Abstract
Background:
The incidence of small renal mass (SRM) increases, and the prognosis of SRM is poor once metastasized. Therefore, we conducted this study to assess the clinical and pathological characteristics of SRM to determine the risk factors that influence the metastasis and prognosis of SRM.
Methods:
A small renal mass is defined as a solid tumor mass with the largest diameter of 4 cm or less on the pathological diagnosis. The metastasis is confirmed by imaging or pathological examination. We retrospectively included 40 patients with metastatic SRM (mSRM) treated in the department of urology of Peking University Third Hospital from October 2002 to October 2020. Meanwhile, 358 patients with nonmetastatic SRM treated in our hospital from January 2015 to December 2017 were selected as controls. Clinicopathologic features were compiled.
Results:
Multivariate logistic regression analysis showed that age (P = .027, odds ratio [OR] = 1.037, 95% confidence interval [CI] 1.004-1.070), clinical symptoms (P < .001, OR = 4.311, 95% CI 1.922-9.672), World Health Organization/International Society of Urological Pathology (WHO/ISUP) nuclear grade 3/4 (P = .004, OR = 7.637, 95% CI 1.943-30.012; P = .004, OR = 20.523, 95% CI 2.628-160.287), and lymphatic invasion (P = .030, OR = 15.844, 95% CI 1.314-191.033) were risk factors for distant metastasis of SRM. Once metastasis occurs, the prognosis of SRM is poor. Multivariate Cox regression analysis of the prognosis of mSRM showed that age (P = .016, hazard ratio [HR] = 1.125, 95% CI 1.022-1.239), preoperative serum creatinine (P = .041, HR = 1.003, 95% CI 1.000-1.005), vascular invasion (P = .041, HR = 1.003, 95% CI 1.000-1.005), and metastasis (P < .001, HR = 24.069, 95% CI 4.549-127.356) were risk factors for overall survival (OS), and only metastasis (P < .001, HR = 9.52, 95% CI 5.43-16.7) was a risk factor for progression-free survival (PFS) of SRM.
Conclusions:
SRM with advanced age, clinical symptoms, high pathological nuclear grade, and lymphatic invasion are more likely to have distant metastasis. And SRM with older age, poor preoperative basic renal function, pathological vascular invasion, and metastasis have worse OS.
Introduction
Renal cancer is a common malignant tumor of the urinary system, with approximately 76 000 new cases in the United States every year, accounting for about 4% of all cancer types. 1 A small renal mass (SRM) is defined as a solid tumor mass with the largest diameter of 4 cm or less on the pathological diagnosis. 2 At present, the increase in SRM detection is associated with ultrasound and other screening technologies. 3 Robot-assisted or laparoscopic partial nephrectomy is an accepted standard of care for SRM. For patients with renal sinus invasion, renal pelvis invasion, or renal vein cancer embolism, robot-assisted or laparoscopic radical nephrectomy is usually adopted. 4
However, about 2% to 7% of SRM will metastasize.5 -8 Patients with mSRM are treated with systemic therapies, including tyrosine kinase inhibitor (TKI)-targeted therapies or immunotherapies. 4 The prognosis is generally poor in those patients. The 5-year specific survival rate is approximately 10%, and the median overall survival (OS) is 10 to 15 months. 9 However, research on the clinicopathological characteristics and prognosis of mSRM is limited. Therefore, we conducted this study to assess the clinical and pathological characteristics of SRM to determine the risk factors that influence the metastasis and prognosis of SRM.
Population and Methods
Patients
We retrospectively included 40 patients with mSRM treated in the department of urology of Peking University Third Hospital from October 2002 to October 2020. Meanwhile, 358 patients with nonmetastatic SRM treated in our hospital from January 2015 to December 2017 were selected as controls.
All included patients met the following inclusion criteria: (1) with histologically confirmed renal cell carcinoma, (2) maximum tumor diameter of 4 cm or less. Metastasis can be examined during initial diagnosis or follow-up. Different examinations were used to confirm potential metastases at specific sites: lung metastasis usually was identified by chest computerized tomography (CT); liver metastasis and adrenal metastasis used abdominal ultrasound or abdominal CT; bone scan was received if there was an abnormal increase of alkaline phosphatase or symptoms of bone metastasis such as bone pain; patients with signs of the acute nervous system would receive immediate CT or magnetic resonance image (MRI) scan on the head or spinal cord based on the corresponding segmental symptoms; for patients with good economic conditions or systemic symptoms, positron emission tomography-computed tomography (PET-CT) was performed; pathological examination would be selected when necessary.
For this study, demographic characteristics (age, gender, height, weight, body mass index [BMI]) of each patient and whether there was any clinical symptom (including systemic symptoms such as fever, fatigue, weight loss, cachexia, and local symptom such as pain, local mass, hematuria) were collected. The operative time and intraoperative blood loss were used to represent the difficulty of operation, and the American Society of Anesthesiologists (ASA) score was used to evaluate the risk of anesthesia and operation. Detailed histologic characteristics, including pathologic type, tumor diameter, World Health Organization/International Society of Urological Pathology (WHO/ISUP) nuclear grade, invasion of renal sinus, vascular invasion, lymphatic invasion, presence of sarcomatoid component or rhabdoid component, were also recorded.
Outpatient check-ups and telephone interviews strictly followed up patients after surgery. The follow-up evaluation comprises physical examination, renal function, and abdominal ultrasound performed every 3 to 6 months for the first 2 years after surgery, every 6 to 12 months from postoperative 3 to 5 years, and every 12 months after that.
Statistical Methods
Progression-free survival (PFS) is defined as the time from diagnosis to first disease progression or death from any cause; OS is defined as the time from diagnosis to death from any cause.
SPSS IBM 18.0 software was used for analysis. Normal distribution was tested with the Kolmogorov-Smirnov test for continuous variables. For the normal distribution, the mean ± standard deviation was used. The clinical and pathological features of patients with and without metastases were analyzed using two independent samples t test. For nonnormal distribution data, the median with the interquartile range was used to represent, and nonparametric tests were used to compare the differences between the two groups of distributions. The classification data were represented by frequency (percentage), and the chi-square test compared the difference of proportion between groups. Those who did not meet the chi-square test were compared by Fisher’s exact probability method. P < .05 was considered statistically significant. Multivariate logistic regression analysis was used to identify risk factors for metastasis of SRM. Univariate and multivariate Cox proportional hazards analyses were applied in the analysis of OS and PFS. Variables achieving P < .05 in the univariate analysis were incorporated in the multivariable model to determine independent prognostic factors.
Results
A total of 398 patients were retrospectively collected from October 2002 to October 2020 by searching electronic medical records. Forty patients in the group of mSRM, and 358 patients with nonmetastatic SRM were included in the control group. The clinical and pathological data of the patients were shown in Tables 1 and 2. Univariate analysis of the clinical and pathological features between the two groups showed that there were significant differences in age (P = .002), clinical symptoms (P < .001), operation approach (P < .001), operative time (P = .033), intraoperative blood loss (P = .001), ASA score (P = .008), tumor diameter (P = .016), WHO/ISUP nuclear grade (P < .001), lymphatic invasion (P < .001), and perirenal fat invasion (P = .009). Multivariate logistic regression analysis showed that age (P = .027, odds ratio [OR] = 1.037, 95% confidence interval [CI] 1.004-1.070), clinical symptoms (P < .001, OR = 4.311, 95% CI 1.922-9.672), WHO/ISUP nuclear grade3/4 (P = .004, OR = 7.637, 95% CI 1.943-30.012; P = .004, OR = 20.523, 95% CI 2.628-160.287), and lymphatic invasion (P = .030, OR = 15.844, 95% CI 1.314-191.033) were risk factors for distant metastasis of SRM (Table 3).
Comparison of preoperative characteristics between all SRM and those associated with distant metastatic disease.

Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; SRM, small renal mass.
Comparison of intraoperative and postoperative features between all SRM and those associated with distant metastatic disease.

Abbreviations: SRM, small renal mass; WHO/ISUP, World Health Organization/International Society of Urological Pathology.
Multivariate logistic regression analysis of metastatic risk factors for patients with SRM.

Abbreviations: ASA, American Society of Anesthesiologists; CI, confidence interval; OR, odds ratio; WHO/ISUP, World Health Organization/International Society of Urological Pathology.
Among the 40 patients with metastatic SRM, 16 cases had metastasis at initial diagnosis, and 24 cases had metastasis during follow-up. Lung metastasis was found in 14 cases (35%), bone metastasis in 10 cases (25%), adrenal metastasis in 4 cases (10%), other site metastasis in 4 cases (10%), and 8 cases (20%) had metastasized in more than 2 metastatic sites. A total of 50 metastatic lesions were found in 40 patients, including 17 (34%) pulmonary metastases, 16 (32%) bone metastases, 2 (4%) liver metastases, 1 (2%) brain metastases, 7 (14%) adrenal metastases, and 7 (14%) other metastases (Figure 1).

Venn diagram representing the distribution of distant metastatic sites.
Multivariate Cox regression analysis of the prognosis of SRM showed that age (P = .016, hazard ratio [HR] = 1.125, 95% CI 1.022-1.239), preoperative serum creatinine (SCR; P = .041, HR = 1.003, 95% CI 1.000-1.005), vascular invasion (P = .041, HR = 1.003, 95% CI 1.000-1.005), and metastasis (P < .001, HR = 24.069, 95% CI 4.549-127.356) were risk factors for OS (Table 4), and only metastasis (P < .001, HR = 9.52, 95% CI 5.43-16.7) was a risk factor for PFS of SRM (Table 5).
Univariate and multivariate regression analysis of prognostic risk factors (overall survival) for patients with SRM.
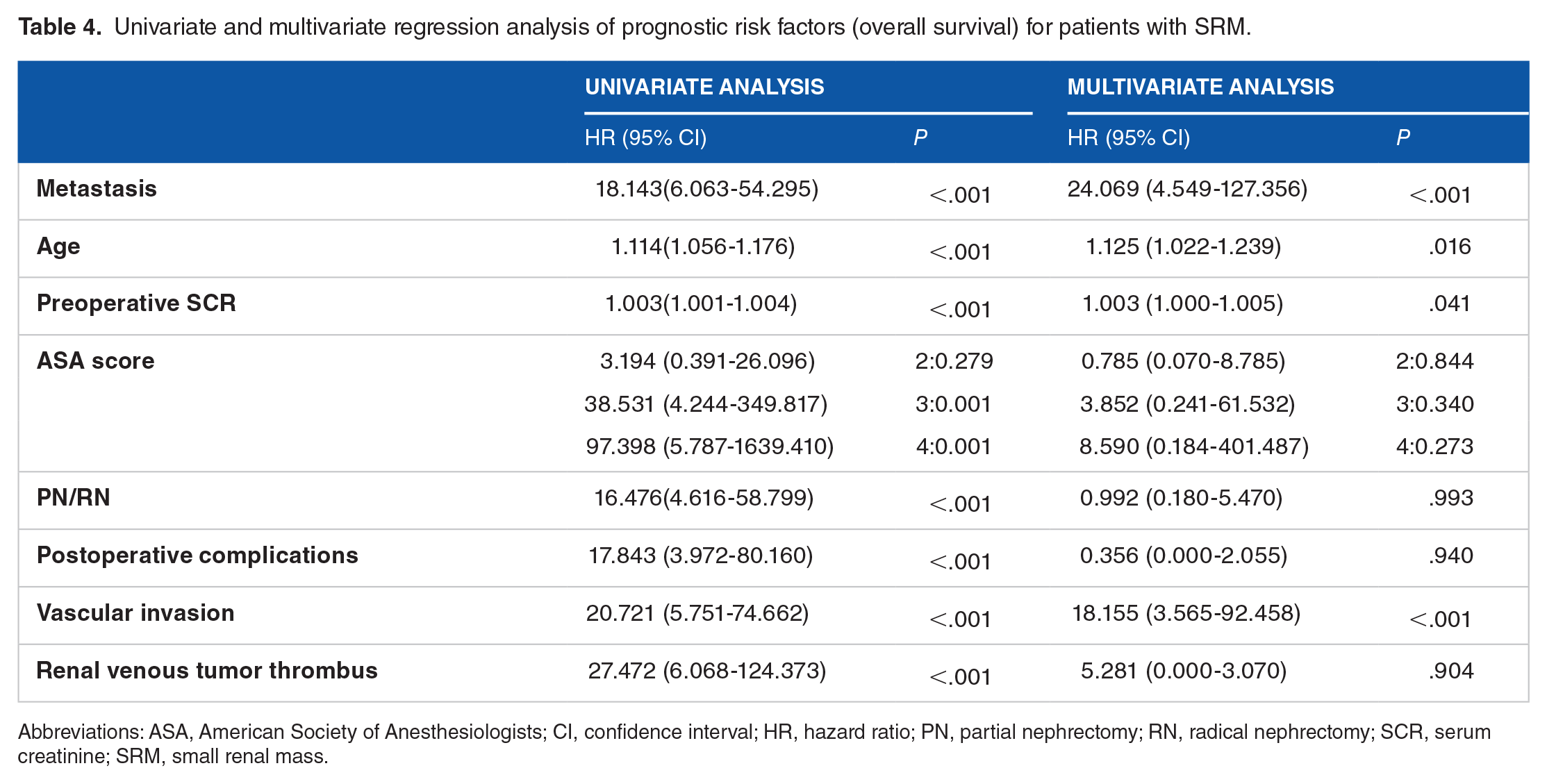
Abbreviations: ASA, American Society of Anesthesiologists; CI, confidence interval; HR, hazard ratio; PN, partial nephrectomy; RN, radical nephrectomy; SCR, serum creatinine; SRM, small renal mass.
Univariate and multivariate regression analysis of prognostic risk factors (progression-free survival) for patients with SRM.

Abbreviations: ASA, American Society of Anesthesiologists; CI, confidence interval; HR, hazard ratio; PN, partial nephrectomy; RN, radical nephrectomy; SRM, small renal mass; WHO/ISUP, World Health Organization/International Society of Urological Pathology.
In our study, the median follow-up time was 38 months, and the median survivorship time was not achieved because of the short follow-up period. The OS rate (P = .002, HR = 14.128, 95% CI 2.698-73.967 & P < .001, HR = 19.721, 95% CI 6.296-61.767) and PFS rate (P < .001, HR = 20.22, 95% CI 7.205-56.75) of mSRM were significantly shortened; meanwhile, there was no statistical difference between the prognosis of patients with synchronous metastasis and that of patients with metachronous metastasis (P = .762) (shown in Figures 2 and 3).

Differences in overal survival among nonmetastatic SRM, synchronous metastatic SRM and metachronous metastatic SRM.
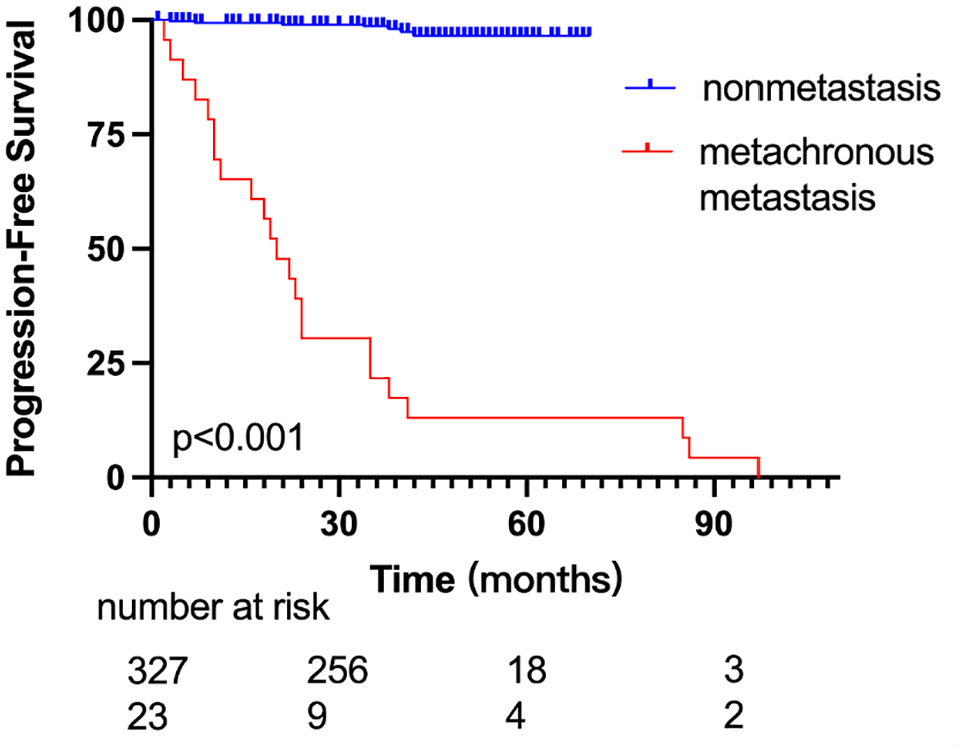
Differences in progression-free survival between nonmetastatic SRM and metachronous metastatic SRM.
Discussion
With the popularity of ultrasound, CT, and other imaging examinations, the incidence of SRM is gradually increasing. 10 For SRM, the guidelines recommend that a biopsy be performed before treatment. Of all surgically resected SRM, approximately 25% are benign, and the other 25% are indolent tumors with limited metastatic potential. 11 Generally, we believe that the larger the tumor size, the higher the risk of metastasis. 12 Some studies have shown that the progression of SRM is slow and the risk of metastasis is low, active monitoring surgery is an option for elderly patients and patients with more complications that are not suitable for surgery.13,14 However, about 2% to 7% of SRM with metastasis. For SRM with metastasis at initial diagnosis, or patients with metastasis during follow-up, the treatment is based on metastatic renal cell carcinoma guidelines, and the prognosis is poor. 4
Our study found that mSRM was easier to choose open surgery and radical nephrectomy; we considered that there were more malignant manifestations found in the preoperative imaging evaluation, such as suspected invasion of the renal pelvis and calyx, or the presence of cystic components that may lead to local implantation, as well as intraoperative reasons. Besides, longer operation time and more intraoperative blood loss suggest that the operation is more difficult. The existing literature shows that patients with metastatic renal cell carcinoma (mRCC) can still benefit from cytoreductive surgery than patients with systemic therapy alone, and patients who undergo cytoreductive surgery after systemic treatment also show benefits in OS time and time to treatment failure.15,16 Under the premise of immunotherapy, cytoreductive surgery can also benefit patients. 17 All of the patients we included have undergone surgery, so they cannot be compared with patients who have received systemic treatment alone. Moreover, patients with mSRM may be older and have obvious clinical manifestations because of treatment-seeking delay. At present, few studies are predicting the metastatic risk of SRM. Zastrow et al analyzed 2058 cases of RCC and found that tumor size and ASA score were important prognostic factors of synchronous metastasis. 18 Takayama et al studied 451 cases of T1a RCC, of which 32 cases were associated with metastasis. They reported that symptoms at diagnosis, CRP ⩾ 0.4 mg/dL, tumor size ⩾ 3.0 cm, histological grade 3, sarcomatoid component, and microvascular invasion were independent risk factors for metastasis. 19 Another study has found that diabetes mellitus is an independent predictor of metastasis in T1a RCC patients. 20 Tan et al found that age, tumor size, symptoms, and ipsilateral synchronous tumors were independent risk factors for predicting metastasis. 21 Several works of the literature suggest that tumor size is a risk factor for metastasis. Tumor size has a statistical difference in the univariate analysis of our results, but it loses significance in the multivariate analysis may be due to the small number of cases. We observed that there was a difference in renal sinus invasion between the two groups, but did not meet the standard of statistical difference in univariate analysis. And, there was a statistic difference in perirenal fat invasion, but it lost its significance after multivariate analysis, which may be due to the greater weighting of other factors. In addition, high nuclear grade and lymphatic invasion are risk factors for metastasis, while in Tan’s study, they are independent predictors for disease recurrence. 21
At present, limited by the number of cases, few studies are still on the prognosis of metastatic SRM. As early as 2001, Yaycioglu et al developed a prognostic model using only preoperative clinical variables after analyzing 296 cases of open radical nephrectomy. 22 Kattan summarized the data of 601 cases of nephrectomy and constructed a nomogram, including histological type, tumor size, Tumor-node-metastasis (TNM) classification, and clinical manifestations, to evaluate the possibility of recurrence after a 5-year follow-up. 23 In 2002, Igor et al proposed a system (the tumor stage, size, grade and necrosis [SSIGN] score) based on tumor staging, size, nuclear grade, and histological tumor necrosis. 24 Memorial Sloan-Kettering Cancer Center (MSKCC) score, developed by Motzer et al, is one of the most widely used predictive models for mRCC patients. Patients are divided into three risk groups (good, moderate, and poor) according to the number of adverse factors (for example, less than 1 year from diagnosis to initiation of systemic treatment, corrected elevated serum calcium, elevated lactate dehydrogenase [LDH], low hemoglobin levels and low Karnofsky PS score). 25 Another well-known University of California Los Angeles Integrated Staging System (UISS) scoring system for RCC combined TNM classification, Eastern Cooperative Oncology Group (ECOG) PS score, and the Fuhrman score. In this system, patients are divided into 3 categories (low, medium, and high risk) to predict OS. 26 Kume et al retrospectively analyzed the surgical treatment of 165 cases with SRM ⩽ 3 cm, and concluded that microvascular infiltration was an important prognostic factor for patients with a less than 3 cm tumor. 27 Although we did not form a predictive model because of the limited number of cases, we also found the risk factors affecting the PFS and OS of SRM. Judging from our result, patients with metastasis were more likely to progress, while patients with older age, poor preoperative basic renal function, pathological vascular invasion, and metastasis had worse overall prognoses.
The 5-year OS rate of localized RCC can reach 92.7%, but it will directly drop to 13.9% once metastasis occurs. 28 The 5-year cancer-specific survivorship after nephrectomy of stage I renal cell carcinoma can reach 97.1% to 98.1%. 29 For SRM, current studies have shown that the 5-year OS rate can reach 92%, and the 5-year tumor-specific mortality rate is about 5.3%.30,31 The median survival time of mSRM is about 37 to 38 months. 32 In this result, we can also see a significant decrease in the OS of mSRM. A meta-analysis found that the local progression rate after localized partial nephrectomy was about 0.4%. 33 Our data found that the tumor PFS of mSRM was significantly shortened, and the median PFS was about 20 months. The literature reported that the longer the interval between initial diagnosis and metastasis, the better the prognosis. 34 When we reviewed all mRCC patients in our center, we found that the survival of synchronous metastatic patients was worse than that of metachronous metastatic patients. However, no statistical difference was found in patients with SRM which may be due to the small number of samples.
The present study has several limitations:
The main limitation of the present study is the relatively short follow-up time that may underestimate the potential of metastasis of nonmetastatic SRM, and potentially affect the outcome of the study. Although the median follow-up lasted more than 36 months, longer follow-up is required to validate the current findings.
The metastasis of some cases depends on imaging diagnosis and has no pathological basis.
The definition of SRM depends on the maximum diameter of the pathological specimen, but it may be smaller than the actual size because of the fixation and sampling of the specimen.
This study is a retrospective study, the number of cases is limited, and it still needs to be further verified by a large sample of research.
Conclusion
SRM with advanced age, clinical symptoms, high pathological nuclear grade, and lymphatic invasion are more likely to have distant metastasis. Moreover, SRM with older age, poor preoperative basic renal function, pathological vascular invasion, and metastasis have worse OS. Of course, further research is needed to find a more accurate method to judge the metastatic potential of SRM.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
WH, ZL, and YT contributed equally to this work and were responsible for project development, data collection, data analysis and interpretation, and manuscript writing. YL, CX, RX, PH, ST, LG, XZ, GZ, and HZ contributed to data collection and language revision. CL and LM were responsible for project development and manuscript editing.
Statement of Ethics
The study was approved by the Peking University Third Hospital Medical Science Research Ethics Committee. All of the procedures were performed in accordance with the Declaration of Helsinki and relevant policies in China (IRB:00006761-M2021003).
