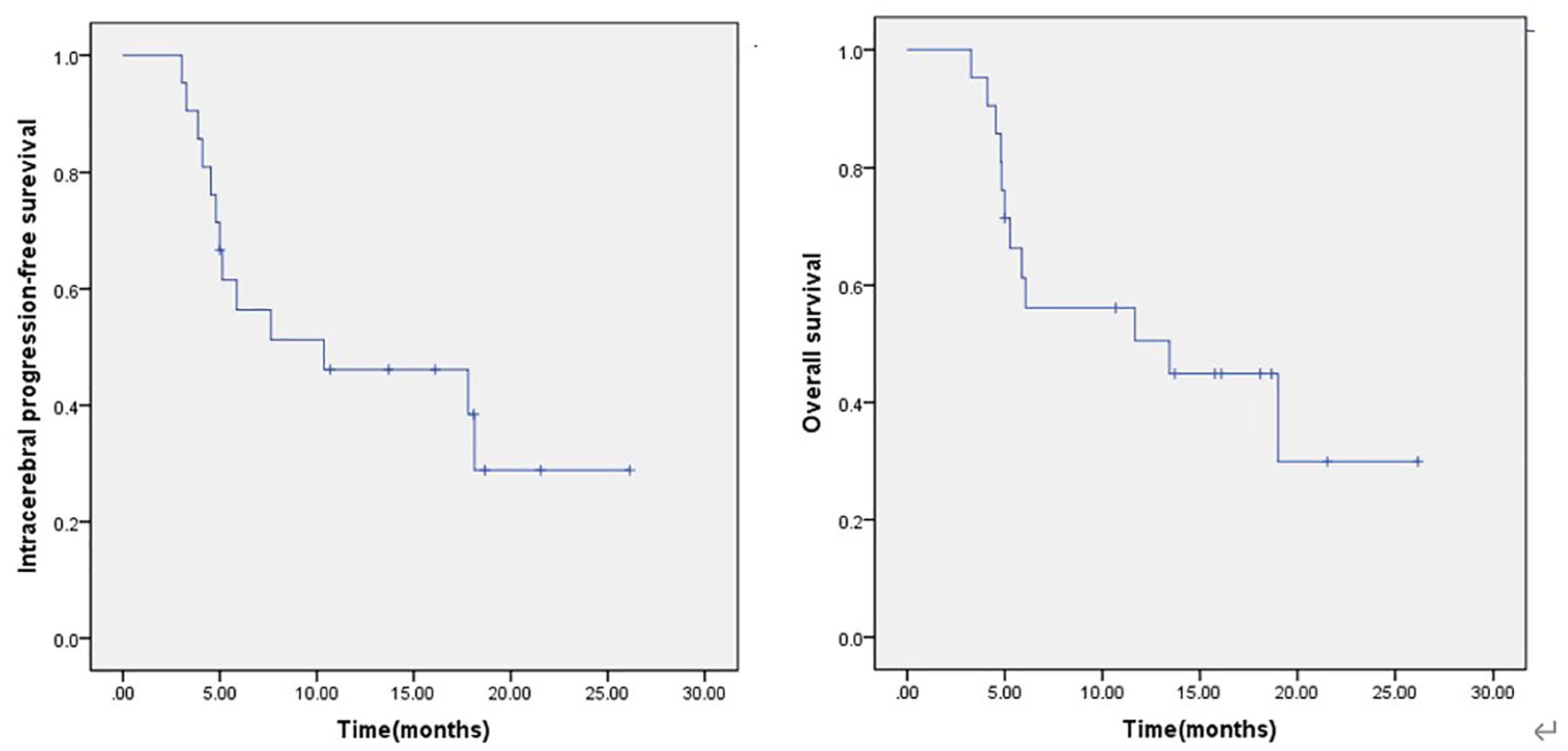
iPFS and OS of WBRT + anlotinib. The median iPFS and OS were 10.3 months (range: 0-24.8 months) and 13.4 months (range: 0-27.9 months), respectively. iPFS indicates intracerebral progression-free survival; OS, overall survival; WBRT, whole-brain radiotherapy.
Acute toxicity and treatment compliance
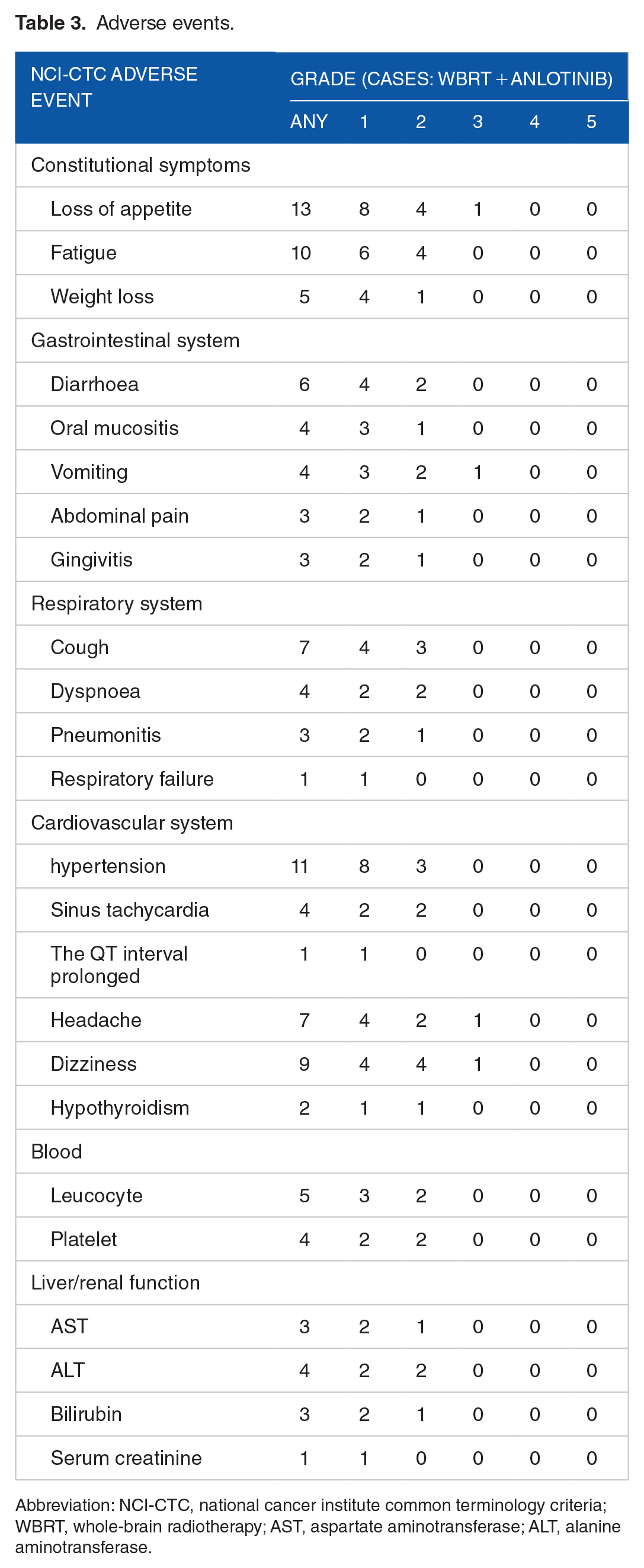
The different acute toxicities are summarised in Table 3. None of the patients developed grade 4 or higher grade adverse reactions, such as gastrointestinal, respiratory, and cardiovascular system problems; hypothyroidism; headache; nausea; and haematological, hepatic, and renal dysfunction. The following adverse reactions were prevalent: loss of appetite, fatigue, diarrhoea, vomiting, dizziness, and headache. All adverse reactions were well tolerated, and none of the patients required anlotinib dose reduction.
Adverse events.
Abbreviation: NCI-CTC, national cancer institute common terminology criteria; WBRT, whole-brain radiotherapy; AST, aspartate aminotransferase; ALT, alanine aminotransferase.
Discussion
The treatment for BM includes surgery, WBRT, SRS, chemotherapy, targeted therapy, and immunotherapy. Regarding the treatment options for patients with a limited number of BM (⩽3), SRT alone has a survival advantage over WBRT alone and can better preserve the cognitive function, 15 and local therapy including neurosurgical resection or SRS with or without WBRT is considered. 16 Currently, WBRT is less frequently used in patients with limited BM. 17 However, it is still recommended in patients with multiple metastases (>3 BMs). 18 WBRT is considered the standard of care in patients (>3 BMs) not eligible for receiving local treatments, with a median OS of 8 to 10 months, and with a 1-year OS rate of 16% to 28%. 19 WBRT with a wide range of exposure can control subclinical intracranial lesions and reduce the number of new brain lesions. However, due to the limited tolerance of normal brain tissues, the total radiation dose is also limited. More than one-third of BMs cannot be controlled locally after radiotherapy, and 50% of patients with BM die of intracranial disease progression. 16
In the era of targeted therapy, dramatic intracranial response rates have been reported in NSCLC patients with EGFR mutations or ALK rearrangements, and TKIs, such as icotinib, are recommended as the first-line standard treatment for NSCLC with EGFR mutation and multiple BM, and can significantly improve the intracranial and extracranial PFS rates of these patients. 20 However, there is currently no effective systemic targeted therapy for NSCLC with multiple BMs without targeted driving mutations. Due to the limited penetration of a wide range of drugs across the BBB, 21 WBRT combined with chemotherapy has little benefit. Several chemotherapeutic regimens have been investigated concurrently with WBRT as treatment for NSCLC with multiple BMs, but they showed disappointing results.22,23
How to improve the control rate of intracranial metastases from NSCLC without target-driven mutations is a clinical problem worthy of study.
Tumour angiogenesis is 1 of the tumour’s malignant characteristics. Peritumoural oedema due to BM is the main cause of intracranial hypertension symptoms and death. Antiangiogenic drugs can not only alleviate brain oedema caused by tumours and shrink tumours by blocking abnormal blood supply but also normalise the tumour vasculature and enhance radiosensitivity by improving hypoxia. 9
As a novel small-molecule multitarget TKI, anlotinib inhibits tumour angiogenesis to achieve antitumour angiogenesis as well as tumour growth. In vitro studies of the H520 cell cycle and apoptosis by anlotinib combined with radiotherapy showed that anlotinib had an inhibitory effect on lung cancer H520 cell proliferation. A higher rate of apoptosis and G2/M phase block was observed in the anlotinib-radiotherapy combined group. Anlotinib combined with radiotherapy synergistically inhibits the growth of tumour cells. 24 Cranial radiotherapy can increase the permeability of the BBB, which may increase anlotinib content in brain tissue; thus, the curative effect of cranial radiotherapy combined with anlotinib may be superior to that of cranial radiotherapy alone for NSCLC patients with no specific gene mutation. 25 A previous retrospective study 26 and 2 prospective studies27,28 suggested that anlotinib combined with radiotherapy for BM has satisfactory efficacy with tolerable toxicity. A phase III trial, ALTER 0303, showed that anlotinib improved the PFS and OS as a second- or third-line therapy for advanced NSCLC. 11 Post hoc analysis of the ALTER 0303 trial showed that anlotinib has intracranial activity and plays a potential role in tumour control at intracranial sites. The intracranial ORR and DCR were 14.3% and 85.7%, respectively, in patients with BM who were treated with anlotinib. 29
Recently, a phase II study of anlotinib combined with WBRT reported significant therapeutic efficacy of this regimen and manageable AEs in patients with advanced NSCLC with multiple BMs. 28 Among the 10 patients, 6 achieved PR and 3 had SD during the intracranial evaluation. The 6-month iPFS rate was 87.5%. The ORR and DCR were 60.0% and 90.0%, respectively, during the intracranial evaluation.
In our study, the ORRs of patients with cerebral and extracerebral lesions were 66.7% and 33.3%, respectively. The DCRs of patients with cerebral and extracerebral lesions were 90.5% and 81.0%, respectively. The median iPFS and OS were 10.3 months (95% confidence interval [CI]: 0-24.8 months) and 13.4 months (95% CI: 0-27.9 months), respectively. Results showed that the remission rate and control rate of intracranial tumours improved, and the increased intracranial control achieved by WBRT combined with anlotinib resulted in an increase in OS. This may be due to the good synergy between radiotherapy and antiangiogenic agents. Anlotinib has a radiosensitisation effect that enhances the tumour cell killing effect, improves the local curative effect, reduces brain oedema caused by intracranial tumours, and improves the symptoms of patients. Second, during radiotherapy combined with anlotinib, the extracranial lesions are also well controlled, which is related to the systemic antitumour effect of anlotinib, inhibiting tumour angiogenesis and delaying tumour progression. In addition, 18 of the 21 patients in this trial were histologically diagnosed with adenocarcinoma. Patients with adenocarcinoma showed a significantly higher median survival time than those with other histological types of NSCLC with BM 30 ; moreover, the limited number of cases and follow-up time increased the statistical bias of the survival time.
Of safety, grade 3 or higher grade adverse reactions in this study mainly included vomiting (1 of 21), loss of appetite (1 of 21), headache (1 of 21), and dizziness (1 of 21), which improved after drug withdrawal. The other adverse reactions were mainly grade 1 to 2, which improved after symptomatic treatment. The overall incidence of adverse reactions was low, which may be related to the dose adjustment during treatment and symptomatic support therapy.
Conclusions
In conclusion, our study showed that anlotinib combined with WBRT is effective and well tolerated in patients with multiple BM (⩾3) from NSCLC without targetable driver mutations. The application has a higher ORR, higher DCR, and lower incidence of adverse events, which can prolong the survival time of patients NSCLC with BM. However, our study was a small-scale, single-arm phase II study with a potent bias. Therefore, further randomised trials are needed.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Health Science and Technology Plan Project of Zhejiang (2021KY1149) and the Bethune Medical Science Research Foundation (B19083FT).
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
DW designed the study project. DW and JL drafted article. JX and JL performed data analysis. JL, JX, WY, WZ, XZ, and JM contributed to data collection. All authors approved the final article.
