Abstract
Background:
Dipeptidyl peptidase-4 (DPP-4) inhibitors are oral hypoglycemic agents widely prescribed in India despite safety concerns. However, studies focused on their safety profile are scarce, especially in South India.
Objective:
To evaluate the prevalence and predictors of adverse events (AEs) with DPP-4 inhibitors in patients with type 2 diabetes mellitus (T2DM).
Research design and methods:
This retrospective cross-sectional study analyzed data from medical records of T2DM patients prescribed DPP-4 inhibitors admitted to the medicine department from 2019 to 2021 at a South Indian tertiary care hospital. The causality of AEs was assessed using the WHO-Uppsala Monitoring Centre (WHO-UMC) criteria and the Naranjo scale, and severity using the Modified Hartwig and Seigel scale. We applied a Generalized model with a binary response and logit-link function to understand the factors that best explain the AE. The best-fit models were chosen based on least Akaike’s information criterion and highest PseudoR2 and presented the odds ratio (OR) with a 95% confidence interval. The analyses were performed in R software version 4.2.1.
Results:
Among the 796 patients included in the study, 26% experienced AEs. A total of 212 AEs were observed, and Saxagliptin-associated AEs were the most prevalent (66.6%). Hepatic AEs were predominant (37.7%), followed by gastrointestinal events (16.5%) and electrolyte imbalances (12.3%). Most AEs were possible based on WHO-UMC criteria (78.7%) and the Naranjo scale (86.7%), with 58% being of moderate severity and 42% mild. In the multivariate analysis, aspartate transaminase [OR: 1.013 (0.006–1.020)], alkaline phosphatase [OR: 1.004 (1.001–1.007)] and patients already on DPP-4 inhibitors [OR 1.191(1.012–1.366)] were significant predictors for AEs with DPP-4 inhibitors.
Conclusion:
The study highlighted a high prevalence of AEs with DPP-4 inhibitors and identified significant predictors of these AEs. These findings underscore the necessity of vigilant monitoring and risk assessment while prescribing DPP-4 inhibitors to the Indian population.
Plain Language Summary
Introduction:
DPP-4 inhibitors are a class of drugs used to manage type 2 diabetes mellitus. These drugs are commonly prescribed regardless of their safety issues in the Indian population. The studies focusing on the side effects or adverse events associated with these drugs and the contributing factors are limited in India. Understanding how common these adverse events are is vital to providing better patient care and management.
Aim:
To assess the frequency of adverse events with DPP-4 inhibitors and the contributory factors to these events.
Method:
A retrospective study analyzing the medical records of diabetic patients on DPP-4 inhibitors admitted to a major hospital in South India between 2019 and 2021 was conducted. The frequency, severity and potential causes of adverse events were identified through descriptive analysis.
Result:
A total of 796 diabetic patients on DPP-4 inhibitors were included in the study, out of which 26% (212 adverse events) experienced adverse events. Most common adverse events were related to liver (37.7%) followed by gastrointestinal (16.5%) and electrolyte imbalance (12.3%). The severity of the events was moderate (58%) and mild (42%). Elevated liver enzymes aspartate transaminase and alkaline phosphatase, and patients already on DPP-4 inhibitors were at high odds of developing adverse events.
Conclusion:
The study identifies DPP-4 inhibitor-associated adverse events and contributory factors that should be addressed when prescribing these drugs to diabetic patients.
Introduction
India, the most populous country in the world, is a major diabetes hub, with 77.4 million adults with diabetes, constituting 14% of the global diabetes population. 1 About 90% of all diabetic cases account for type 2 diabetes mellitus (T2DM), a chronic progressive condition characterized by insulin insensitivity. 2 Insulin insensitivity is related to insulin resistance and pancreatic beta-cell dysfunction. The insulin levels are initially maintained within the normal range through the compensatory increase in insulin secretion. As the disease progresses, alterations in the beta cells and the decline in insulin secretion result in hyperglycemia. 3
The effective management of diabetes involves a comprehensive strategy typically encompassing lifestyle changes, pharmacological approaches, and diligent monitoring to attain and maintain optimal blood sugars while mitigating potential adverse outcomes. 4 Dipeptidyl peptidase-4 (DPP-4) has gained attention due to its ability to improve glycemic control with favorable safety and tolerability profile among the various antidiabetic agents available. 5
The rationale for the initial investigation of DPP-4 inhibitor as a potential antidiabetic drug is due to the strong validation of glucagon-like peptide-1 (GLP-1) for managing diabetes, coupled with the discovery that the enzyme DPP-4 plays a critical role in regulating GLP-1. DPP-4 inhibitors in animal models have demonstrated elevated levels of active GLP-1 in the bloodstream, supporting the hypothesis that DPP-4 blockade results in higher GLP-1 levels. 6 DPP-4 inhibitors act by preventing the degradation of incretin hormones GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) by the DPP-4 enzyme, increasing levels of active GLP-1. They stimulate insulin release and block glucagon production in a glucose-dependent manner, reducing hypoglycemia risk. 7
Evidence suggested that DPP-4 inhibitors may be more effective in Asians than Caucasians,8 -10 potentially attributed to the differences in the underlying pathophysiology of T2DM. Asian diabetic patients often exhibit a phenotype characterized by altered pancreatic beta cell function and lower body weight (emaciation), while Caucasians have a phenotype marked by insulin resistance and obesity. 11
DPP-4 inhibitors were marketed in 2006 and have gained much popularity in recent years, establishing themselves as a better second-line oral hypoglycemic agent. 12 Sitagliptin was the first US-Food and Drug Administration(US-FDA) approved DPP-4 inhibitor, followed by Saxagliptin (2009), Alogliptin (2010), and Linagliptin (2011), while Vildagliptin was approved by European Medicines Agency (EMA) in 2007. Other gliptins like Anagliptin, Gemigliptin, and Teneligliptin were approved in Japan, South Korea, and India and released in 2012. Evogliptin, Omarigliptin, Trelagliptin and Gosogliptin are newer DPP-4 inhibitors released in 2015 and approved in South Korea, Japan, and the Russian Federation, respectively. 13 Currently, Sitagliptin, Saxagliptin, Linagliptin, Vildagliptin, teneligliptin and Gemigliptin are the DPP-4 inhibitors available in India. 14
While clinical trials have stated the safety and efficacy of DPP-4 inhibitors, real-world evidence is crucial to understanding their performance in general clinical practice. Adverse drug reactions (ADR) are a major public health concern that may result in increased hospital stays, morbidity, mortality, and economic burden. 15 Studies have reported that the average cost incurred for a patient for hospitalization due to ADR was INR 4915 (USD 115), 16 and the median hospital stay was 5 days (95% CI: 5.37-7.11) 17 in India. There are reports of serious AEs such as bullous pemphigoid, hypoglycemia, acute pancreatitis, hepatic disease, intestinal obstruction, interstitial lung disease, arthralgia, and acute renal injury with DPP-4 inhibitors. 18 Nevertheless, data on the safety aspects of these agents are limited based on population studies in the Indian setup.
Reports on ADRs with DPP-4 inhibitors in India are scarce, and it is challenging to generalize the safety data from clinical trials due to individual patient variations. Assessing the prevalence of ADRs with DPP-4 inhibitors will enhance treatment outcomes, safeguard patient well-being, and empower healthcare providers to tailor treatment approaches. This study will help in the optimal use of DPP-4 inhibitors by identifying the most prevalent ADR among Indian diabetic patients. Additionally, recognizing the risk factors precipitating these ADRs will aid in appropriate prescription practices among vulnerable populations. Hence, the present study aimed to determine the prevalence of AEs with DPP-4 inhibitors and the factors contributing to these AEs in diabetic patients in South India.
Methodology
Study design
This retrospective cross-sectional study was carried out in a South Indian tertiary care teaching hospital. The data of type 2 diabetic patients admitted to the general medicine department between 2019 and 2021 were collected from the medical records department. The study was initiated after the Institutional Ethics Committee approval (IEC-764/2021) and adhered to the Declaration of Helsinki. Informed consent from the patients was not necessary due to the retrospective study design. The study report follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, 19 and the checklist is attached as Supplemental File 1.
Patients and data collection
All type 2 diabetic patients [International Classification of Diseases (ICD)-10 coding E11] aged >18 years treated with DPP-4 inhibitors admitted to the hospital between 2019 and 2021 were included in the study. Patients with type 1 diabetes mellitus, ADRs confirmed with drugs other than DPP-4 inhibitors, pregnant women and lactating mothers, and immunocompromised patients were excluded. The sample size (n) was calculated based on the pilot study conducted to understand the prescribing pattern of DPP-4 inhibitors in our hospital setting. We used the single proportion population formula: n = [z2 × p(1 − p)]/d2, where z is the standard score (2.576 for 99% CI), p is the proportion of DPP-4 inhibitor prescribed (.439), d is the absolute precision (.05) to calculate the sample size and was found to be 653 [(2.5762 × 0.439 × 0.561)/0.0025]. We included an additional 20% sample size to address the missing or incomplete data. The minimum sample size required was found to be 784.
The patient data was collected in a predesigned data collection form after obtaining the ethical committee’s approval. The data were collected from the hospital medical records retrospectively, including the patient demographic characteristics such as age, gender, body mass index (BMI), social habits, diabetes duration, medical history, medication history, comorbidities, diabetic complications, current medications and so on. BMI was classified according to the revised consensus guidelines for India as follows: underweight (<18.5 kg/m2), normal or lean BMI (18.5-22.9 kg/m2), overweight (23.0-24.9 kg/m2), and obese (⩾25 kg/m2). 20 Clinical parameters like fasting blood sugar (FBS), random blood sugar (RBS), glycated hemoglobin (HbA1c), renal function test, urine analysis lipid profile, and liver function test were collected.
Identification and characterization of suspected AEs
The ADRs were identified based on any event reported as ADR in the patient’s medical record or was considered as an AE for any abnormal symptoms or abnormal changes in the laboratory values suspected for which either the drug was unchanged, withheld or changed in the prescription orders. The details on ADR/AE were collected, including the suspected type of ADR/AE, the onset of ADR/AE, DPP4 inhibitor used, dose and duration of the drug, abnormal laboratory investigations, changes in the reported symptoms, date of drug withdrawal and concomitant use of other medications. The causality of the AEs between the suspected AE and drug was assessed using the WHO-Uppsala Monitoring Centre Criteria (WHO-UMC) 21 and the Naranjo et al 22 algorithm. The WHO-UMC criteria contain 6 terms to evaluate the ADRs: certain, probable/likely, possible, unlikely, unclassified/ conditional, and unclassifiable/unassessable. This includes 4 critical factors for assessing causality: assessing the temporal relation between the drug and the suspected AEs, evaluating the presence of any other potential causes like other drugs or underlying medical conditions, monitoring the response to dose reduction or drug withdrawal (dechallenge) and observing the response to drug readministration (rechallenge). The Naranjo scale is a questionnaire designed to determine whether the drug is more likely to cause ADR than other causative factors. It consists of 10 questions about the drug involved and the type of reaction observed. Each question is scored separately (−1, 0, +1, +2), and the total score ranges from −4 to +13. Based on the total score, the likelihood of the drug being associated with the reaction is categorized into 4 levels: definite (total score > 9), probable (total score 5-8), possible (total score 1-4), and doubtful (total score ⩽ 0). The study focused on AE reports that are classified as certain, probable or possible in causality assessment. The severity of the AEs was assessed using the modified Hartwig et al 23 and Seigel scale. It categorizes ADRs into 7 levels (from 1 to 7) based on their severity, ranging from mild (levels 1 and 2) to moderate (levels 3, 4a, and 4b) and severe and life-threatening reactions (levels 5, 6, and 7).
Outcomes measured
The prevalence of AEs among DPP-4 inhibitor users was calculated using the given formula,
We used logistic regression to assess the factors causing AEs with DPP-4 inhibitors.
Statistical analysis
We used descriptive statistics, employing mean and standard deviation or median and interquartile range for continuous variables and frequencies or numbers with percentages for categorical variables. The missing values were handled using the linear trend at point method. We applied a Generalized Linear Model (GLM) with a binary response and logit-link function to understand the factors that best explain the AEs. To address multicollinearity, we used Pearson’s correlation for continuous variables and the chi-squared test for categorical variables to assess the relationships between independent variables. Covariates with a correlation coefficient (r) greater than .5 were considered correlated and removed from the candidate models to prevent overfitting. Prior to analysis, continuous variables were standardized to z-scores. Initial univariate analyses were performed to identify factors that contributed to AE preliminarily. Subsequently, significant factors (P < .25) identified in the univariate analyses were incorporated into multivariate regression analyses, along with other potential predictors. The best-fit models were chosen based on the least Akaike’s Information Criterion (AIC) 24 and the highest Pseudo R 2 25 and presented the odds ratio (OR) of the affected variables with a 95% confidence interval. The P-value of <.05 was considered statistically significant. All the analyses were performed in R software version 4.2.1. R Foundation for Statistical Computing, Vienna, Austria: https://www.R-project.org/.
Results
A total of 4125 case records of diabetic patients admitted over 2 years were reviewed from the medical records department. Even though the minimum sample size required for our study was 784, we collected data from 898 diabetic patients receiving DPP-4 inhibitors. Following the exclusion of 102 patients due to incomplete records, 796 eligible type 2 diabetic patients on DPP-4 inhibitors were included in the study.
Demographics and clinical characteristics of the study population
The study population were predominantly males, that is, 462(58%), with a mean age of 59.56 ± 9.53 years. The median duration of diabetes was found to be 9.74 years (IQR: 4-15). The demographic details of the patients are provided in Table 1.
Demographic details of the study participants.

Comorbidities and complications associated with T2DM
Of the 796 patients, 513 (64.4%) patients had hypertension as the major comorbidity, followed by 81 (10.2%) patients with hypothyroidism, 37 (4.6%) with dyslipidemia, 28 (3.5%) with bronchial asthma, 24 (3%) with pulmonary tuberculosis and so on. Among the microvascular complications, 107 (13.4%) had diabetic kidney disease(DKD), followed by 31 (3.9%) with diabetic neuropathy and 17 (2.1%) with diabetic retinopathy. About 43 (5.4%) patients had cardiovascular complications, 28 (3.5%) had cerebrovascular complications, 12 (1.5%) had diabetic foot infections as macrovascular complications. And 3 (0.4%) patients had diabetic ketoacidosis. The percentage of diabetic complications among the study population is given in Supplemental File 2.
Prescribing pattern of DPP-4 inhibitors with other antidiabetic agents among the study population
Our study population observed an average of 6.08 ± 2.4 drugs per prescription, and among these, the mean number of antidiabetic agents prescribed was 3.03 ± 1.01. About 333(41.83%) of the patients were on triple antidiabetic therapy, followed by 196 (24.63%) patients on quadruple therapy, 191(24%) on dual therapy, 48(6%) on monotherapy, 25 (3.14%) patients on quintuple therapy and 3 (0.4%) patients on sextuple therapy. About 48(6%) patients had DPP-4 inhibitor monotherapy, and 106 (13.3%) had a combination therapy of DPP4 inhibitor with metformin. The majority of the patients were on combination therapy of DPP-4 inhibitor + metformin + sulfonylurea, that is, 209 patients (26.3%) followed by 106 (13.3%) patients on DPP-4 inhibitor + metformin + sulfonylurea + insulin, 106 (13.3%) patients on DPP-4 inhibitor + metformin, 86 (10.8%) patients on DPP-4 inhibitor + metformin + insulin, and so on. The various antidiabetic drug combinations given to the study population are presented in Supplemental File 3.
Among the DPP-4 inhibitors, Teneligliptin 530 (66.6%) was the most prescribed, followed by Vildagliptin 179 (22.5%), Sitagliptin 63 (7.9%), Linagliptin 21 (2.6%), and Saxagliptin 3 (0.4%). About 634 (79.6%) of the patients were on metformin along with DPP-4 inhibitors followed by 462 (58.04%) on sulfonylureas, 317 (39.8%) on insulins, 101 (12.7%) on alpha-glucosidase inhibitors, 27 (3.4%) on thiazolidinediones, 17 (2.1%) on SGLT-2 inhibitors and 1 (0.1%) on GLP-1 receptor agonist.
Prevalence of adverse events among the study population
In 796 T2DM patients, 212 AEs were observed in 207 patients. Of the 207 patients with AEs, 202 had a single AE, whereas 5 patients developed 2 AEs. The prevalence of AEs was 26% in our study population. The organ system most affected by the AEs was the hepatic system 80 (37.7%), followed by the gastrointestinal (GI) system 35 (16.5%), electrolyte imbalance 26 (12.3%), hypoglycemia 19 (8.9%) and so on. Most of the AEs were associated with Saxagliptin (66.6%), followed by Sitagliptin (31.7%), Linagliptin (28.6%), Vildagliptin (26.8%) and Teneligliptin (25.7%). AEs were observed more in males [121 (58.6%)] than in females [86 (41.5%)]. The patients aged <60 years 111(53.6%) were having more AEs compared to those aged ⩾60 years 96(46.4%). The majority of AEs were observed in patients on polypharmacy (those taking 5 or more drugs), accounting for 141 (68.1%) cases. The number of AEs related to individual DPP-4 inhibitors and the organ system affected is detailed in Table 2.
The number of adverse events associated with individual DPP-4 inhibitors and the organ system affected.

Increased creatinine phosphokinase and pedal edema.
The AEs were categorized based on causality according to the WHO-UMC criteria and Naranjo scale, and the severity was assessed based on the modified Hartwig and Seigel scale. Most AEs were considered possible based on WHO-UMC criteria [167 (78.7%)] and the Naranjo scale [184 (86.7%)]. About 45 events were probable based on WHO-UMC criteria and 28 based on the Naranjo scale. No definite AE was identified. The severity assessment of the AEs using the modified Hartwig and Seigel scale showed no severe AEs. 123 (58.1%) AEs were moderately severe [Level 3: 59 (27.8%), Level 4a: 23 (10.8%), Level 4b: 41 (19.3%)] and 89 (41.9%) were of mild severity [Level 1: 3 (1.41%), Level 2: 86 (38.7%)]. The frequency of AEs with the abnormal laboratory parameters and their causality and severity assessment is detailed in Supplemental File 4.
Factors contributing to DPP-4 inhibitor-induced adverse events
We explored variables that were predictive of having an AE with DPP-4 inhibitors. In the univariate analysis, aspartate transaminase [AST] (P = .0005), triglycerides [TG] (P = .0254), high-density lipoprotein [HDL](P = .0658), total cholesterol/HDL[TC/HDL] Ratio (P = .0395), alkaline phosphatase [ALP ] (P = .00588), alanine transaminase [ALT] (P = .0064), thyroid stimulating hormone [TSH] (P = .0744), high-density lipoprotein] LDL(P = .1), sodium (P = .105), serum creatinine (P = .114), systolic blood pressure (P = .127), serum potassium (P = .1454), estimated glomerular filtration rate (P = .232) were found statistically significant (P < .25). In the multivariate analysis, the model with the lowest AIC and highest Pseudo R 2 values suggested AST [OR: 1.013 (0.006–1.020)], ALP [OR: 1.004 (1.001–1.007)], and DPP-4 inhibitor use [OR: 1.191 (1.012–1.366)] as the statistically significant predictors for the occurrence of AE with DPP-4 inhibitors. The OR for alcohol use was 1.410 (0.985–1.698) but was found to be non-significant (P = .0575). The top 5 models with lower AIC and other parameters that explain the factors causing AEs with DPP-4 inhibitors are depicted in Table 3.
Top 5 models with lower AIC and associated parameters that explain the factors contributing to adverse events with DPP-4 inhibitors.
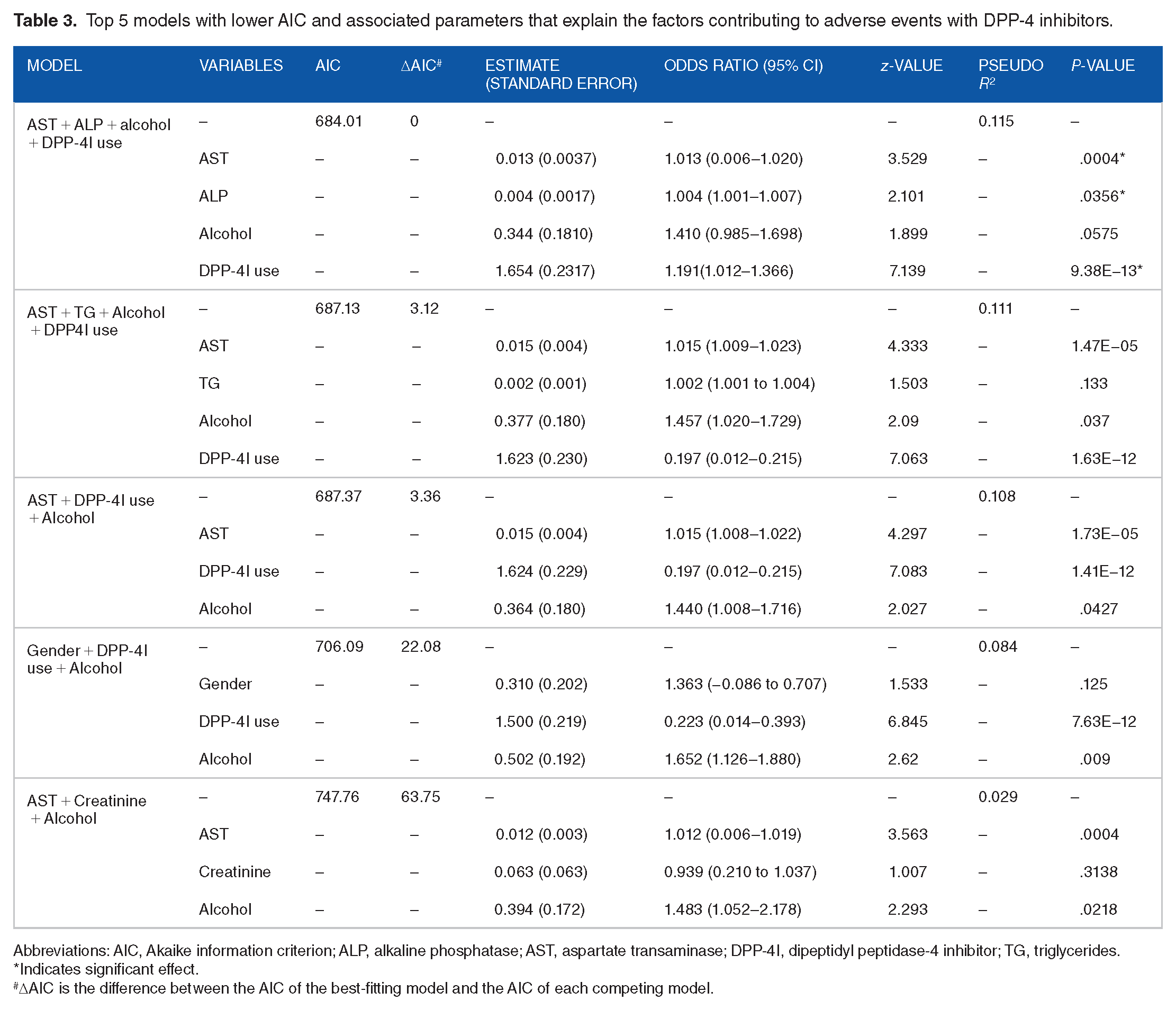
Abbreviations: AIC, Akaike information criterion; ALP, alkaline phosphatase; AST, aspartate transaminase; DPP-4I, dipeptidyl peptidase-4 inhibitor; TG, triglycerides.
Indicates significant effect.
ΔAIC is the difference between the AIC of the best-fitting model and the AIC of each competing model.
Discussion
DPP-4 inhibitors were widely prescribed as antidiabetic agents and were regarded as the primary choice following metformin until recently. They effectively lowered blood glucose, had relatively low hypoglycemia risk, neutral body weight effects, and a favorable safety profile. 26 As ADRs significantly contribute to increased hospitalizations and prolonged hospital stays, it is crucial to address them. Our study emphasizes the prevalence of AEs associated with DPP-4 inhibitors and identifies significant contributing factors to these AEs.
In our study, 58% were males having type 2 diabetes on DPP-4 inhibitors, similar to other studies where male predominance was observed.27 -29 The average age of our study participants was 59.56 years, which aligns with findings from a South Indian study by Gill et al 30 that investigated the safety profile of second-line agents in uncomplicated type 2 diabetes.
Hypertension emerged as the predominant comorbidity in our study population, aligning with results reported in other studies.31,32 We observed a lower prevalence of dyslipidemia among our study population, inconsistent with other Indian studies. ICMR-INDIAB study reported an 81.2% weighted prevalence of dyslipidemia among Indians. 33 Similarly, Parikh et al 34 reported a higher prevalence of 97.8% among females and 85.5% among males. Previous studies indicate that in the Asian population, dyslipidemia is more prevalent in urban areas compared to rural ones.35 -37 The ICMR-INDIAB study revealed significant variability in dyslipidemias across different states and regions and was more prevalent in urban areas. South Indian states like Karnataka, Tamil Nadu, and Andhra Pradesh had lower hypercholesterolemia (15%-24.9%) and high LDL (0%-14.9%) prevalence in the rural areas. 33 A South Indian study by Mohanraj et al 38 also showed a lower prevalence of dyslipidemia among rural and sub-urban areas compared to urban populations. The variation in dyslipidemia prevalence in our study could be due to factors such as the study setting, which attracts a higher number of patients from rural areas in Karnataka for disease management, along with demographic changes, lifestyle, and dietary practices. This regional variation may warrant further research on how the difference in lipid disorders might influence AEs and treatment outcomes in future. The majority of patients exhibited DKD as the primary complication associated with diabetes, followed by diabetic retinopathy, cardiovascular complications (IHD), diabetic neuropathy, and so forth. A prospective study conducted by Swami TN in South India found that ischemic heart disease was the most reported diabetic complication in contrast to our findings. 39
The average number of drugs per prescription in our study was 6.08 ± 2.4, and previous studies reported an average of 2 to 5 drugs per prescription.40,41 The average antidiabetic drug prescribed in our study was found to be 3.03 ± 1.01. Similarly, Tiwari et al 42 and Agarwal et al 43 reported an average of antidiabetic drugs per prescription as 2.89 and 1.4, indicating strict control of polypharmacy. Most of our patients were on antidiabetic combination therapy, where triple therapy was the most prominent, followed by quadruple and dual antidiabetic therapy, aligning with the findings by Das et al 44 in East India. In our study, Teneligliptin emerged as the most prescribed DPP-4 inhibitor, whereas Elnaem et al 45 reported that Sitagliptin was the most frequently prescribed among the Malaysian population. The increased prescription of Teneligliptin may be attributed to its cost-effectiveness compared to other DPP-4 inhibitors. 46
ADRs are a significant concern impacting patient safety and treatment outcomes. Comprehending the causality and severity of ADRs is essential for optimizing medication management and reducing potential harm. Our methodology included causality and severity assessments using validated tools. We used the WHO-UMC criteria and the Naranjo scale for causality assessment and the modified Hartwig and Seigel scale for severity assessment. Most of the AEs observed were possible and had moderate severity. No permanent harm or death due to AEs was identified. The prevalence of AEs found in the current study was 26%, where 207 patients had at least 1 AE. This was higher compared to the long-term post-marketing surveillance studies on the efficacy and safety of Vildagliptin, Sitagliptin, and Teneligliptin, with a prevalence of 13.6%, 47 6.3%, 48 3.85% 49 respectively.
Hepatic AEs were the most observed, followed by GI AEs, electrolyte abnormalities and hypoglycemia. Studies have suggested that DPP-4 inhibitors were well tolerated in liver disease; however, they reported elevated liver enzymes with Sitagliptin, Vildagliptin and Saxagliptin therapy.50,51 Contrary to most studies, we reported GI AEs as the second most common AE among our study population. The GI intolerance associated with DPP-4 inhibitors appears to rely partially on the motility effects of GLP-1 and GIP, along with other gastric hormones like pituitary adenylate cyclase-activating peptide and oxyntomodulin. 52 Acute pancreatitis, a rare ADR associated with DPP-4 inhibitors, was observed only in 1 patient on Vildagliptin therapy. The occurrence of electrolyte imbalance, namely, hyponatremia and hyperkalemia, was observed in our study participants. Diabetes is recognized as one of the conditions linked to a higher likelihood of electrolyte abnormalities. This association is often attributed to several factors commonly found in individuals with diabetes, such as impaired renal function, malabsorption syndromes, acid-base disorders, and the utilization of multidrug regimens. 53 Previous reports have documented cases of hyponatremia linked to sulfonylureas, thiazolidinediones, and GLP-1 receptor agonists,54 -56 but there is no known prior incidence of DPP-4 inhibitor-related hyponatremia. A recent study comparing the risk of hyperkalemia between SGLT-2 inhibitors and DPP-4 inhibitors showed an increased risk of hyperkalemia with DPP-4 inhibitors. 57 DPP-4 inhibitors are less likely to cause hypoglycemia than other antidiabetic agents due to their mechanism of action, which involves prolonging the action of native GLP-1, thereby stimulating glucose-dependent insulin secretion from pancreatic beta-cells. 28 We observed an incidence of 8.9% of hypoglycemic AEs in our study. Most of these events can be attributed to the concurrent use of sulfonylureas or insulin in our study population. Anemia was an adverse event in 7.6% of our study population, with half of the participants having DKD, where anemia is a common complication. A retrospective cohort study suggested that the DPP-4 inhibitor ameliorates hemoglobin decline in DKD. However, reports of anemia with DPP-4 inhibitors have not been reported, but a case report of thrombocytopenia with Vildagliptin was previously reported. 58
Although infections like upper respiratory tract infections and urinary tract infections were reported with DPP-4 inhibitors, we observed only 1.9% of cases in our study population. The mechanism by which infections are associated with DPP-4 inhibitors is connected to immune regulation by DPP-4. Specifically, DPP-4 inhibition can stimulate T cells to produce transforming growth factor-β1, inhibiting T cells from producing inflammatory cytokines and impacting cell development, differentiation, and death, potentially altering immune responses and increasing the susceptibility to infections. 59 The incidence of infections can be attributed not only to the use of DPP-4 inhibitors but also to the underlying diabetes. 60 Similarly, only 2 cases of bullous pemphigoid, a rare ADR of DPP-4 inhibitors, with Teneligliptin were identified among our study participants. DPP-4 expression is widespread in the dermis, and inhibition of DPP-4 is linked to various skin conditions like bullous pemphigoid, psoriasis, and atopic dermatitis. 61 The development of bullous pemphigoid associated with DPP-4 inhibitor use can be attributed to the role of DPP-4 in converting plasminogen to plasmin. One proposed mechanism suggests that DPP-4 inhibitors prevent plasmin from cleaving collagen XVII, leading to the failure of immunotolerance against collagen and the production of autoantibodies against specific collagen epitopes. 62 Additionally, DPP-4 inhibition may enhance the proinflammatory chemokine activity, such as CCL11/exotoxin, contributing to blister formation. DPP-4 inhibition could also interfere with keratinocyte migration and delay wound healing. 61
Our study also focused on identifying the factors causing AEs with DPP-4 inhibitors. Most of the AEs were observed in males in our study population. In contrast, studies have suggested that the incidence of ADRs was high in females,63,64 connected to various physiological factors like their lower body weight and size, higher fat content, decreased gastric emptying and gastric pH, and reduced glomerular filtration that will impact the kinetics of the drug. 65 Nevertheless, the study by Watson et al, 66 which examined ADR reports among females and males using data from the WHO global database, VigiBase, indicated a higher occurrence of severe and fatal ADR reports among male populations. We observed that patients <60 years had more AEs compared to those ⩾60 years, in contrast to the long-term study by Kadowaki et al, 67 where patients more than 65 years experienced higher ADR incidence. Additionally, AEs were more prevalent among patients undergoing multiple drug therapies in our population, highlighting the impact of polypharmacy on the occurrence of ADRs. 68 The findings from the scoping review by Reghunath et al 69 suggested that the main risk factors for DPP-4 inhibitor-associated ADRs were liver disease, age over 65, female gender, grade 4 and 5 renal impairment, current DPP-4 inhibitor use, concurrent insulin therapy, and disease duration (more than 10 years).
In the univariate analysis, AST, TG, TC/HDL ratio, ALP, ALT, and TSH exhibited a strong association with ADR occurrence. We employed a model selection approach in the multivariate analysis, considering the AIC and Pseudo R 2 values. The model with the lowest AIC and highest Pseudo R 2 suggested AST, ALP, and patients already on DPP-4 inhibitors as the significant predictors of DPP-4 inhibitor-induced AEs. The AST and ALP levels as risk factors can be owing to the fact that hepatic AEs with DPP-4 inhibitors were the most observed among our study population. A study by Nishida et al 70 found that DPP-4 inhibitors (Alogliptin, Sitagliptin, and Linagliptin) resulted in a substantial decrease in ALT and AST levels over a 3-month period. Similarly, Kusunoki et al 71 demonstrated a substantial reduction in AST, ALT and ɣ-glutamyl transpeptidase(ɣ-GT) concentrations with combination therapy of DPP-4 inhibitors and SGLT-2 inhibitors. These studies indicate that certain gliptins may have a hepatoprotective effect, contrasting our findings regarding ALT and ALP as risk factors for DPP-4 inhibitor-induced AEs. However, further studies are warranted to understand the relationship between DPP-4 inhibitors and hepatic function and to determine the clinical implications of these findings.
The old use (who were already taking the drug) of DPP-4 inhibitors was another significant risk factor associated with AE incidence. Studies have shown that the incidence of arthralgia 72 and acute liver injury 73 with DPP-4 inhibitors was higher in DPP-4 inhibitor users for more than 1 year. Since the details on the duration of DPP-4 inhibitor therapy were unavailable, we couldn’t confirm the effects of chronic or long-term use. Also, alcohol intake was found to be a contributing factor but was not statistically significant. Evidence suggests that the incidence of ADR is high among current users of alcohol, with intake of 40 g of alcohol per day. 74
Our study had some limitations as the ADR-related data were taken from medical records of a tertiary care center; the potential for under-reporting of suspected ADRs and other patient characteristics can’t be neglected. There were challenges in determining the causality of the suspected reactions as the rechallenge test was undertaken rarely. So, we considered the suspected adverse reactions as AEs rather than ADRs. Also, we could not perform the time-to-event analysis due to the unavailability of the details on DPP-4 inhibitor initiation. Additionally, due to the single-center approach, our study’s external validity and generalizability to a wider population are limited. The power analysis of the study was not performed, assuming a larger sample size. Despite the retrospective nature of our study, a significant segment of the study population can still provide valuable insights for drawing appropriate conclusions. The study results can help healthcare practitioners identify patients at higher risk of experiencing ADRs with DPP-4 inhibitors, allowing for more personalized and effective clinical care. Long-term prospective multicentre studies are warranted to assess further the safety profile of DPP-4 inhibitors in Indian diabetic patients. In future, machine learning-based risk prediction models using large electronic health records or databases to identify patients at higher risk of developing AEs or ADRs with DPP-4 inhibitors can be developed and validated.
Conclusion
Our study identified a 26% prevalence of AEs with DPP-4 inhibitors, with hepatic AEs being the most observed, followed by GI AEs, electrolyte abnormalities, and hypoglycemia. Most AEs were considered possible and had moderate severity. Factors such as AST, ALP, and patients who were already on DPP-4 inhibitors were statistically significant predictors for ADRs, whereas alcohol intake was statistically non-significant. These findings underscore the necessity of vigilant monitoring and risk assessment while using these drugs in the Indian population, ensuring safer prescription practices.
Supplemental Material
sj-docx-1-end-10.1177_11795514241288645 – Supplemental material for Prevalence and Predictors of Adverse Events Associated With Dipeptidyl Peptidase-4 (DPP-4) Inhibitors in Type 2 Diabetic Patients: A Cross-sectional Study
Supplemental material, sj-docx-1-end-10.1177_11795514241288645 for Prevalence and Predictors of Adverse Events Associated With Dipeptidyl Peptidase-4 (DPP-4) Inhibitors in Type 2 Diabetic Patients: A Cross-sectional Study by Swetha R Reghunath, Ashna Chackochan, Girish Thunga, Dinesh U Acharya, Kaniyoor Nagri Shivashankara, Attur Ravindra Prabhu and Leelavathi D Acharya in Clinical Medicine Insights: Endocrinology and Diabetes
Supplemental Material
sj-docx-2-end-10.1177_11795514241288645 – Supplemental material for Prevalence and Predictors of Adverse Events Associated With Dipeptidyl Peptidase-4 (DPP-4) Inhibitors in Type 2 Diabetic Patients: A Cross-sectional Study
Supplemental material, sj-docx-2-end-10.1177_11795514241288645 for Prevalence and Predictors of Adverse Events Associated With Dipeptidyl Peptidase-4 (DPP-4) Inhibitors in Type 2 Diabetic Patients: A Cross-sectional Study by Swetha R Reghunath, Ashna Chackochan, Girish Thunga, Dinesh U Acharya, Kaniyoor Nagri Shivashankara, Attur Ravindra Prabhu and Leelavathi D Acharya in Clinical Medicine Insights: Endocrinology and Diabetes
Supplemental Material
sj-docx-3-end-10.1177_11795514241288645 – Supplemental material for Prevalence and Predictors of Adverse Events Associated With Dipeptidyl Peptidase-4 (DPP-4) Inhibitors in Type 2 Diabetic Patients: A Cross-sectional Study
Supplemental material, sj-docx-3-end-10.1177_11795514241288645 for Prevalence and Predictors of Adverse Events Associated With Dipeptidyl Peptidase-4 (DPP-4) Inhibitors in Type 2 Diabetic Patients: A Cross-sectional Study by Swetha R Reghunath, Ashna Chackochan, Girish Thunga, Dinesh U Acharya, Kaniyoor Nagri Shivashankara, Attur Ravindra Prabhu and Leelavathi D Acharya in Clinical Medicine Insights: Endocrinology and Diabetes
Supplemental Material
sj-docx-4-end-10.1177_11795514241288645 – Supplemental material for Prevalence and Predictors of Adverse Events Associated With Dipeptidyl Peptidase-4 (DPP-4) Inhibitors in Type 2 Diabetic Patients: A Cross-sectional Study
Supplemental material, sj-docx-4-end-10.1177_11795514241288645 for Prevalence and Predictors of Adverse Events Associated With Dipeptidyl Peptidase-4 (DPP-4) Inhibitors in Type 2 Diabetic Patients: A Cross-sectional Study by Swetha R Reghunath, Ashna Chackochan, Girish Thunga, Dinesh U Acharya, Kaniyoor Nagri Shivashankara, Attur Ravindra Prabhu and Leelavathi D Acharya in Clinical Medicine Insights: Endocrinology and Diabetes
Footnotes
Acknowledgements
The authors would like to acknowledge Manipal Academy of Higher Education, Manipal College of Pharmaceutical Sciences, Department of Pharmacy Practice, Manipal, India, for providing all the support and facilities for the best possible completion of this work.
Declarations
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

