Abstract
Objective:
This study was designed to reevaluate and improve the quality and safety of the chemotherapy preparation in a Central Chemotherapy Preparation Unit of a Public Hospital.
Methods:
A failure modes, effects, and criticality analysis (FMECA) was conducted by a multidisciplinary team. All potential failure modes at each stage of the chemotherapy preparation were recorded, and the associated risks were scored for their severity, occurrence, and detectability with a risk priority number (RPN). Corrective actions were suggested, and new RPNs were estimated for the modified process.
Results:
Failure modes, effects, and criticality analysis and priority matrix construction, revealed that the partial compliance of Unit’s premises with international standards (RPNstage: 307), the human errors throughout the compounding (RPNstage: 223)—labeling (RPNstage: 216)—prescribing (RPNstage: 198) steps, and the violation of working protocols by employees (RPNstage: 215), were the most important risks for which either urgent or immediate corrective actions had to be taken. Modifying the procedure through the proposed corrective actions is expected to lead to a significant (71.3%) risk containment, with a total RPNpreparation process reduction from 2102 to 604.
Conclusions:
Failure modes, effects, and criticality analysis and priority matrix development identified and prioritized effectively the risks associated with chemotherapy preparation allowing for the improvement of health services to cancer patients.
Introduction
The acute serious adverse events experienced by cancer patients, as well as the potential long-term mutagenic and teratogenic effects of cytotoxic drugs for both patients and health professionals, require the careful planning and efficient organization of the three basic steps of chemotherapy treatment: prescription, preparation, and administration.
Many errors that have led to ineffective treatment or serious damage to patient’s health have been documented and apply to all steps following chemotherapy prescription, including incorrect medication or doses administration, errors in infusion times and rates, wrong administration route, omission of medication or lack of hydration and inappropriate drug solutions preparation.1-3
Health care workers who prepare or administer chemotherapy solutions, may be exposed to hazardous agents either by inhaling or by contact on work surfaces, contaminated clothing and medical equipment. Several studies have connected workplace exposures to hazardous drugs with significant health problems, such as skin rashes and adverse reproductive outcomes (including infertility, spontaneous abortions, and congenital malformations) and possibly leukemia or other neoplasms.4,5
In order to reduce errors and minimize the risk for both patients and health care professionals, strategies have been developed, such as the development of dedicated central units in the Hospitals for the preparation of cytotoxic solutions, the assumption of the central compounding responsibility by the Hospital Pharmacists, the use of Class II biological safety cabinets (BSCs) or isolators, the use of closed systems so as to limit surface contamination and occupational exposure of workers, the limitation of the personnel handling cytotoxic solutions and its full training.6-13
Reduction of clinical risk through all stages of chemotherapy preparation and application is of great importance 14 and should be the primary goal for all hospitals. Clinical risk management could be achieved through the implementation of risk analysis. One of the most widespread risk analysis model is the failure modes, effects, and criticality analysis (FMECA), which is a tool for systematically assessing a complex process to identify risk-sensitive items.15,16
Implementation of risk analyses in health systems worldwide seems to be spreading in all sectors of the health care system, including the parenteral nutrition unit and the unit for the preparation of intravenous cytotoxic solutions.14,17-20 Previous studies using FMECA indicated that the centralization to the pharmacy was associated with a strong improvement, but additional developments involving information technologies also contributed to a major risk reduction for patients. 17
In our Public General University Hospital (PGHG), intravenous cytotoxic solutions for cancer patients are compounded in the Central Unit of Sterile Cytotoxic Solutions Preparation (PGHG Unit), which has been working under the responsibility of the Hospital Pharmacy since 2006, when a new process for the preparation of cytotoxic solutions for cancer patients has been launched, based on relative guidelines.21-23 The new procedure was clearly better but issues with regard to the quality of the preparations and the safety of patients, and personnel were not completely eliminated. These issues included the preservation of the sterility of the preparations, errors in the prescriptions and in their control, and rejection of prepared medicines due to compounding errors. This has prompted us to conduct an FMECA risk analysis to identify and assess the risks of our chemotherapy preparation process and to propose optimization measures for each identified failure mode. Our data highlight the importance of applying such risk assessment analyses for improving standard hospital procedures for the benefit of both patients and health care professionals.
Methods
Setting
This work was carried out at the Central Sterile Cytotoxic Solution Preparation Unit of a PGHG. Public General University Hospital in Greece Unit has been functioning under the responsibility of the Hospital Pharmacy since 2006, it is temporarily located within the Oncology Department (Satellite Pharmaceutical Unit) and serves annually approximately 15 000 oncology patients.
Implementation of FMECA in the PGHG Unit
The basic steps followed for FMECA analysis were as follows:
Recording of the stages of the cytotoxic drug solution production process, (Figure 1).
Establishment of a multi and interdisciplinary team to assess the risks associated with each of the stages of the preparation process. The team consisted of the Clinical Pharmacist-Head of the PGHG Unit, a Hospital Pharmacist—Head of the Hospital Pharmacy, two Pharmacists—University Members, two Oncologists, an Oncology Nurse and a Pharmacist—Postgraduate student who worked in the PGHG Unit, performing the diploma work required for the master’s thesis.
Failure modes, effects, and criticality analysis table construction: All possible failure modes that could occur in each step of the chemotherapy preparation process, were identified by the members of the working team and were depicted on a cause-effect plot (Isikawa/Fish bone diagram, Figures 2 and 3). Cause-effect diagram’s data were used to construct the FMECA table and formed part of it. Failure modes, effects, and criticality analysis table was filled with all the consequences of the identified failures and their possible causes. The same table outlined both the protective measures that were already in place when this study began and the new proposed measures aimed at improving the current process. Due to its large extent, the FMECA table has not been included in this article, but its important contents, such as the causes of errors, their consequences and the corrective measures proposed to improve the current production process, have been included in the Results and the Discussion sections.
Assessment of the current and the proposed preparation process. The quantification of the risk of each failure mode was based on the rating of three basic parameters, on a scale from 1 to 5. 24 That is: severity (S: Severity) of each failure mode’s consequences, probability to happen (O: Occurrence), and possibility to be early detected (D: Detectability).
The S, O, and D values were used to calculate the risk priority number (RPN) according to the equation:
Risk Priority Number = Severity × Occurrence × Detection
The maximum RPN for each failure mode is 125 and the minimum is 1. 16 The failure modes that require attention are those with RPN values equal to or greater than the half of the evaluation scale (>62.5). However, due to the FMECA subjectivity, the analysts may highlight any other point estimated to be important.
In this study, the final RPN value corresponding to each failure mode is the average given by all collaborating researchers and is referred to as RPNfailure mode (RPNfm). The total RPN value corresponding to each separate work stage (step) of the cytotoxic drug solution production process was derived from the sum of the individual values of RPNfm and is referred as RPNstage (RPNs). Finally, the sum of all values of RPNs of all stages of each preparation process gives a final RPN that characterizes the specific process and is referred as RPNpreparation process (RPNpp). Corrective measures were then proposed to address critical points, and the safety and quality of the modified process was assessed by applying the same as the aforementioned method.
Priority matrix construction: this matrix has been made to prioritize the actions needed to be implemented to improve and/or solve the problems associated with the identified failure modes.16,19 The priority matrix was divided into four color areas, corresponding to different priority levels. The red area signifies activities that require urgent action, the orange area immediate action, the yellow area includes activities for which measures must be implemented while the green corresponds to actions that warrant a simple check. Only failure modes scored with an RPN value greater than or equal to 40 (20% of the total number of failure modes) were placed in the priority matrix. 16

Brief description of the current process for the production of sterile cytotoxic drug solutions in PGHG Unit.
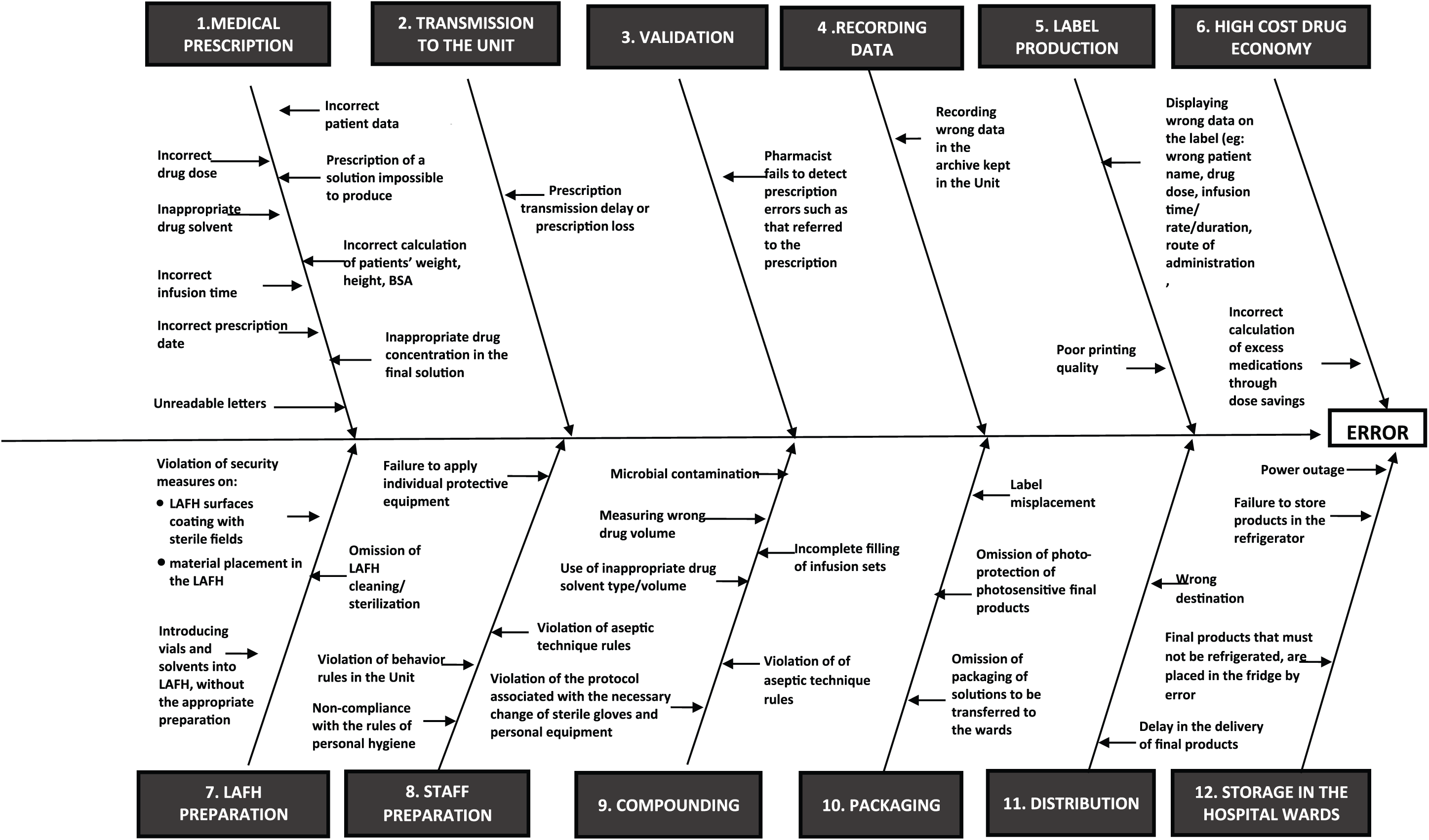
Isikawa cause-effect diagram for the main failure modes that may occur during all stages of chemotherapy preparation process in PGHG Unit

Isikawa cause-effect diagram for the the main failure modes associated with current premises/equipment and personnel working in PGHG Unit
Results
FMECA table construction
The preparation process stages and the related main potential failure modes are presented in Figures 1 and 2, respectively. Failure modes related to Unit’s premises/equipment, and problems related to employees and their exposure to hazardous substances are presented in Figure 3. Failure modes’ identification was followed by the recording of their consequences and related risks. The consequences were divided into two categories. The first category concerns the patient under chemotherapy, and the second records the staff working in the cytotoxic drug dialysis unit.
The consequences for the patient could be life-threatening bacteremia-septicemia, over- or under-dosing the patient and infusion of wrong medication solution. The incomplete filling of the infusion devices with the solution to be administered may lead to the entry of large amounts of air into the patient’s circulation resulting in pulmonary embolism and death. 25 The incidence of other failure events (Figure 2) with rather increased possibility to occur during the intravenous administration of cytotoxic solutions was associated with the infusion rate, the infusion duration, and the route of administration. These failure events might result in consequences that include induction of serious adverse events such as increased toxicity, extravasation, and alteration of the drug pharmacokinetic profile.
The consequences for the employees could be their exposure to hazardous substances. The risk of employees’ exposure to hazardous agents is related to staff shortage and the consequent stress of employees, and was assigned an RPNstage of 115 (Table 1). Main recommendations to decrease this risk have been the generation of standard operation procedures (SOPs) with regard to the handling of the medicines and the preparations by the staff, the education of the employees, the actions when such an accident happens, and the recruitment of adequate number of employees. If the recommendations were implemented, the risk is expected to be significantly reduced (new RPNstage 44, reduction 61.74%; Table 1).
Stages of the chemotherapy solutions preparation process, comparative criticality indicators (RPN) for the current and the new proposed preparation process, and risk reduction rates after taking additional corrective measures.
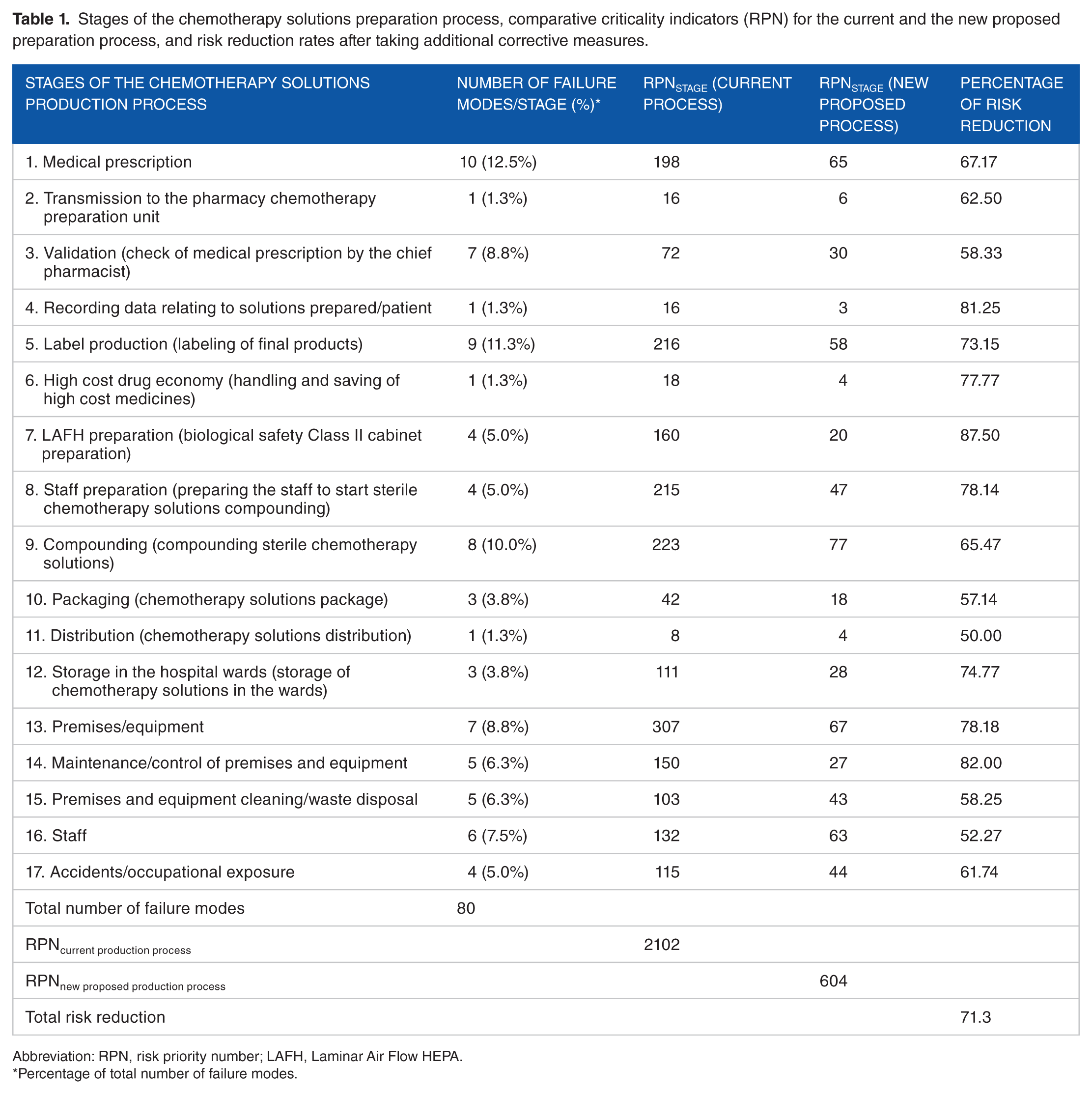
Abbreviation: RPN, risk priority number; LAFH, Laminar Air Flow HEPA.
Percentage of total number of failure modes.
Failure modes, effects, and criticality analysis table construction was completed by recording the measures that were already in place when the study began, which include the recheck of medical prescriptions by the Responsible Clinical Pharmacist, the use of written tables that include data for the proper dilution and storage of drugs, the implementation of aseptic techniques for compounding sterile solutions, tests of final products sterility, adequate staff training, the best possible communication between health care professionals, and the personal effort of each one to cope with the difficulties and to carry out his duties (Supplemental Table S1).
Among the new proposed corrective actions was the involvement of a second health professional to recheck critical steps of the procedure that could be the cause of chain errors, written protocols for each task in the unit (SOPs), lifelong training of personnel, and their skills assessment at regular intervals based on worldwide guidelines. Careful selection of employees and installation of video surveillance systems (cameras) in the workplace to monitor and verify whether the employees’ behavior comply with the given SOPs recommendations, were also proposed. Full compliance of the Unit’s facilities and equipment with international standards is also required to reduce the derogations that take place in the Unit and to improve the safety and efficiency of the preparation process (Supplemental Table S1).
Risk assessment
The most serious problem was related to the Preparation Unit’s area, which was not organized according to the international recommendations and scored RPNs value 307. Important problems and risks were also identified in (1) sterile cytotoxic drug solutions compounding (RPNs: 223), (2) final products labeling (RPNs: 216), (3) preparation of personnel for compounding solutions (RPNs: 215), and (4) medical prescription (RPNs: 198). The problems related to BSC preparation, control of premises and equipment, personnel, accidents at work, storage/preservation of final products, and cleaning facilities/waste disposal were scored RPNs values 160, 150, 132, 115, 111, and 103, respectively, with the working team recognizing significant risks in these stages for both the staff and the patients (Table 1).
The number of failure modes per stage of work, the percent proportion of all identified failure modes, RPNs values for both the current and proposed (prospective, not yet realized) preparation processes, in which the recommendations of first FMECA (Supplemental Table S1) have been implemented, as well as the cumulative RPNpp values for each of the evaluated processes are shown in Table 1. The same table presents the percent risk reduction for each one of the preparation stages under the scenario of taking additional safety measures. The current preparation process has reached an RPNpp value equal to 2102. After the implementation of the proposed corrective measures (Supplemental Table S1), the RPN value for the new preparation process is expected to receive the value 604, corresponding to a risk reduction of 71.3% (Table 1). Risk reduction is expected to occur in all stages of the current preparation process (Figure 4) in percentages ranging from 50% to 87.5% (Table 1). Most of the failure modes were identified in the stages of writing the medical prescription (13.5%), checking the medical prescription by pharmacist (9.5%), final products labeling (10.8%), and sterile cytotoxic solutions compounding (8.1%).
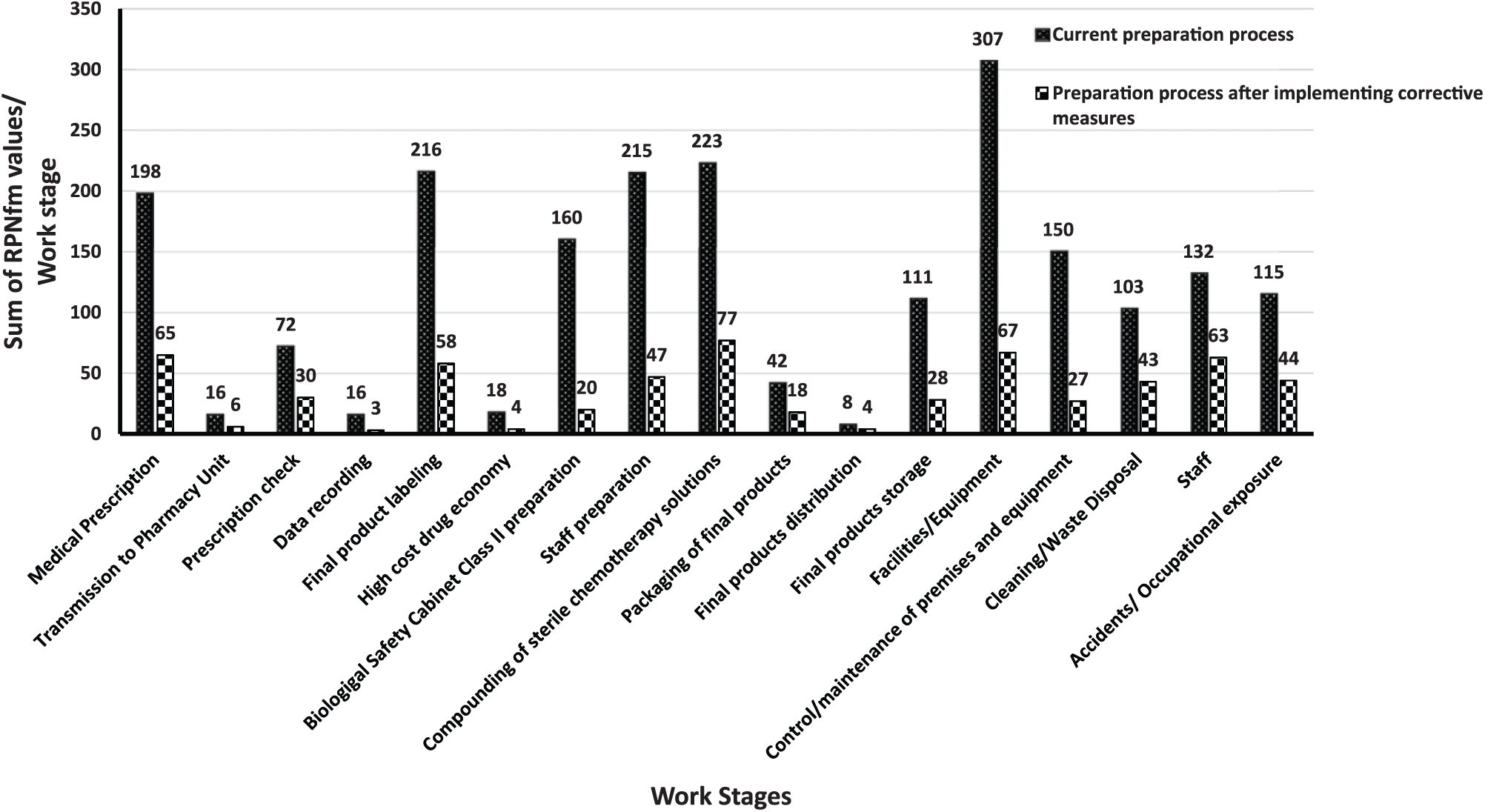
Comparison of the current chemotherapy preparation process in PGHG Unit with the process that will emerge after the implementation of new corrective measures.
Based on the above, the constructed priority matrix is presented in Figure 5, where the numbered circles correspond to the RPNs of 17 selected failure modes scored with an RPN value greater than or equal to 40 (20% of the total number of failure modes). The implementation of corrective measures (Supplemental Table S1) led to a significant shift of failure modes toward less critical areas of the matrix, indicating a reduction of the overall risk associated with the chemotherapy preparation process (Figure 4).

Priority matrix: failure modes (numbered circles) scored with RPN values ⩾40, have been positioned according to their severity, occurrence, and detectability in the four color regions of the matrix.
Discussion
The results of the FMECA risk analysis highlighted a number of failures that may occur during all stages of chemotherapy preparation process in a public hospital setting (Figures 2 and 3).
According to priority matrix data (Figure 5), actions that needed to be launched immediately and in priority are those related to the Unit’s premises and equipment, the computerization of procedures, the creation of SOPs and training of the staff. The chemotherapy preparation unit needs to be reconstructed according to international standards. Since the quality of final products manufactured in the Unit depends on stable and repeatable operating procedures, written SOPs for each process that takes place in the Unit were considered as absolutely necessary and proposed as an improvement measure. Immediate actions are required for all of the rest failure modes which have been placed in the orange area of the priority matrix. Thus, retesting of all mentioned critical preparation processes by a second health professional was also proposed and is expected to minimize the probability of errors which, if they were to occur could lead to domino effect errors in subsequent processes.
If the new proposed measures are implemented in the near future, they are expected to lead to a significant shift of failure modes toward less critical areas of the matrix (Figure 5). Although this indicates a clear improvement of quality of service, it appears that in complex processes such as that of producing chemotherapy solutions, dysfunctions do not completely disappear (Figures 4 and 5) and must always be handled with care regardless of the organization level of the Unit.
Given the strong contribution of the human factor to all stages of chemotherapy, the training of health professionals involved in prescription-preparation and chemotherapy administration is one of the most important strategies to minimize errors and it should not be overlooked by any health care provider.9-13 In this study, it is clearly shown that although training of all health professionals was already in place, its improvement was still highlighted as a corrective measure. Besides continuous and proper training, a sufficient number of employees must work in the unit to ensure smooth and uninterrupted operation, as well as prevention of professional staff burnout.
According to the FMECA results (Table 1), the most serious problem of the preparation process in the PGHG Unit was related to the partial compliance of the Unit to international standards. Although all chemotherapy solutions in PGHG Unit, are currently compounded in a certified biological safety Class II cabinet to preserve both the sterility of final products and workers safety, the area in which this cabinet is placed do not comply with the space air-purity of Grade ISO 7. 21 Furthermore, other problems have been related to the access of unauthorized hospital personnel in the unit and the daily heavy workload. Unfortunately, although the organization and operation of central units for sterile cytotoxic solutions preparation according to international standards has been a topic of discussion and a major goal of future planning in many Greek hospitals for several years, the high cost of these facilities’ construction poses a serious and almost insurmountable obstacle. This, greatly endangers the quality of all stages of the preparation process and the ability to deliver final products certified for their safety and effectiveness.
Many of the aforementioned measures are standard corrective interventions in any attempt to improve a sterile cytotoxic drug preparation unit. Thus, the investigation of the compliance of Centro di Riferimento Oncologico Unit in Aviano of Italy with the international guidelines, as well as the study of the risks associated with their preparation process, revealed that between the measures needed to be taken to avoid accidents were the computerization of medical prescriptions, the quality control of the final products, and the regular health check of the employees. 26 In the same context, Bonnabry et al 17 studied five different types of chemotherapy drug preparation unit organizations with respect to the risks involved in each work step, using FMECA. The research focused on improving the preparation process using electronic media, such as computers and special programs to perform calculations, protocol intersections, and other tasks, without detailing all dysfunctional points. Two of the studied units approximate the current state of the PGHG Unit and similarly to our work, errors relating to the compounding of sterile solutions, medical prescription, and final product labeling were scored with high RPN values.
The application of risk analyses is constantly gaining ground in the health sector. The FMECA method allows for a quantitative estimation of the relative risk for the patients and the health personnel of the different activities involved in the preparation of chemotherapy mixtures, as well as for a quantitative estimation of the risk reduction if measures indicated by the risk analysis are implemented.
Conclusions
The FMECA method and a priority matrix development have been found to be powerful tools to assess the risks associated with possible errors in the process of chemotherapy preparation in a public hospital and prioritize the actions needed to reduce such risks, allowing for the improvement of the safety of the process for both the patients and health professionals.
Supplemental Material
TABLE_S1_REVISED – Supplemental material for Improvement of Chemotherapy Solutions Production Procedure in a Hospital Central Chemotherapy Preparation Unit: A Systematic Risk Assessment to Prevent Avoidable Harm in Cancer Patients
Supplemental material, TABLE_S1_REVISED for Improvement of Chemotherapy Solutions Production Procedure in a Hospital Central Chemotherapy Preparation Unit: A Systematic Risk Assessment to Prevent Avoidable Harm in Cancer Patients by Klio Bourika, Angelos Koutras, Haralambos Kalofonos, Anna Vicha, Ekaterini Tsiata, Evangelia Papadimitriou, Konstantinos Avgoustakis and Zoi Panagi in Clinical Medicine Insights: Oncology
Footnotes
Funding:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed equally to this work.
Ethical approval
Hospital research ethics committee (reference no: 388/18.10.2016)
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
