Abstract
Introduction:
Reduction in bone mineral density (BMD) is a common side effect of androgen deprivation therapy (ADT). We aimed to examine the cross-sectional and longitudinal variation in BMD and associated bone markers in patients with nonmetastatic prostate cancer (PCa) managed with and without ADT.
Methods:
Bone mineral density of the total body, lumbar spine, femoral neck, ultradistal forearm, and one-third distal radius was measured in 88 patients with PCa without bone metastases at baseline and at 6 months. Patients were categorized into 4 groups: (1) acute ADT (≤6 months), (2) chronic ADT (>6 months), (3) former ADT, and (4) no ADT (controls). Serum levels of bone metabolism markers, procollagen type I N-terminal propeptide (PINP) and C-terminal cross-linking telopeptide of type I collagen (CTX), were also measured.
Results:
In the cross-sectional analysis, men receiving chronic ADT had significantly lower total body BMD as compared with former ADT users and men with no ADT. In longitudinal analysis, a significant reduction in ultradistal forearm BMD was observed in both acute and chronic ADT users after 6 months (4.08% and 2.7%, P = .012 and .026, respectively). A significant reduction in total body BMD was observed in acute ADT users (2.99%, P = .032). Former ADT users had a significant increase in both lumbar spine and femoral neck BMD (2.84% and 1.59%, P = .008 and .002, respectively). The changes in BMD were not significantly different between acute and chronic ADT users. In the cross-sectional analysis, higher levels of PINP and CTX were observed in acute and chronic ADT users than former ADT users or PCa controls. In longitudinal analysis, the level of serum PINP and CTX did not change significantly from baseline to 6 months in acute, chronic, and former ADT users, or PCa controls, and the percentage change did not differ among the 4 groups.
Conclusions:
Men on acute ADT had a similar rate of bone loss to men on chronic ADT. Reversibility in ADT-induced bone loss was observed in those who discontinued ADT. Serum levels of PINP and CTX were higher in acute and chronic ADT users and levels returned to the range of PCa controls when treatment was withdrawn.
Keywords
Introduction
Androgen deprivation therapy (ADT) is the standard treatment for metastatic prostate cancer (PCa), which can be achieved by medical castration (gonadotropin-releasing hormone [GnRH] agonist or GnRH antagonists) and, less frequently, surgical castration (bilateral orchiectomy), with or without antiandrogens. 1 It is also common practice to integrate ADT into the treatment of nonmetastatic PCa. 2 Androgen deprivation therapy is frequently used in combination with radiotherapy for locally advanced or high-risk localized disease or for those with rising prostate-specific antigen (PSA) after primary treatment.
The use of ADT has been associated with deleterious effect on bone health, resulting in accelerated loss of bone density and increased risk of bone fracture. 3 Most studies examining the association between ADT and bone mineral density (BMD) were cross-sectional and have demonstrated that patients who received ADT had significantly lower BMD at multiple skeletal sites than patients who did not receive ADT or healthy age-matched individuals.4–8 Fewer prospective longitudinal studies have been conducted to assess the effect of ADT on bone loss. Some studies showed a greater bone loss in the first 6 to 12 months of ADT use, and decline in bone density was much less prominent beyond the first year.2,9–11 In contrast, a large longitudinal study of 618 men with newly diagnosed advanced PCa showed that those with normal BMD (n = 124) or osteopenia (n = 241) at baseline had declining BMD for up to 7 years with GnRH agonist treatment, even though the latter received daily vitamin D (400 IU) and calcium (500 mg) supplements. 12 Thus, this indicates that the commonly prescribed vitamin D and calcium doses seem to be insufficient to prevent ADT-induced bone loss. Those with osteoporosis (n = 253) treated with bicalutamide and receiving vitamin D and calcium supplements showed no change in BMD over the 7-year period. 12 In another study, Lee et al 13 analyzed 12-month prospective data from 65 men with nonmetastatic PCa (42 men initiated GnRH agonist treatment at study entry, and 23 had received GnRH agonist treatment before study entry for a mean of 35 months). The authors found that total hip BMD declined steadily regardless of whether they started the study with no previous GnRH agonist treatment or having GnRH agonist treatment for ≤18 months or >18 months at study entry. 13
In a small study of 15 men on GnRH agonist who developed osteoporosis, BMD decreased further after 12 months of discontinuation of GnRH agonist, as a result of continued suppression of testosterone. 14 Recent studies indicated that intermittent ADT, which enables testosterone recovery, appeared to attenuate ADT-induced bone loss as the levels of testosterone recovered.15,16
The aim of this study was to examine the effect of acute ADT or long-term ADT on BMD and associated bone markers and to measure changes in these parameters over 6 months in a PCa cohort in New Zealand. A secondary aim was to compare bone loss in patients who discontinued GnRH agonist to patients on acute or chronic androgen suppression, as well as patients managed without ADT options.
Methods
Study participants
The participants were 88 men diagnosed with PCa without known bone metastases. They had received or were receiving various treatment options such as ADT (GnRH agonist alone or in combination with antiandrogen), radiation therapy, radical prostatectomy, and active surveillance. They were recruited between October 2014 and September 2015 and were diagnosed and/or treated in Auckland and Waikato regions. Patients aged 50 years or older and without bone metastases were eligible to participate in the study. Potential participants were contacted through specialist urology nurses and urologists from Auckland, Waitemata, and Waikato District Health Boards and private practices from these areas. A total of 250 patients with PCa registered with the Urology Department’s database at North Shore Hospital managed under the Waitemata District Health Board were invited to participate. A total of 46 patients identified from patient’s medical records from Waikato District Health Board database or attending private practices in Hamilton were also invited to take part in the study. In addition, 206 patients who have previously taken part in Urology studies at the University of Auckland were also invited to participate in this study.
Participants were excluded if they had any disease that may affect their bone health (eg, Paget’s disease, rheumatoid arthritis, hyperthyroidism, hyperparathyroidism, severe hepatic disease, or renal failure) or using medicines that may cause poor bone health or had been treated with antiosteoporotic therapy in the last 12 months (eg, bisphosphonate, estrogen receptor modulator, calcitonin, parathyroid hormone [PTH], or glucocorticoid). The flow of participants through the study is presented in Figure 1.

Flowchart of study participants.
Study design
In the first part of this study, a cross-sectional analysis was conducted to examine the association between ADT and BMD. A longitudinal analysis was then conducted to examine the association between ADT and changes in BMD. All participants from the Auckland region and one participant from the Waikato region had dual-energy X-ray absorptiometry (DXA) scans done at the University of Auckland Body Composition Laboratory located in the Auckland City Hospital. Fasting blood samples were collected before or after the scan at the Clinical Research Centre at the Faculty of Medical and Health Sciences, University of Auckland. Participants from Waikato region had their DXA scans done at Waikato Bone Density Services in Hamilton or Coromandel Bone Density Service at Thames Hospital. Their fasting blood samples were collected at Meade Clinic Centre at Waikato Hospital or Laboratory at Thames Hospital. Blood samples were collected in serum separation tubes (Becton Dickinson).
The participants were asked to attend a follow-up clinic visit 6 months later to undergo the same procedures. Fifteen participants were not considered for a follow-up visit due to various reasons. These included 7 who were placed on bisphosphonate, 1 diagnosed with bone metastases, 1 death, 1 placed on abiraterone and prednisone, and 3 withdrawals due to personal reasons arising since their baseline visit. Thus, these patients were excluded from the longitudinal analyses of the study.
The participants were categorized into 4 groups: (1) patients who were treated with GnRH agonists for 6 months or less at study entry, (2) patients who were treated with GnRH agonists for more than 6 months at study entry, (3) patients who had previously been treated with GnRH agonists but were no longer receiving the treatment or patients on intermittent ADT at study entry, and (4) patients who had not received ADT.
One participant commenced on GnRH agonist shortly after his first clinical appointment and therefore was moved to group 1 for longitudinal analysis. Two patients in group 3 received intermittent ADT, and 1 patient had received a single 6-month GnRH agonist depot once a year. One patient had antiandrogen for only 1 month and the recruitment took place 1 year after the treatment and therefore was classified as having no ADT. All the participants provided written informed consent to participate in this study. This study received ethical approval from the Northern Y Regional Ethics Committee (ethics ref: NTY/05/06/037).
Data collection
Clinical information, such as PSA at diagnosis, biopsy or postsurgery Gleason score, and tumor stage, were obtained from hospital medical records. Participants completed a self-administered questionnaire which consisted of 2 parts: first part was to record the PCa management options they have received and the details on treatment duration (data were verified with hospital records) and the second part included information specifically on their bone health, such as fracture history, family history of fracture, and whether they have any medical condition or use of medication which could affect their bone health. This part of the questionnaire also inquired about use of calcium and vitamin D supplements and is adapted from the National Osteoporosis Society, Camerton, Bath, UK.
Measurement
Bone mineral density of the L2-L4 lumbar spine, femoral neck, ultradistal forearm, one-third distal radius, and total body was measured at baseline and 6 months by DXA, using iDXA (GE Lunar, Madison, WI, USA) (for patients from Auckland region), Discovery W (Hologic Inc, Bedford, MA, USA) (for patients from Gisborne), and XR-800 or XR-600 densitometer (Norland, Cooper Surgical Company, Fort Atkinson, WI, USA) (for patients from Waikato region). As T score of the one-third distal radius was not available for participants who had their DXA scans done at Hamilton, this study used the T score of the lumbar spine or femoral neck (whichever was lower) to classify patients into normal (greater than −1 SD), osteopenic (between −1 and −2.5 SD), and osteoporotic (−2.5 SD or less) groups, according to the World Health Organization criteria.
Testosterone, follicle-stimulating hormone (FSH), luteinizing hormone (LH), estradiol (E2), PTH, and 25-hydroxyvitamin D were measured on serum samples by automated immunoassays on the Roche Cobas e411 (Hitachi High-Technologies Corporation, Tokyo, Japan) at the Liggins Institute, University of Auckland. Intra-assay coefficients of variation (CVs) were 2.50% to 2.74% for testosterone, 1.16% to 1.18% for FSH, 1.57% to 1.64% for LH, 2.15% to 2.75% for estradiol, and 2.20% to 2.45% for PTH. Serum procollagen type I N-terminal propeptide (PINP) and serum C-terminal telopeptide of type I collagen (CTX) were also measured on the automated Cobas e411 analyzer (Hitachi High-Technologies Corporation) at the Liggins Institute, the University of Auckland. Intra-assay CVs were 3.23% to 3.64% for PINP and 1.56% to 2.22% for CTX.
Statistical analysis
Baseline participant characteristics, BMD parameters, the levels of sex steroid hormones (estradiol and testosterone), gonadotropins (LH and FSH), 25-hydroxyvitamin D, PTH, and bone turnover markers (PINP, CTX) were compared between acute or chronic ADT users, former ADT users, and PCa controls. Continuous variables were expressed as median and range because they were not normally distributed. Comparisons of continuous variables among groups were performed by the Kruskal-Wallis test. If there was a significant difference, testing was followed with the Wilcoxon 2-sample test for pairwise comparison. Categorical variables were expressed as frequencies and percentages and were compared between all ADT users (including former ADT users) and PCa controls using the Fisher exact test. Proportion test was used to compare differences in proportion of osteoporosis, osteopenia, and normal BMD among the groups.
Least squares (LS) mean values with corresponding standard error of the means were used to assess the percentage change in BMD from baseline to 6 months. Changes in BMD within each group were analyzed using paired Student t test. The percentage of change for each parameter was compared across groups using the Kruskal-Wallis test and followed, if significant, by Wilcoxon 2-sample test for pairwise comparison. A P value of ≤.05 was considered significant. All statistical analyses were conducted using SAS (v9.4 SAS Institute, Cary, NC, USA).
Results
Baseline characteristics of participants are presented in Table 1. There were no significant differences in age, height, weight, and body mass index across the 4 groups at study entry. The average duration of GnRH agonist treatment in acute ADT group was 4 months (range: 3-6) and that of chronic ADT group was 22.2 months (range: 7-74). For the men on acute ADT, 3 were on GnRH agonist monotherapy and 4 were on combined androgen blockade (GnRH agonist with antiandrogen). For the men on chronic ADT, 19 were on GnRH agonist monotherapy and 11 were on combined therapy. For former ADT users, 15 had GnRH agonist monotherapy and 6 had combined therapy. Most of the men on ADT or former ADT users had also undergone radiation therapy (71.4% in acute ADT group, 73.3% in chronic ADT group, and 90.5% in former ADT users). In the PCa control group, 10 patients had undergone radical prostatectomy, 9 patients had received radiation therapy, 3 patients had received both prostatectomy and radiation therapy, and 8 patients were on active surveillance.
Baseline characteristics of the 4 groups of patients with prostate cancer.

Abbreviations: ADT, androgen deprivation therapy; BMI, body mass index; GnRH, gonadotropin-releasing hormone; PCa, prostate cancer.
Comparison of clinical and lifestyle factors between ADT users (including former ADT users) and PCa controls is presented in Table 2. At baseline, 7 patients were on calcium supplementation and 14 patients were on vitamin D supplementation. There were no significant differences in calcium and vitamin D supplement use between ADT users (including former ADT users) and PC a controls. At baseline, 15 patients had prior fragility fractures (1 in acute ADT group, 4 in chronic ADT group, 5 in former ADT users, and 5 in control group). Most of these fractures had occurred before the diagnosis of PCa (n = 11, 73.3%), and the proportion of fractures occurred after diagnosis of PCa was 26.7% (n = 4). The median PSA levels at diagnosis and Gleason scores were significantly higher in ADT users than PCa controls (P < .0001).
Clinical and lifestyle factors of ADT users and PCa controls.

Abbreviations: ADT, androgen deprivation therapy; PCa, prostate cancer; PSA, prostate-specific antigen.
Baseline BMD and prevalence of osteoporosis and osteopenia
There was a significant difference in total body BMD across the groups (Table 3). Bone mineral density of the total body was significantly lower in the chronic ADT group than in PCa controls and former ADT users (median 1.096 g/cm2 vs 1.214 and 1.188, respectively). There was weak evidence for differences in BMD at the lumbar spine and one-third distal radius across the groups (P = .094 and P = .098, respectively). There were no significant differences in BMD of the femoral neck and ultradistal forearm across the 4 groups (P = .242 and P = .17).
Baseline BMD of the 4 groups of patients with prostate cancer.

Abbreviations: ADT, androgen deprivation therapy; BMD, bone mineral density; PCa, prostate cancer.
Results are presented as median (range).
At baseline, the rates of osteoporosis and osteopenia were 28.6% and 28.6% in acute ADT group, 20% and 60% in chronic ADT group, 23.8% and 42.9% in former ADT users, and 10% and 36.7% in men with no ADT, respectively, at the site of worst BMD (Table 4). The differences in rates of osteoporosis between the groups were not significant. However, men on chronic ADT had a significantly lower rate of having normal BMD than hormone-naïve men (20% vs 53.3%, P = .007), at the site with worst BMD.
Frequency of osteoporosis, osteopenia, and normal BMD of the 4 groups of patients with prostate cancer.

Abbreviations: ADT, androgen deprivation therapy; BMD, bone mineral density.
Serum hormone levels
There were significant differences in baseline testosterone, estradiol, LH, and FSH levels across the groups (all P < .0001) (Table 5). As expected, testosterone levels were significantly lower in acute and chronic ADT users compared with former ADT users and PCa controls (0.048 and 0.063 ng/mL vs 3.78 and 4.34 ng/mL, respectively). Baseline levels of estradiol were significantly lower in acute and chronic ADT users than former ADT users and PCa controls (5 and 5 pg/mL vs 24.40 and 23.24 pg/mL). Serum LH and FSH levels were significantly lower in acute and chronic ADT users compared with former ADT users and patients with PCa with no ADT. However, there were no significant differences in serum PTH levels across the 4 groups. Serum 25-hydroxyvitamin D levels also did not differ significantly among patients who were or were not receiving ADT (24.1-32.55 ng/mL, P = .305).
Baseline levels of hormones and bone turnover markers.

Abbreviations: ADT, androgen deprivation therapy; CTX, C-terminal telopeptide of type I collagen; FSH, follicle-stimulating hormone; LH, luteinizing hormone; PINP, procollagen type I N-terminal propeptide.
Results are presented as median (range). Testosterone: 1 ng/mL = 3.5 nmol/L.
Serum levels of bone turnover markers—PINP and CTX
There were significant differences in serum PINP (P = .0005) and CTX (P = .0057) levels across the groups (Table 5). Serum PINP levels were significantly higher at baseline in acute and chronic ADT users than in PCa controls (76.78 and 70.61 ng/mL vs 48.48 ng/mL). Chronic ADT users also had a significantly higher level of PINP compared with former ADT users (70.61 vs 46.07 ng/mL). Serum CTX levels were significantly higher at baseline in men receiving acute or chronic ADT as compared with former ADT users or PCa controls (0.72 or 0.60 ng/mL vs 0.39 or 0.46 ng/mL, respectively).
Changes in BMD
Of the 88 patients at baseline, 15 patients discontinued during the 6-month study, including 1 patient on acute ADT, 5 patients on chronic ADT, 5 former ADT users, and 4 PCa controls. After 6 months, former ADT users had a significantly increased BMD at lumbar spine (+2.84%, P = .0076). There was no significant change from baseline in lumbar spine BMD in acute ADT users (−0.17%, P = .87), chronic ADT users (−0.27%, P = .72), and PCa controls (+0.18%, P = .75). The BMD changes at lumbar spine differed significantly across the groups (P = .0412). The increase in lumbar spine BMD in former ADT users was significantly different compared with other 3 groups (Figure 2). The femoral neck BMD increased significantly from baseline in former ADT users (+1.59%, P = .002), whereas no significant change from baseline was observed in acute ADT users (−1.52%, P = .42), chronic ADT users (−1.40%, P = .16), and PCa controls (+0.49%, P = .46). The femoral neck BMD changes was differed significantly across the groups (P = .0435). The increase in femoral neck BMD in former ADT users was significantly different compared with chronic ADT users (Figure 2).
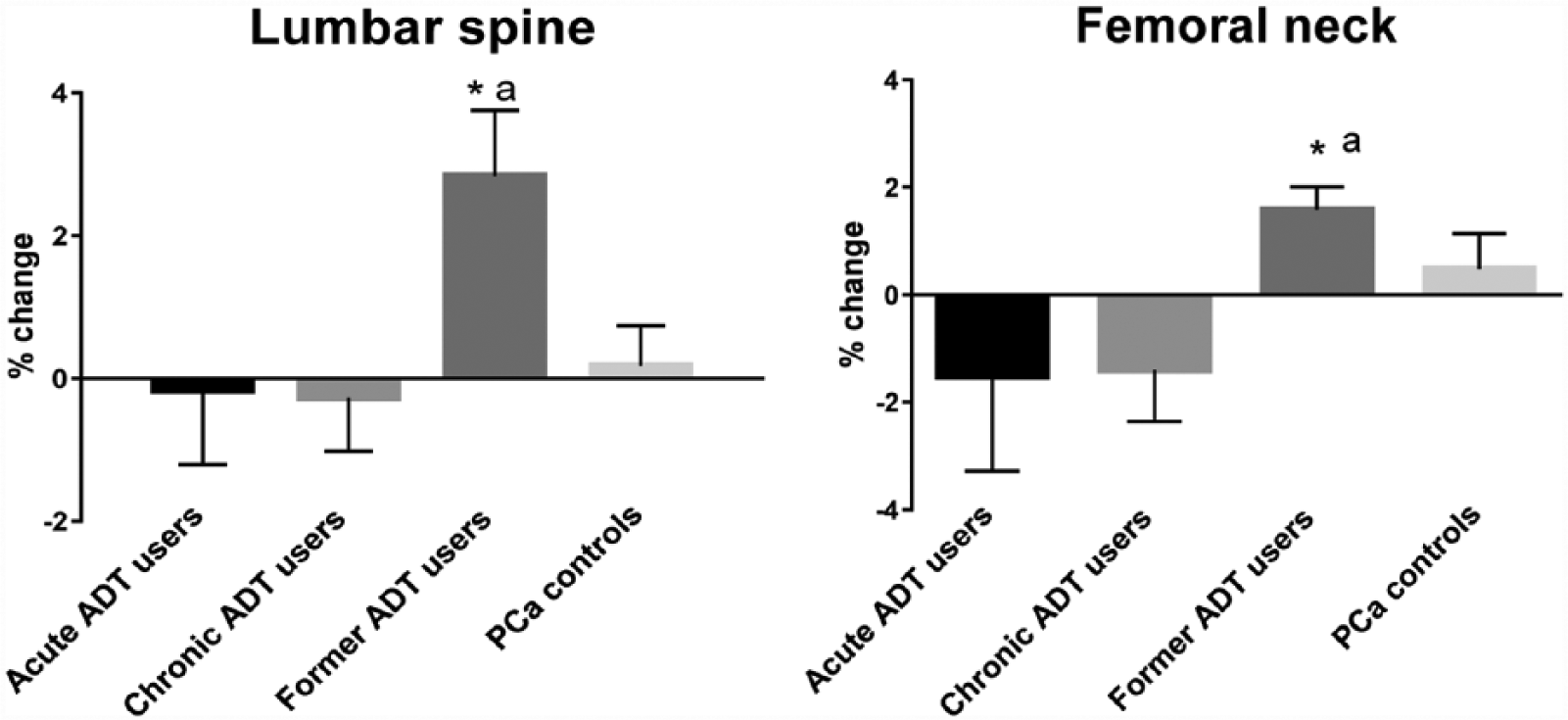
Least squares mean percentage change (with SE) from baseline in lumbar spine and femoral neck BMD. ADT indicates androgen deprivation therapy; BMD, bone mineral density; PCa, prostate cancer.
A trend for increased BMD at one-third distal radius was observed in PCa controls (+2.06%, P = .094), whereas no significant change from baseline was observed in acute ADT users (−0.86%, P = .43), chronic ADT users (−0.37%, P = .55), and former ADT users (+0.23%, P = .78). There was no significant difference in the BMD changes at one-third distal radius across the groups (P = .313) (Figure 3). Men on acute and chronic ADT had a significant reduction in BMD at ultradistal forearm (−4.08%, P = .012 and 2.70%, P = .026, respectively) from the baseline. A reduction in ultradistal forearm BMD was also observed in PCa controls, but the change did not reach statistical significance (−2.7%, P = .26). A borderline significant difference in the ultradistal forearm BMD changes was observed across the group (P = .071) (Figure 3). Men on acute ADT had a significant reduction in total body BMD (−2.99%, P = .032). There was a borderline significant reduction in BMD of the total body in PCa controls (−0.643%, P = .053), and no significant change from baseline was observed in chronic ADT users (−1.14%, P = .19) and former ADT users (+0.13%, P = .82). There was a borderline significant difference in the total body BMD changes across the groups (P = .0623) (Figure 3).

Least squares mean percentage change (with SE) from baseline in one-third distal radius, ultradistal forearm, and total body BMD. ADT indicates androgen deprivation therapy; BMD, bone mineral density; PCa, prostate cancer.
Changes in serum levels of bone turnover markers—PINP and CTX
The LS mean percentage changes in serum PINP and CTX are shown in Figure 4. Serum levels of PINP and CTX did not change significantly from baseline to 6 months in all 4 groups. The LS mean changes in PINP were −8.30%, +1.62%, +11.91%, and −3.40% among acute ADT users, chronic ADT users, former ADT users, and PCa controls, respectively. For serum CTX, the corresponding LS mean changes from baseline were −10.75%, +3.56%, −6.53%, and +2.07%, respectively. The percentage change in serum PINP and CTX levels did not differ significantly across the groups (P = .967 and P = .833, respectively).

Least squares mean percentage change from baseline in serum PINP and CTX. ADT indicates androgen deprivation therapy; CTX, C-terminal telopeptide of type 1 collagen; PCa, prostate cancer; PINP, procollagen type I N-terminal propeptide.
Discussion
The results from the cross-sectional analysis in this study demonstrated that men receiving chronic ADT have significantly decreased total body BMD than former ADT users and men with no ADT. This is consistent with previous cross-sectional BMD studies with androgen suppression in patients with PCa.5,6,17 In addition, we also observed that former ADT users had a significant increased BMD, indicating reversibility in ADT-induced bone loss.
In this study, the rate of osteoporosis at baseline was lower in men who did not receive ADT than men in the other 3 groups at baseline, but the differences were not significant. This is consistent with earlier cross-sectional studies which compared the prevalence of osteoporosis between ADT-treated patients and patients who did not receive ADT.18–20 In the cross-sectional study conducted by Morote et al, 18 a significant association between ADT and osteoporosis occurred only when comparing hormone-naïve patients with patients who were treated with GnRH agonist for 6 years or more. 18 The average duration of ADT in this study was limited (approximately 22 months for the chronic ADT users), and in most of the earlier cross-sectional studies, durations were shorter; this may be a reason why a significant association between ADT and osteoporosis was not observed in these studies including ours. Wei et al 19 examined bone density in 8 patients with PCa who were about to commence ADT and 24 patients who received ADT for more than 1 year. The authors detected osteoporosis in 38% of the ADT-treated patients and 25% in patients who had not yet started on ADT. 19 Other studies in Western populations reported the prevalence of osteoporosis to be more than 40% in men receiving ADT, which is higher than the prevalence reported in this study. For example, the study by Morote et al 18 reported that 42.9% of the patients who had been treated with ADT for 2 years (n = 112) had osteoporosis, whereas 35.4% of hormone-naïve patients had osteoporosis (n = 124). In another study, Morote et al 20 detected osteoporosis in 41.5% of the patients who received ADT for at least 12 months (n = 53) and 28.1% in patients who did not receive ADT (n = 57). In this study, 53.3% of the patients with no ADT had normal BMD, which was significantly higher than the rate in men on chronic ADT (20%). In concordance with our study, Morote et al 20 also reported a significantly higher rate of normal BMD in patients with no ADT than those receiving ADT (28.1% vs 13.2%, P = .035). In a retrospective study, Bruder et al 21 determined the prevalence of osteoporosis and osteopenia in 89 patients with PCa undergoing ADT for a mean duration of 2.7 ± 2.5 years. With BMD measurement of the spine and hip, 26.9% and 50.6% of the patients had osteoporosis and osteopenia, respectively. 21 Similarly, in this study, 20% and 60% of the men on chronic ADT and 23.8% and 42.9% of the former ADT users had osteoporosis and osteopenia, respectively.
The longitudinal analysis from this study demonstrated that reduction in BMD at the measured sites ranged from 0.17% to 4.08% after 6 months in men receiving acute ADT. However, only the reduction in BMD at ultradistal and total body reached significance. The reduction in BMD ranged from 0.27% to 2.70% in chronic ADT users, and only the BMD reduction at ultradistal forearm reached significance. The greatest bone loss occurred in ultradistal forearm in both acute and chronic ADT users. In the 12-month longitudinal study conducted by Mittan et al, 22 bone loss was also maximal at forearm (5.3% in ultradistal radius) in men treated with GnRH agonist, which is consistent with the present finding. It has been shown that forearm BMD is the strongest predictor of osteoporotic fracture in men. 23 The ultradistal region of the forearm has a greater proportion of trabecular bone (65%) than femoral neck (25%) or total hip (50%).12,22 As trabecular bone is more metabolically active than cortical bone, it therefore may be more responsive to ADT-related effects. Lumbar spine has a similar proportion of trabecular bone (66%) to the ultradistal forearm, but in this study, significant reduction in lumbar spine BMD was not observed in acute or chronic ADT users. This may be due to higher prevalence of spinal osteoarthritis or aortic calcification in older patients, which may falsely elevate BMD at lumbar spine and mask bone loss due to aging or ADT. Taken together, forearm is a preferred site for monitoring bone loss in patients with PCa.
In this study, the changes in BMD were not statistically different across the groups. In contrast, the 12-month longitudinal study conducted by Greenspan et al 9 showed that BMD reduction at the total body, total hip, and trochanter in acute ADT users was significantly different compared with men on chronic ADT, men with PCa not receiving ADT, and healthy controls. The differences become statistically significant as early as 6 months for total body BMD. The relatively shorter term of follow-up (~6 months) and small number of acute ADT users in this study may explain the differences in the results observed.
Unexpectedly, BMD at lumbar spine and femoral neck increased significantly over 6 months in former ADT users, and the increase in lumbar spine BMD was significantly different compared with other groups. This indicated that ADT-induced bone loss appears to be reversed in patients whose testosterone levels recovered. Indeed, BMD recovery has been demonstrated in men on intermittent ADT. In a small longitudinal study of 56 men with nonmetastatic, hormone-sensitive PCa, Yu et al 16 investigated BMD changes during intermittent ADT. In their study, patients experienced the greatest change/decline in BMD during early treatment periods of intermittent therapy (ie, during the first on-treatment period), and the changes were smaller thereafter. The authors observed that BMD recovery at the lumbar spine was significant during the first off-treatment period (+1.2%, P = .001). 16 Another study investigated changes in BMD over 3 years of intermittent ADT in 72 patients with PCa without bone metastases. The authors showed that BMD declined during the 9 months of initial treatment, but further decline in the off-treatment period was attenuated due to testosterone recovery. Failure of recovery of testosterone to more than 5 nmol/L was associated with worse final BMD. 15
Several observational studies have assessed markers of bone turnover in patients with PCa treated with ADT and most have observed an increase in both bone formation and resorption markers in ADT-treated patients compared with nontreated men.4,5,24–26 The cross-sectional analysis of baseline data in this study demonstrated that both markers of bone formation and bone resorption (PINP and CTX, respectively) were significantly higher in men on acute or chronic ADT as compared with former ADT users and PCa controls, indicating an increased bone turnover in those with ongoing ADT. Our observation is consistent with several other cross-sectional studies in which men treated with ADT for at least 6 months had higher levels of bone formation markers, such as bone-specific alkaline phosphatase (BSAP), and osteocalcin and bone resorption marker, such as urinary N-terminal propeptide type 1 collagen (NTX), than PCa controls or healthy controls.4,5,8,24,26
Varsavsky et al 27 assessed 2 bone formation markers, BSAP and osteocalcin, and 2 bone resorption markers, CTX and tartrate-resistant acid phosphatase isoform 5b (TRAP-5b), in 42 PCa patients treated with ADT in comparison with 41 patients with PCa not on ADT. The authors observed higher CTX and BSAP levels in patients receiving ADT as compared with patients with no ADT after age adjustment. 27 Another cross-sectional study conducted by the same group measured 1 bone formation marker, BSAP, and 2 bone resorption markers, CTX and TRAP-5b, in 25 patients with PCa treated with ADT, 34 patients with PCa without ADT, and 22 healthy controls. In that study, Garcia-Fontana et al 28 observed an increased serum CTX levels in ADT-treated patients compared with healthy controls and increased serum BSAP levels in PCa patients with and without ADT compared with healthy controls.
In this study, the level of serum PINP did not change significantly from baseline to 6 months in acute, chronic, and former ADT users, or PCa controls, and the percentage change did not differ among the 4 groups. In contrast, the 12-month longitudinal study by Greenspan et al 9 observed an elevation in PINP in acute ADT users after 6 months compared with patients with PCa not on ADT and healthy controls, but not compared with chronic ADT users. According to the authors, PINP showed a difference between acute ADT users and chronic ADT users after 12 months. 9 The longitudinal analysis of this study did not indicate elevated markers of bone formation and bone resorption in acute ADT users as reported by Greenspan et al. 9 In another 12-month longitudinal study, Mittan et al 22 measured serum and urinary levels of bone turnover markers in patients with PCa initiating GnRH agonist therapy and compared these levels with age-matched healthy controls. In their study, levels of a marker of bone resorption, urinary NTX, were increased at 6 or 12 months compared with baseline in patients with PCa receiving GnRH agonist compared with controls. The authors assessed BSAP and osteocalcin as bone formation markers over 12 months but did not detect significant changes in these makers of bone formation. 22 In this study, there was a nonsignificant reduction in serum PINP and CTX observed in acute ADT users after 6 months of follow-up. However, the small sample size in this group means low statistical power for these analyses. We observed a significant increase in lumbar spine and femoral neck BMD from baseline in former ADT users, which was reflected by a nonsignificant increase in PINP and reduction in CTX after 6 months of follow-up.
This study had some limitations that should be mentioned. First, patients were not randomized to receive GnRH agonist. A second limitation was the small sample size, particularly the small number of acute ADT users. This may have led to missing statistically significant changes in BMD, particularly in acute ADT users. However, even with such a small sample size, we were able to detect significant changes in BMD at the ultradistal forearm and total body. Third, the follow-up study period was relatively short. A fourth limitation is that BMD was measured by DXA instruments from different manufacturers. However, each patient had their repeat BMD measurements performed on the same scanner.
In conclusion, men receiving ADT had a similar rate of bone loss during initial and long-term ADT, and the greatest decline occurred in the ultradistal forearm. This study also demonstrated that ADT-induced bone loss appeared to be reversed in patients who discontinued the treatment. Serum levels of PINP and CTX were higher in acute and chronic ADT users, and levels returned to those seen in PCa controls when treatment was withdrawn. The results suggested that it is important to monitor testosterone recovery after ADT.
Footnotes
Acknowledgements
The authors would like to thank Ms Bobo Hu and Ms Ying Huang for statistical advice and Mr Eric Thorstensen for facilitating with the immunoassays.
Peer review:
Three peer reviewers contributed to the peer review report. Reviewers’ reports totaled 1295 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A.W. is funded by a scholarship provided by the Sarah FitzGibbons Trust. The study was funded through a research grant from the Prostate Cancer Foundation New Zealand.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AW, NK, and RL conceived and designed the experiments and jointly developed the structure and arguments for the paper. AW analyzed the data and wrote the first draft of the manuscript. AW, NK, LP, SZ, SO, KB, CB, TS, JM, MH, RH, CK, LF, and RL agree with manuscript results and conclusions and made critical revisions and approved final version. All authors reviewed and approved the final manuscript.
Disclosures and Ethics
As a requirement of publication, author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality, and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
