Abstract
Background:
The risk factors for head and neck cancers (HNC) vary in different parts of the world.
Objectives:
To identify the risk factors for HNC and the correlation between these factors and the involved anatomical sites.
Methods:
We retrieved and analyzed health records of patients that met the inclusion criteria for HNC managed at our facility in a 10-year period using the International Classification of Diseases (ICD) version 10.
Results:
We studied 122 patients with a male to female ratio of 2.1:1 aged 13 years to 85 years (mean = 51 years). Alcohol (P = .02), cigarette smoking (P = .01), and cooking wood smoke (P = .01) were associated with advanced tumor stage.
Conclusions:
The strongest risk factors for HNC are alcohol, tobacco, HIV, agricultural chemicals, and cooking wood fumes in both sexes in their sixth and seventh decades.
Introduction
The primary risk factors for head and neck cancers (HNC) include the use of tobacco, ingestion of alcohol. 1 Infection with Epstein-Barr and human papilloma viruses (EBV and HPV) are independent risk factors particularly for cancers in the nasopharynx and oropharynx, respectively.1,2 The biological significance of high-risk HPV genotypes such as HPV-16 and 18 in the pathogenesis of HNC is further buttressed by the fact that patients with HPV-positive HNC exhibit better prognosis than patients with HPV-negative HNC.3,4
Without the traditional risk factors of smoking and alcohol consumption, other factors documented are radiation and occupational exposures, periodontal disease, vitamin deficiencies, dietary habits, and genetic factors.5-7
Chronic exposure of the upper aerodigestive tract mucosa to these carcinogens leads to dysplastic and premalignant changes that ultimately result in HNC. Heavy cigarette smoking and exposure to other forms of tobacco with alcohol consumption are the 2 strongest known risk factors. Acting both independently and synergistically, they cause genetic damage to squamous cells. A known tobacco carcinogen called benzopyrene diol epoxide (BPDE) induces genetic damage by forming covalently bound DNA adducts throughout the genome including cytochrome p53 mutations. 8
The prognosis of HNC will therefore depend on the exposure to these risk factors, the stage of the tumor at presentation, comorbidities, access to appropriate health care, and tumor biology. 9
However, there are worldwide geographical variations in the risk factors and the outcomes for HNC. The incidences of cancer and the death rates vary among racial and ethnic groups with a higher incidence in blacks than whites. 10 The causes for these differences are largely undefined but tumor biology and socioeconomic differences have been implicated. 11
The association between risk factors and the cause of HNC therefore signifies that the clinical courses of these cancers are modifiable following early detection and these can be achieved by screening and prevention. 12
The risk factors for HNC in Nigeria have not been adequately studied. Therefore, the primary aim of this study is to determine the risk factors for morbidity and mortality in patients with HNC in our environment with the hope that modifying identifiable risk factors from health education will help to reduce risks. The secondary aim is to determine the correlation between these factors and the head and neck anatomical sites involved.
Patients and Methods
Study design and setting
We performed a retrospective cohort study of patients with histologically confirmed HNC surgically treated at the Jos University Teaching Hospital from May 1, 2007 to April 30, 2017. This hospital is a 520-bed space tertiary referral center in north-central Nigeria sub-serving cancer management for patients in Plateau state and 6 neighboring states including the Federal Capital Territory of Abuja.
Ethical considerations
Approval for this study was sought and granted by the Institutional Health Research Ethics Committee (IHREC) of the Jos University Teaching Hospital (ethical approval number JUTH/DCS/ADM/127/XIX/6610).
Data collection
All health records of patients that met the inclusion criteria for HNC in the study period were retrieved by 4 trained health record clerks using standardized codes in the International Classification of Diseases, tenth revision (ICD-10). These records were cross checked by a head and neck oncologist to ensure reliability of the collected data.
A predesigned structured format was used to collect the data which included age, sex, occupation, duration of symptoms, exposure to risk factors, diagnosis and stage at presentation, site, histology, treatment, and outcomes of treatment.
The TNM staging system developed by the American Joint Committee on Cancer (AJCC) was used to stage the tumors. 13
Patients with incomplete health records, those who refused admission at time of presentation and who sought discharge against medical advice were excluded from the study.
Data analysis
Data generated was analyzed using EPI Info statistical software version 7.2.2.1 (EPI Info, Center for Disease Control, Atlanta, Georgia, 2017).
The independent variables considered were stage of disease at presentation (stages I to IV), the anatomical site of disease, risk factor exposure, the type of treatment received (surgery, radiotherapy, chemotherapy, or in combination), and the site of metastasis. The dependent variable was the outcome of treatment grouped into disease-free periods, recurrence-locoregional or distant spread, residual disease, or lost to follow-up.
Descriptive analysis of means and standard deviations was used to summarize the data.
The associations between variables were modeled using logistic regression analysis which was used to calculate odds ratios (ORs) and 95% confidence intervals (CIs). Statistical significance was deduced at P value of ⩽.05. We present the results in simple descriptive forms.
Result
Biodemographic characteristics
From a total 487 presentations with head and neck neoplasms (benign and malignant) in the study period, 129 (26.5%) were malignant, of which 122 (25.1%) health records met the criteria for analysis with 7 (5.4%) missing records. A total of 83 (68.0%) men and 39 (32.0%) women giving a male to female ratio of 2.1:1 were studied. Patients were aged 13 to 85 years (mean = 51. years; standard deviation = 16 years). The peak age incidence was in the sixth decade followed closely by the seventh decade of life (Table 1). Farmers (n = 45; 36.9%) were the commonest group of individuals who presented.
Sociodemographic characteristics of patients (n = 122).

Clinical presentation and site
Nasal obstruction (n = 47; 38.5%) was the commonest presenting feature. Others were neck mass (n = 29; 23.8%), hoarseness (n = 25; 20.5%), upper airway obstruction (n = 21; 17.2%), muffled voice (n = 10; 8.2%), and dysphagia (n = 6; 4.9%). Cervical lymphadenopathy with lymph node diameter >6 cm was seen in 17 (14%) patients.
The commonest tumor sites irrespective of sex were the nasopharynx (n = 34; 27.9%) (Table 2) larynx (n = 25; 20.5%), sinonasal (n = 15; 12.3%) and oropharynx (n = 15; 12.3%). The other sites are shown in Figure 1.
Tumor site distribution by sex.

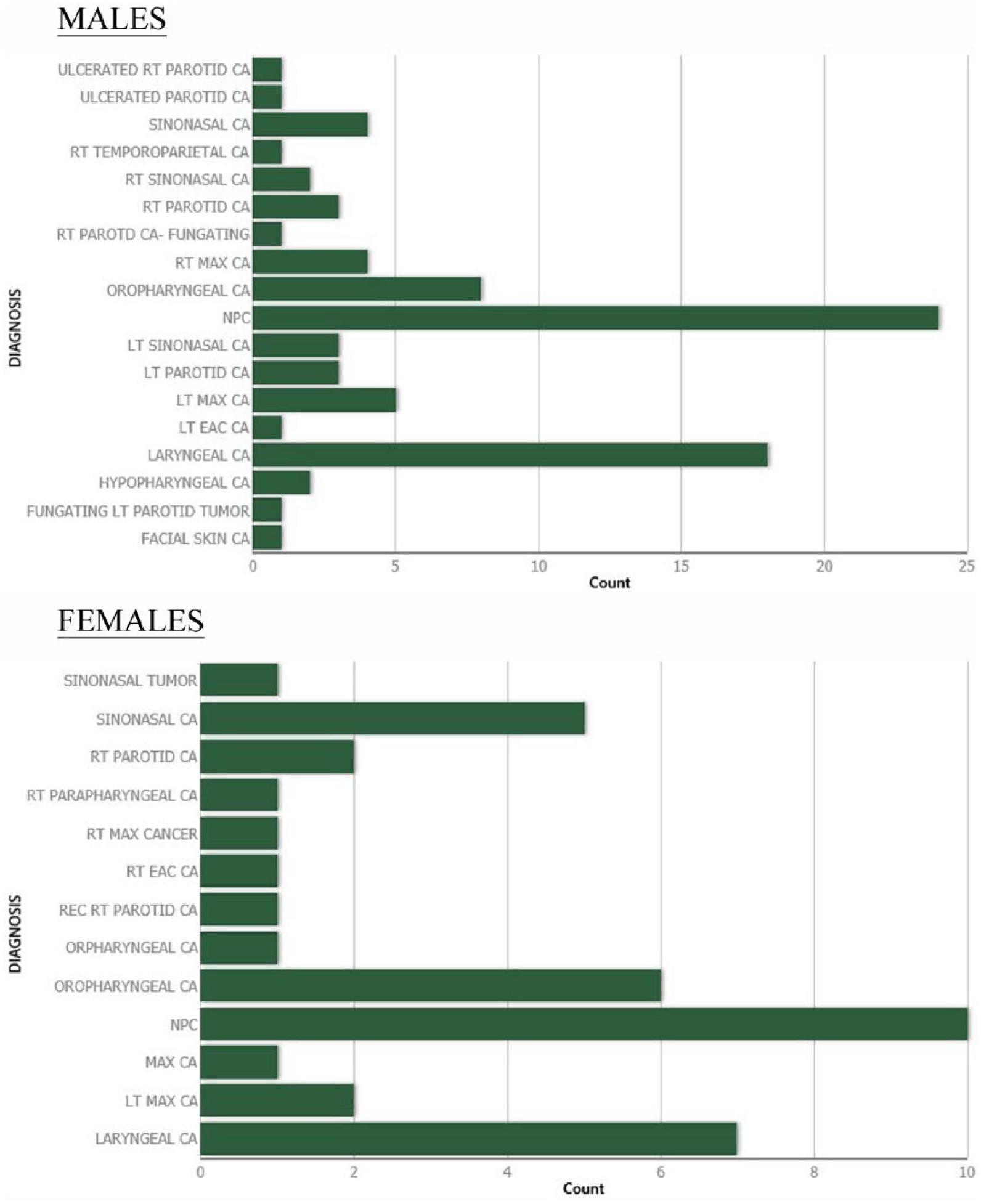
Distribution of head and neck cancer diagnosis according to sex.
The duration of symptoms ranged from 8 to 24 months (mean = 13.3 months; standard deviation = ±4.9) with a negative statistical correlation between symptom duration and tumor stage at presentation (P= .76).
Four (3.3%) patients were seropositive for the human immunodeficiency virus (HIV).
Risk factors
We recorded alcohol consumption in 49 (40.2%) patients of which 48 (98.0%) were men. In all, 27 (22.1%) patients were exposed to tobacco in various forms—19 (15.6%) smoked cigarettes and 6 (4.9%) inhaled snuff, and 2 (1.6%) chewed tobacco. No woman was exposed to tobacco. A total of 19 (15.6%) patients, all men, consumed alcohol and tobacco concomitantly. The duration of alcohol consumption was 4 to 17 years (mean = 8.1 years; standard deviation = ±11.7). The duration of cigarette smoking was 3 to 42 years (mean = 3.4 years; standard deviation = ±9.0). The cigarette pack years ranged from 2.25 to 31.5 (mean = 14.7 years; standard deviation = ±8.4).
After adjustment, logistic regression analysis revealed alcohol consumption alone was associated with advanced tumor stage at presentation (OR = 1.99; 95% CI = 1.08-3.69; P = .02). Increased cigarette pack years was associated with advanced tumor stage (OR = 3.07; 95% CI = 1.32-7.16; P = .01). In addition, concomitant alcohol and cigarette smoke exposure was also significantly associated with advanced tumor stage at presentation (OR = 1.48; 95% CI = 0.70-3.14; P < .0001). Alcohol and cigarette smoking were more associated with the development of laryngeal cancers in men but not tumors involving other sites.
There was positive family history of cancer in 9 (7.4%) patients, 6 of which were unrelated to the primary tumor. A positive family history had no statistical correlation with the stage of tumor at presentation (OR = 0.39; 95% CI = 0.10-1.28; P = .11).
Periodontal disease was recorded in 31 (25.4%) patients and 2 (1.6%) patients were exposed to wood dust of which one presented with sinonasal cancer. This, however, did not confer an association with tumor stage at presentation (OR = 0.92; 95% CI = 0.47-1.78; P = .8). Exposure to smoke from cooking wood was noted in 17 (13.9%) patients but the duration of exposure could not be established. There was a significant statistical correlation between this type of exposure in patients with advanced stage of nasopharyngeal and sinonasal tumors (OR = 3.39; 95% CI = 1.29-6.85; P = .01) in women. None of these risk factors was associated with tumors in other sites.
Serological tests for antigens to viruses such as EBV and HPV and tests for tumor biomarkers were not conducted as facilities for analysis are lacking in our hospital.
Stage and histological types
Most of patients the presented in stages II and III disease (Figure 2). Squamous cell carcinoma was the commonest histological type in 69 (56.6%) patients followed by adenoid cystic carcinoma in 30 (24.6%).

Stage of disease at presentation.
Treatment and complications
All patients had multimodal treatment consisting of examination under anesthesia and biopsy for histological diagnosis, tracheostomy, surgical excisions with cosmetic repair, and postoperative radiotherapy and/or chemotherapy.
A posttreatment complication rate of 47.5% was recorded with the commonest complication being mucositis in 29 (23.8%).
Follow-up
A total of 68 (55.7%) patients never showed up following initial treatment. The follow-up duration for those who presented was between 5 and 68 months (mean = 22.1 months; standard deviation = ±14.1).
Disease recurrence was witnessed in 23 (18.9%) patients, of which 17 (14%) were distant spread and 6 (5%) were locoregional. Recurrence was noted in patients who presented with stages III and IV diseases. The commonest site of distant metastasis was the lungs in 11 (9.0%) patients mostly from sinonasal cancers in 4 (3.3%) patients.
A total of 22 (18.0%) deaths were recorded—2 (1.6%) from complications of retroviral disease and the others were not recorded.
Discussion
The sex ratio for HNC in the developed world has narrowed over the years owing to the changes in lifestyle where heavy tobacco and alcohol use have been reported among women, a factor also responsible for the increase in the incidence of HNC worldwide. 14 A meta-analysis in Nigeria revealed a sex ratio ranging from 1:1 to 2.3:1 for which our study results of 2.1:1 male to female ratio falls within range and is similar to a report from Tanzania.15,16 The male preponderance in our society is because smoking and alcohol ingestion are more common among men than women. This is shown by the relative nonassociation of smoking and alcohol consumption and the female sex in our study. However, concomitant alcohol and tobacco use both showed statistically significant association with advanced tumor stage in laryngeal cancers at presentation in men in this study. A mean cigarette pack year of 14.7 years was reported in this study with a positive statistical correlation between it and an advanced stage of tumor presentation. Heavy tobacco use with alcohol consumption is the 2 strongest known risk factors for HNC. 8 They both act independently and synergistically to cause genetic damage to squamous cells in the mucosa of the aerodigestive tract by the covalent bonding of the carcinogen BPDE and DNA adducts throughout the genome including cytochrome p53 mutations. 8
Variations occur in the anatomic sites and subsites in HNC in the Nigerian and international population. The nasopharynx is the commonest site reported in this study. This compares favorably with the reports from most cancer registries in Nigeria according to the GLOBOCAN estimate of 2008. 17 The prevalence rate of HNC of 25.1% obtained in this study is less than the reports from studies in the southern part of Nigeria and India.18,19 These disparities may be due to the differences in the exposure to differing risk factors from one geographical region to the other.
It has been reported that HNC affects Africans at a younger age than whites with tumor biology, socioeconomic factors, and earlier exposure to risk factors given as reasons for this disparity.10,11 However, our study shows a higher occurrence in the sixth and the seventh decades of life.
Occupational exposure is a known risk factor for HNC and could explain why farmers were the largest occupational group affected in our study. It has been reported that long exposure to the sun and inhalation of the chemical components of agricultural materials which are known to contain carcinogens may be associated with the high prevalence in this group. 20 This may explain why most of the tumors in our series were of epithelial origin occurring more in the nasopharynx and the larynx.
This study did not establish an association between a positive family history of cancer and an increased risk of HNC at presentation. Negri et al 21 in their study stated that positive family history confers a 1.68-fold increased risk for HNC and shared risk factors such as lifestyle have been implicated in causing this increased risk in individuals with a positive family history of HNC. 22 Periodontal disease 23 and exposure to wood dust are other established risk factors for HNC especially oropharyngeal and sinonasal cancers but this study did not find a relationship between these risk factors and any form of HNC.
The commonest type of cooking fuel available in low- and middle-income countries is the wood which can be used in enclosures. Indoor pollution occurring from the use of these solid fuels in cooking and heating has been associated with several types of cancers, HNC inclusive with some estimated 4 million annual deaths worldwide in these countries. 24 This is a significant risk factor in our study as exposure to cooking wood is shown to be statistically correlated to advancing stage of tumor at presentation especially in women who are more exposed to the smoke from cooking wood in our environment. The exposure to exhaust fumes from diesel engines and other fossil fuels and fine particulate materials classed as carcinogens by the International Agency for Research on Cancer (IARC) are prevalent in our environment and government regulations on air pollution are nonexistent. These are factors individuals in our society are exposed to daily that are risk factors whose role in the cause of HNC have not been studied. Therefore, studies are required to establish this role and the epidemiologic characteristics thereof.
The biological significance of infectious agents such as HPV, eg, high-risk strains such as HPV-16 and 18 as risk factors in HNC especially in oral and oropharyngeal cancers, is well established. 4 However, in contrast to results from other parts of the world which show an increase in the prevalence of HPV-related HNC, studies from Africa have indicated a low prevalence.25,26 Prospective population-based studies are required to establish true figures of HPV-related HNC in Nigeria. This will also enable the appropriate commencement of vaccination for HPV in endemic regions once established. The fact that HPV testing is done in Nigeria now is a positive indication that molecular staging for HNC would be soon be available to help with early diagnosis, risk stratifying patients, and assist in the appropriate direction of treatment based on disease behavior and prognosis. 27
There are evidence in literature supporting EBV in the pathogenesis of nasopharyngeal cancer and lymphoma supported by the detection of multiple copies of EBV genome in nasopharyngeal cancer and serum antibodies to EBV antigens.28,29 Serological tests for HPV and EBV were not conducted in our study as our hospital lacks the facilities for such tests. This may have shown an association between EBV and nasopharyngeal cancer in this study apart from the probable inhalation of environmental carcinogens from the combustion of cooking wood and hydrocarbon fuels. A call therefore is made here for the availability of facilities for epigenetic studies in our hospital for these analyses which would be helpful in screening and monitoring of treatment.
The HIV infection is known to increase the risk of development of HNC. The HIV prevalence rate in HNC of 3.3% obtained in our study is lower than the report from Tanzania. 16 This low prevalence may be due to lack of adequate facilities required for the diagnosis of cancers in patients with HIV. Despite the aforementioned, this study further shows the significance of HIV in the clinical course of HNC.
Late hospital presentation is a significant factor in worsening the prognosis of HNC. 12 Most of our patients presented relatively early and this statistically was not a factor that determined patient outcome in our study.
The major limitation of this retrospective study is the poor patient health record documentation which might have introduced selection, recall, and miscalculation biases. Documentation of other risk factors such as exposure to radiation, dietary deficiencies, and genetic factors was not available in the patients’ records for this study. Despite these limitations, this study has been able to demonstrate the risk factors for morbidity and mortality in HNC in our region and the influence on the anatomic sites involved.
Conclusions
The strongest risk factors for HNC in our environment are alcohol, tobacco, HIV, agricultural chemicals, and fumes from cooking wood especially nasopharyngeal and laryngeal cancers. Alcohol, tobacco, and agricultural chemicals are risk factors in men of the sixth and seventh decade age bracket, whereas fumes from cooking wood is a significant risk factor in women of the same age group.
Facilities for epigenetic studies are needed to establish the relationship between infectious agents and HNC in our region.
Community-based studies to determine other significant risk factors are required.
Footnotes
Acknowledgements
The authors are grateful to all the staff of the Records Department of the hospital for their invaluable assistance with patients’ health records.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors reviewed and approved the final manuscript.
